On track for full commercial launch in the U.S.
in late June
Sage Therapeutics (NASDAQ: SAGE), a biopharmaceutical company
developing novel medicines to treat life-altering central nervous
system (CNS) disorders, today announced that the U.S. Drug
Enforcement Administration (DEA) has placed ZULRESSO™ (brexanolone)
injection into Schedule IV of the Controlled Substances Act.
ZULRESSO, which was approved by the U.S. Food and Drug
Administration (FDA) on March 19, 2019, is the first and only
treatment specifically approved for postpartum depression (PPD),
the most common medical complication of childbirth.
This press release features multimedia. View
the full release here:
https://www.businesswire.com/news/home/20190614005243/en/
Product shot (white) (Photo: Business
Wire)
With this decision, the product label for ZULRESSO will be
finalized. The Company expects to launch ZULRESSO in late June as
planned. ZULRESSO is administered via continuous intravenous (IV)
infusion for 2.5 days under the supervision of healthcare providers
in sites of care certified under the ZULRESSO Risk Evaluation and
Mitigation Strategy (REMS) program. The one-time infusion offers
the potential for rapid resolution of depressive symptoms of
PPD.
“Historically, women suffering from PPD may have avoided seeking
help because of the stigma, lack of a specifically approved
treatment and the complicated journey to care – challenges that
have been difficult for women with PPD and their families to
overcome,” said Mike Cloonan, chief business officer at Sage. “Sage
is taking on those challenges and creating a family-centric
approach to ZULRESSO availability by enabling pathways to care
including building our patient support organization, where we will
provide a range of meaningful support resources to women with PPD
and their families to help navigate the ZULRESSO treatment journey.
We also continue to activate Centers of Excellence across the
country with the goal of supporting a positive patient experience
and broad availability to ZULRESSO for women with PPD.”
The Company continues to focus on identifying and activating
Centers of Excellence (COE) and other healthcare settings capable
of administering ZULRESSO to women with PPD. The key criteria for
identifying a potential COE or other sites of care include: a PPD
healthcare provider champion; the ability to secure appropriate
payer reimbursement for ZULRESSO; and the capability to meet the
requirements for certification under the ZULRESSO REMS, which
includes monitoring by qualified trained staff, adherence to
specific protocols and the maintenance of a patient registry. The
Company continues to execute against its planned go-to-market
strategy with a focus on the commercial launch of ZULRESSO in late
June.
For more information on ZULRESSO, including the final product
label, visit www.sagerx.com.
About Postpartum DepressionPostpartum depression (PPD) is
the most common medical complication of childbirth. PPD is a
distinct and readily identified major depressive episode that can
occur during pregnancy or after giving birth. Expert opinions vary
as to the timing of the onset of PPD, ranging from onset during
pregnancy up to 4-weeks postpartum and onset during pregnancy up to
12-months postpartum. PPD may have devastating consequences for a
woman and for her family, which may include significant functional
impairment, depressed mood and/or loss of interest in her newborn,
and associated symptoms of depression such as loss of appetite,
difficulty sleeping, motor challenges, lack of concentration, loss
of energy and poor self-esteem. PPD can be a life-threatening
condition due to the risk of suicide, a leading cause of maternal
death following childbirth. PPD is estimated to affect
approximately one in nine women who have given birth in the U.S.
and 400,000 women annually. More than half of these cases may go
undiagnosed without proper screening.
About ZULRESSO™ (brexanolone) injectionZULRESSO, the
first medicine specifically approved by the U.S. Food and Drug
Administration (FDA) for the treatment of postpartum
depression (PDD) in adults, is a positive allosteric modulator of
both synaptic and extrasynaptic GABAA receptors. Allosteric
modulation of neurotransmitter receptor activity results in varying
degrees of desired activity rather than complete activation or
inhibition of the receptor.
What is ZULRESSO?
ZULRESSO™ (brexanolone) CIV is a prescription medicine used in
adults to treat a certain type of depression called Postpartum
Depression.
IMPORTANT SAFETY INFORMATION
What is the most important information I should know about
ZULRESSO?
ZULRESSO can cause serious side effects, including:
- Excessive sedation and sudden loss
of consciousness. ZULRESSO may cause you to feel very sleepy
(excessive sedation) or pass out (loss of consciousness). Your
healthcare provider should check you for symptoms of excessive
sleepiness every 2 hours while you are awake.
