DALLAS, Dec. 14, 2021 /PRNewswire/ -- Lantern Pharma
(NASDAQ: LTRN), a clinical stage biopharmaceutical company using
its proprietary RADR® artificial intelligence ("A.I.")
platform to transform the cost, pace, and timeline of oncology drug
discovery and development, today announced that Lantern Pharma
presented positive data on the effectiveness of LP-284 in
hematologic cancers at the 63rd American Society of
Hematology (ASH) Annual Meeting, which was held in-person and
virtually from December 11 – 14,
2021. This poster presentation can be viewed on Lantern Pharma's
website at: https://www.lanternpharma.com/about/publications.
LP-284 is a fully synthetic molecule belonging to the new
generation of acylfulvenes, a family of naturally derived
anti-cancer drug candidates. LP-284 is the stereoisomer
(enantiomer) of LP-184 and has the potential for development as
monotherapy and also as a synergistic agent in combination with
other drugs. LP-284 is currently being evaluated for activity in a
wide spectrum of hematological cancers. Earlier this year, Lantern
filed multi-national patent applications directed to both the
composition and manufacture of LP-284.
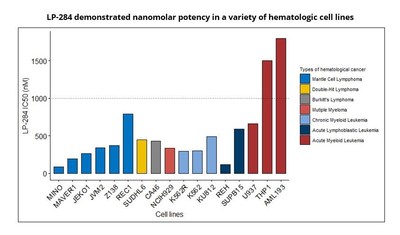
The study demonstrated LP-284's broad in vitro anti-tumor
activity in lymphoma, multiple myeloma, and leukemia cells.
Notably, the enantiomer pair, LP-184 and LP-284, exhibit distinct
patterns of anti-tumor activities. As a result, the novel
enantiomer LP-284 may provide a targeted therapy option for
hematologic cancers with compromised DNA repair, supporting further
targeted development plans for LP-284.
"Approximately every 3 minutes, one person in the U.S. is
diagnosed with leukemia, lymphoma or myeloma and approximately
every 9 minutes someone in the U.S. dies from a blood
cancer1," commented Panna
Sharma, CEO and President of Lantern Pharma Inc. "There is
an urgent need to develop new, targeted therapies; however, the
current industry approach is time consuming and expensive. This
latest data provides further validation of Lantern's
RADR® platform, which leverages A.I. and machine
learning with the aim of dramatically shortening the timeline and
reducing costs associated with drug discovery and development.
Specifically, these findings support our hypothesis that LP-284 has
the potential to become a targeted therapy option for hematologic
cancers with compromised DNA repair. We plan to apply the insights
obtained from this work to advance the development of LP-284 in
rare blood cancer indications. This data is very encouraging, and
we look forward to advancing LP-284 towards the clinic, while
leveraging our RADR® platform to develop new
cutting-edge treatments for rare blood cancers and other
indications."
Lantern's proprietary RADR® A.I. platform
leverages over 10 billion data points, machine learning, genomics,
and computational biology to accelerate the discovery of potential
mechanisms of action, and biomarker signatures that correlate to
drug response in rare blood cancers.
About Lantern Pharma
Lantern Pharma (NASDAQ: LTRN) is
a clinical-stage oncology-focused biopharmaceutical company
leveraging its proprietary RADR® A.I. platform and
machine learning to discover biomarker signatures that identify
patients most likely to respond to its pipeline of
genomically-targeted therapeutics. Lantern is currently developing
four drug candidates and an ADC program across eight disclosed
tumor targets, including two phase 2 programs. By targeting drugs
to patients whose genomic profile identifies them as having the
highest probability of benefiting from the drug, Lantern's approach
represents the potential to deliver best-in-class outcomes. More
information is available at: www.lanternpharma.com and Twitter
@lanternpharma.
