By Thomas Barnard
Originally Published 18 December 2018
Summary
Merck collaboration on Phase II cancer trial against triple
negative breast cancer and Shenzhen collaboration for Phase II
cancer trial against prostate cancer.
Buccal delivery system for insulin inside the cheek of the mouth
has been concentrated so that what took 6-20 sprays only requires
2-5 sprays.
Purchased Veneto Holdings, an MSO (medical services
organization) with sales in the tens of millions.
Purchased 51% of Regentys Corp. whose patent for ulcerative
colitis just received a European patent.
Purchased 51% Olaregen Therapeutix, whose product Excellagen can
be used to treat diabetic wounds, surgical wounds, and wounds.
The principal research of Generex Biotechnology (OTCQB:GNBT) for years has been
its buccal delivery system. The buccal tissue is the tissue on the
inside of the cheek and under the tongue. Generex buccal delivery
sprays a solution under a high gas aerosol formulation containing a
drug on to the blood-rich tissue inside the cheek and under the
tongue. The solution does not go into the lungs.
In 2011, Generex completed the Phase III trial of its buccal
delivery of insulin. The trial was neither positive nor negative,
but the FDA wanted more trials (see more under interview with James
Anderson). The company was tapped out on money, and the company and
the stock went into coma. There was little activity, and in 2015,
the entire workforce was officially laid off.
The Challenger Deep
Generex's stock drifted down, and it ended with a final
1-for-1,000 reverse stock split, at which point it found itself at
the bottom of the Mariana Trench, a place called the Challenger
Deep, and there it sat with thousands of pounds of atmospheric
pressure on top of it, by which I mean a ton of negative sentiment.
This is a hard thing to overcome, and we will address this again at
the end of this article. When the stock finally reached bottom, the
stock was within about $2 million of disappearing. It is a place so
low that it is just a graveyard of whale bones lying on top of the
ocean silt. Usually, when stocks reach such a low level, they
disappear. Generex did not disappear thanks to the efforts of
Joseph Moscato and his day-by-day efforts to formulate a solid
management team, and by the scientists who wanted to see their
technologies through to realization.
The Changes Begin
Joseph Moscato was elected CEO on January 17, 2017. He came in
with the Hema Diagnostics acquisition. But he didn't just come in
out of the blue; he has been involved with Generex since 2009 as a
consultant only, never management, and never a board member, just a
voice sometimes heard above the din. In the SEC filings, you can
find this on his background on the 10K:
Mr. Moscato has over 30 years of experience in healthcare, sales
and marketing, distribution management, and finance. Mr. Moscato
brings his marketing and advertising acumen to drug discovery and
diagnostic & treatment development and commercialization. Since
2009, Mr. Moscato has been working as an exclusive consultant to
the Company. He has advised on equity financings totaling over $300
million, and has implemented the broad strategic vision for the
Company. Mr. Moscato has worked and consulted for Pfizer in several
capacities from sales and marketing to new drug discovery &
development. He has worked with other biopharmaceutical companies
such as GlaxoSmithKline, Johnson & Johnson, Parke-Davis, Amgen
and others. Mr. Moscato has consulted for several healthcare
focused private equity, hedge funds and family offices.
Mr. Moscato also owned several advertising and marketing
agencies focused on media, entertainment, and healthcare with
clients ranging from Motorola, Chadmoore Wireless, Nextel, Cannon,
Sharp, GlaxoSmithKline, Pfizer, and other biopharmaceutical
companies.
He comes from marketing side, with experience in financing, a
good match for Generex, which was basically without this kind of
talent. Previous management lacked these kinds of skill sets and
connections and was not able to bring its important insulin
delivery trial to a successful conclusion.
As I read over the SEC filings, especially in retrospect, I
think you could spot Joe Moscato behind the scenes even before his
election as CEO in certain financing parties, and acquisitions that
went to completion (Hema Diagnostic Systems), and some that did not
(Emmaus, Core Tech). The deals that did not go to completion went
sideways. "Emmaus would have ended up owning Generex, and Core Tech
lost both of its Big Pharma partnerships, rendering both of these
deals undoable." Joe's consulting work for Generex began in 2009.
And my first contact with Joe was when he called me in 2010 to
enlist support for a reverse split. This was not popular with
shareholders, so it wasn't until 2017 that it was finally
accomplished, but it was a necessary prerequisite before further
steps could be taken. Moscato: "A stock with billions of shares and
no revenues and earnings cannot be taken seriously."d
New Focus
Joseph Moscato's focus is broader than the Generex he took over,
which was essentially a small biotech research firm. As I have
said, he comes from the marketing side of things. He has developed
financial connections that would only be available to Generex
through him. And as was revealed in the January 2018 conference
call, he has already looked at over 100 firms for acquisition.
