Philips starts repair and replacement program of first-generation DreamStation devices in the US in relation to earlier announced recall notification*
September 01 2021 - 2:00AM

September 1, 2021
Amsterdam, the Netherlands – Royal Philips
(NYSE: PHG; AEX: PHIA) today announced an update in connection with
the June 14, 2021 recall notification* for specific Philips sleep
and respiratory care devices that was issued to address potential
health risks related to the polyester-based polyurethane (PE-PUR)
sound abatement foam component in these devices. More than half of
the affected devices in use globally are in the US. The vast
majority (>80%) of the registered affected devices in the US to
date are in the first-generation DreamStation product family.
Philips received authorization from the US Food and Drug
Administration (FDA) for the rework of the affected
first-generation DreamStation devices [1], which consists of
replacement of the PE-PUR sound abatement foam with a new material.
Philips anticipates rework to commence in the course of September
2021. In addition to the rework, the company has already started
replacing certain affected first-generation DreamStation CPAP
devices in the US with DreamStation 2 CPAP devices. Philips remains
in dialogue with the FDA with respect to other aspects of the
recall notification and mitigation plan in the US [2].
Philips is initiating the repair and replacement programs in
other countries as well and expects to have these underway in the
majority of its markets by the end of September 2021. The company
intends to complete the repair and replacement programs within
approximately 12 months.
“We fully recognize that the timeframe for remediation of the
affected devices places patients in a difficult situation,” said
Frans van Houten, CEO of Royal Philips. “We are mobilized to
deliver a solution to them as fast as possible. We have
significantly increased our production, service and rework
capacity, and further intensified our outreach to our customers and
their patients. We urge patients with affected active devices to
register these on the dedicated recall notification website.”
More information on the recall notification, as well as
instructions for customers, users, and physicians can be found at
www.philips.com/src-update. Patients with affected devices
currently in use are requested to register their products on this
website to facilitate the repair and replacement program.
* This is a
recall notification for the US only, and a field safety notice for
the rest of the world. In the US, the recall notification has been
classified by the FDA as a Class I
recall.[1] This
includes DreamStation CPAP, Auto CPAP; Dream Station Bi-Level PAP;
DreamStation ASV; and DreamStation ST, AVAPS
devices.[2] The
remaining affected devices for remediation in the US can be found
at www.philips.com/src-update.
For media
information, please contact:
Ben ZwirsPhilips Global Press OfficeTel.: +31 6 15213446E-mail:
ben.zwirs@philips.com
Derya GuzelPhilips Investor RelationsTel.: +31 20 59
77055E-mail: derya.guzel@philips.com
About Royal PhilipsRoyal Philips (NYSE: PHG,
AEX: PHIA) is a leading health technology company focused on
improving people's health and enabling better outcomes across the
health continuum from healthy living and prevention, to diagnosis,
treatment and home care. Philips leverages advanced technology and
deep clinical and consumer insights to deliver integrated
solutions. Headquartered in the Netherlands, the company is a
leader in diagnostic imaging, image-guided therapy, patient
monitoring and health informatics, as well as in consumer health
and home care. Philips generated 2020 sales of EUR 17.3 billion and
employs approximately 77,000 employees with sales and services in
more than 100 countries. News about Philips can be found at
www.philips.com/newscenter.
Forward-looking statements This release
contains certain forward-looking statements with respect to the
financial condition, results of operations and business of Philips
and certain of the plans and objectives of Philips with respect to
these items. Examples of forward-looking statements include
statements made about the strategy, estimates of sales growth,
future EBITA, future developments in Philips’ organic business and
the completion of acquisitions and divestments. By their nature,
these statements involve risk and uncertainty because they relate
to future events and circumstances and there are many factors that
could cause actual results and developments to differ materially
from those expressed or implied by these statements.
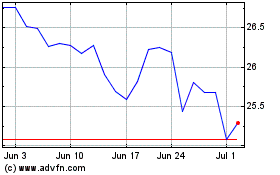
Koninklijke Philips NV (NYSE:PHG)
Historical Stock Chart
From Aug 2024 to Sep 2024
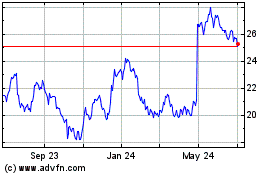
Koninklijke Philips NV (NYSE:PHG)
Historical Stock Chart
From Sep 2023 to Sep 2024