About RADR®
RADR® or Response
Algorithm for Drug Positioning & Rescue,
is Lantern's proprietary integrated A.I. platform for large-scale
biomarker and drug-tumor interaction data analytics that leverages
machine-learning. RADR® is used to provide mechanistic
insights about drug-tumor interactions, predict the potential
response of cancer types and subtypes to existing drugs and drug
candidates, and uncover patient groups that may respond to
potential therapies being developed by Lantern and its
collaborators.
Forward-looking Statements
This press release contains
forward-looking statements within the meaning of Section 27A of the
Securities Act of 1933, as amended, and Section 21E of the
Securities Exchange Act of 1934, as amended. These forward-looking
statements include, among other things, statements relating to:
future events or our future financial performance; the potential
advantages of our RADR® platform in identifying drug
candidates and patient populations that are likely to respond to a
drug candidate; our strategic plans to advance the development of
our drug candidates and antibody drug conjugate (ADC) development
program; estimates regarding the development timing for our drug
candidates and ADC development program; our research and
development efforts of our internal drug discovery programs and the
utilization of our RADR® platform to streamline the
drug development process; our intention to leverage artificial
intelligence, machine learning and genomic data to streamline and
transform the pace, risk and cost of oncology drug discovery and
development and to identify patient populations that would likely
respond to a drug candidate; estimates regarding potential markets
and potential market sizes; sales estimates for our drug candidates
and our plans to discover and develop drug candidates and to
maximize their commercial potential by advancing such drug
candidates ourselves or in collaboration with others. Any
statements that are not statements of historical fact (including,
without limitation, statements that use words such as "anticipate,"
"believe," "contemplate," "could," "estimate," "expect," "intend,"
"seek," "may," "might," "plan," "potential," "predict," "project,"
"target," "objective," "aim," "upcoming," "should," "will,"
"would," or the negative of these words or other similar
expressions) should be considered forward-looking statements. There
are a number of important factors that could cause our actual
results to differ materially from those indicated by the
forward-looking statements, such as (i) the impact of the COVID-19
pandemic, (ii) the risk that our research and the research of our
collaborators relating to LP-284 may not be successful, (iii) the
risk that none of our product candidates has received FDA marketing
approval, and we may not be able to successfully initiate, conduct,
or conclude clinical testing for or obtain marketing approval for
our product candidates, (iv) the risk that no drug product based on
our proprietary RADR A.I. platform has received FDA marketing
approval or otherwise been incorporated into a commercial product,
and (v) those other factors set forth in the Risk Factors section
in our Annual Report on Form 10-K for the year ended December 31, 2020, filed with the Securities and
Exchange Commission on March 10,
2021. You may access our Annual Report on Form 10-K for the
year ended December 31, 2020 under
the investor SEC filings tab of our website at
www.lanternpharma.com or on the SEC's website at www.sec.gov.
Given these risks and uncertainties, we can give no assurances that
our forward-looking statements will prove to be accurate, or that
any other results or events projected or contemplated by our
forward-looking statements will in fact occur, and we caution
investors not to place undue reliance on these statements. All
forward-looking statements in this press release represent our
judgment as of the date hereof, and, except as otherwise required
by law, we disclaim any obligation to update any forward-looking
statements to conform the statement to actual results or changes in
our expectations.
CONTACT:
Investor Relations
David
Waldman/Natalya Rudman,
Crescendo Communications, LLC
IR@lanternpharma.com
212-671-1021
1
https://www.lls.org/facts-and-statistics/facts-and-statistics-overview
 View original content to download
multimedia:https://www.prnewswire.com/news-releases/lantern-pharma-presents-positive-data-on-the-effectiveness-of-lp-284-in-hematologic-cancers-at-the-63rd-american-society-of-hematology-ash-annual-meeting-301444227.html
View original content to download
multimedia:https://www.prnewswire.com/news-releases/lantern-pharma-presents-positive-data-on-the-effectiveness-of-lp-284-in-hematologic-cancers-at-the-63rd-american-society-of-hematology-ash-annual-meeting-301444227.html
SOURCE Lantern Pharma