- During your ZULRESSO infusion, tell
your healthcare provider right away if you feel like you cannot
stay awake during the time you are normally awake or if you feel
like you are going to pass out. Your healthcare provider may lower
your dose or stop the infusion until symptoms go away.
- You must have a caregiver or family
member with you to help care for your child(ren) during your
ZULRESSO infusion.
- Because of the risk of serious harm
resulting from excessive sedation or sudden loss of consciousness,
ZULRESSO is only available through a restricted program called the
ZULRESSO REMS.
Before receiving ZULRESSO, tell your healthcare provider
about all your medical conditions, including if you:
- drink alcohol
- have kidney problems
- are pregnant or think you may be
pregnant. It is not known if ZULRESSO will harm your unborn baby.
- There is a pregnancy registry for
females who are exposed to ZULRESSO during pregnancy. The purpose
of the registry is to collect information about the health of
females exposed to ZULRESSO and their baby. If you become pregnant
during treatment with ZULRESSO, talk to your healthcare provider
about registering with the National Pregnancy Registry for
Antidepressants at 1-844-405-6185 or visit
https://womensmentalhealth.org/clinical-and-research-programs/pregnancyregistry/antidepressants/
- are breastfeeding or plan to
breastfeed. ZULRESSO passes into breast milk. Talk to your
healthcare provider about the risks and benefits of breastfeeding
and about the best way to feed your baby while receiving
ZULRESSO.
Tell your healthcare provider about all the medicines you
take, including prescription and over-the-counter medicines,
vitamins, and herbal supplements.
ZULRESSO and some medicines may interact with each other and
cause serious side effects.
Especially tell your healthcare provider if you take
other antidepressants, opioids, or Central Nervous System (CNS)
depressants (such as benzodiazepines).
Know the medicines you take. Keep a list of them to show your
healthcare provider and pharmacist when you get a new medicine.
Your healthcare provider will decide if other medicines can be
taken with ZULRESSO.
How will I receive ZULRESSO?
ZULRESSO is given to you by continuous intravenous (IV) infusion
into your vein. The infusion will last for a total of 60 hours (2.5
days).
What should I avoid while receiving ZULRESSO?
- ZULRESSO may make you feel dizzy and
sleepy. Do not drive a car or do other dangerous activities after
your ZULRESSO infusion until your feeling of sleepiness has
completely gone away. See “What is the most important
information I should know about ZULRESSO?”
- Do not drink alcohol while receiving
ZULRESSO.
What are the possible side effects of ZULRESSO?
ZULRESSO can cause serious side effects, including:
- See “What is the most important
information I should know about ZULRESSO?”
- Increased risk of suicidal thoughts
or actions. ZULRESSO and other antidepressant medicines may
increase suicidal thoughts and actions in some people 24 years of
age and younger. Depression or other serious mental illnesses are
the most important causes of suicidal thoughts or actions.
How can I watch for and try to prevent suicidal thoughts and
actions?
- Pay close attention to any changes,
especially sudden changes in mood, behavior, thoughts, or feelings,
or if you develop suicidal thoughts or actions.
- Tell your healthcare provider right
away if you have any new or sudden changes in mood, behavior,
thoughts, or feelings.
- Keep all follow-up visits with your
healthcare provider as scheduled. Call your healthcare provider
between visits as needed, especially if you have concerns about
symptoms.
Tell your healthcare provider right away if you have any of
the following symptoms, especially if they are new, worse, or worry
you:
- Attempts to commit suicide, thoughts
about suicide or dying, new or worse depression, other unusual
changes in behavior or mood
The most common side effects of ZULRESSO include:
- Sleepiness, dry mouth, passing out,
flushing of the skin or face.
These are not all the side effects of ZULRESSO.
Call your doctor for medical advice about side effects. You may
report side effects to FDA at 1-800-FDA-1088.
Please see Full Prescribing Information including Boxed
Warning and Medication Guide for ZULRESSO™ and discuss
any questions you may have with your healthcare provider.
