Brazil Authorizes Use of Two Covid-19 Vaccines -- Update
January 17 2021 - 2:06PM
Dow Jones News
By Samantha Pearson and Luciana Magalhaes
Brazilian health regulator Anvisa voted Sunday to approve the
use of the country's first Covid-19 vaccines, China's CoronaVac and
the shot developed by AstraZeneca PLC and Oxford University,
raising spirits in a nation that is struggling to cope with a surge
in infections.
The ruling follows a tug of war between President Jair Bolsonaro
and São Paulo's powerful state governor, João Doria, to secure the
first vaccines for the country of more than 210 million people.
Gov. Doria, who is expected to run for president next year, has
led a partnership with Chinese company Sinovac Biotech Ltd. to test
and produce CoronaVac in São Paulo, criticizing what he has called
a "genocidal" lack of action by Mr. Bolsonaro.
The dispute comes as the country's average daily death toll
approaches 1,000 people, with scientists warning that a new variant
that likely emerged in the Amazon could be partly responsible for a
recent surge in infections. For the past week, Brazil has
registered an average of more than 50,000 new cases of the disease
each day -- the highest since the pandemic began.
Write to Samantha Pearson at samantha.pearson@wsj.com and
Luciana Magalhaes at Luciana.Magalhaes@wsj.com
(END) Dow Jones Newswires
January 17, 2021 13:51 ET (18:51 GMT)
Copyright (c) 2021 Dow Jones & Company, Inc.
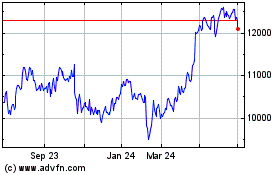
Astrazeneca (LSE:AZN)
Historical Stock Chart
From Mar 2024 to Apr 2024
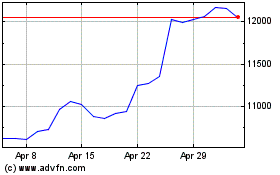
Astrazeneca (LSE:AZN)
Historical Stock Chart
From Apr 2023 to Apr 2024