false
0001425205
0001425205
2024-02-16
2024-02-16
iso4217:USD
xbrli:shares
iso4217:USD
xbrli:shares
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM 8-K
Current Report
Pursuant to Section 13 or 15(d)
of the Securities Exchange Act of 1934
Date of Report (date of earliest event reported): February 16, 2024
IOVANCE BIOTHERAPEUTICS, INC.
(Exact Name of Registrant as Specified in
Charter)
| Delaware |
| (State of Incorporation) |
| |
| 001-36860 |
|
75-3254381 |
| Commission File Number |
|
(I.R.S. Employer Identification No.) |
| |
|
|
| 825
Industrial Road, Suite 400 |
|
|
| San Carlos, CA |
|
94070 |
| (Address of Principal Executive Offices) |
|
(Zip Code) |
| |
|
|
| (650) 260-7120 |
| (Registrant’s Telephone Number, Including Area Code) |
| |
Check the appropriate box below if the Form 8-K filing is intended
to simultaneously satisfy the filing obligation of the registrant under any of the following provisions:
|
¨ |
Written communications pursuant to Rule 425 under the Securities Act (17 CFR 230.425). |
|
¨ |
Soliciting material pursuant to Rule 14a-12 under the Exchange Act (17 CFR 240.14a-12). |
|
¨ |
Pre-commencement communications pursuant to Rule 14d-2(b) under the Exchange Act (17 CFR 240.14d-2(b)). |
|
¨ |
Pre-commencement communications pursuant to Rule 13e-4(c) under the Exchange Act (17 CFR 240.13e-4(c)). |
Indicate
by check mark whether the registrant is an emerging growth company as defined in as defined in Rule 405 of the Securities Act
of 1933 (§230.405 of this chapter) or Rule 12b-2 of the Securities Exchange Act of 1934 (§240.12b-2 of this chapter).
Emerging growth company ¨
If
an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for
complying with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act. ¨
Securities registered pursuant to Section 12(b) of the Act:
| Title of each class |
Trading
Symbol(s) |
Name of each exchange on which
registered |
| Common stock, par value $0.000041666 per value |
IOVA |
The Nasdaq Stock Market, LLC |
Item 2.02. Results of Operations
and Financial Condition.
Iovance
Biotherapeutics, Inc. (the “Company”) reports that as of December 31, 2023, the Company had approximately $346.3 million in
cash, cash equivalents, investments, and restricted cash ($114.9 million of cash and cash equivalents, $165.0 million in short-term investments,
and $66.4 million in restricted cash).
The
foregoing information reflects the Company's preliminary estimates with respect to cash, cash equivalents, investments, and restricted
cash. This announcement is not a comprehensive statement of the Company’s financial results and is subject to completion of an audit
by the Company’s independent registered public accounting firm. The Company’s final financial results will be issued upon
completion of such audit and may vary from these preliminary estimates.
Item 8.01. Other Events.
On
February 16, 2024, the Company issued a press release announcing that AMTAGVI® (lifileucel) has received U.S. Food and
Drug Administration accelerated approval for the treatment of advanced melanoma. The Company also updated its corporate presentation that
it uses for presentations at healthcare conferences and to analysts, current stockholders, and others.
A
copy of the press release and the Company’s corporate presentation are attached hereto as Exhibit 99.1 and Exhibit 99.2, respectively,
and are incorporated by reference herein.
The
information contained in Item 2.02 of this Current Report on Form 8-K is incorporated by reference herein.
Item 9.01. Financial
Statements and Exhibits.
(d) Exhibits.
SIGNATURES
Pursuant
to the requirements of the Securities Exchange Act of 1934, as amended, the registrant has duly caused this report to be signed on its
behalf by the undersigned hereunto duly authorized.
| |
Iovance Biotherapeutics, Inc. |
| |
|
|
| |
|
|
| Dated: February 20, 2024 |
By: |
/s/ Frederick G. Vogt |
| |
Name: |
Frederick G. Vogt, Ph.D., J.D. |
| |
Title: |
Interim CEO and General Counsel |
Exhibit 99.1

