Ironwood Pharmaceuticals Appoints Michael Shetzline, M.D., Ph.D. as Chief Medical Officer
January 06 2019 - 5:00PM
Business Wire
– Dr. Shetzline will lead global product
development for Ironwood following separation –
Ironwood Pharmaceuticals, Inc. (NASDAQ: IRWD) today announced
that Michael Shetzline, M.D., Ph.D., will join Ironwood as chief
medical officer, senior vice president and head of drug
development, effective January 28, 2019. Dr. Shetzline will lead
global product development for Ironwood following its planned
separation into two independent, publicly-traded companies. In his
role, Dr. Shetzline will focus on driving innovation designed to
enhance the value of the company’s existing GI assets and creating
new GI product opportunities. He will report to Thomas McCourt, who
will become President of Ironwood following the separation.
This press release features multimedia. View
the full release here:
https://www.businesswire.com/news/home/20190106005100/en/
Michael Shetzline (Photo: Business
Wire)
Mr. McCourt commented, “Mike is one of the most experienced drug
developers in GI and a vital addition to the Ironwood team as we
focus on building a leading GI healthcare company. His vast
experience leading global clinical development, regulatory and
commercial strategy across multiple therapeutic areas including GI
will be pivotal for us as we advance our late-stage development
portfolio – IW-3718 for persistent GERD and MD-7246 for abdominal
pain – and seek to bring new innovative medicines to patients.”
“Ironwood is at a transformative point in its trajectory, one
that’s built on an incredibly solid foundation of GI expertise,”
said Dr. Shetzline. “From an industry perspective, it is clear that
Ironwood’s focus on GI positions it strongly for long-term growth
and I am honored and excited to lead development and expansion of
its promising pipeline.”
Dr. Shetzline is a gastroenterologist and internist, bringing
exceptional expertise from more than 25 years in the
biopharmaceutical industry and academia. His proven track record is
derived from his leadership and vast experience in all facets of
drug development – including discovery research, translational
medicine and clinical development – across many areas of GI science
including functional GI disorders, inflammatory bowel disease,
microbiome, rare diseases, and acid related disorders. Dr.
Shetzline has been involved in several successful approved drug
development programs in the U.S. and globally. He also has
extensive experience in managing collaborations, including serving
on the joint development committees for early- and late-stage
development ventures. Before joining Ironwood, Dr. Shetzline served
as vice president and head of gastroenterology clinical sciences at
Takeda Pharmaceuticals International Co., where he led global
clinical development for all GI assets. Prior to his role at
Takeda, he served as vice president and global head of
gastroenterology at Ferring International Pharmascience Center
U.S., Inc, during which he led the largest clinical development
program in functional GI disorders. Before that, he was vice
president and global program head, integrated hospital care,
critical care and cardiovascular and metabolism, and head of
translational medicine GI discovery at Novartis Pharmaceuticals AG.
Dr. Shetzline also played a key role on the U.S. GI franchise team
supporting ZELNORM while at Novartis. Dr. Shetzline had a
successful career within academia serving as gastroenterology
program director and assistant professor of medicine at Duke
University Medical Center in the U.S. He has published over 40 full
papers and book chapters and acted as a reviewer for a range of
medical journals, including authoring a chapter in Sleisenger and
Fordtran’s Gastrointestinal and Liver Disease on Gastrointestinal
Hormones. Dr. Shetzline earned his M.D. and Ph.D. at The Ohio State
University in physiology and medicine. He completed his internal
medicine residency and fellowship in gastroenterology as well
as serving on the faculty as a National Institutes of
Health (NIH) supported physician scientist at Duke University
Medical Center. Dr. Shetzline is a Fellow of the American College
of Physicians, the American College of Gastroenterology, and
American Gastroenterological Association.
About Ironwood Pharmaceuticals
Ironwood Pharmaceuticals (Nasdaq: IRWD) is a commercial
biotechnology company focused on creating medicines that make a
difference for patients, building value for our fellow
shareholders, and empowering our passionate team. We discovered,
developed and are commercializing linaclotide, the U.S. branded
prescription market leader for adults with irritable bowel syndrome
with constipation (IBS-C) or chronic idiopathic constipation (CIC).
