Karuna Therapeutics Announces Topline Data From Phase 1b Trial Evaluating KarXT on Experimentally Induced Pain in Healthy Vol...
August 03 2020 - 4:30PM
Business Wire
Topline results of exploratory trial do not
provide conclusive evidence of an analgesic benefit of KarXT on
experimentally induced pain in healthy volunteers
The Company will not move forward to evaluate
KarXT for the treatment of pain
The safety and tolerability profile of KarXT
was consistent with previous trials
Karuna Therapeutics, Inc. (NASDAQ: KRTX), an innovative
clinical-stage biopharmaceutical company committed to developing
novel therapies with the potential to transform the lives of people
with disabling and potentially fatal neuropsychiatric disorders,
today announced results from KAR-020, its Phase 1b trial evaluating
the analgesic effects of KarXT on experimentally induced pain in
healthy volunteers. Topline results of this exploratory trial were
inconclusive and do not provide sufficient evidence of an analgesic
benefit of KarXT compared to placebo. The Company will not move
forward to develop KarXT in pain.
The KAR-020 trial employed an intra-individual cross-over design
to evaluate objective and subjective pain measurements of KarXT and
placebo in three different skin conditions meant to model
nociceptive (normal skin), inflammatory (UVB-irradiated skin) and
neuropathic (capsaicin-treated skin) pain in 24 healthy volunteers.
The safety and tolerability profile of KarXT was consistent with
previous clinical trials of KarXT. There were no serious adverse
events observed in the trial, and no clinically meaningful changes
in vital signs or clinical laboratory values.
“Today’s results do not provide compelling evidence of the
analgesic benefit observed in preclinical trials,” said Steve Paul,
M.D., chief executive officer, president and chairman of Karuna
Therapeutics. “While this data does not support the continued
development of KarXT in pain, we will continue to evaluate data
from this trial to better assess the potential role of
muscarinic-targeted receptor treatments in pain management.”
Dr. Paul continued: “Looking ahead, we remain focused on
developing KarXT for the treatment of severe neuropsychiatric
disorders, including schizophrenia and dementia-related psychosis,
where robust evidence from previous clinical trials supports the
potential of KarXT to provide a new, unique and mechanistically
differentiated therapeutic for the treatment of these disorders. We
remain on track to initiate Phase 3 development in schizophrenia
later this year.”
About KarXT
KarXT, a proprietary oral modulator of muscarinic cholinergic
receptors, is Karuna’s lead product candidate that combines
xanomeline, a novel muscarinic agonist, with trospium, an
FDA-approved muscarinic antagonist that does not appreciably cross
the blood-brain-barrier, to preferentially stimulate muscarinic
receptors in the central nervous system (CNS). This novel product
candidate, if approved, has the potential to usher in a new
treatment paradigm and dramatically impact patients with
schizophrenia and other psychotic disorders by providing a
differentiated mechanism of action relative to current D2 dopamine
and serotonin receptor-targeting antipsychotic drugs. KarXT is
currently being evaluated in clinical development as a potential
treatment for serious neuropsychiatric disorders, including
schizophrenia and dementia-related psychosis.
About Karuna
Karuna is a clinical-stage biopharmaceutical company committed
to developing and delivering first-in-class therapies with the
potential to transform the lives of people with CNS disorders –
which remain among the most disabling and potentially fatal
disorders worldwide. Galvanized by the understanding that today’s
neuropsychiatric patients deserve better, Karuna’s mission is to
harness the untapped potential of the brain’s complex biology in
pursuit of novel therapeutic pathways that will advance the
standard of care. For more information, please visit
karunatx.com.
Forward Looking Statements
This press release contains forward looking statements within
the meaning of Section 27A of the Securities Act of 1933, as
amended, and Section 21E of the Securities Exchange Act of 1934, as
amended, including statements regarding the timing of advancing of
our planned clinical trials and regulatory filings, interim trial
results, our goals to develop and commercialize our product
candidates, and other statements identified by words such as
“could,” “expects,” “intends,” “may,” “plans,” “potential,”
“should,” “will,” “would,” or similar expressions and the negatives
of those terms. Forward-looking statements are not promises or
guarantees of future performance, and are subject to a variety of
risks and uncertainties, many of which are beyond our control, and
which could cause actual results to differ materially from those
contemplated in such forward-looking statements. These factors
include risks related to our limited operating history, our ability
to generate positive clinical trial results for our product
candidates, the timing and scope of regulatory approvals, changes
in laws and regulations to which we are subject, competitive
pressures, our ability to identify additional product candidates,
risks relating to business interruptions resulting from the
coronavirus (COVID-19) pandemic, and other risks set forth under
the heading “Risk Factors” of our Annual Report on Form 10-K for
the year ended December 31, 2019. Our actual results could differ
materially from the results described in or implied by such
forward-looking statements. Forward-looking statements speak only
as of the date hereof, and, except as required by law, we undertake
no obligation to update or revise these forward-looking
statements.
View source
version on businesswire.com: https://www.businesswire.com/news/home/20200803005741/en/
Investor Contact Alexis Smith 518-338-8990
asmith@karunatx.com
Karuna Therapeutics (NASDAQ:KRTX)
Historical Stock Chart
From Aug 2024 to Sep 2024
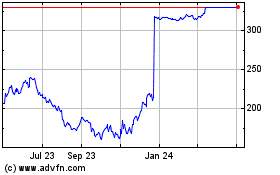
Karuna Therapeutics (NASDAQ:KRTX)
Historical Stock Chart
From Sep 2023 to Sep 2024