Iovance’s AMTAGVITM (lifileucel)
Receives U.S. FDA Accelerated Approval
for Advanced Melanoma
AMTAGVI is the first FDA-approved T cell therapy
for a solid tumor cancer and first treatment option for advanced melanoma after anti-PD-1 and targeted therapy
AMTAGVI deploys patient-specific immune cells
that recognize and fight cancer
SAN CARLOS, Calif., February 16, 2024 -- Iovance
Biotherapeutics, Inc. (NASDAQ: IOVA), a biotechnology company focused on innovating, developing and delivering novel polyclonal tumor
infiltrating lymphocyte (TIL) cell therapies for patients with cancer, today announced that the U.S. Food and Drug Administration (FDA)
has approved AMTAGVITM (lifileucel) suspension for intravenous infusion. AMTAGVI is a tumor-derived autologous T cell immunotherapy
indicated for the treatment of adult patients with unresectable or metastatic melanoma previously treated with a PD-1 blocking antibody,
and if BRAF V600 mutation positive, a BRAF inhibitor with or without a MEK inhibitor. This indication is approved under an accelerated
approval based on overall response rate (ORR) and duration of response. Iovance is also conducting TILVANCE-301, a Phase 3 trial to confirm
clinical benefit.
AMTAGVI is the first and the only one-time, individualized T cell
therapy to receive FDA approval for a solid tumor cancer. The proposed mechanism for AMTAGVI offers a new cell therapy approach that
deploys patient-specific T cells called TIL cells. When cancer is detected, the immune system creates TIL cells to locate, attack, and
destroy cancer. TIL cells recognize distinctive tumor markers on the cell surface of each person’s cancer. When cancer develops
and prevails, the body’s natural TIL cells can no longer perform their intended function to fight cancer.
AMTAGVI is manufactured using a proprietary process to collect and
expand a patient’s unique T cells from a portion of their tumor. AMTAGVI returns billions of the patient’s T cells back to
the body to fight their cancer.* Authorized Treatment Centers (ATCs) will administer AMTAGVI to patients as part of a treatment regimen
that includes lymphodepletion and a short course of high-dose PROLEUKIN® (aldesleukin).
“The accelerated approval of AMTAGVITM is the first
step in realizing Iovance’s ambition to usher in the next generation of cell therapy by bringing this breakthrough to patients
with advanced solid tumors,” said Frederick Vogt, Ph.D., J.D., Interim Chief Executive Officer and President of Iovance. “Given
the significant unmet needs in the advanced melanoma community, we are proud to offer a personalized, one-time therapeutic option for
these patients. We are continuing our development efforts to address additional unmet medical needs in patients with solid tumor cancers,
making our novel cell therapies available to more patients with melanoma and other types of cancers.”
Each year, approximately 8,000 people in the U.S. die from melanoma.1
Until now, there have been no FDA-approved treatment options for patients with advanced melanoma whose disease progressed following
initial treatment with an immune checkpoint inhibitor and, if appropriate, targeted therapy.
“The approval of AMTAGVITM offers hope to those with
advanced melanoma, who have progressed following initial standard of care therapies, as the current treatment options are not effective
for many patients,” said Samantha R. Guild, J.D., President, AIM at Melanoma Foundation. “This one-time cell therapy represents
a promising innovation for the melanoma community, and we are excited by its potential to transform care for patients who are in dire
need of additional therapeutic options.”
The FDA approval is based on safety and efficacy results from the
C-144-01 clinical trial. C-144-01 is a global, multicenter trial investigating AMTAGVI in patients with advanced melanoma previously
treated with anti-PD-1 therapy and targeted therapy, where applicable. AMTAGVI demonstrated deep and durable responses. The primary efficacy
analysis set included 73 patients from Cohort 4 who received the recommended AMTAGVI dose from an approved manufacturing facility. Among
the 73 patients, 31.5% achieved an objective response by Response Evaluation Criteria in Solid Tumors (RECIST 1.1) with a median duration
of response not reached at 18.6 months follow up,2 (43.5% of responses had a duration greater than 12 months). Additionally,
the supporting pooled efficacy set included a total of 153 patients from Cohort 4 and Cohort 2. Among these 153 patients, 31.4% achieved
an objective response by RECIST 1.1 with a median duration of response not reached at 21.5 months follow up,2 (54.2% of responses
had a duration greater than 12 months). The detailed results of clinical trial C-144-01 are published in The Journal for ImmunoTherapy
of Cancer (Chesney 2022).
AMTAGVI is for autologous use only. AMTAGVI has a boxed warning for
treatment-related mortality, prolonged severe cytopenia, severe infection, and cardiopulmonary and renal impairment. Warnings and precautions
include treatment-related mortality, prolonged severe cytopenia, internal organ hemorrhage, severe infection, cardiac disorder, respiratory
failure, acute renal failure, and hypersensitivity reactions. Please see Important Safety Information and Prescribing Information below.
“This landmark FDA approval reflects significant advancements
in TIL cell therapy since we initially showed that TIL cells isolated from patients with metastatic melanoma could be expanded in the
lab and returned to the patient to mediate cancer regression,” said Steven Rosenberg, M.D., Ph.D., Chief, Surgery Branch, National
Cancer Institute, and a TIL and immunotherapy pioneer. “This approval is transformative for the entire research field and supports
continued investigation of TIL cell therapy across additional types of solid tumors.”
“One-time treatment with AMTAGVITM offered clinically
meaningful and deep, durable responses in the Phase 2 clinical trial, and I am excited by its potential as a much-needed new treatment
option for the many advanced melanoma patients who progress on the current standard of care,” said Dr. Alexander N. Shoushtari,
Melanoma Oncologist & Cellular Therapist at Memorial Sloan Kettering Cancer Center. “This welcome news represents an important
step forward in harnessing cell therapy to treat solid tumors,” added Dr. Jae Park, Chief of Cellular Therapy Service at Memorial
Sloan Kettering Cancer Center.
AMTAGVI will be manufactured in Philadelphia at the Iovance Cell Therapy
Center (iCTC), with capacity for up to several thousand patients annually, including a nearby contract manufacturer. Additional expansion
at iCTC is underway which will significantly increase this capacity over the next few years. iCTC is the first FDA-approved, centralized,
and scalable manufacturing facility dedicated to producing TIL cell therapies for patients with solid tumors. AMTAGVI must be administered
in an ATC, and more than 30 ATCs are prepared to collect and ship tumor tissue from patients for AMTAGVI manufacturing.
Iovance is dedicated to providing access to AMTAGVI for patients with
advanced melanoma. A comprehensive support program, IovanceCaresTM, is now available for patients and ATCs throughout the
treatment journey. IovanceCares will also offer copay support, financial assistance, and travel and lodging assistance for eligible patients
during AMTAGVI therapy. For more information, physicians and patients may call 833-400-IOVA (4682) or visit www.iovancecares.com.
Iovance is investigating AMTAGVI in frontline advanced melanoma in
the Phase 3 confirmatory trial, TILVANCE-301, as well as additional solid tumor types, which represent 91% of the cancers in the U.S.1
For more information, please visit: https://www.iovance.com/clinical-trials/.
1National Cancer Institute Surveillance, Epidemiology and
End Results (SEER) Program. 2023 Estimates. https://seer.cancer.gov accessed February 2024.
2Kaplan-Meier estimate of median potential follow-up for
duration of response.
*A single dose of AMTAGVI contains 7.5 × 109 to 72
× 109 viable cells.
Webcast and Conference Call
Iovance
will host a conference call and live audio webcast to discuss the FDA approval of AMTAGVI today, details will be shared in a subsequent
announcement.
About the C-144-01 Clinical Trial
C-144-01 is a global, multicenter Phase 2 study in which patients
received treatment with lifileucel. The study enrolled patients with metastatic melanoma who were previously treated with at least one
systemic therapy, including a PD-1 blocking antibody, and if BRAF V600 mutation-positive, a BRAF inhibitor or BRAF inhibitor with MEK
inhibitor. Efficacy was established on the basis of objective response rate (ORR), and duration of response (DOR) by Independent Review
Committee (IRC) per Response Evaluation Criteria in Solid Tumors (RECIST) version 1.1. The pivotal Cohort 4 and supportive Cohort 2 of
Study C-144-01 enrolled patients that met the same primary eligibility criteria, had the same assessments, and had received the same
regimen and AMTAGVI that was produced using the same manufacturing process, and product formulation. The detailed results of C-144-01
were published in the Journal for ImmunoTherapy of Cancer in 2022.
What is AMTAGVI (lifileucel)?
AMTAGVI is a prescription medicine used to treat adults with a type
of skin cancer that cannot be removed surgically or has spread to other parts of the body called unresectable or metastatic melanoma.
AMTAGVI is used when your melanoma has not responded or stopped responding
to a PD-1 blocking drug either by itself or in a combination, and if your cancer is BRAF mutation positive, a BRAF inhibitor drug with
or without a MEK inhibitor drug that has also stopped working.
The approval of AMTAGVI is based on a study that measured response
rate. Continued approval for this use may depend on the results of an ongoing study to confirm benefit.
Important Safety Information
What is the most important information that I should know about
AMTAGVI?
You will likely be in a hospital prior to and after receiving AMTAGVI.
Before taking AMTAGVI, tell your healthcare provider about all
of your medical conditions, including if you:
| · | Have
any lung, heart, liver or kidney problems |
| · | Have
low blood pressure |
| · | Have
a recent or active infection or other inflammatory conditions including cytomegalovirus (CMV)
infection, hepatitis B or C or human immunodeficiency virus (HIV) infection |
| · | Are
pregnant, think you may be pregnant, or plan to become pregnant |
| · | Are
breastfeeding |
| · | Notice
the symptoms of your cancer are getting worse |
| · | Have
had a vaccination in the past 28 days or plan to have one in the next few months |
| · | Have
been taking a blood thinner |
Tell your doctor about all the medications you take, including
prescription and over-the-counter medicines, vitamins, and herbal supplements.
How will I receive AMTAGVI?
| · | AMTAGVI
is made from your surgically removed tumor. Tumor derived T cells are grown in a manufacturing
center at the end of which they number in the billions of cells. |
| · | Your
tumor tissue is sent to a manufacturing center to make AMTAGVI. It takes about 34 days from
the time your tumor tissue is received at the manufacturing center until AMTAGVI is available
to be shipped back to your healthcare provider, but the time may vary. Your AMTAGVI will
be provided in 1-4 patient-specific infusion bag(s) containing 100 mL to 125 mL of viable
(alive) cells per bag. |
| · | After
your AMTAGVI arrives at your treating institution, your healthcare provider will give you
lymphodepleting chemotherapy to prepare your body. |
| · | Approximately
30 to 60 minutes before you are given AMTAGVI, you may be given other medicines including: |
| o | Medicines for an allergic reaction (anti-histamines) |
| o | Medicines for fever (such as acetaminophen) |
| · | Your
AMTAGVI will be provided in 1 to 4 infusion bag(s) containing 100 mL to 125 mL of viable
cells per bag. When your body is ready for AMTAGVI infusion, your healthcare provider will
give AMTAGVI to you by intravenous infusion. This usually takes less than 90 minutes. |
After getting AMTAGVI
Beginning 3 to 24 hours after AMTAGVI is given, you may be given up
to 6 doses of IL-2 (aldesleukin) every 8 to 12 hours via intravenous infusion. Your doctor may discontinue IL-2 (aldesleukin) infusion
any time if you have severe side effects.
You will have to stay in the hospital until you have completed the
IL-2 (aldesleukin) treatment and you have recovered from any serious side effects associated with the AMTAGVI treatment.
You should plan to stay within 2 hours of the location where you received
your treatment for several weeks after getting AMTAGVI. Your healthcare provider will check to see if your treatment is working and help
you with any side effects that occur.
What are the possible side effects of AMTAGVI?
The most common side effects of the AMTAGVI treatment include chills,
fever, low white blood cell count (may increase risk of infections), fatigue, low red blood cell count (may cause you to feel tired or
weak), fast or irregular heartbeat, rash, low blood pressure, and diarrhea.
These are not all the possible side effects of the AMTAGVI treatment.
Talk with your healthcare provider for more information about AMTAGVI. You can ask your healthcare provider for information about AMTAGVI
that is written for healthcare professionals.
You may report side effects to Iovance at 1-833-400-4682, or to the
FDA, at 1-800-FDA-1088 or at www.fda.gov/medwatch.
Please see Full Prescribing Information and Patient Information, including
Boxed Warning, for additional Important Safety Information.
About Iovance Biotherapeutics, Inc.
Iovance Biotherapeutics aims to be the global leader in innovating,
developing and delivering tumor infiltrating lymphocyte (TIL) cell therapies for patients with cancer. We are pioneering a transformational
approach to cure cancer by harnessing the human immune system’s ability to recognize and destroy diverse cancer cells in each patient.
The Iovance TIL platform has demonstrated promising clinical data across multiple solid tumors. Iovance’s AMTAGVI™
is the first FDA-approved T cell therapy for a solid tumor indication. We are committed to continuous innovation in cell therapy, including
gene-edited cell therapy, which may be a promising option for patients with cancer. For more information, please visit www.iovance.com.
AMTAGVITM and its accompanying
design marks, PROLEUKIN®, IOVANCE®, and IOVANCECARESTM are trademarks and registered trademarks of Iovance Biotherapeutics,
Inc. or its subsidiaries. All other trademarks and registered trademarks are the property of their respective owners.
Forward-Looking Statements
Certain matters discussed in this press release are “forward-looking
statements” of Iovance Biotherapeutics, Inc. (hereinafter referred to as the “Company,” “we,” “us,”
or “our”) within the meaning of the Private Securities Litigation Reform Act of 1995 (the “PSLRA”). All such
written or oral statements, including, without limitation, the statements by Frederick Vogt, Ph.D., J.D., Samantha R. Guild, J.D., Steven
Rosenberg, M.D., Ph.D., Dr. Alexander N. Shoushtari, and Dr. Jae Park, made in this press release, other than statements of historical
fact, are forward-looking statements and are intended to be covered by the safe harbor for forward-looking statements provided by the
PSLRA. Without limiting the foregoing, we may, in some cases, use terms such as “predicts,” “believes,” “potential,”
“continue,” “estimates,” “anticipates,” “expects,” “plans,” “intends,”
“forecast,” “guidance,” “outlook,” “may,” “could,” “might,” “will,”
“should,” or other words that convey uncertainty of future events or outcomes and are intended to identify forward-looking
statements. Forward-looking statements are based on assumptions and assessments made in light of management’s experience and perception
of historical trends, current conditions, expected future developments, and other factors believed to be appropriate. Forward-looking
statements in this press release are made as of the date of this press release, and we undertake no duty to update or revise any such
statements, whether as a result of new information, future events or otherwise. Forward-looking statements are not guarantees of future
performance and are subject to risks, uncertainties, and other factors, many of which are outside of our control, that may cause actual
results, levels of activity, performance, achievements, and developments to be materially different from those expressed in or implied
by these forward-looking statements. Important factors that could cause actual results, developments, and business decisions to differ
materially from forward-looking statements are described in the sections titled "Risk Factors" in our filings with the U.S.
Securities and Exchange Commission, including our most recent Annual Report on Form 10-K and Quarterly Reports on Form 10-Q, and include,
but are not limited to, the following substantial known and unknown risks and uncertainties inherent in our business: the risks related
to our ability to successfully commercialize our products, including AMTAGVI, for which we obtain U.S. Food and Drug Administration (“FDA”),
European Medicines Agency (“EMA”), or other regulatory authority approval; the risk that the EMA or other regulatory authorities
may not approve or may delay approval for our biologics license application (“BLA”) submission for lifileucel in metastatic
melanoma; the acceptance by the market of our products, including AMTAGVI, and their potential pricing and/or reimbursement by payors,
if approved (in the case of our product candidates), in the U.S. and other international markets and whether such acceptance is sufficient
to support continued commercialization or development of our products, including AMTAGVI, or product candidates, respectively; our ability
or inability to manufacture our therapies using third party manufacturers or at our own facility may adversely affect our commercial
launch; the results of clinical trials with collaborators using different manufacturing processes may not be reflected in our sponsored
trials; the risk regarding the successful integration of the recent Proleukin acquisition; the risk that the successful development or
commercialization of our products, including AMTAGVI, may not generate sufficient revenue from product sales, and we may not become profitable
in the near term, or at all; the risk that future competitive or other market factors may adversely affect the commercial potential for
AMTAGVI; the risks related to the timing of and our ability to successfully develop, submit, obtain, or maintain FDA, EMA, or other regulatory
authority approval of, or other action with respect to, our product candidates; whether clinical trial results from our pivotal studies
and cohorts, and meetings with the FDA, EMA, or other regulatory authorities may support registrational studies and subsequent approvals
by the FDA, EMA, or other regulatory authorities, including the risk that the planned single arm Phase 2 IOV-LUN-202 trial may not support
registration; preliminary and interim clinical results, which may include efficacy and safety results, from ongoing clinical trials or
cohorts may not be reflected in the final analyses of our ongoing clinical trials or subgroups within these trials or in other prior
trials or cohorts; the risk that enrollment may need to be adjusted for our trials and cohorts within those trials based on FDA and other
regulatory agency input; the risk that the changing landscape of care for cervical cancer patients may impact our clinical trials in
this indication; the risk that we may be required to conduct additional clinical trials or modify ongoing or future clinical trials based
on feedback from the FDA, EMA, or other regulatory authorities; the risk that our interpretation of the results of our clinical trials
or communications with the FDA, EMA, or other regulatory authorities may differ from the interpretation of such results or communications
by such regulatory authorities (including from our prior meetings with the FDA regarding our non-small cell lung cancer clinical trials);
the risk that clinical data from ongoing clinical trials of AMTAGVI will not continue or be repeated in ongoing or planned clinical trials
or may not support regulatory approval or renewal of authorization; the risk that unanticipated expenses may decrease our estimated cash
balances and forecasts and increase our estimated capital requirements; the effects of the COVID-19 pandemic; and other factors, including
general economic conditions and regulatory developments, not within our control.
CONTACTS
Iovance Biotherapeutics, Inc:
Sara Pellegrino, IRC
Senior Vice President, Investor Relations & Corporate Communications
650-260-7120 ext. 264
Sara.Pellegrino@iovance.com
Jen Saunders
Senior Director, Investor Relations & Corporate Communications
267-485-3119
Jen.Saunders@iovance.com
Exhibit 99.2

1 © 2024, Iovance Biotherapeutics, Inc. © 2024, Iovance Biotherapeutics, Inc. Corporate Overview February 16, 2024 1

