Genprex Announces Formation of Clinical Advisory Board
February 08 2021 - 3:30PM
Business Wire
World reknowned medical and clinical experts to
guide Genprex’ novel gene therapy clinical development programs in
cancer and diabetes, including its two upcoming clinical trials in
lung cancer
Genprex, Inc. (“Genprex” or the “Company”) (NASDAQ: GNPX), a
clinical-stage gene therapy company focused on developing
life-changing therapies for patients with cancer and diabetes,
today announced the formation of a Clinical Advisory Board (CAB) to
support its oncology and diabetes development programs. Comprised
of preeminent clinical specialists, the CAB will lead and advise
Genprex as it advances its REQORSA™ immunogene therapy program,
including its Acclaim clinical trials in non-small cell lung
cancer, and its preclinical diabetes gene therapy program. Some of
the members also serve in additional roles at the Company.
“We are pleased to bring together this group of seasoned
practitioners in the field of oncology and diabetes to guide and
advise Genprex in advancing our clinical development programs,”
said Genprex’s Chief Executive Officer, Rodney Varner. “The
Clinical Advisory Board’s experience in clinical trials of cancer
and diabetes therapies, combined with their expertise in consulting
with small and large biotechnology companies, is invaluable as our
drug candidates progress into the clinic.”
The members of Genprex’ Clinical Advisory Board include:
- Michael Morse, MD, MHS, FACP – Dr. Morse is a Professor
of Medicine in the Division of Medical Oncology and Professor in
the Department of Surgery at Duke University Medical Center,
Durham, NC. His clinical expertise includes management of
gastrointestinal malignancies including colon, hepatobiliary,
gastroesophageal and pancreatic cancer. His research expertise
includes the development of targeted therapies including
immunotherapies for cancer. He has published extensively on topics
in cancer immunotherapy, targeted therapies and gastrointestinal
malignancies, including textbooks on Cancer Vaccines and Liver
Tumors. Dr. Morse is Editor for Current Opinions in Biologic
Therapies. He has been the principal investigator of a number of
investigator-initiated and industry-supported phase 1 and 2
clinical trials of cancer immunotherapy and therapies for
gastrointestinal malignancies. He is a member of the American
Association of Cancer Research, the American Society of Clinical
Oncology, the American Society for Clinical Investigation, American
College of Physicians, and the Society for the Immunotherapy of
Cancer.
- Col. George E. Peoples, MD, FACS– Dr. Peoples served 30
years of active duty as a surgeon and research scientist in the
military. He is the Founder and Director of the Cancer Vaccine
Development Program (CVDP), which has been focused on the
discovery, development, and clinical testing of cancer vaccines for
more than 20 years. Four of the program’s cancer vaccines have been
licensed for commercial development. Dr. Peoples currently serves
as the CEO of Cancer Insight, LLC, CVDP’s commercial counterpart,
which is a boutique cancer immunotherapy Contract Research
Organization conducting multiple Phase 1 and 2 clinical trials. He
also serves as Professor of Surgery at the Uniformed Services
University of the Health Sciences and Professor (adjunct) of
Surgical Oncology at MD Anderson Cancer Center (MDACC). Dr. Peoples
is the past Chair of the Cancer Care Program, San Antonio Military
Medical Center and the past Military Director of the United States
Military Cancer Institute. He is a graduate of the United States
Military Academy, West Point and the Johns Hopkins School of
Medicine. Dr. Peoples completed his surgical training at Harvard’s
Brigham and Women’s Hospital and a surgical oncology fellowship at
MDACC.
- Andrew B. Becker, MD, PhD – Dr. Becker is President and
Founder of Becker Pharmaceutical Consulting, a market research,
competitive intelligence and strategic planning consulting firm
that provides analytic and strategy services to companies ranging
from small biotechnology and medical device companies up to large
multinational pharmaceutical companies on a global basis. Dr.
Becker received both his medical degree and PhD in Molecular
Pharmacology from Stanford University. At Stanford, Dr. Becker's
research focused on signaling pathways for the insulin and IGF–1
receptors, deciphering the structure and function of the insulin
degrading enzyme and its role in insulin processing.