Joe's thought was that if firms with existing income could be
combined with Generex's promising technologies, Generex would not
have to keep looking for funds to finish the research. Those deals
just become more and more onerous and hellish for shareholders. So,
the idea arose to create a holding company to organize the new
acquisitions with the existing businesses, hence a new name for the
firm, NuGenerex Biotechnology Holdings, Inc., and a new mission:
direct-to-patient, end-to-end healthcare solutions for patients,
healthcare professionals, and payors. But let's give a look at the
company's research.
2018, The Year The Nobel Prize in Medicine Goes to
Cancer Vaccine Research
This is the year that the Nobel committee gave the Medicine award to
researchers in cancer vaccines. It is the year of checkpoint
inhibitors. The problem is that cancer cells disguise themselves as
the body's own cells. And the killer T-cells will not attack cells
it recognizes as those of the body. Checkpoint inhibitors are what
prevent T-cells from attacking "self" cells. James T. Allison
studied one of them, called CTLA-4, which is one of the checkpoint
proteins that brake T-cells. And Tasuku Honjo discovered another
checkpoint inhibitor, PD-1. And if the brakes are put on both
checkpoint inhibitors, even better results can be obtained.
It turns out this is exactly where the Generex subsidiary,
Antigen Express, and its primary researcher, Eric von Hofe, have
been working on lo these many years. It has done studies both
against on breast cancer and prostate cancer with success. These
are studies in which patients have been treated to the point where
no cancer can be detected; the vaccine is given to prevent
recurrence. The breast cancer study was done against triple
negative cancer, the worst. Merck (MRK), having some luck with its
Keytruda, a checkpoint inhibitor, looked around for drugs that it
might use in a combination attack. It found the Antigen Express
study on triple negative breast cancer and decided in July of 2017
to partner on a Phase II study.
The stock market took this news in stride. That is to say, it
reacted not at all.
In January 2018, Shenzhen BioScien, a Chinese biotech firm,
announced it is going to test Antigen's AE37 against prostate
cancer and paid $700,000 for the right to do a Phase II study, and
will pay more if it decides to do a Phase III study. Moscato said
to me, "They pay for everything. They pay at the given milestones.
And if the data is good, Generex can use it for partnering
everywhere else other than China. It's a win-win for Generex."
The stock market took this news in stride. That is to say, again
it reacted not at all.
A Few Puffs - Revisiting Its Drug Delivery
System
The happy thing that has occurred since the company's last visit
to the FDA is that the formula for its insulin aerosol has been
strengthened so that what took 6 to 20 sprays can now be
accomplished with 2 to 5 sprays. Users will have to test themselves
to see how many sprays inside the cheek they will need, but they
will be able to save themselves from the needle, even if
advertisers try to camouflage the pain by calling the needle a
pen.
But insulin may not be the only drug that can use this buccal
delivery system. As the CEO Joseph Moscato told me, it will depend
on the size of the molecule and the taste. He reported that it was
tried with opium-based painkillers, but the users found the taste
so abhorrent that they would rather take it with a needle. But for
those situations where it works, Big Pharma may be able to extend
the life of their patents, which could be another boon for Generex.
So, the company expects to be finding applications for its buccal
drug delivery system for years to come.

Veneto
With the acquisition of the medical services organization (MSO)
of Veneto Holdings LLC ("Veneto Group"), Generex continues the
effort to diversify and locate revenue and income producing
properties begun in January. The Veneto Group will be folded into
the recent created subsidiary, NuGenerex Distribution Solutions,
Inc. Veneto is headquartered in Dallas, with offices in Texas,
Oklahoma and Arizona. A lot of its business comes from a
specialized pharmacy, but the business provides comprehensive
support for doctors including - testing, lab work, scanning,
insurance adjudication, strategic planning, marketing, accounting,
and legal and regulatory guidance. The plan is to take the company
from the current 5 states to 27 states around the country.
Generex suddenly becomes a company with more than 100 employees
and sales in the tens of millions. Since Generex bought the
meaningful assets, no audits are needed, other than going forward
because Veneto has completed three years of audits. The company was
started in 2014, so growth has been exceptional.
What seems to be obvious is that the announcement of the
acquisition of a company with significant sales is what has
activated the stock.
Diagnostic Products
The company acquired Hema Diagnostics in 2017, renamed NuGenerex
Diagnostics, Inc., which has in the pipelines point-of-care
diagnostic tests for syphilis, tuberculosis, HIV, and sepsis.
Sadly, syphilis, which had been pretty much knocked down, is now in
a worldwide resurgence. Sepsis is a very tricky thing to diagnose
since it is an immune response, a syndrome, and not necessarily
caused by a particular bacteria or virus. The tests are cleverly
designed to be inexpensive with relatively easy to use test strips,
and management is paying attention to the matter of how it will be
paid.