2 © 2024, Iovance Biotherapeutics, Inc. Forward - Looking Statements Certain matters discussed in this press release are “forward - looking statements” of Iovance Biotherapeutics, Inc. (hereinafter r eferred to as the “Company,” “we,” “us,” or “our”) within the meaning of the Private Securities Litigation Reform Act of 1995 (the “PSLRA”). Without limiting the foregoing, we may, in so me cases, use terms such as “predicts,” “believes,” “potential,” “continue,” “estimates,” “anticipates,” “expects,” “plans,” “intends,” “forecast,” “guidance,” “outlook,” “may,” “c ould,” “might,” “will,” “should,” or other words that convey uncertainty of future events or outcomes and are intended to identify forward - looking statements. Forward - looking stateme nts are based on assumptions and assessments made in light of management’s experience and perception of historical trends, current conditions, expected future development s, and other factors believed to be appropriate. Forward - looking statements in this press release are made as of the date of this press release, and we undertake no duty to update or re vise any such statements, whether as a result of new information, future events or otherwise. Forward - looking statements are not guarantees of future performance and are subject to risks, uncertainties, and other factors, many of which are outside of our control, that may cause actual results, levels of activity, performance, achievements, and developments to be materially different from those expressed in or implied by these forward - looking statements. Important factors that could cause actual results, developments, and business decisions to dif fer materially from forward - looking statements are described in the sections titled "Risk Factors" in our filings with the U.S. Securities and Exchange Commission, including ou r m ost recent Annual Report on Form 10 - K and Quarterly Reports on Form 10 - Q, and include, but are not limited to, the following substantial known and unknown risks and uncertainties i nherent in our business: the risks related to our ability to successfully commercialize our products, including AMTAGVI, for which we obtain U.S. Food and Drug Administration (“FDA”), Eu rop ean Medicines Agency (“EMA”), or other regulatory authority approval; the risk that the EMA or other regulatory authorities may not approve or may delay approval for our biolo gic s license application (“BLA”) submission for lifileucel in metastatic melanoma; the acceptance by the market of our products, including AMTAGVI, and their potential pricing and/or reim bur sement by payors, if approved (in the case of our product candidates), in the U.S. and other international markets and whether such acceptance is sufficient to support continu ed commercialization or development of our products, including AMTAGVI, or product candidates, respectively; our ability or inability to manufacture our therapies using third par ty manufacturers or at our own facility may adversely affect our commercial launch; the results of clinical trials with collaborators using different manufacturing processes may not be refle cte d in our sponsored trials; the risk regarding the successful integration of the recent Proleukin acquisition; the risk that the successful development or commercialization of our product s, including AMTAGVI, may not generate sufficient revenue from product sales, and we may not become profitable in the near term, or at all; the risk that future competitive or other m ark et factors may adversely affect the commercial potential for AMTAGVI; the risks related to the timing of and our ability to successfully develop, submit, obtain, or maintain FDA, EMA, or ot her regulatory authority approval of, or other action with respect to, our product candidates; whether clinical trial results from our pivotal studies and cohorts, and meetings with th e F DA, EMA, or other regulatory authorities may support registrational studies and subsequent approvals by the FDA, EMA, or other regulatory authorities, including the risk that the pl anned single arm Phase 2 IOV - LUN - 202 trial may not support registration; preliminary and interim clinical results, which may include efficacy and safety results, from ongoing c lin ical trials or cohorts may not be reflected in the final analyses of our ongoing clinical trials or subgroups within these trials or in other prior trials or cohorts; the risk that enrollment ma y need to be adjusted for our trials and cohorts within those trials based on FDA and other regulatory agency input; the risk that the changing landscape of care for cervical cancer patients may im pact our clinical trials in this indication; the risk that we may be required to conduct additional clinical trials or modify ongoing or future clinical trials based on feedback from the FDA , EMA, or other regulatory authorities; the risk that our interpretation of the results of our clinical trials or communications with the FDA, EMA, or other regulatory authorities may di ffer from the interpretation of such results or communications by such regulatory authorities (including from our prior meetings with the FDA regarding our non - small cell lung cancer clinical trials); the risk that clinical data from ongoing clinical trials of AMTAGVI will not continue or be repeated in ongoing or planned clinical trials or may not support reg ulatory approval or renewal of authorization; the risk that unanticipated expenses may decrease our estimated cash balances and forecasts and increase our estimated capital requirements ; t he effects of the COVID - 19 pandemic; and other factors, including general economic conditions and regulatory developments, not within our control.
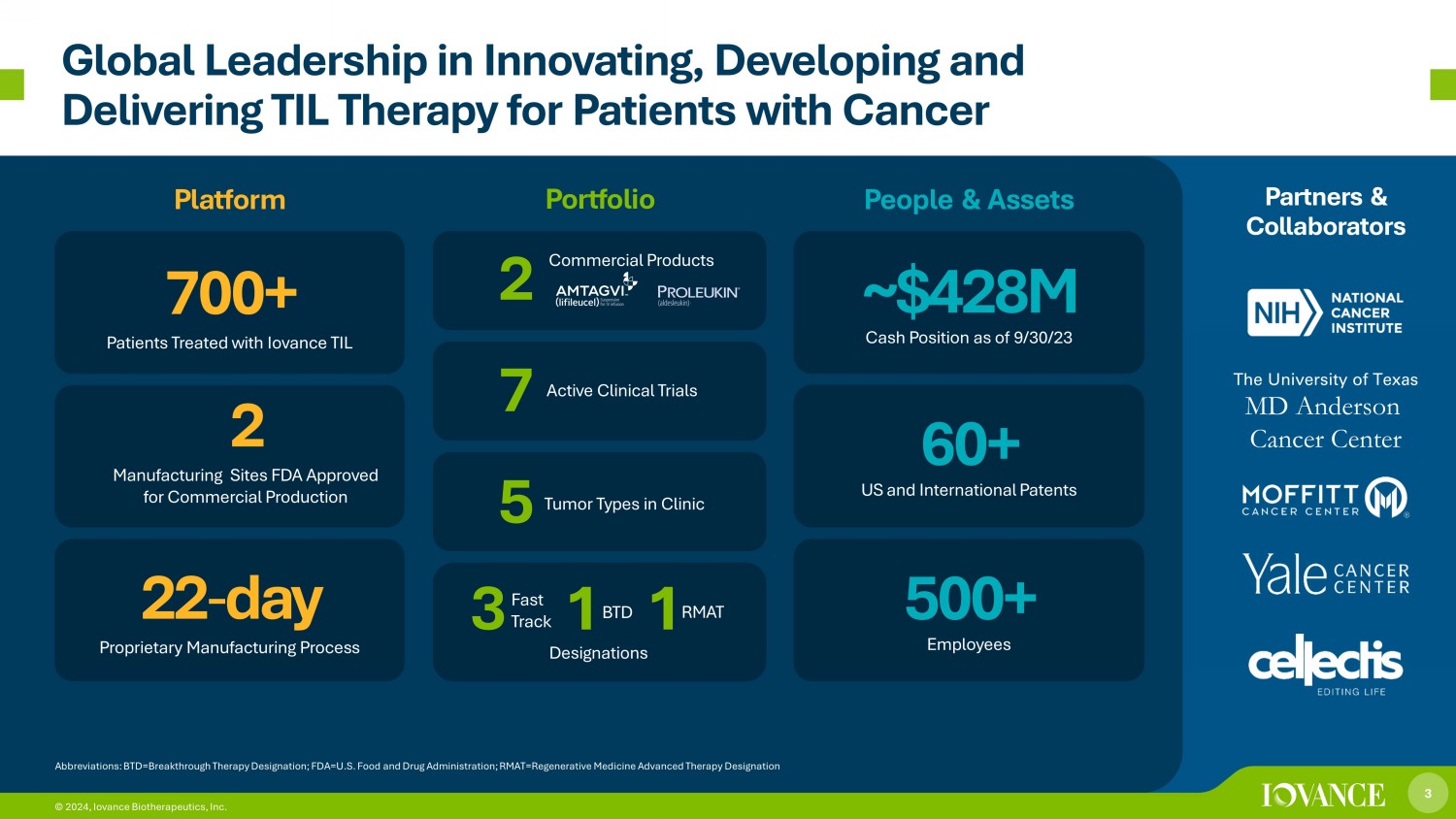
3 © 2024, Iovance Biotherapeutics, Inc. Global Leadership in Innovating, Developing and Delivering TIL Therapy for Patients with Cancer Platform Portfolio People & Assets Partners & Collaborators Abbreviations: BTD=Breakthrough Therapy Designation; FDA=U.S. Food and Drug Administration; RMAT =Regenerative Medicine Advanced Therapy Designation 22 - day Proprietary Manufacturing Process 2 Manufacturing Sites FDA Approved for Commercial Production 7 00+ Patients Treated with Iovance TIL ~$ 428 M Cash Position as of 9/30/23 60+ US and International Patents 500+ Employees The University of Texas MD Anderson Cancer Center 3 © 2024, Iovance Biotherapeutics, Inc. 3 1 1 Designations Active Clinical Trials 7 Tumor Types in Clinic 5 Commercial Products Fast Track BTD RMAT 2

4 © 2024, Iovance Biotherapeutics, Inc. *Enrollment complete Abbreviations: 1L=first line; 2L=second line; 4L=fourth line; BTD=Breakthrough Therapy Designation; FTD=Fast Track Designatio n; ipi/nivo=ipilimumab/nivolumab; NSCLC=non - small cell lung cancer; ODD=Orphan Drug Designation; PD - 1=programmed cell death protein - 1; RMAT=Regenerative Medicines Advanced Therapy; TIL=tumor infiltrating lymphocy tes Iovance Solid Tumor Portfolio Highlights Confirmatory, FTD BTD, ODD AMTAGVI treatment regimen A dvanced melanoma, renal cell carcinoma Post - anti - PD - 1 advanced melanoma NOW APPROVED CANDIDATE INDICATIONS PHASE 1 PHASE 2 PHASE 3 Registration - Directed Lifileucel + pembro Frontline advanced melanoma TILVANCE - 301 Phase 3 LN - 145 2L post - chemo & post - anti - PD - 1 advanced NSCLC IOV - LUN - 202: Cohorts 1&2 Lifileucel Post - chemo & post - anti - PD - 1 cervical C - 145 - 04: Cohort 2 Additional Pipeline LN - 145 + pembro 1L chemo and anti - PD - 1 naïve cervical C - 145 - 04: Cohort 3* Lifileucel 2L post - chemo & post - anti - PD - 1 endometrial Planned Phase 2 LN - 145, LN - 145 + ICI 2 - 4L incl. post - anti - PD - 1 advanced NSCLC IOV - COM - 202: Cohorts 3A, 3B*,3C LN - 145 + ICI 1L advanced melanoma IOV - COM - 202: Cohort 1A Next Generation PD - 1 Inactivated TIL (IOV - 4001) Post anti - PD1 advanced melanoma IOV - GM1 - 201: Cohort 1 PD - 1 Inactivated TIL (IOV - 4001) 2 - 4L incl. post - anti - PD - 1 advanced NSCLC IOV - GM1 - 201: Cohort 2 LN - 145 Gen 3 + core biopsy 2L post - chemo & post - anti - PD - 1 advanced NSCLC IOV - LUN - 202: Cohort 3
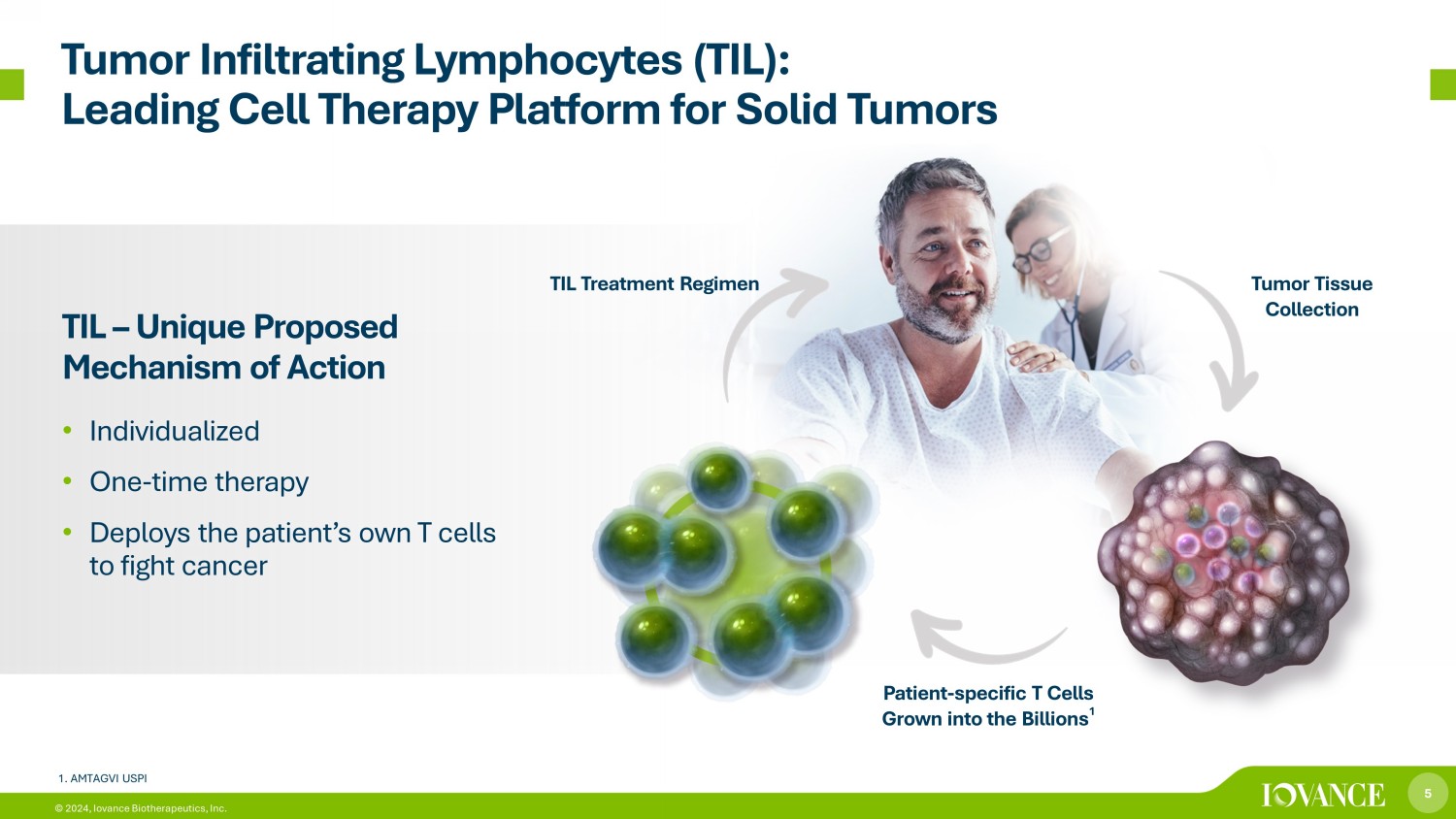
5 © 2024, Iovance Biotherapeutics, Inc. Tumor Infiltrating Lymphocytes (TIL): Leading Cell Therapy Platform for Solid Tumors TIL – Unique Proposed Mechanism of Action • Individualized • One - time therapy • Deploys the patient’s own T cells to fight cancer Tumor Tissue Collection Patient - specific T Cells Grown into the Billions 1 TIL Treatment Regimen 1. AMTAGVI USPI

6 © 2024, Iovance Biotherapeutics, Inc. 6 AMTAGVI ¯ (lifileucel): First and only one - time, individualized T cell therapy approved by FDA for a solid tumor cancer

7 © 2024, Iovance Biotherapeutics, Inc. 1. AMTAGVI USPI

8 © 2024, Iovance Biotherapeutics, Inc. U.S. Unmet Medical Need for Metastatic Melanoma Therapy AMTAGVI is th e First FDA Approved Treatment Option After Progression on ICI (Anti - PD - 1) Therapy and BRAF/MEK inhibitors More than half of patients progress within 12 months on current 1L ICIs , regardless of BRAF mutation status 4 High unmet need for patients who progress after immune checkpoint inhibitors 9.9K 6.3K 4.8K 1L 2L 3L-4L Melanoma Drug - Treated Population in 2021 Unresectable / Metastatic (US) 3 Abbreviations: 1L=first line therapy, 2L=second line therapy, 3L=third line therapy, 4L=fourth line therapy; ICI=immune check poi nt inhibitor; PD - 1=programmed cell death protein - 1 15k Annual new cases of advanced melanoma in U.S. 1 8k Annual deaths in U.S. 2 1. Estimate of US incidence (2021) of unresectable or metastatic melanoma based on secondary and primary market research 2. National Cancer Institute Surveillance, Epidemiology and End Results (SEER) Program. 2023 Estimates. https://seer.cancer.gov accessed February 2024 3. Clarivate DRG Disease Landscape (2021) 4. Larkin et al, NEJM 2019; Robert et al, Lancet Oncology 2019; Tawbi et al, NEJM 2022