- William E. Gannon, Jr., MD, MBA– Dr. Gannon serves as
Genprex’s Vice President of Regulatory Affairs. He is responsible
for managing the Company’s regulatory affairs for its upcoming
clinical trials. He also will serve as the Medical Monitor,
interfacing with medical personnel at trial sites and assisting
with FDA communication throughout the clinical trials. Dr. Gannon
has more than 30 years of experience in the biotech and
pharmaceutical industries, with expertise in clinical development,
regulatory affairs and commercialization of products, and a strong
background in oncology and gene therapy.
About Genprex, Inc.
Genprex, Inc. is a clinical-stage gene therapy company focused
on developing life-changing therapies for patients with cancer and
diabetes. Genprex’s technologies are designed to administer
disease-fighting genes to provide new therapies for large patient
populations with cancer and diabetes who currently have limited
treatment options. Genprex works with world-class institutions and
collaborators to develop drug candidates to further its pipeline of
gene therapies in order to provide novel treatment approaches. The
Company’s lead product candidate, REQORSA™ (quaratusugene
ozeplasmid), is being evaluated as a treatment for non-small cell
lung cancer (NSCLC). REQORSA has a multimodal mechanism of action
that has been shown to interrupt cell signaling pathways that cause
replication and proliferation of cancer cells; re-establish
pathways for apoptosis, or programmed cell death, in cancer cells;
and modulate the immune response against cancer cells. REQORSA has
also been shown to block mechanisms that create drug resistance. In
January 2020, the U.S. Food and Drug Administration granted Fast
Track Designation for REQORSA for NSCLC in combination therapy with
osimertinib (AstraZeneca’s Tagrisso®) for patients with EFGR
mutations whose tumors progressed after treatment with osimertinib
alone.
For more information, please visit the Company’s web site at
www.genprex.com or follow Genprex on Twitter, Facebook and
LinkedIn.
Forward-Looking Statements
Statements contained in this press release regarding matters
that are not historical facts are "forward-looking statements"
within the meaning of the Private Securities Litigation Reform Act
of 1995. Because such statements are subject to risks and
uncertainties, actual results may differ materially from those
expressed or implied by such forward-looking statements. Such
statements include, but are not limited to, statements regarding
the effect of Genprex’s product candidates, alone and in
combination with other therapies, on cancer and diabetes, regarding
potential, current, regarding the Company’s future growth and
financial status and regarding our commercial partnerships and
intellectual property licenses. Risks that contribute to the
uncertain nature of the forward-looking statements include the
presence and level of the effect of our product candidates, alone
and in combination with other therapies, on cancer; the timing and
success of our clinical trials and planned clinical trials of
REQORSA™ immunogene therapy drug, alone and in combination with
targeted therapies and/or immunotherapies, and whether our other
potential product candidates, including GPX-002, our gene therapy
in diabetes, advance into clinical trials; the success of our
strategic partnerships, including those relating to manufacturing
of our product candidates; the timing and success at all of
obtaining any FDA approvals of REQORSA and our other potential
product candidates including whether we receive necessary approvals
to commence clinical trials or benefit from fast track or similar
regulatory designations; costs associated with developing our
product candidates, whether we identify and succeed in acquiring
other technologies and whether patents will ever be issued under
patent applications that are the subject of our license agreements
or otherwise. These and other risks and uncertainties are described
more fully under the caption “Risk Factors” and elsewhere in our
filings and reports with the United States Securities and Exchange
Commission. All forward-looking statements contained in this press
release speak only as of the date on which they were made. We
undertake no obligation to update such statements to reflect events
that occur or circumstances that exist after the date on which they
were made.
View source
version on businesswire.com: https://www.businesswire.com/news/home/20210208005812/en/
Genprex, Inc. (877) 774-GNPX (4679)
Investor Relations GNPX Investor Relations (877) 774-GNPX
(4679) ext. #2 investors@genprex.com
Media Contact Genprex Media Relations Kalyn Dabbs (877)
774-GNPX (4679) ext. #3 media@genprex.com
Genprex (NASDAQ:GNPX)
Historical Stock Chart
From Aug 2024 to Sep 2024
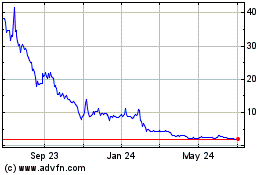
Genprex (NASDAQ:GNPX)
Historical Stock Chart
From Sep 2023 to Sep 2024