J&J Says Covid-19 Vaccine Batch Didn't Meet Quality Standards -- Update
March 31 2021 - 7:42PM
Dow Jones News
By Peter Loftus and Thomas M. Burton
Johnson & Johnson said one batch of its new Covid-19 vaccine
didn't meet quality standards at a contract manufacturer, and the
doses weren't distributed.
J&J said Wednesday the batch never advanced to the filling
and finish stages of its manufacturing process, and that the
quality lapse wouldn't affect its ability to supply the U.S. with
100 million doses by the end of May.
J&J didn't disclose the nature of the quality lapse or how
many doses were affected, but said it arose from quality checks
during the start-up phase of manufacturing. The company said it
shared information about the issue with the U.S. Food and Drug
Administration.
The FDA is investigating, according to a person familiar with
the matter.
The New York Times reported Wednesday that the J&J doses
were ruined due to an accidental mix-up of ingredients at Emergent
BioSolutions Inc., the contract manufacturer working with J&J.
The Times reported that about 15 million doses were ruined.
Emergent declined to comment.
J&J's vaccine was the third to be authorized for use against
Covid-19, after shots from Pfizer Inc. and its partner BioNTech SE
and from Moderna Inc. Health authorities especially welcomed the
addition of the J&J vaccine because it requires just one dose
and is easier to store.
Supplies in the U.S. were expected to increase as J&J's
manufacturing network ramped up production, accelerating a mass
vaccination campaign that has been gaining steam.
J&J said it is providing additional experts in
manufacturing, technical operations and quality to be on-site at
Emergent to oversee all manufacturing of the J&J vaccine
there.
Emergent, a contract manufacturer based in Gaithersburg, Md.,
has been making the main ingredient for J&J's vaccine at an
Emergent plant in Baltimore.
At the same plant, Emergent also makes the main ingredient for
AstraZeneca PLC's Covid-19 vaccine, which hasn't yet been
authorized for use in the U.S.
J&J's own plant in the Netherlands has been making the main
ingredient for the initial U.S. supply of its vaccine -- including
the nearly four million doses that were distributed immediately
after it was authorized in late February.
J&J said it was able to meet a target of delivering a total
of 20 million vaccine doses by the end of March. The company said
it expects to deliver an additional 24 million doses in April, and
plans to have delivered a total of 100 million by the end of
May.
Write to Peter Loftus at peter.loftus@wsj.com and Thomas M.
Burton at tom.burton@wsj.com
(END) Dow Jones Newswires
March 31, 2021 19:27 ET (23:27 GMT)
Copyright (c) 2021 Dow Jones & Company, Inc.
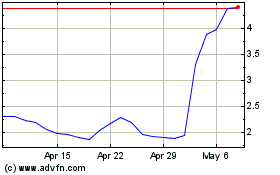
Emergent Biosolutions (NYSE:EBS)
Historical Stock Chart
From Apr 2024 to May 2024
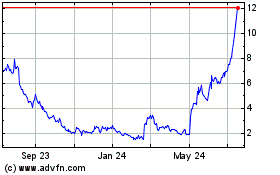
Emergent Biosolutions (NYSE:EBS)
Historical Stock Chart
From May 2023 to May 2024