9 © 2024, Iovance Biotherapeutics, Inc. AMTAGVI ¯ Delivered Deep and Durable Responses ORR 31.5 % mDOR Not Reached (95% CI: 21.1, 43.4) mDOR Not Reached ORR 31.4 % (95% CI: 24.1, 39.4) (Range: 1 .4 +, 45.0+) (Range: 1.4+, 26.3+; 95% CI: 4.1, NR ) 18.6 months follow up 21.5 months follow up 2 1. AMTAGVI USPI 2. Data on file. CI, confidence interval; mDOR, median duration of response; NR, not reached. Cohort 4 Pivotal 1 (N=73) Supportive Pooled Data 1 (n=153)
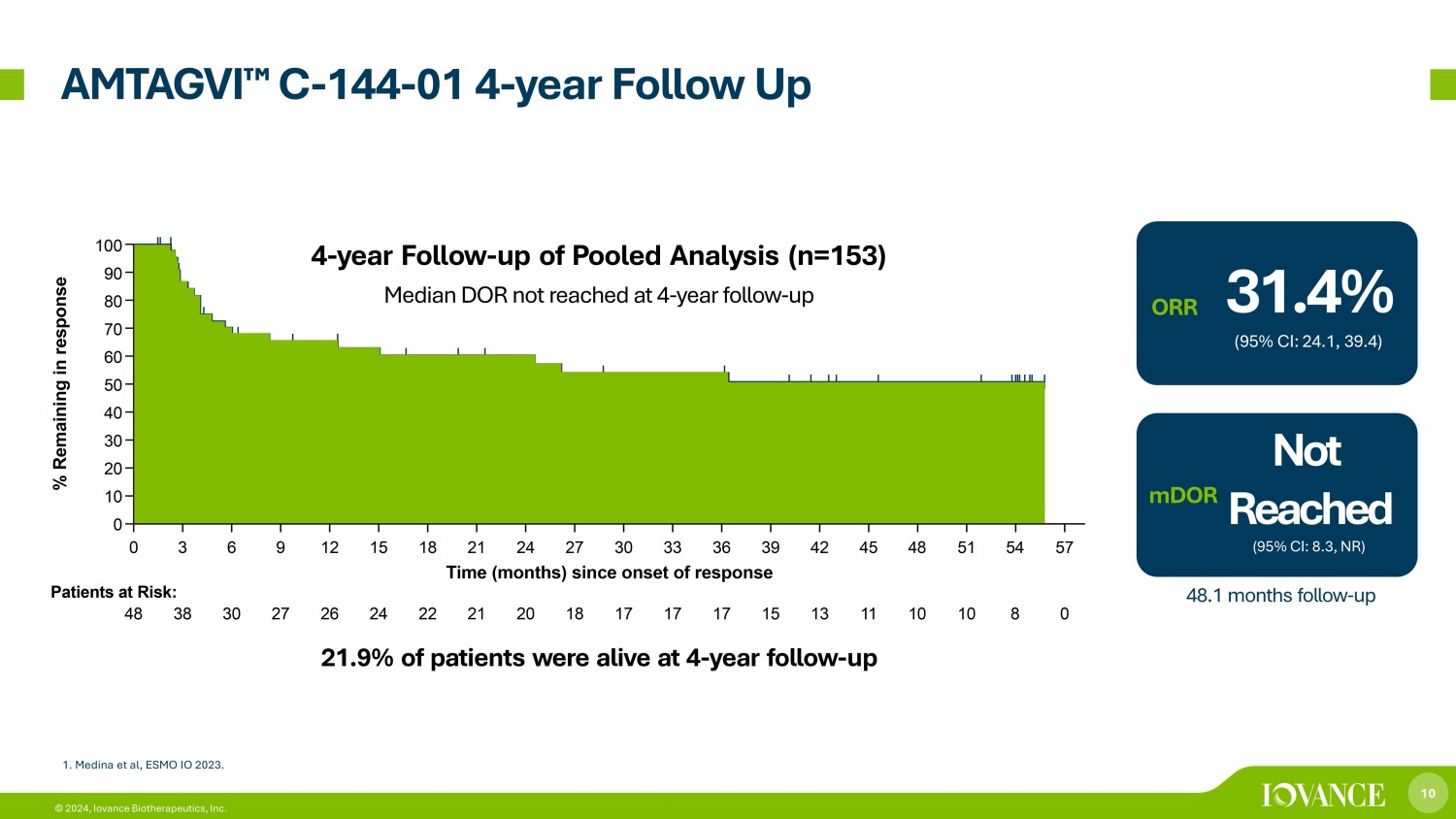
10 © 2024, Iovance Biotherapeutics, Inc. AMTAGVI ¯ C - 144 - 01 4 - year Follow Up Median DOR not reached at 4 - year follow - up 4 - year Follow - up of Pooled Analysis (n=153) 31.4% (95% CI: 24.1, 39.4) ORR Not Reached (95% CI: 8.3, NR) mDOR 48.1 months follow - up 21.9% of patients were alive at 4 - year follow - up 1. Medina et al, ESMO IO 2023 .

11 © 2024, Iovance Biotherapeutics, Inc. AMTAGVI ¯ Patient Journey AMTAGVI Autologous T Cell Therapy ) AMTAGVI starts with a piece of the patient’s tumor tissue • Lymphodepletion • AMTAGVI 䉼 (lifileucel) • Short - Course Proleukin® Scheduling & Tumor Tissue Procurement Treatment Regimen & Monitoring Treatment Decision & Reimbursement Approval T Cell Therapy Manufacturing & Release Testing TIL cells are grown into the billions in a manufacturing facility

12 © 2024, Iovance Biotherapeutics, Inc. Iovance Cell Therapy Center: i CTC • Built - to - suit custom facility in Navy Yard Philadelphia • Annual capacity for up to several thousand patients as built • Expansion underway for additional capacity within iCTC over next few years • Additional CDMO capacity • Control to optimize capacity, quality & COGS FDA - Approved Cell Therapy Manufacturing Facility Dedicated to Commercial and Clinical TIL Cell Therapies

13 © 2024, Iovance Biotherapeutics, Inc. Iovance Cell Therapy Center ( i CTC): Capacity Expansion Plans Pre - Approval (Complete) 100s of patients/year Launch Prep in core suites for commercial 4 separate flex suites for clinical Today (as built) up to 2,000 + patients/year 1 12 core suites for commercial 4 separate flex suites for clinical Site Expansion (in progress) 2 5,000+ patients/year 24 core suites for commercial 4 separate flex suites for clinical i CTC + Future Site(s) 10,000+ patients/year i CTC Adjacent and new sites 3 Automation 13 © 2024, Iovance Biotherapeutics, Inc. 1. Ongoing staffing 2. Expansion within existing shell 3. Option to build on adjacent parcel