oe felt I was devoting too much time on him, and so he provided
with contact information on other members of the team.
The Team
Terry Thompson, B.B.A. University of Memphis, resides in
Nashville; Chief Operating Officer
I begin with Terry Thompson, Joe's COO, and the man to head the
medical services organization, NuGenerex Distribution Services,
Inc., which where Veneto will end up, and where all the stock
market value lies at present. Terry has really, really extensive
experience, and that lies beyond the scope of this piece, but I
invite readers to check out his resume on LinkedIn, and be sure to open all the
"see more" buttons to appreciate the full bouquet of this seasoned
operating executive. But to be sure, he has a long history of
experiences making him ideal to lead Generex's new pharmacy
initiative. Here are the highlights:
He spent fifteen years at FedEx, starting at 17 years old in
Memphis when the firm had less than 100 employees, before any IPO,
in a company everyone today knows changed the way the world did
business. This early bird was there for the explosive growth, and
that led to good jobs in Chicago, Boston, and Memphis where he led
some of the largest mission critical day-in-and-day-out operations
in the industry.
After FedEx, he was recruited into a long second career in
healthcare, where he experienced a second explosive growth
situation when Paid Prescriptions and National Pharmacy were
acquired to form Medco Containment Services, Inc., which grew like
wildfire, and got sold to Merck for $6 billion. He was happy to
move on post-acquisition because he felt he been working in a world
class company to help lower and contain the cost of healthcare in a
transparent way but then felt that being bought out by Big Pharma
"clouded the appearances of the mission."
He led several other ventures since and did a lot of consulting,
and among other things, he visited Generex and met with a previous
CEO, Anna Gluskin, in Toronto, as a possible acquisition or
investment opportunity for one of his clients, but decided "there
was nothing there." It was during this time that he met Joe
Moscato. And he would hear from Joe from time to time. "And I would
think: Joe is still there. Why is he still interested?" Joe would
ask if he would like to join him, but Thompson demurred, though it
should be noted that he admired Joe's persistence. And finally, one
of Joe's calls found Thompson's situation changed, "Talk about
timing. My first wife had just died from triple negative breast
cancer at a very young age, which, of course, is what Generex's
drug AE37 is being tested for together with Merck's Keytruda. Joe
laid out his whole plan and I liked it."
I asked: there is no money, is anyone being paid? "No. That's
one of the amazing things."
He is keen on the Veneto acquisition because with his Medco
experience he is comfortable in the pharmacy area. Veneto is
focused on alternative pre- and post-operative pain management in
the face of an opioid epidemic. It is a timely business to be in.
Inventory is not large, "only 30 to 40 SKUs, and there will be many
turns." Meaning: the inventory will turnover rapidly. "Physicians
and patients are operating with a lower risk of addiction by
limiting opioid days' supply and providing alternative therapies to
relieve pain. The model is compliant with all laws and regulatory
issues, and I had not seen a private company to this date that had
spent so much on its legal opinions and compliance programs." They
are only operating in five states now, but he is gearing up for a
national footprint with internal growth, acquisitions and sales
initiatives all in the mix.
Jason Terrell, M.D. University of Texas at Austin, resides in
Austin, TX; Chief Science Officer, Chief Medical Officer
Jason Terrell met Joe Moscato on a road show for a small biotech
start up over six years ago. "Joe was the only one I spoke with who
told me the truth - that we would never get funding the way we were
going about it. I greatly appreciated his honest, direct approach
and we got along really well. So, I kept in touch with Joe, and he
would run this idea by me and that idea." Moscato needed a medical
sciences expert to bounce off of when he came across new
technologies.
Jason is an unabashed supporter of the company's technologies.
He thinks James Anderson, who heads the buccal delivery for
insulin, is one of the "world leaders" in diabetes and insulin.
When I said, "It's a miracle to have such a team." Jason came back,
"Miracle. I think that's a good word."
Jason and Joe have looked over 200 different companies (or
technologies). And they have some in mind to pursue, if
circumstances permit. So, if acquisitions happen, and note recent
acquisitions at the end of this piece, it should be no surprise to
anyone.
I expressed my dismay that such a promising technology as the
Ii-key peptide cancer vaccine adds no value whatsoever to the stock
market valuation. He was similarly dismayed, but he said, "The
trial with Merck will produce the first new data in years and I
expect this will reinvigorate interest." When I told him I was
having difficulty assigning a value to a research business like
this without revenue, he brought up Juno Therapeutics bought by
Celgene (CELG) for about $9 billion and
Kite. "Last year Kite Pharma was acquired by Gilead (GILD) for around $12 billion.
