Current Report Filing (8-k)
July 16 2020 - 4:16PM
Edgar (US Regulatory)
UNITED
STATES
SECURITIES
AND EXCHANGE COMMISSION
Washington,
D.C. 20549
FORM
8-K
CURRENT
REPORT
Pursuant
to Section 13 or 15(d) of the Securities Exchange Act of 1934
Date
of Report (Date of earliest event reported): July 10, 2020
Lantern
Pharma Inc.
(Exact
name of registrant as specified in its charter)
|
Delaware
|
|
001-39318
|
|
46-3973463
|
|
(State
or Other Jurisdiction
of
Incorporation)
|
|
(Commission
File Number)
|
|
(IRS
Employer
Identification
No.)
|
|
1920
McKinney Avenue, 7th Floor
Dallas,
Texas
|
|
75201
|
|
(Address
of Principal Executive Offices)
|
|
(Zip
Code)
|
(972)
277-1136
(Registrant’s
telephone number, including area code)
Check
the appropriate box below if the Form 8-K filing is intended to simultaneously satisfy the filing obligation of the registrant
under any of the following provisions (see General Instruction A.2. below):
|
☐
|
Written
communications pursuant to Rule 425 under the Securities Act (17 CFR 230.425)
|
|
☐
|
Soliciting
material pursuant to Rule 14a-12 under the Exchange Act (17 CFR 240.14a-12)
|
|
☐
|
Pre-commencement
communications pursuant to Rule 14d-2(b) under the Exchange Act (17 CFR 240.14d-2(b))
|
|
☐
|
Pre-commencement
communications pursuant to Rule 13e-4(c) under the Exchange Act (17 CFR 240.13e-4(c))
|
Securities
registered pursuant to Section 12(b) of the Act: Common Stock
|
Title of each class
|
|
Trading Symbol
|
|
Name of each exchange on which registered
|
|
Common Stock, $0.0001 par value
|
|
LTRN
|
|
The Nasdaq Stock Market
|
Indicate
by check mark whether the registrant is an emerging growth company as defined in Rule 405 of the Securities Act of 1933 (§230.405
of this chapter) or Rule 12b-2 of the Securities Exchange Act of 1934 (§240.12b-2 of this chapter).
Emerging
growth company þ
If
an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for
complying with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act. ☐
Item
1.01 Entry into a Material Definitive Agreement.
On
July 10, 2020, we entered into an agreement with Patheon API Services, Inc. (“Patheon”) for the manufacture and supply
of cGMP material to support our planned Phase II clinical trial for our product candidate LP-300.
In
addition to producing LP-300 API (active pharmaceutical ingredient) under cGMP (current Good Manufacturing Practices) conditions,
Patheon will transfer previously validated manufacturing processes and analytical methods for LP-300 and will produce non-GMP
material that can be used to support non-clinical studies for LP-300. The agreement provides for payments in stages as specified
process and manufacturing milestones are achieved.
Patheon,
a part of Thermo Fisher Scientific, has previously developed and/or manufactured more than 700 pharmaceuticals for biopharma clients
and has more than 55 locations around the world, providing access to a fully integrated global network of facilities.
SIGNATURES
Pursuant
to the requirements of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf
by the undersigned hereunto duly authorized.
|
|
Lantern
Pharma Inc.,
|
|
|
A
Delaware Corporation
|
|
|
|
|
Dated:
July 16, 2020
|
By:
|
/s/
David R. Margrave
|
|
|
|
David
R. Margrave, Chief Financial Officer
|
2
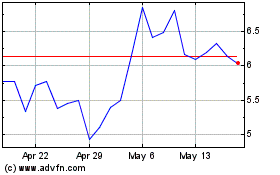
Lantern Pharma (NASDAQ:LTRN)
Historical Stock Chart
From Jun 2024 to Jul 2024
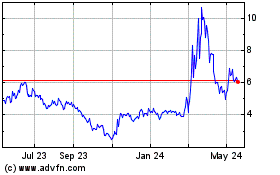
Lantern Pharma (NASDAQ:LTRN)
Historical Stock Chart
From Jul 2023 to Jul 2024