14 © 2024, Iovance Biotherapeutics, Inc. Proleukin® (aldesleukin) Strategic Benefits • Significant revenue anticipated with AMTAGVI ¯ launch • IL - 2 supply chain secured for AMTAGVI regimen • Lower clinical trial costs and COGS anticipated £167.7M Upfront investment Short course Proleukin® is administered after AMTAGVI® to promote T cell growth in the body Key Transaction Figures £41.7M Following first lifileucel approval Global Rights Acquired in May of 2023
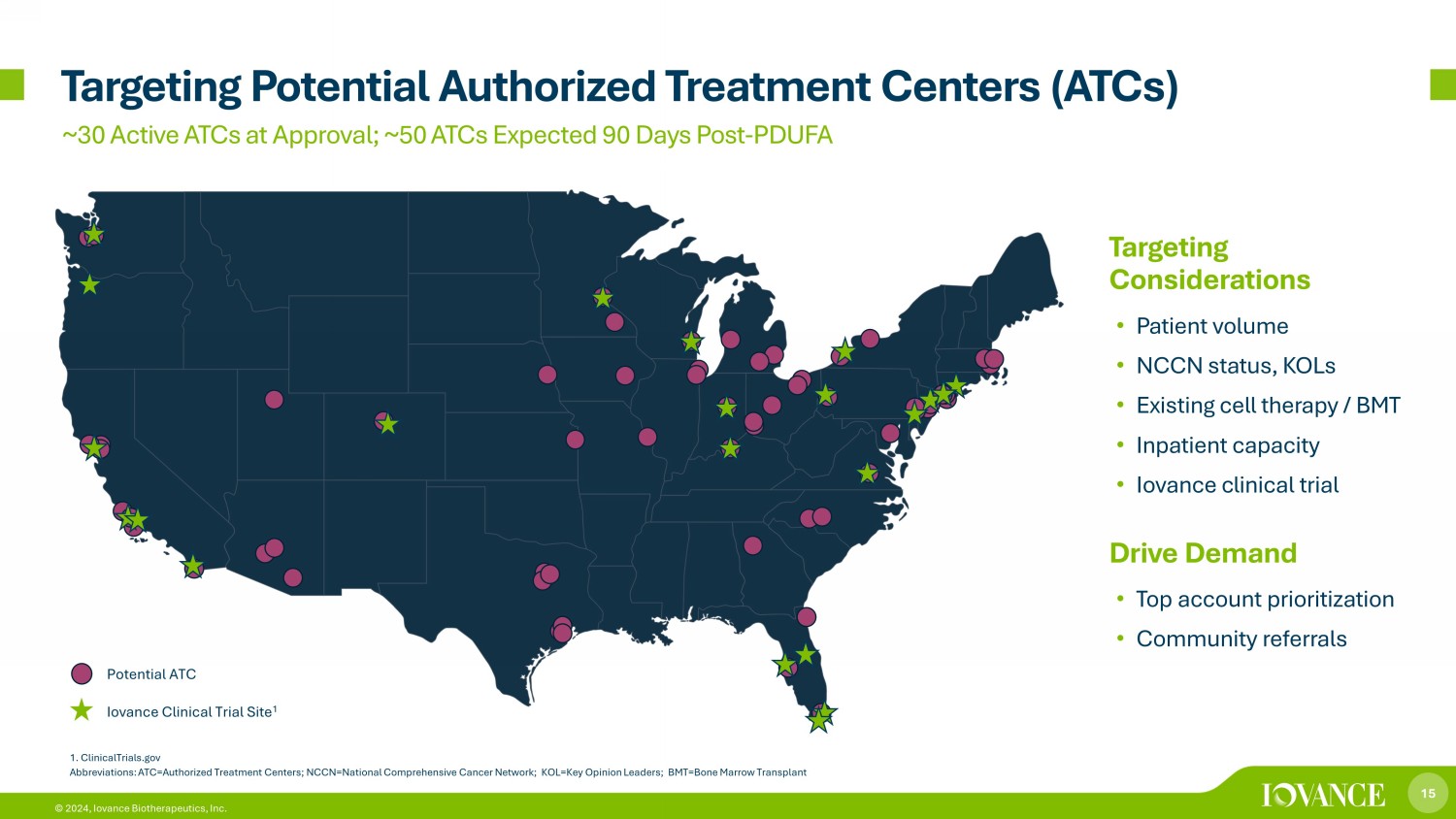
15 © 2024, Iovance Biotherapeutics, Inc. Targeting Potential Authorized Treatment Centers (ATCs) Potential ATC Iovance Clinical Trial Site 1 1. ClinicalTrials.gov Abbreviations: ATC=Authorized Treatment Centers; NCCN=National Comprehensive Cancer Network; KOL=Key Opinion Leaders; BMT=Bone Marrow Transplant Targeting Considerations • Patient volume • NCCN status, KOLs • Existing cell therapy / BMT • Inpatient capacity • Iovance clinical trial (s) Drive Demand • Top account prioritization • Community referrals ~30 Active ATCs at Approval; ~50 ATCs Expected 90 Days Post - PDUFA
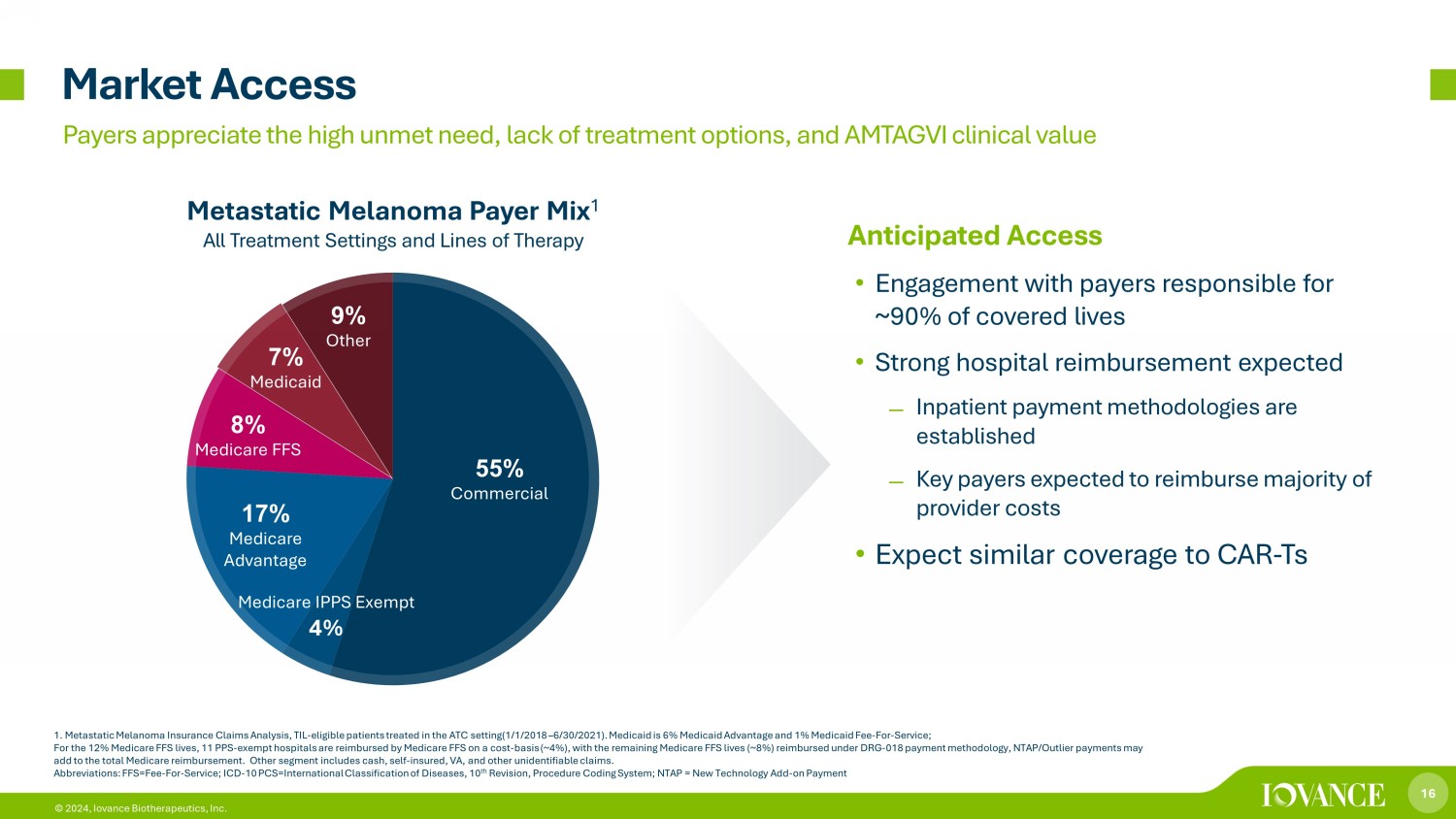
16 © 2024, Iovance Biotherapeutics, Inc. 55% Commercial Medicare IPPS Exempt 4% 17% Medicare Advantage 8% Medicare FFS 7% Medicaid 9% Other Market Access Metastatic Melanoma Payer Mix 1 All Treatment Settings and Lines of Therapy 1. Metastatic Melanoma Insurance Claims Analysis, TIL - eligible patients treated in the ATC setting(1/1/2018 – 6/30/2021). Medicai d is 6% Medicaid Advantage and 1% Medicaid Fee - For - Service; For the 12% Medicare FFS lives, 11 PPS - exempt hospitals are reimbursed by Medicare FFS on a cost - basis (~4%), with the remaining Medicare FFS lives (~8%) reimbursed under DRG - 018 payment methodology, NTAP/Outlier payments may add to the total Medicare reimbursement. Other segment includes cash, self - insured, VA, and other unidentifiable claims. Abbreviations: FFS=Fee - For - Service; ICD - 10 PCS=International Classification of Diseases, 10 th Revision, Procedure Coding System; NTAP = New Technology Add - on Payment Payers appreciate the high unmet need, lack of treatment options, and AMTAGVI clinical value Anticipated Access • Engagement with payers responsible for ~90% of covered lives • Strong hospital reimbursement expected ̶ Inpatient payment methodologies are established ̶ Key payers expected to reimburse majority of provider costs • Expect similar coverage to CAR - Ts
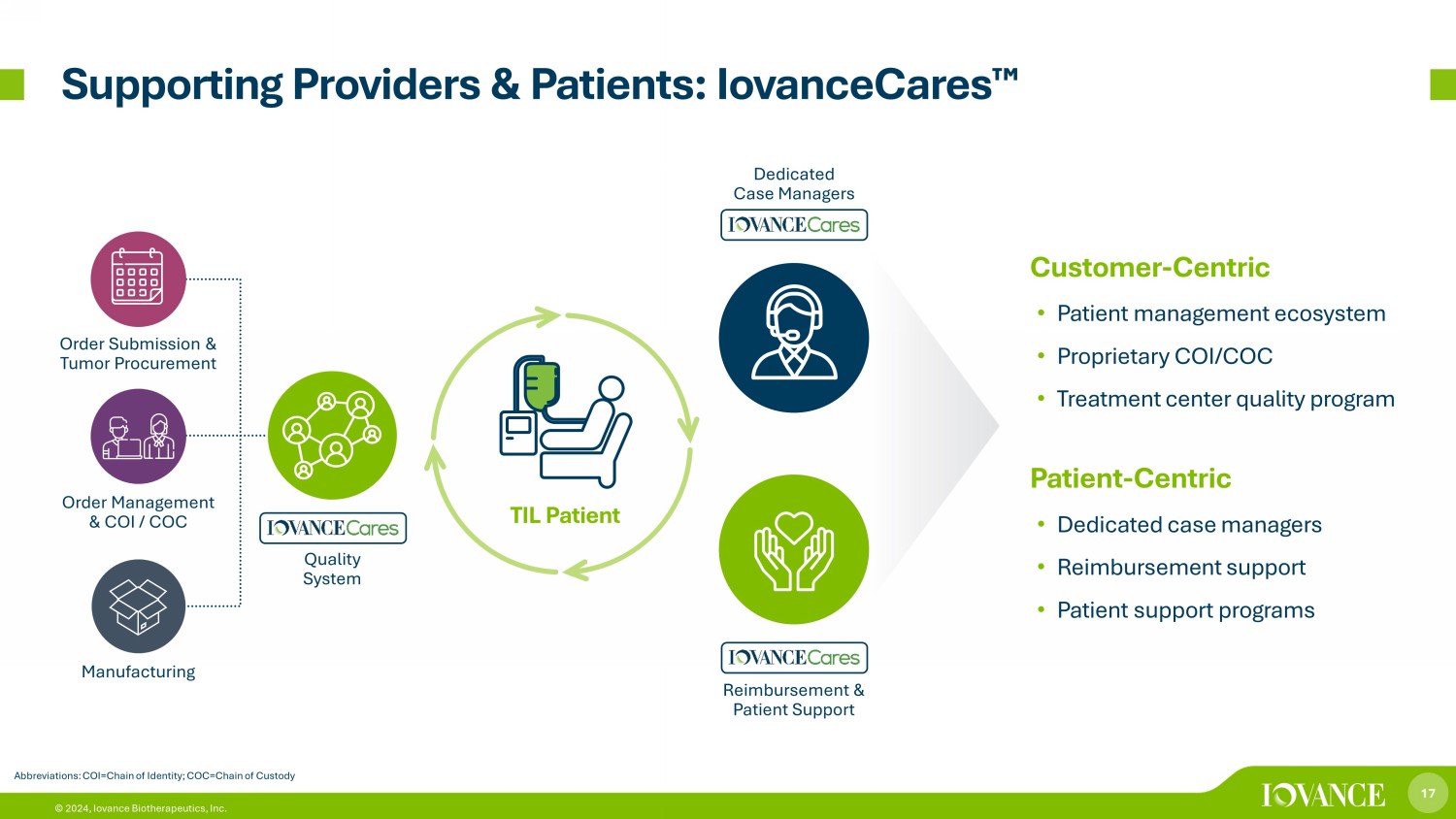
17 © 2024, Iovance Biotherapeutics, Inc. Supporting Providers & Patients: IovanceCares ¯ Abbreviations: COI=Chain of Identity; COC=Chain of Custody Customer - Centric • Patient management ecosystem • Proprietary COI/COC • Treatment center quality program Patient - Centric • Dedicated case managers • Reimbursement support • Patient support programs Quality System Dedicated Case Managers Reimbursement & Patient Support Order Submission & Tumor Procurement Order Management & COI / COC Manufacturing TIL Patient

