QuidelOrtho Receives 510(K) Clearance for Savanna® Multiplex Molecular Platform and Savanna® HSV 1+2/VZV PCR Assay
December 20 2023 - 4:05PM
Business Wire
Savanna delivers results in 25 minutes or
less, improves clinical care, provides cost savings and delivers
sample-to-result molecular testing across all laboratory
settings
QuidelOrtho Corporation (Nasdaq: QDEL) (“QuidelOrtho”), a
global provider of innovative in vitro diagnostic technologies
designed for point-of-care settings, clinical labs and transfusion
medicine, has received 510(K) clearance from the U.S. Food and Drug
Administration for its innovative Savanna PCR platform and Savanna
HSV 1+2/VZV in vitro diagnostic test for the detection and
differentiation of Herpes simplex virus type 1 (HSV-1), Herpes
simplex virus type 2 (HSV-2) and Varicella-zoster virus (VZV)
nucleic acids isolated and purified from swabs obtained from
cutaneous or mucocutaneous lesion specimens obtained from
symptomatic patients. The clearance allows QuidelOrtho to market
and sell the Savanna multiplex molecular platform and the Savanna
HSV 1+2/VZV assay to laboratories performing moderate or high
complexity diagnostic testing in the United States.
“Our relentless pursuit of revolutionizing testing across the
healthcare continuum has resulted in the achievement of another
major milestone – the clearance of our Savanna platform and the
Savanna HSV 1+2/VZV assay,” said Douglas Bryant, President and
Chief Executive Officer of QuidelOrtho. “We’re truly
differentiating diagnostics in the marketplace and allowing labs
and healthcare workers to provide the quick diagnoses needed in
critical situations.”
The Savanna multiplex molecular platform enables professional
customers to analyze up to 12 pathogens or targets, plus up to four
controls, from a single test run in roughly 25 minutes, depending
on the assay. Savanna is a fully integrated, sample-to-result,
automated in vitro diagnostic platform that performs real-time
polymerization chain reaction (“PCR”) tests by using the Savanna
instrument and Savanna assay cartridges. After inserting the
customizable test panel cartridge, the instrument performs sample
and reagent preparation, nucleic acid extraction and amplification,
real-time detection of RNA or DNA target sequence, and qualitative
or quantitative result interpretation from a variety of sample
types. The Savanna platform is intended to aid in the diagnosis of
related diseases, ensuring patients get the right diagnosis quickly
and accurately.
“The Savanna platform’s small footprint hides a powerful set of
features, such as integrated sample prep combined with rapid
real-time PCR amplification and detection technologies, making it a
perfect fit for syndromic testing in hospitals and
moderate-complexity labs, with the goal of eventually accessing
physician offices, urgent care clinics and other point-of-care
locations. Savanna is a truly compelling product, our next flagship
platform and an important growth driver,” said Bryant.
The Savanna HSV 1+2/VZV test panel is intended to aid in the
differential diagnosis of infections with these viruses. Savanna
will be available to professional customers on December 20,
2023.
About QuidelOrtho Corporation
QuidelOrtho Corporation (Nasdaq: QDEL) is a world leader in in
vitro diagnostics, developing and manufacturing intelligent
solutions that transform data into understanding and action for
more people in more places every day.
Offering industry-leading expertise in immunoassay and molecular
testing, clinical chemistry and transfusion medicine, bringing
fast, accurate and reliable diagnostics when and where they are
needed – from home to hospital, lab to clinic. So that patients,
clinicians and health officials can spot trends sooner, respond
quicker and chart the course ahead with accuracy and
confidence.
Building upon its many years of groundbreaking innovation,
QuidelOrtho continues to partner with customers across the
healthcare continuum and around the globe to forge a new diagnostic
frontier. One where insights and solutions know no bounds,
expertise seamlessly connects and a more informed path is
illuminated for each of us.
QuidelOrtho is advancing diagnostics to power a healthier
future.
For more information, please visit www.quidelortho.com.
Source: QuidelOrtho Corporation
View source
version on businesswire.com: https://www.businesswire.com/news/home/20231220372257/en/
Investor Contact: Juliet Cunningham Vice President,
Investor Relations IR@QuidelOrtho.com
Media Contact: Stephanie Kleewein Communications Manager
media@QuidelOrtho.com
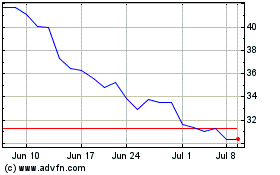
QuidelOrtho (NASDAQ:QDEL)
Historical Stock Chart
From Mar 2024 to Apr 2024
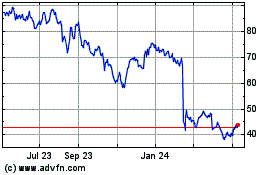
QuidelOrtho (NASDAQ:QDEL)
Historical Stock Chart
From Apr 2023 to Apr 2024