But it reached a value of a billion dollars years before their
first drug received FDA approval. So, in the immuno-oncology space
of research and development, a lack of revenue isn't the hindrance
one would expect."
Jason explained that Juno and Kite are CAR T technologies, where
they take an individual's cancer cells, do some work in the lab and
then put them back in the patient's body. He said it was a very
expensive process. So, naturally, I asked if Generex's Ii-key
peptide technology would be less expensive than those, and he said
that the Ii-key technology was different, but in many ways should
be much more specific and more powerful. And yes, he felt it would
be far less expensive. "Could it supersede the CAR T technologies?"
Jason responded, "Ii-key is a beautiful technology, and with that I
think anything is possible."
But like all scientists, he will not say anything absolutely,
positively definitive; he's waiting for the data like everyone
else. Though he does feel the team at Generex is 100% world
class.
"On the medical side with me, we have people like Craig Eagle.
James Anderson. Gary Lyman. For example, Dr. Lyman, currently at
Fred Hutchinson [Cancer Research Center], is one of the most
respected and accomplished oncologists in the country. He helped
steer the field as a leader on the board of directors for
ASCO."
What's that? "American Society of Clinical Oncology." When I
looked Lyman up, I found that he had chaired the Guideline
Methodology Committee, which harkens back to point #2 of Joe
Moscato's 1-2-3. So, if a top notch team counts for anything,
Generex has it.
James Anderson, M.D., Louisiana State University School of
Medicine, resides near Indianapolis; Director
I had already spoken with James Anderson in 2014 during the
Ebola scare. I knew that Generex's peptide vaccine technology could
be ramped up relatively quickly, and that was what he confirmed. He
is in a singular position to know such diseases. Anderson was one
of the last of the drafted doctors in 1971. Ultimately, he became
Chief of Medicine at the U.S. Army's biological defense facility at
Ft. Detrick (think The Hot Zone). I asked him about
that book, and he felt it was pretty accurate, the
movie Outbreak he felt was less so. He has been
involved in designing the equipment to contain these virulent
diseases. Addressing the issue of the length of time needed to make
vaccines, he informed me that the flu vaccine takes about a year
and a heck of a lot of eggs. He disabused me of the fear of
vaccines. But what of the mercury used in vaccines, I asked? "I
don't like the term fake news. But that is fake news. They haven't
used mercury since the 1950s."
But it turns out his training was in endocrinology, and
principal among endocrine diseases is diabetes. He is at the top of
his field. He was one of the first to use DNA to make insulin. I
asked him how that was accomplished, and he said, "The DNA is
implanted in E. coli, and the E. coli bacteria produce the insulin.
The E. coli is then killed and the insulin harvested."
It is hardly surprising that he was a founding editorial board
member of two journals for diabetes and serves as a reviewer for 5
other diabetes or endocrine journals.
He said his first exposure to Generex was in 1984. "I went to
New Haven with Bruce Frank, who was the best insulin chemist in the
world, also at Lilly. We were intrigued by buccal delivery. At that
time, only a little insulin got into the blood stream, but it was
something." So he kept tabs on the technology and Generex. Later,
he set Generex up with Eli Lilly (NYSE:LLY)
in the early 2000s for a study, but then left for Japan for three
years to complete work on human analogs of insulin. When he came
back from Japan, the deal with Lilly had been terminated. He was
drawn back to Generex years later by Joe Moscato who met him at an
ADA Conference. Afterwards, he had called Anderson to see if he
would be interested in working with Generex, he passed the lead
over to Mark Fletcher, who had taken the reins from the founders,
and Anderson came back to work on buccal delivery.
Anderson said one of the concerns with Pfizer's (NYSE:PFE)
Exubera was that it went into the lung where it was difficult to
see if tumors were developing. Although there have been no tumors
with Ora-lyn, an advantage of Generex's Ora-lyn is that it is easy
to inspect the inside of the mouth for problems, and it can't go in
the lungs.
We discussed the problems of the trials of Ora-lyn that led the
FDA to require more testing. He reported that testing was done in
the European Union, and there were language problems, incomplete
surveys by patients, a weak drug that still required as many as 20
puffs, and it was not a well-designed study, which failed to meet
statistical requirements. And that was what led to the FDA
requiring more testing.
Since that FDA review, studies in Toronto had shown that the
number of puffs can be reduced to 2 to 5, and that in dog studies,
it appeared that the insulin was getting into the blood stream.
When I said I suspected that fear of needles resulted in poor
compliance by patients, he said, "Fear of needles results in the
delayed start of insulin, and the 1/7th of all medical costs in the
past year are due to complications of diabetes."
He reminded me that the main product was not insulin, but a
delivery system that could be used with other drugs. A timely
possibility he noted would be the use of the delivery system with
cannabis oils, which may have neurological possibilities - pain
reduction.