Our pipeline priorities for linaclotide include a Phase IIIb trial
evaluating its efficacy and safety on multiple abdominal symptoms,
including abdominal bloating, pain, and discomfort in adult
patients with IBS-C, as well as research into a formulation of
linaclotide designed to relieve abdominal pain associated with
IBS.
We are also advancing a pipeline of innovative product
candidates in areas of significant unmet need, including persistent
gastroesophageal reflux disease, diabetic nephropathy, heart
failure with preserved ejection fraction and sickle cell disease.
Ironwood was founded in 1998 and is headquartered
in Cambridge, Mass. For more information, please
visit www.ironwoodpharma.com
or www.twitter.com/ironwoodpharma; information that may be
important to investors will be routinely posted in both these
locations.
Forward-Looking Statements
This press release contains forward-looking statements.
Investors are cautioned not to place undue reliance on these
forward-looking statements, including statements about Ironwood's
leadership team, the strength and value thereof, and leadership’s
impact on the company and its business, growth, business strategy,
pipeline advancement, productivity and the potential of its
products and product candidates and their impact; the completion
and timing of the planned separation of each of the Company
following the separation; and statements about the timing of any of
the foregoing. Each forward‐looking statement is subject to risks
and uncertainties that could cause actual results to differ
materially from those expressed or implied in such statement.
Applicable risks and uncertainties include those related to
leadership appointments; those related to the possibility that we
may not complete the separation of our business on the terms or
timeline currently contemplated, if at all, achieve the expected
benefits of the separation, and that the separation could harm our
business, results of operations and financial condition; the risk
that the transaction might not be tax-free; the risk that we may be
unable to make, on a timely or cost-effective basis, the changes
necessary to operate as independent companies; Cyclerion’s lack of
independent operating history and the risk that its accounting and
other management systems may not be prepared to meet the financial
reporting and other requirements of operating as an independent
public company; the risk that a separation may adversely impact our
ability to attract or retain key personnel; the effectiveness of
development and commercialization efforts by us and our partners;
preclinical and clinical development, manufacturing and formulation
development; the risk that findings from our completed nonclinical
and clinical studies may not be replicated in later studies;
efficacy, safety and tolerability of our products and product
candidates; decisions by regulatory authorities; the risk that we
may never get sufficient patent protection for our products and our
product candidates or that we are not able to successfully protect
such patents; the outcomes in legal proceedings to protect or
enforce the patents relating to our products and product
candidates, including ANDA litigation; developments in the
intellectual property landscape; challenges from and rights of
competitors or potential competitors; the risk that our planned
investments do not have the anticipated effect on our company
revenues, our products or product candidates; the risk that we are
unable to manage our operating expenses or cash use for operations,
or are unable to commercialize our products, within the guided
ranges or otherwise as expected and those risks listed under the
heading "Risk Factors" and elsewhere in Ironwood's Quarterly Report
on Form 10-Q for the quarter ended September 30, 2018, and in our
subsequent SEC filings. These forward-looking statements (except as
otherwise noted) speak only as of the date of this press release,
and Ironwood undertakes no obligation to update these
forward-looking statements.
View source
version on businesswire.com: https://www.businesswire.com/news/home/20190106005100/en/
Media and Investors:Meredith Kaya, 617-374-5082Vice
President, Investor Relations and Corporate
Communicationsmkaya@ironwoodpharma.com
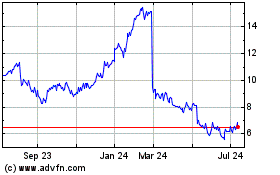
Ironwood Pharmaceuticals (NASDAQ:IRWD)
Historical Stock Chart
From Aug 2024 to Sep 2024
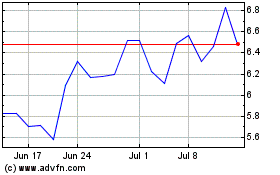
Ironwood Pharmaceuticals (NASDAQ:IRWD)
Historical Stock Chart
From Sep 2023 to Sep 2024