Xenios AG receives approval in China for ECMO devices
July 13 2021 - 3:51AM
Business Wire
Xenios AG, a Fresenius Medical Care company, has received
approval from the National Medical Products Administration (NMPA)
in China for the Xenios console and patient kits for ECMO
therapy.
In early May, Xenios AG, a Fresenius Medical Care company,
received approval for two patient kits in China. It follows NMPA’s
approval of the Xenios console back in December 2020. As a result,
a complete Xenios system is now permitted for ECMO therapy in
China.
This press release features multimedia. View
the full release here:
https://www.businesswire.com/news/home/20210713005495/en/
Xenios launch event in China (Photo:
Business Wire)
Xenios received the approval through a process called “Fast
Registration”. This expedited process is only offered for products
that, for example, are urgently needed for clinical use and can
treat serious, life-threatening diseases. This Xenios ECMO complete
system is certified in China for nine days application period. It
is also the first ever “Fast Registration” for new imported medical
devices in China.
“We are particularly proud to have received this approval!” says
J�rg Buschbell, CEO of Xenios AG, and adds, “In just over a year,
we have gained access not only to the US market, but also to the
Chinese market for our ECMO therapy products. This marks another
milestone for Xenios AG and Fresenius Medical Care.”
“The expedited approval was a joint effort by Xenios and
Fresenius Medical Care China Team. Since the beginning of the
pandemic, they strived to make the advanced critical care products
available in China for treating the critically ill patients.” says
Harry De Wit, President and CEO of Fresenius Medical Care Asia
Pacific. “The approval means that we will be able to support the
Chinese critical care community further with our advanced
developments, ultimately benefitting the patients.”
About Xenios AG:
Xenios AG - a company of the Fresenius Medical Care Group - is a
pioneer in extracorporeal heart and lung support. The Novalung and
Medos products offer a wide range of heart and lung support
products via a single platform, the Xenios console.
Xenios’ mission is to improve patient well-being and care. The
Novalung therapy aims at a paradigm shift in lung support: enabling
the treatment of patients who are more awake, more self-determined
and more mobile.
Xenios AG has been part of Fresenius Medical Care, the world's
leading provider of products and services for people with kidney
disease since the end of 2016. Xenios complements the group's
portfolio in the area of holistic multi-organ support of Fresenius
Medical Care.
www.xenios-ag.com
About Fresenius Medical Care
Fresenius Medical Care is the world's leading provider of
products and services for individuals with renal diseases of which
around 3.7 million patients worldwide regularly undergo dialysis
treatment. Through its network of 4,110 dialysis clinics, Fresenius
Medical Care provides dialysis treatments for 344,476 patients
around the globe. Fresenius Medical Care is also the leading
provider of dialysis products such as dialysis machines or
dialyzers. Along with its core business, the Renal Care Continuum,
the company focuses on expanding in complementary areas and in the
field of critical care. Fresenius Medical Care is listed on the
Frankfurt Stock Exchange (FME) and on the New York Stock Exchange
(FMS).
For more information, please visit the company’s website at
www.freseniusmedicalcare.asia.
Disclaimer
This release contains forward-looking statements that are
subject to various risks and uncertainties. Actual results could
differ materially from those described in these forward-looking
statements due to various factors, including, but not limited to,
changes in business, economic and competitive conditions, legal
changes, regulatory approvals, results of clinical studies, foreign
exchange rate fluctuations, uncertainties in litigation or
investigative proceedings, and the availability of financing. These
and other risks and uncertainties are detailed in Fresenius Medical
Care AG & Co. KGaA's reports filed with the U.S. Securities and
Exchange Commission. Fresenius Medical Care AG & Co. KGaA does
not undertake any responsibility to update the forward-looking
statements in this release.
View source
version on businesswire.com: https://www.businesswire.com/news/home/20210713005495/en/
Dr. Alexandra Villar Vice President Corporate Communications and
Branding Asia Pacific Fresenius Medical Care Northpoint, 100
Miller Street North Sydney, NSW 2060, Australia
www.freseniusmedicalcare.asia T +61 2 9466 8020
Alexandra.Villar@fmc-asia.com
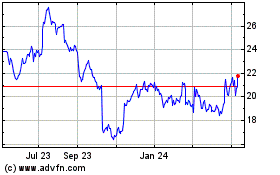
Fresenius Medical Care (NYSE:FMS)
Historical Stock Chart
From Jun 2024 to Jul 2024
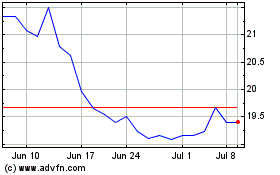
Fresenius Medical Care (NYSE:FMS)
Historical Stock Chart
From Jul 2023 to Jul 2024