Caribou Biosciences Gets FDA Clearance for CB-011 IND Application
November 21 2022 - 9:50AM
Dow Jones News
By Chris Wack
Caribou Biosciences Inc. said Monday that it has received U.S.
Food and Drug Administration clearance of its investigational new
drug application for CB-011, a genome-edited allogeneic anti-BCMA
CAR-T cell therapy with immune cloaking.
The company said the Phase 1 clinical trial, an open-label study
to evaluate the safety and efficacy of a single dose of CB-011 in
adult patients with relapsed or refractory multiple myeloma, is
expected to initiate patient enrollment for treatment at dose level
1 in early 2023.
CB-011 is the second allogeneic cell therapy advancing into
clinical development from Caribou's CAR-T cell platform targeting
hematologic malignancies.
Caribou Biosciences shares were up 5% to $9.55 in premarket
trading.
Write to Chris Wack at chris.wack@wsj.com
(END) Dow Jones Newswires
November 21, 2022 09:35 ET (14:35 GMT)
Copyright (c) 2022 Dow Jones & Company, Inc.
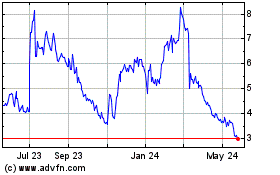
Caribou Biosciences (NASDAQ:CRBU)
Historical Stock Chart
From Jun 2024 to Jul 2024
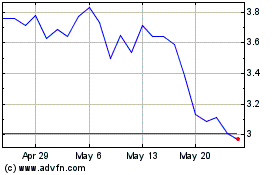
Caribou Biosciences (NASDAQ:CRBU)
Historical Stock Chart
From Jul 2023 to Jul 2024