18 © 2024, Iovance Biotherapeutics, Inc. 18 AMTAGVI ¯ Expansion Plans in Advanced Melanoma
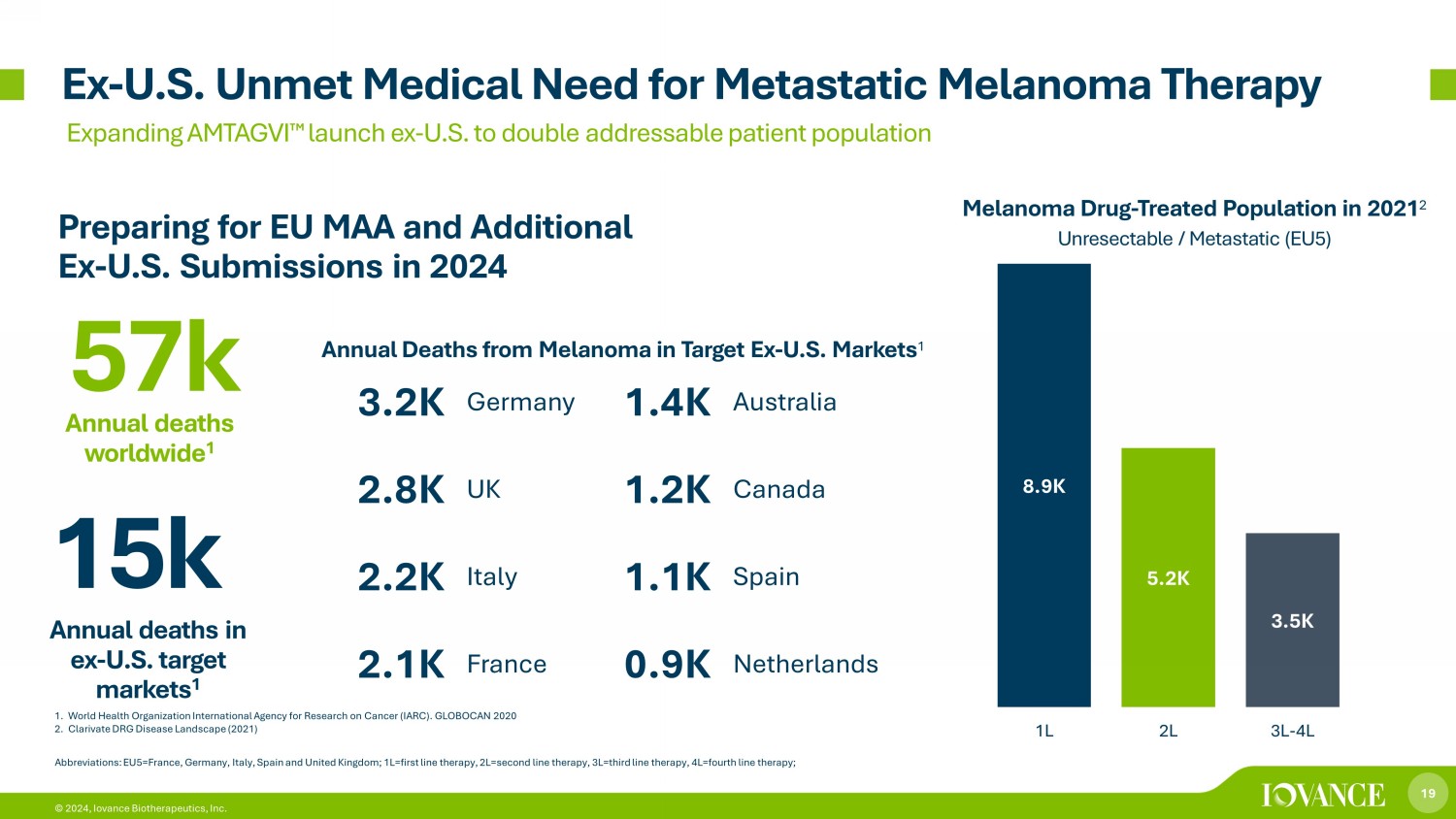
19 © 2024, Iovance Biotherapeutics, Inc. 1. World Health Organization International Agency for Research on Cancer (IARC). GLOBOCAN 2020 2. Clarivate DRG Disease Landscape (2021) Ex - U.S. Unmet Medical Need for Metastatic Melanoma Therapy Expanding AMTAGVI ¯ launch ex - U.S. to double addressable patient population 8.9K 5.2K 3.5K 1L 2L 3L-4L Melanoma Drug - Treated Population in 2021 2 Unresectable / Metastatic (EU5) Abbreviations: EU5=France, Germany, Italy, Spain and United Kingdom; 1L=first line therapy, 2L=second line therapy, 3L=third lin e therapy, 4L=fourth line therapy; 15k Annual deaths in ex - U.S. target markets 1 3.2K Germany 1.4K Australia 2.8K UK 1.2K Canada 2.2K Italy 1.1K Spain 2.1K France 0.9K Netherlands 57k Annual deaths worldwide 1 Annual Deaths from Melanoma in Target Ex - U.S. Markets 1 Preparing for EU MAA and Additional Ex - U.S. Submissions in 2024
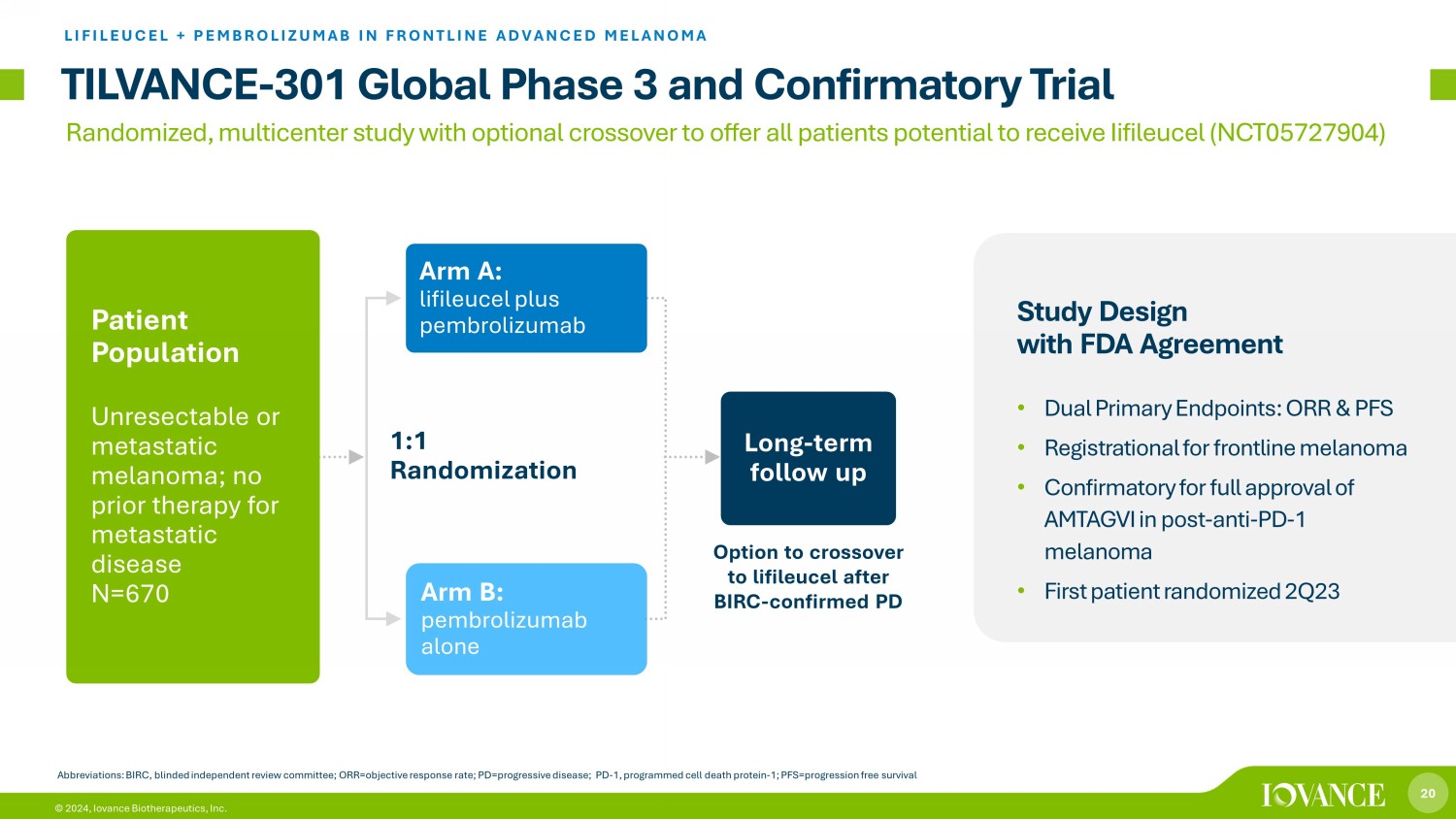
20 © 2024, Iovance Biotherapeutics, Inc. 1:1 Randomization TILVANCE - 301 Global Phase 3 and Confirmatory Trial LIFILEUCEL + PEMBROLIZUMAB IN FRONTLINE ADVANCED MELANOMA Abbreviations: BIRC, blinded independent review committee; ORR=objective response rate; PD=progressive disease; PD - 1, programme d cell death protein - 1; PFS=progression free survival Arm A: lifileucel plus pembrolizumab Long - term follow up Patient Population Unresectable or metastatic melanoma; no prior therapy for metastatic disease N=670 Arm B: pembrolizumab alone Study Design with FDA Agreement • Dual Primary Endpoints: ORR & PFS • Registrational for frontline melanoma • Confirmatory for full approval of AMTAGVI in post - anti - PD - 1 melanoma • First patient randomized 2Q23 Option to crossover to lifileucel after BIRC - confirmed PD Randomized, multicenter study with optional crossover to offer all patients potential to receive Iifileucel (NCT05727904)

21 © 2024, Iovance Biotherapeutics, Inc. Lifileucel in combination with anti - PD - 1/PD - L1 therapy in ICI - naïve patients (IOV - COM - 202 Cohort 1A, N=12 ) 1 Iovance TIL Clinical Highlights in Combination with Pembrolizumab in Metastatic Melanoma IOV - COM - 202 COHORT 1A MELANOMA COMBINATION (TIL+PEMBROLIZUMAB) • 8 / 12 patients had a confirmed objective response per RECIST v1.1 (3 CRs & 5 PRs) • 6 / 8 responders had ongoing response • 5 responders had DOR >1 year • FDA Fast Track Designation 66.7% ORR 1. As assessed by investigator using RECIST 1.1 (January 20, 2022 data cutoff) 2. Each bar is presented for each patient starting from date of TIL infusion up to date of new anti - cancer therapy, end of assessme nt, death, or data cutoff date, whichever occurs earlier. Abbreviations: CR=complete response; ICI=immune checkpoint inhibitor; ORR=objective response rate; PR=partial response; SD=st abl e disease; pembro=pembrolizumab; RECIST=Response Evaluation Criteria in Solid Tumors Time (months) since TIL Infusion 1A-12 1A-11 1A-04 1A-07 1A-06 1A-05 1A-01 1A-03 S u b j e c t s 0 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 26 27 28 29 30 31 32 Time (months) since TIL Infusion Pembro Infusion Progression Ongoing on Study PR Start CR Start Time (months) since TIL Infusion 1A - 03 1A - 01 1A - 05 1A - 06 1A - 07 1A - 04 1A - 11 1A - 12 Patient ID Time to Response for Responders 2 Patient ID CRPRSD Best Overall Response -30 1A-09 1A-02 1A-08 1A-01 1A-11 1A-12 1A-04 1A-05 1A-03 1A-06 1A-07 -100 -80 -60 -40 -20 0 20 % C h a n g e f r o m B a s e l i n e N=0 N=11 Cohort 1A % Change from Baseline SD PR CR 1A - 09 1A - 02 1A - 08 1A - 01 1A - 11 1A - 12 1A - 04 1A - 05 1A - 03 1A - 06 1A - 07 Patient ID Best Overall Response for Evaluable Patients

22 © 2024, Iovance Biotherapeutics, Inc. 22 TIL Therapy Pipeline
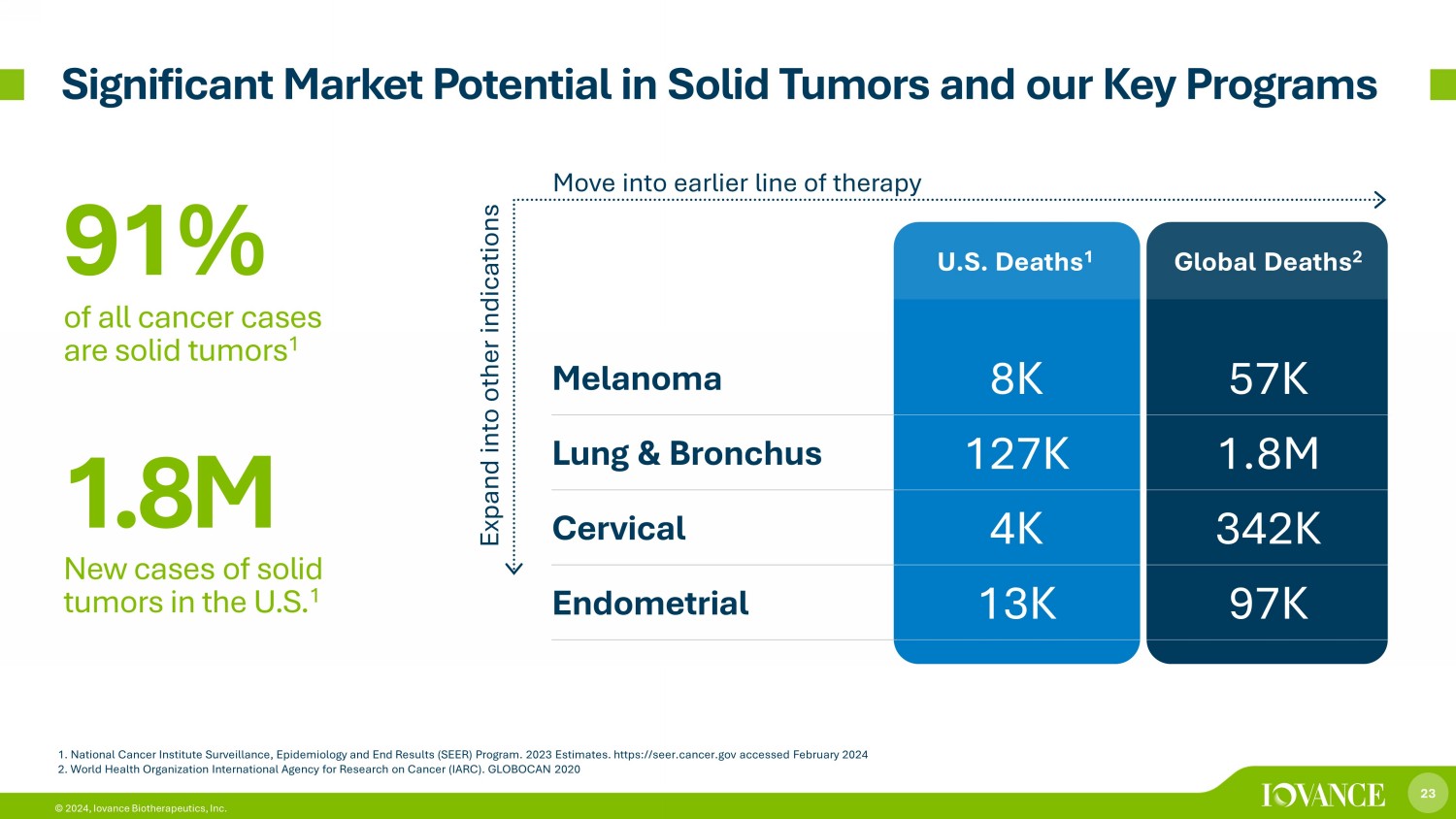
23 © 2024, Iovance Biotherapeutics, Inc. 1. National Cancer Institute Surveillance, Epidemiology and End Results (SEER) Program. 2023 Estimates. https://seer.cancer.g ov accessed February 2024 2. World Health Organization International Agency for Research on Cancer (IARC). GLOBOCAN 2020 U.S. Deaths 1 Global Deaths 2 Melanoma 8K 57K Lung & Bronchus 127K 1.8M Cervical 4K 342K Endometrial 13K 97K 91% of all cancer cases are solid tumors 1 1 .8 M New cases of solid tumors in the U.S. 1 Expand into other indications Move into earlier line of therapy Significant Market Potential in Solid Tumors and our Key Programs

24 © 2024, Iovance Biotherapeutics, Inc. 1. National Cancer Institute Surveillance, Epidemiology and End Results (SEER) Program. 2023 Estimates. https://seer.cancer.gov acc essed February 2024 2. American Cancer Society, Lung Cancer. https://www.cancer.org/cancer/types/lung - cancer/about.html accessed July 2023 3. National Cancer Database, NSCLC survival from >1 million patients assessed. Lou Y et al. Survival trends among non - small - cell lu ng cancer patients over a decade: impact of initial therapy at academic centers. Cancer Med. 2018. 4. Clarivate DRG Disease Landscape (2021) Abbreviations: EU5=France, Germany, Italy, Spain and United Kingdom; 1L=first line therapy, 2L=second line therapy, 3L=third lin e therapy, 4L=fourth line therapy; mOS=median overall survival Potential Market for Advanced Non - Small Cell Lung Cancer (NSCLC) Addressing a Substantial Unmet Need in Metastatic NSCLC Iovance TIL clinical program: • 6 cohorts across 3 trials • Multiple treatment regimens • Various populations and stages of disease Annual Deaths in U.S. 2 127K annual deaths in U.S. 1 107K 51.6K 19.0K 116K 46.2K 12.3K 1L 2L 3L US EU5 NSCLC Drug - Treated Population in 2022 Stage IV (US and EU5) 4 Leading cause of U.S. cancer deaths, accounting for ~1 in 5 cancer - related deaths 2 9% 5 - year survival rate 2 and real - world overall survival <6 months 3 in U.S.