I said that I felt that Big Pharma was missing an opportunity by
not funding studies, he agreed, and suggested it might take another
safety study with the new concentrated formula to entice them.
Anderson brightened up at the thought that the current leadership
under Joe Moscato might be able to find the money to do that
study.
Eric von Hofe, Ph.D. University of South California, post-doc
University Hospital of Zurich, Switzerland, post-doc Harvard;
President, Antigen Express
Eric von Hofe has spent his whole career in cancer oncology,
potentially a depressing area to be in. While he was an Assistant
Professor at the University of Massachusetts Medical School, "I
became acquainted with Bob Humphreys, who also did a post-doc at
Harvard, who was making key discoveries." Eric then moved on to
biotech, first at an antisense company in Worcester and then moving
on to Millennium Pharmaceuticals. Meanwhile, Humphreys was
continuing to get by with SBIR (Small Business Innovation Research)
grants, and making progress. Eric consulted with him for a while in
1999, and kept in email contact. Eventually, Humphreys went on to
start Antigen Express, and Eric joined Humphreys. In 2003, Antigen
Express was bought out by Generex.
Eric informed me that Merck with its Keytruda drug had made
progress with checkpoint inhibitors, which stop the immune system
from going after the body's own cells. If you can get past the
checkpoints, then you can kill the cancer cells, which employ a
human disguise. He said, "It's a blunt tool. Merck noticed that it
only worked in about 25% or 30% of patients, so they started
looking around for other drugs that could work with it." Merck
culled the studies, and found that Antigen Express had had some
luck in triple negative breast cancer. AE37 vaccine stimulates the
immune system to recognize a protein expressed on cancer cells
(HER2); more importantly, the immune system can recognize this
protein at lower levels than even the very successful drug
Herceptin, which targets the same protein. And a key advantage of
AE37 is that it is practically non-toxic. And this was the basis
for the partnership with Merck. It was
similar story with Shenzhen and the trial for
prostate cancer. Antigen Express had done a Phase I study on
prostate cancer that looked promising.
As Jason Terrell explained above, a lot of the work in the immune
space has been in the CAR T cell area, where T cells are taken from
the patient's body and tinkered with. Then they grow a lot of them,
and then re-introduce them into the body. Von Hofe explained to me
that this was a very expensive procedure that can cost as much as
half a million per patient per year.
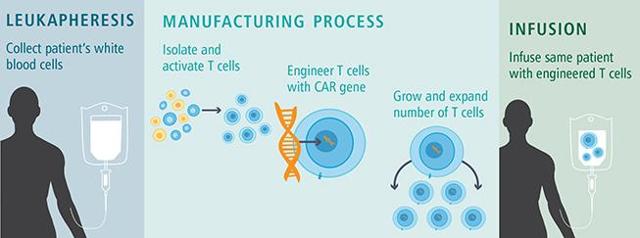
Source: Dana-Farber Cancer Institute
I asked him if the Ii-key would be any less expensive, and this
is what he told me: "Yes, the fragment of the protein or pathogen
only needs to be around 15 amino acids long, to which the Ii-Key (4
amino acids) are added. This peptide can be produced synthetically
by a machine that is fed the appropriate amino acids; it simply
adds one amino acid after the other to produce the peptide. When
produced in bulk it's very cost effective. Anything that is not an
off-the-shelf product and needs to be biologically produced, like
CAR T cells, are much more labor intensive and costly."
I asked him if it would be more effective, and he replied that
it would probably work better on solid tumors than for leukemias
and lymphomas, where CAR T has been "amazingly" successful.
For in-depth investors, I recommend that they look for the
October 2011 issue of Scientific America and
Eric von Hofe's article there entitled "A New Ally Against Cancer."
I would create a link, but Scientific America insists, perhaps
rightfully so, that you must have a subscription. In the article,
von Hofe gives a history of cancer and immune response, and brings
it from the 19th century all the way up to the date of the
article.
I had spoken with James Anderson about how AE37 vaccine
technology (Ii-key) might work against Ebola and other potentially
life threatening viruses like a pandemic flu strain, and I got
clarification from von Hofe. He said the company could make a
vaccine, and relatively quickly. The main difference from a
traditional viral vaccine is that the Antigen Express technology is
more of a cellular immune response than an antibody response. So,
while one may not get total protective immunity, someone who
contracted a potentially lethal virus like a pandemic influenza or
some other disease might still get sick even with the vaccine, "but
they probably won't die." I suggested that this might allow the
body time to produce a proper anti-body that would bring the
patient to health. He said, "Yes, that's it."