About Sage TherapeuticsSage Therapeutics is a
biopharmaceutical company committed to developing novel medicines
to transform the lives of patients with life-altering CNS
disorders. ZULRESSO™ (brexanolone) injection is a rapidly acting
GABA modulator now approved by the U.S. Food and Drug
Administration as the first and only treatment specifically
indicated for postpartum depression. Sage is developing a portfolio
of novel product candidates targeting critical CNS receptor
systems, including SAGE-217, which is in Phase 3 development in
major depressive disorder and postpartum depression. For more
information, please visit www.sagerx.com.
Forward-Looking StatementsVarious statements in this
release concern Sage's future expectations, plans and prospects,
including without limitation: our expectations regarding the timing
of availability and launch of ZULRESSO in the treatment of PPD; our
plans regarding commercial activities and patient support; our
expectations regarding availability of REMS-certified sites of care
for the administration of ZULRESSO; our expectations regarding
access to treatment for women with PPD; our statements regarding
the potential benefit of ZULRESSO in the treatment of PPD; our
estimates as to the number of women who suffer from PPD; and other
statements regarding the potential and our plans and expectations
for ZULRESSO and our other programs and business. These statements
constitute forward-looking statements as that term is defined in
the Private Securities Litigation Reform Act of 1995. These
forward-looking statements are neither promises nor guarantees of
future performance, and are subject to a variety of risks and
uncertainties, many of which are beyond our control, which could
cause actual results to differ materially from those contemplated
in these forward-looking statements, including the risks that:
availability and launch of ZULRESSO may not occur on the timelines
we expect; we may encounter issues or other challenges in launching
and commercializing ZULRESSO, including issues related to market
acceptance by healthcare providers, healthcare settings and women
with PPD, issues related to availability of sites of care,
challenges with reimbursement, other issues related to limitations
on the site of administration of ZULRESSO to REMS-certified
supervised healthcare settings and the other requirements of the
REMS, and challenges associated with execution of our marketing,
sales and patient support activities, which in each case could
limit the potential of ZULRESSO; results achieved with use of
ZULRESSO in the treatment of PPD once we have launched the product
may be different than observed in clinical trials, and may vary
among patients; the number of women with PPD or the unmet need for
additional treatment options may be significantly smaller than we
expect; we may encounter unexpected safety or tolerability issues
with ZULRESSO or any of our product candidates; we may encounter
supply issues with respect to ZULRESSO or any of our product
candidates; we may not be successful in our development of any of
our current or future product candidates in any indication we are
currently pursuing or may in the future pursue; success in early
stage clinical trials may not be repeated or observed in ongoing or
future studies of any of our product candidates; ongoing and future
clinical results may not support further development or be
sufficient to gain regulatory approval of our product candidates;
we may decide that a development pathway for one of our product
candidates in one or more indications is no longer feasible or
advisable or that the unmet need no longer exists; the FDA may
decide that the development program for any of our product
candidates, even if positive, is not sufficient for a new drug
application filing or approval; and we may encounter technical and
other unexpected hurdles in the commercialization of ZULRESSO or in
the development of our product candidates; as well as those risks
more fully discussed in the section entitled "Risk Factors" in our
most recent report filed with the Securities and Exchange
Commission (SEC), and discussions of potential risks,
uncertainties, and other important factors in our subsequent
filings with the SEC. In addition, any forward-looking statements
represent our views only as of today, and should not be relied upon
as representing our views as of any subsequent date. We explicitly
disclaim any obligation to update any forward-looking
statements.
View source
version on businesswire.com: https://www.businesswire.com/news/home/20190614005243/en/
Investor Contact:Maren Killackey,
617-949-4113maren.killackey@sagerx.comMedia Contact:Alexis
Smith, 617-588-3740alexis.smith@sagerx.com
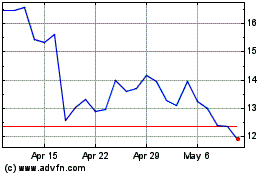
Sage Therapeutics (NASDAQ:SAGE)
Historical Stock Chart
From Aug 2024 to Sep 2024
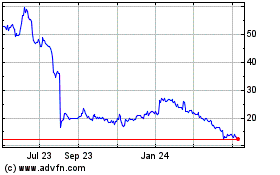
Sage Therapeutics (NASDAQ:SAGE)
Historical Stock Chart
From Sep 2023 to Sep 2024