25 © 2024, Iovance Biotherapeutics, Inc. IOV - LUN - 202 Trial Design Phase 2 Multicenter Study of LN - 145 † in Patients Post - Anti - PD - 1 NSCLC (NCT04614103)* Endpoints • Primary: ORR by IRC • Secondary: Safety * U.S. FDA placed a partial clinical hold on the IOV - LUN - 202 trial on December 22, 2023 . Enrollment for new patients is paused. Patients previously treated continue to be monitored and followed. Patients who have already undergone tumor resection will continue to receive the LN - 145 TIL treatment regimen with additional pr ecautions and risk mitigations.. † Gen 2 TIL product † † Cohort 3 patients unable to undergo surgical harvest, TIL grown from core biopsy Abbreviations: Anti - PD - 1, anti - programmed cell death inhibitor; IRC, independent review committee; NSCLC, non - small - cell lung ca ncer; ORR, objective response rate; TPS, tumor proportion score Iovance TIL Therapy LN - 145 in NSCLC IOV - LUN - 202 is designed to enroll patients with advanced NSCLC with a high unmet medical need, but limited prior lines of therapy post anti - PD - 1 treatment Patient Population Unresectable or metastatic NSCLC with progression on or after prior anti - PD - 1 treatment and chemotherapy ~40 sites and expanding in U.S., Canada, Europe Cohort 1: NSCLC Patients with < 1% or unknown TPS LN - 145 Cohort 2: NSCLC Patients with ≥ 1% TPS LN - 145 Cohort 3: Core Biopsy and Gen3 †† LN - 145 Cohort 4: LN - 145, pre - PD tumor harvest Retreatment Cohort Exploratory Cohorts Registrational Cohorts

26 © 2024, Iovance Biotherapeutics, Inc. Objective Response Rate of 26.1% by RECIST 1.1, Regardless of PD - L1 Status Preliminary Clinical Results for IOV - LUN - 202 Cohorts 1 and 2 IOV - LUN - 202 COHORTS 1 AND 2, POST - ANTI - PD - 1 NSCLC, JULY 6, 2023 Data cut: July 6, 2023. 21 evaluable patients for response. Abbreviations: CR, complete response; NSCLC, non - small - cell lung cancer; PR, partial response; SD=stable disease; SOD, sum of di ameters; TPS, tumor proportion score. n

27 © 2024, Iovance Biotherapeutics, Inc. All Patients Progressed on or After Anti - PD - 1 Therapy and Chemotherapy Preliminary Clinical Results for IOV - LUN - 202 Cohorts 1 and 2 1. Data cut: July 6, 2023. Responses were assessed by investigator. 2. Patients who have progressed on or after chemotherapy and anti - PD - 1 therapy for advanced (unresectable or metastatic) NSCLC with out EGFR, ROS or ALK genomic mutations and had received at least one line of an FDA - approved targeted therapy if indicated by other actionable tumor mutations. Abbreviations: AE, adverse event; CI, confidence interval; CR, complete response; ICI, immune checkpoint inhibitor; NE, not e val uable; NMA - LD, non - myeloablative lymphodepletion; NSCLC, non - small - cell lung cancer; ORR, objective response rate; PD, progressive disease; PR, partial response; SD, stable disease; TEAE, trea tme nt - emergent AE. Iovance TIL Therapy LN - 145 in NSCLC Cohort 1 + 2 (n=23) 2 Objective Response Rate, n (%) 1 6 (26.1) (95% CI) (10.2, 48.4) Best overall response, n (%) CR 1 (4.3) PR 5 (21.7) SD 13 (56.5) PD 2 (8.7) NE 2 (8.7) IOV - LUN - 202 COHORTS 1 AND 2, POST - ANTI - PD - 1 NSCLC, JULY 6, 2023 TEAEs were consistent with the underlying disease and known AE profiles of NMA - LD and IL - 2
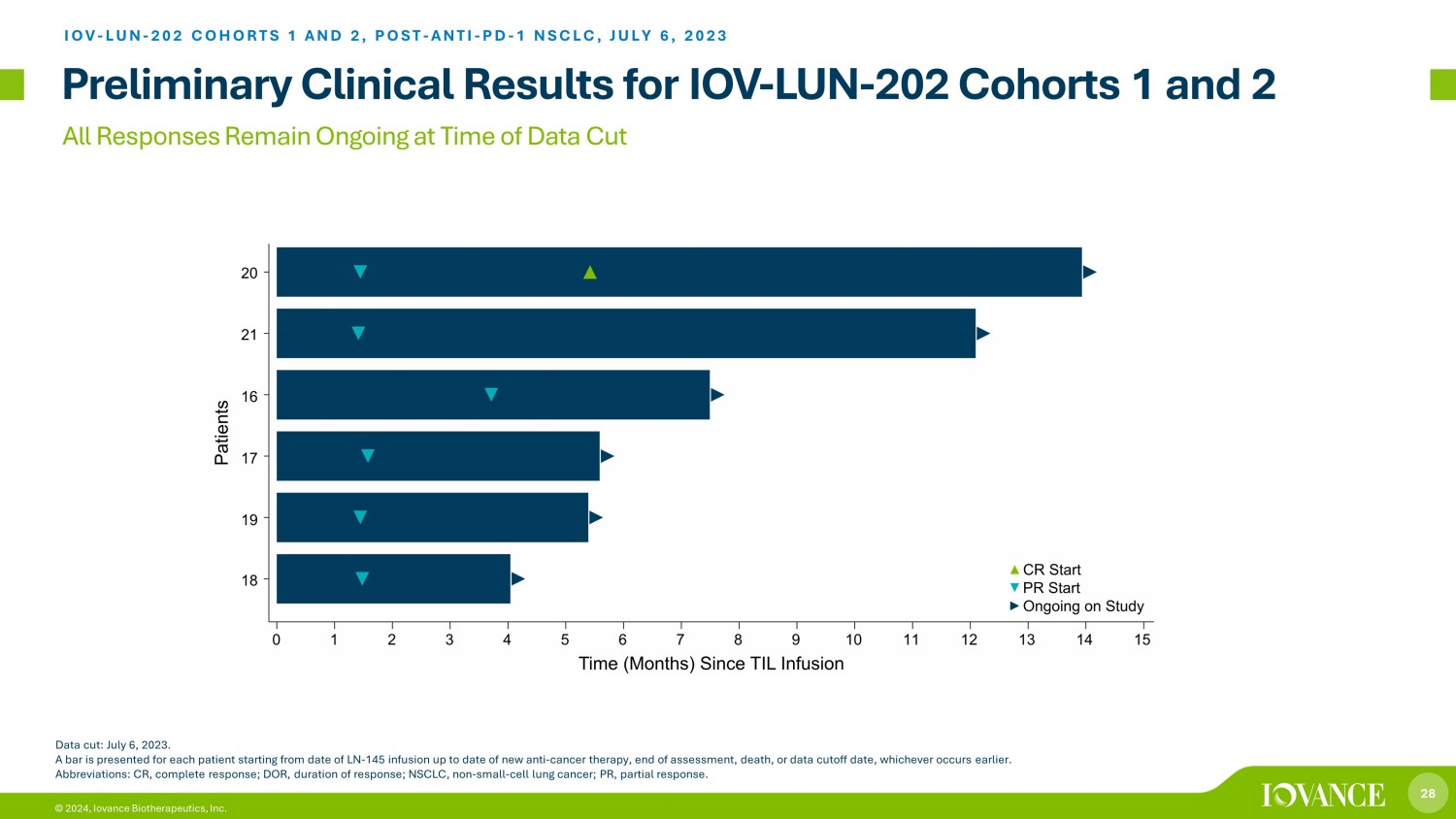
28 © 2024, Iovance Biotherapeutics, Inc. All Responses Remain Ongoing at Time of Data Cut Data cut: July 6, 2023. A bar is presented for each patient starting from date of LN - 145 infusion up to date of new anti - cancer therapy, end of assessme nt, death, or data cutoff date, whichever occurs earlier. Abbreviations: CR, complete response; DOR, duration of response; NSCLC, non - small - cell lung cancer; PR, partial response. IOV - LUN - 202 COHORTS 1 AND 2, POST - ANTI - PD - 1 NSCLC, JULY 6, 2023 Preliminary Clinical Results for IOV - LUN - 202 Cohorts 1 and 2

29 © 2024, Iovance Biotherapeutics, Inc. Proof - of - Concept for TIL in ICI - Naïve NSCLC Regardless of PD - L1 Status Cohort 3A Summary IOV - COM - 202 COHORT 3A NSCLC COMBINATION (TIL+PEMBROLIZUMAB ), WCLC ORAL PRESENTATION (JUNE 26, 2023 DATA CUT) Cohort 3A Results Support Adding TIL Therapy to Frontline Pembrolizumab + Chemotherapy Combination Regimens Clinical Activity at 18.2 Months of Follow Up 1 • Activity across ICI naïve subgroups and TPS Scores • 58.3% (7/12) ORR and 3 ongoing responses in NSCLC patients with EGFR WT disease • Safety consistent with Iovance TIL combination studies • Supports proposed registrational trial design in patients with EGFR WT disease in the frontline setting Screening Pembrolizumab Administration (Single Dose) NMA - LD Cy/Flu Lifileucel / LN - 145 Infusion IL - 2 ≤6 Doses Pembrolizumab Q3W or Q6W Efficacy Follow - up Enrollment / Surgical Resection Assessment Period: Day 0 to EOA GMP Manufacturing EOA Progression or New Therapy EOS EOT Long - term Follow - up 1. Schoenfeld, et al. WCLC 2023 Abbreviations: cy/flu, cytarabine/fludarabine; EGFR, epidermal growth factor receptor; ICI, immune checkpoint inhibitor; IL - 2, interleukin - 2; NMA - LD, non - myeloablative lymphodepletion; TPS, tumor proportion score; WT, wild type

30 © 2024, Iovance Biotherapeutics, Inc. Frontline NSCLC Registrational Trial: Design Supported by Cohort 3A Data Long - Term Follow Up SOC Maintenance Treatment +/ - TIL SOC Chemo - IO Treatment* Adding TIL Therapy to Standard - of - Care Therapy TIL Patient Population Treatment naïve metastatic NSCLC EGFR WT Disease All PD - L1 TPS Scores Chemotherapy Platinum Doublet * SOC Chemo - IO is 4 - 6 cycles of pembro + platinum - based chemotherapy doublet 1. KEYTRUDA USPI 2. Ghandi et al, NEJM 2018 Benchmarks EGFR/ALK status ORR mDOR (mos) mPFS (mos.) Prior IO Prior Chemo PD - L1 (%) SQ or NSQ Keynote - 189 1,2 WT 48% 11.2 8.8 No No All NSQ PD - L1 <1 Subgroup 1,2 WT 32% No No <1 NSQ Keynote - 407 1 N/A 58% 7.2 6.4 No No All SQ Experimental Arm Pembrolizumab Comparator Arm Abbreviations: EGFR, epidermal growth factor receptor; mDOR, median duration of response; NSCLC, non - small - cell lung cancer; NSQ, nonsquamous; platinum doublet, pemetrexed and cisplatin or carboplatin; PD - L1, programmed death ligand 1; PR, partial response; Pt, patient; SOC, standard of care; SQ, squamous; TPS, tumor proportion score; WT, wild - type Chemotherapy Platinum Doublet Pembrolizumab • FDA meeting in 2024 to discuss frontline trial to be registrational in treatment naïve EGFR WT NSCLC patients • Confirmatory trial for accelerated approval in post anti - PD - 1 NSCLC