Eric is not put off by the disappointing response of investors
to the announcement of two Phase II studies, he said,
"it was clear to investors that it would take more than these
deals to resurrect Generex and Antigen Express after their period
of extended dormancy and low valuation. In the meantime, Generex
has entirely reorganized its corporate structure, business model
and demonstrated that it has an exciting future ahead; to which the
markets have reacted enthusiastically."
Harold Haines, Ph.D. Tulane University, resides in the Miami
area; President, NuGenerex Diagnostics, Inc.
Hal is a long time Floridian with the bruises to go with it. He
lost a house to Hurricane Andrew in 1992 and lost part of house and
a roof to Katrina before she made her way over to New Orleans. Hal
came with Hema Diagnostics, which is now re-named NuGenerex
Diagnostics (NDG). He was consulting for it before taking over its
management.
The idea was to make tests that could yield results while the
patient is still in the office, or while the nurse is in the room
with the patient, and thus initiate or modulate treatment in a more
timely way. Tests have already been developed for: HIV,
Hepatitis-B, Hepatitis-C, and Malaria.
A test for syphilis is before the European Union awaiting the CE
mark of authorization. This could be a real seller because syphilis
is staging a comeback. Interestingly, it has become prevalent in
the retirement homes. This test could be the first real money-maker
for NGD.
Hal is particularly keen on a test for sepsis. Sepsis is
particularly hard to diagnose. In sepsis there is a trigger,
infection or injury, and that generates an immune response, and
that's the problem. He explained to me, "In influenza, and in other
diseases, you die from the immune response, not from the
virus."
"Most of our tests are qualitative. They tell you if someone has
a disease or not, but in sepsis we will be able to tell whether the
condition is increasing or decreasing." He explained there is a
real need for such a test because it is hard to diagnose, and a
patient can die in 48 to 72 hours.
He thinks that after they get going in Europe, even more sales may
be generated in China. "In Europe and the U.S., you can only
administer these tests in a doctor setting, but in China tests can
be conducted in the home."

What You Can Expect
You can expect dilution. There will be option grants to
employees for services that have been rendered over the years, and
option grants to incentivize new employees. There are and will be
warrants and options in the deal-making, perhaps with limited use
for now by requirements to maintain the net operating loss. And
although some of the share stuff in the 10K has been explained to
me, it is still has not come into bold relief, and it remains to be
seen how all of that will play out, so I am confining myself to
market cap as opposed to share price. Here's what I can tell you.
In the December 2018 conference call, Joseph Moscato
said there are 28 million shares outstanding now, and that would
likely max out at "60 to 75 million shares."
You can expect deal-making (witness the deals of the last few
days at the end of this piece). You can expect Joe to pick up some
new technologies, and to expand the pharmacy-medical services
business. You can expect that some of these technologies to be
great, but others to be a drag on earnings. Expect ups and downs. I
feel Generex might be like Cisco, which grew from one acquisition
to the next.
The Motivations of the Employees
It is not just the kids who want to improve our lives. James
Anderson talks about 1/7th of all disease coming from diabetes.
Eric von Hofe was motivated to improve cancer outcomes even when
Generex went into suspended animation. Terry Thompson left Medco
when he couldn't get the lowest price for his customers and is
motivated to improve opioid addiction outcomes, and to avoid waste
and corruption. Hal Haines wants to detect disease as early as
possible for as little as possible. And Joe Moscato has wanted to
see patients benefit from all of these improvements since he first
began working with the firm. And not to forget, it's all an
all-volunteer army at this point. There is no money for salaries.
Maybe they will get paid something eventually. It's not guaranteed.
But working to solve big problems motivates people.
The Problem Of Valuing A New
Biotechnology
Cancer vaccines are a hot area. The question is: what is a
cancer vaccine company worth before the Phase III trial and FDA
approval? It apparently depends on market sentiment. When the
market is really hot as was the case with Juno, an untested company
could be worth as much as a billion. Other times, the market can be
more skeptical. Then, suddenly, a company can open its pocket book,
witness the move by Bristol-Myers
Squibb (BMY) in February 2018, which
paid $1 billion cash and made an $850 million stock purchase from
Nektar (NKTR).
The Ii-key peptide technology has been tested, and it has shown
good safety and efficacy. And if Generex stock were not in the
Mariana Trench, the announced trials with Merck and Shenzhen might
have added millions to the stock market cap, but as it is, it added
nothing at all. As Jason Terrell has suggested above, new data
might do the trick. But ultimately, high values are possible as the
sales of Juno Therapeutics to Celgene for $9 billion and Kite
Pharma to Gilead for nearly $12 billion shows.
We'll know when the market approves because the stock valuation
will expand suddenly by hundreds of millions, or a billion. In the
meantime, this makes Joseph Moscato's plan of diversification into
medical services - pharmacy and opioid management - looks both
timely and very sensible indeed. The feelings of the leader of that
initiative, Terry Thompson, will not be hurt if things suddenly
improve for the cancer vaccine end of the business, as per his
personal experience cited above. He will rejoice. No matter what,
he will see his end of the business through to growth and
profit.
Let's discuss the case of insulin. The formulation has always
worked to some extent, but initially the formulations were weak,
sometimes requiring as many as 20 puffs. In 2015 research had improved the
formulation to the point where only 2 to 5 puffs were required, and
research in dogs had established its efficacy, and again its
safety. It's my take that this good news in the improvement in
potency is not built into the stock. Generex needs, of course, to
test this in humans, but that requires money, and that is the
current stumbling block. James Anderson thinks maybe a new study
will stir up interest.
What I don't understand is that one of the big insulin players -
Eli Lilly, Novo Nordisk (NVO), or Sanofi (SNY) have not ponied up to get
that research completed. Do they really think needles or pens
(again, a needle) are really satisfactory?
Unfortunately, you cannot necessarily count on the drug
companies to act in the interest of their patients. In 2017 a suit
was brought by a group of diabetes patients against the big three
insulin makers for collusion.
If 60 Minutes were to run a story about how
diabetic patients could be saved a lot of pain, and then interview
doctors on how patients would be much more compliant with doctor's
recommendations, and how their outlooks would be so much better,
I'm not sure even that would move the insulin makers. They would
say to interviewers that the technology is not ready yet. Then I
suppose the interviewers would have to be clever enough to ask a
second question, why aren't you funding such a study? And a third
question: Don't you care about the pain needles cause patients?
Well, my job is not to foment a revolution; my job is to shine a
light on a promising technology. But if patients can sue over
collusion, they might also use their weight to let their fellow
users know that the drug companies are slugs again. There is
something around that could save them a lot of pain. All I know is
that my diabetic neighbor downstairs has injected himself for
decades, but my take is that he would change in a day if he could
just puff a little insulin inside his cheek.
Let's take another tack. You're in the insulin business. Ask
yourself: what would be the best way to sell more insulin? Answer:
make it painless. James Anderson says patients avoid starting
insulin when they need to, sometimes by years, because of the
needle. And doctors fear their patients don't take insulin often
enough, also because of the pain of the needle. Getting these
patients to take insulin when they need to will reduce the medical
bill for the country, but it would also sell a heck of a lot more
insulin.
Do good - and hopefully get something out of it. That seems to
be a theme I heard throughout top management.
Investors must be patient. It seems to me Generex buccal
delivery will own the insulin market. I am personally satisfied
that it will work out at the 90% confidence level. Why? It has been
tested for years and years and has been found to be safe. Insulin
is something the body produces on its own. I don't expect a lot to
go wrong. The fact that the company has had problems with a poorly
designed trial (as per James Anderson above) doesn't mean it won't
work. It means the study was poorly designed (see point #2 under
Joe Moscato's 1-2-3 for Investing in Biotech above). All of that
came before Messrs. Moscato and Anderson. Gerald Bernstein M.D., a
former VP of Generex, and a former President of the American
Diabetic Association, told me in a telephone interview that he had
personally used the product for a long time. And the FDA in 2009
had given approval under its IND program for patients "with serious
or life-threatening Type 1 or Type 2 diabetes who have no
satisfactory alternative treatments."
Pfizer had an idea about the delivery of insulin, and that was
through the lungs. The product they developed was called Exubera.
It turned out patients and doctors were not comfortable with the
continual ingestion of insulin into the lungs. Pfizer decided to
discontinue the product, and wrote off $2.8 billion. But Pfizer
wasn't in it for the write-off; they were in it for a successful
product. So I am valuing Ora-lyn, Generex's product, at twice the
write-off. I think they had the right idea, but the lung was the
wrong target for the drug. It seems they gave up on alternative
means of ingestion, even though they were on the right track.
Mannkind (MNKD) had the same idea. Insulin
which comes in through the lung with a product called Afrezza.
There is no way to describe them (after Pfizer walked off the
field) but as madly persistent. They did develop a product and got
it through the FDA. They made a deal with insulin maker Sanofi for
$150 million, but ultimately, they discovered what Pfizer had
already found out. Patients don't want to put stuff in their lungs,
and the agreement with Sanofi was terminated on January 5,
2016.
As far as I can tell, this technology should be worth hundreds
of millions even as things currently stand. The idea that all
information is built into the stock price is hogwash. But that can
be an advantage for astute investors.
Zample: those of us who invested in DSL (digital subscriber
lines) technology because it would provide the bandwidth necessary
for streaming video through telephone lines - we had to wait a long
time. Meanwhile, we were out there chatting on the bulletin boards:
What's taking the Baby Bells so long? Then sentiment changed, the
technology was adopted, and my DSL stocks started to climb. I
remember one of my winners, Aware, made a ten times move. Given the
architecture of the telephone business, DSL was inevitable.