31 © 2024, Iovance Biotherapeutics, Inc. Phase 1/2 Open - Label First - in - Human Study: IOV - GM1 - 201 Endpoints • Phase 1: Safety • Phase 2: Objective Response Rate (ORR) per RECIST v1.1 as assessed by the investigator • Secondary endpoints include complete response (CR) rate, duration of response (DOR), disease control rate (DCR), progression free survival (PFS), overall survival (OS), safety and tolerability, feasibility Study Updates • 3Q22: first patient treated Cohort 1: Unresectable or metastatic melanoma Post - anti - PD - 1/L1, post - BRAF/MEK inhibitor in patients with BRAF mutations Cohort 2: Stage III or IV NSCLC Post - anti - PD - 1/L1 or post targeted therapy and either chemotherapy or anti - PD - 1/L1 Patient Population Adults with unresectable or metastatic melanoma or advanced NSCLC N=53 Genetically Modified, PD - 1 Inactivated TIL Therapy IOV - 4001 in Previously Treated Metastatic Melanoma and NSCLC (NCT05361174) NEXT - GENERATION TIL THERAPY IOV - 4001 NSCLC=non - small - cell lung cancer
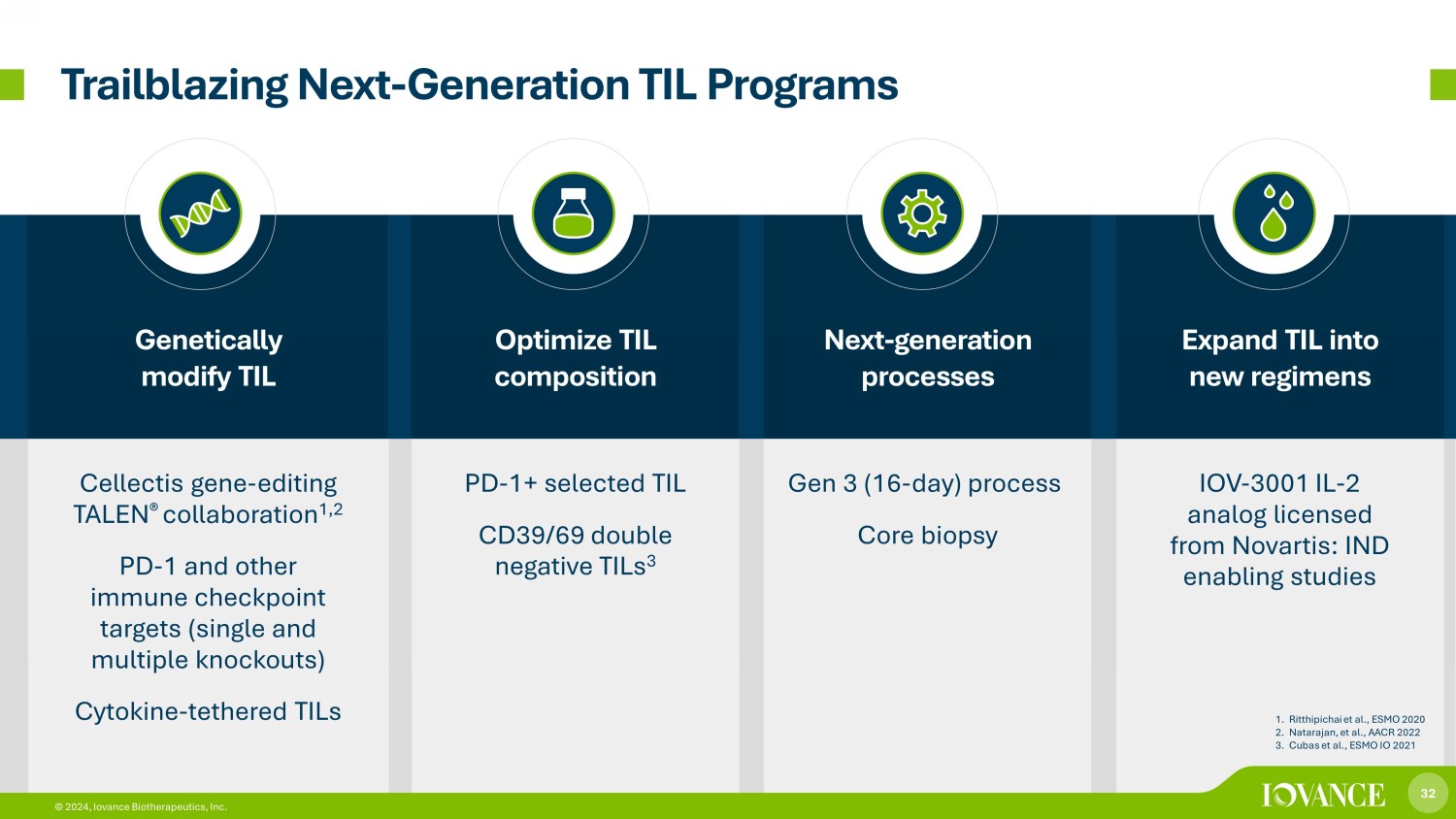
32 © 2024, Iovance Biotherapeutics, Inc. Genetically modify TIL Optimize TIL composition Next - generation processes Expand TIL into new regimens Cellectis gene - editing TALEN ® collaboration 1,2 PD - 1 and other immune checkpoint targets (single and multiple knockouts) Cytokine - tethered TILs PD - 1+ selected TIL CD39/69 double n egative TILs 3 Gen 3 (16 - day) process Core biopsy IOV - 3001 IL - 2 analog licensed from Novartis: IND enabling studies Trailblazing Next - Generation TIL Programs 1. Ritthipichai et al., ESMO 2020 2. Natarajan, et al., AACR 2022 3. Cubas et al., ESMO IO 2021 32 © 2024, Iovance Biotherapeutics, Inc.

33 © 2024, Iovance Biotherapeutics, Inc. 33 Corporate Summary & Milestones

34 © 2024, Iovance Biotherapeutics, Inc. September 30, 2023 (in millions) Cash, cash equivalents, investments, restricted cash $427.8 1 Common shares outstanding 255.8 Preferred shares outstanding 2.9 2 Stock options and restricted stock units outstanding 23.1 1. Includes Restricted Cash of $66.4 million as of September 30, 2023. 2. Preferred shares are shown on an as - converted basis Well - Capitalized in Pursuit of TIL Commercialization Cash runway is sufficient into 2025* * Includes anticipated revenue in 2024 from AMTAGVI ¯ and Proleukin®

35 © 2024, Iovance Biotherapeutics, Inc. REGULATORY □ Obtain FDA approval for lifileucel in advanced melanoma (PDUFA date: February 24, 2024) □ Submit EMA regulatory dossier (1H24) □ Submit additional ex - US dossiers (2H24) □ Meet with FDA to discuss NSCLC registrational path/frontline study PIPELINE □ Report clinical and pre - clinical data □ Resume enrollment in IOV - LUN - 202 □ Initiate Phase 2 trial in endometrial cancer □ Continue to enroll patients in clinical trials for advanced melanoma, NSCLC and gynecological cancers □ Advance new products toward clinic, including additional genetically - modified TIL therapies MANUFACTURING □ Fulfill patient demand for commercial launch and clinical trials □ Further expand capacity to meet US and ex - US demand COMMERCIAL □ Execute commercial launch (1Q24) □ On - board 50 ATCs within 90 days of PDUFA date Anticipated 2024 Milestones

36 © 2024, Iovance Biotherapeutics, Inc. Corporate Highlights Pioneering a Transformational Approach to Cure Cancer Large Market Opportunity in High Unmet Need Cancers First FDA Approved T Cell Therapy for a Solid Tumor Cancer Efficient and Scalable Proprietary Manufacturing Facility Fully - Integrated for Commercial Success • Initial focus in post - ICI solid tumors • Expansion into combinations, earlier lines of therapy and genetic modifications • Key late - stage trials in melanoma, NSCLC and cervical cancer • First - in - human trial of genetically modified PD - 1 inactivated TIL • FDA accelerated approval for AMTAGVI ¯ in advanced melanoma • TILVANCE - 301 Phase 3 confirmatory trial in frontline advanced melanoma (FTD) • Defined registration strategy in NSCLC and cervical cancer (BTD) • Iovance Cell Therapy Center ( i CTC) in - house manufacturing • Additional capacity with contract manufacturers • Rapid 22 - day Gen 2 manufacturing • >700 patients treated with Iovance proprietary process • Experienced cross - functional cell therapy team • TIL service - line capabilities established with leading U.S. cancer centers • IovanceCares ¯ proprietary platform 36 © 2024, Iovance Biotherapeutics, Inc. Abbreviations: BLA, Biologics License Application; BTD, breakthrough therapy designation; FTD, fast track designation; ICI, i mmu ne checkpoint inhibitor; NSCLC, non - small cell lung cancer; PD - 1, programmed cell death protein - 1; RMAT, Regenerative Medicines Advanced Therapy; TIL, tumor inf iltrating lymphocytes.

37 © 2024, Iovance Biotherapeutics, Inc. © 2024, Iovance Biotherapeutics, Inc. Thank You
v3.24.0.1
Cover
|
Feb. 16, 2024 |
| Cover [Abstract] |
|
| Document Type |
8-K
|
| Amendment Flag |
false
|
| Document Period End Date |
Feb. 16, 2024
|
| Entity File Number |
001-36860
|
| Entity Registrant Name |
IOVANCE BIOTHERAPEUTICS, INC.
|
| Entity Central Index Key |
0001425205
|
| Entity Tax Identification Number |
75-3254381
|
| Entity Incorporation, State or Country Code |
DE
|
| Entity Address, Address Line One |
825
Industrial Road
|
| Entity Address, Address Line Two |
Suite 400
|
| Entity Address, City or Town |
San Carlos
|
| Entity Address, State or Province |
CA
|
| Entity Address, Postal Zip Code |
94070
|
| City Area Code |
650
|
| Local Phone Number |
260-7120
|
| Written Communications |
false
|
| Soliciting Material |
false
|
| Pre-commencement Tender Offer |
false
|
| Pre-commencement Issuer Tender Offer |
false
|
| Title of 12(b) Security |
Common stock, par value $0.000041666 per value
|
| Trading Symbol |
IOVA
|
| Security Exchange Name |
NASDAQ
|
| Entity Emerging Growth Company |
false
|
| X |
- DefinitionBoolean flag that is true when the XBRL content amends previously-filed or accepted submission.
| Name: |
dei_AmendmentFlag |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionFor the EDGAR submission types of Form 8-K: the date of the report, the date of the earliest event reported; for the EDGAR submission types of Form N-1A: the filing date; for all other submission types: the end of the reporting or transition period. The format of the date is YYYY-MM-DD.
| Name: |
dei_DocumentPeriodEndDate |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:dateItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe type of document being provided (such as 10-K, 10-Q, 485BPOS, etc). The document type is limited to the same value as the supporting SEC submission type, or the word 'Other'.
| Name: |
dei_DocumentType |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:submissionTypeItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionAddress Line 1 such as Attn, Building Name, Street Name
| Name: |
dei_EntityAddressAddressLine1 |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionAddress Line 2 such as Street or Suite number
| Name: |
dei_EntityAddressAddressLine2 |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- Definition
+ References
+ Details
| Name: |
dei_EntityAddressCityOrTown |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionCode for the postal or zip code
| Name: |
dei_EntityAddressPostalZipCode |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionName of the state or province.
| Name: |
dei_EntityAddressStateOrProvince |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:stateOrProvinceItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionA unique 10-digit SEC-issued value to identify entities that have filed disclosures with the SEC. It is commonly abbreviated as CIK. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b-2
| Name: |
dei_EntityCentralIndexKey |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:centralIndexKeyItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionIndicate if registrant meets the emerging growth company criteria. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b-2
| Name: |
dei_EntityEmergingGrowthCompany |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionCommission file number. The field allows up to 17 characters. The prefix may contain 1-3 digits, the sequence number may contain 1-8 digits, the optional suffix may contain 1-4 characters, and the fields are separated with a hyphen.
| Name: |
dei_EntityFileNumber |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:fileNumberItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionTwo-character EDGAR code representing the state or country of incorporation.
| Name: |
dei_EntityIncorporationStateCountryCode |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:edgarStateCountryItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe exact name of the entity filing the report as specified in its charter, which is required by forms filed with the SEC. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b-2
| Name: |
dei_EntityRegistrantName |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe Tax Identification Number (TIN), also known as an Employer Identification Number (EIN), is a unique 9-digit value assigned by the IRS. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b-2
| Name: |
dei_EntityTaxIdentificationNumber |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:employerIdItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionLocal phone number for entity.
| Name: |
dei_LocalPhoneNumber |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionBoolean flag that is true when the Form 8-K filing is intended to satisfy the filing obligation of the registrant as pre-commencement communications pursuant to Rule 13e-4(c) under the Exchange Act. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 13e
-Subsection 4c
| Name: |
dei_PreCommencementIssuerTenderOffer |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionBoolean flag that is true when the Form 8-K filing is intended to satisfy the filing obligation of the registrant as pre-commencement communications pursuant to Rule 14d-2(b) under the Exchange Act. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 14d
-Subsection 2b
| Name: |
dei_PreCommencementTenderOffer |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionTitle of a 12(b) registered security. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b
| Name: |
dei_Security12bTitle |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:securityTitleItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionName of the Exchange on which a security is registered. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection d1-1
| Name: |
dei_SecurityExchangeName |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:edgarExchangeCodeItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionBoolean flag that is true when the Form 8-K filing is intended to satisfy the filing obligation of the registrant as soliciting material pursuant to Rule 14a-12 under the Exchange Act. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Section 14a
-Number 240
-Subsection 12
| Name: |
dei_SolicitingMaterial |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionTrading symbol of an instrument as listed on an exchange.
| Name: |
dei_TradingSymbol |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:tradingSymbolItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionBoolean flag that is true when the Form 8-K filing is intended to satisfy the filing obligation of the registrant as written communications pursuant to Rule 425 under the Securities Act. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Securities Act
-Number 230
-Section 425
| Name: |
dei_WrittenCommunications |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
Iovance Biotherapeutics (NASDAQ:IOVA)
Historical Stock Chart
From Oct 2024 to Nov 2024
Iovance Biotherapeutics (NASDAQ:IOVA)
Historical Stock Chart
From Nov 2023 to Nov 2024