The inside of the cheek is rich in blood vessels. For a means of
getting insulin inside the body without the pain of a needle, it's
a natural. Investors need to stay the course.
The Ongoing IPO
Veneto was privately owned, so the acquisition is, in effect, a
sort of IPO, especially given Generex's low market cap. An IPO
would produce an instant value for Veneto that might reasonably put
the stock at 20 times adjusted EBITDA of $7.6 million, or $152
million. And the stock did rise very quickly in near IPO style to
the $25 million level, but it still feels light, so I'm not
convinced that the IPO of Veneto is over. In addition, Joseph
Moscato told listeners to the conference call in December
that another privately-owned pharmacy network would be added to
NuGenerex Distribution Solutions (NDS), to which Veneto was the
base acquisition, so the ongoing IPO of these previously untraded
companies could continue for a while. And if other like businesses
become available for sale at an attractive price, yet more private
value might be added to this public company.
Additionally, private value becomes public value for the recent
acquisitions, Regentys and Olaregen. So, there is and will
be a continuing on-going IPO at Generex of at least these four
companies (Veneto, As-Yet-Unnamed Pharmacy, Regentys, and
Olaregen). Generex is paying $35 million for Veneto, $15 million for 51% of
Regentys, $12 million for 51% of
Olaregen.
Regentys acquired a patent on Friday
(12/14/2018), and the stock jumped 24% percent. "Regentys™
Extracellular Matrix Hydrogel ("RECMH") is a proprietary, patented
UC treatment that protects damaged tissue from waste flow and
promotes tissue regeneration and healing rather than suppressing
the immune system as other treatments currently do." I was
intrigued by their description of the product: "The product has
been developed with polymeric characteristics that transform ECMH
from a liquid to gel form upon adherence to the mucosal lining of
the diseased colon, providing a "Bio-Scaffold Bandage" to the
affected area." Then there were the numbers. Two million sufferers
of Crohn's Disease and other inflammatory bowel diseases, and $6
billion global market for these kinds of diseases. The company
thinks it might capture a billion dollars of that market.
Also purchased was Olaregen Therapeutix, which has
a patented collagen-based dermal matrix which can be used in a
variety of different wounds - surgical wounds, trauma wounds,
diabetic ulcers, look to the link at the beginning of the paragraph
for a complete description of all the uses of its product,
Excellagen. The list is extensive. Generex believes this product
together with its product in development, Excellasome, for the
regeneration bones, joints and cartilage, could also reach a
billion dollars.
Circling back to Veneto, Generex is developing an MSO (medical
services organization) network of doctors, offices, pharmacies,
patients. And this network will give them faster than normal access
to patients who can use - not just opioids, and other drug
products, but CBD products (partnership with Puration), their new
products - Excellagen for wounds, and their "scaffold bandage" for
use in the intestinal tract, but also for all manner of other
services mentioned above. So, the network will pay dividends as it
can refer patients of MSO to its other products and services.
So, there is an urgency to get the MSO network setup. Terry
Thompson is the uniquely qualified to pull off this sweep into new
territory with his experience at Medco. Generex thinks that it will
add $5 million of revenue for each state it adds to its network,
and it is looking to go into an additional 22 states. No timetable
has been given for when these states will be added, but I think it
is reasonable to assume that all states will be reached within
three years' time.
A Trip to the Dark Side
Looking at the pitfalls, in the case of the vaccines, I read 25
years ago about a trial where scientists were able to resolve brain
cancer in mice 21 out of 21 times. I thought: this is not chance. I
think this must have been before the internet, and I cannot find
this article anymore. But as I recall, my research led me to Immune
Response Corporation, a company founded or co-founded by Jonas
Salk. A lot of Ph.D.'s on staff. Salk had developed a vaccine for
HIV called Remune. Eventually, funds ran out. They, too, were on a
mission. They had a peptide vaccine for rheumatoid arthritis,
psoriasis, and multiple sclerosis. They had gene therapy for
lowering cholesterol, hemophilia, hepatitis C. They, too, wanted to
change the world. In trying to find out what became of them for
this article, I looked up their website, I got the message that the
domain may be for sale. I had sold my shares for peanuts years
ago.
And the Bristol-Myers Squibb collaboration with Nektar mentioned
above is looking a little iffy after new data came
in, though more data is needed to clarify effectiveness.
Buccal delivery. The non-inferiority tests required by the FDA
can be devastating. I owned shares in a blood substitute maker
Northfield Laboratories. It had to show that its product performed
no less well than blood or blood plasma. It performed pretty well,
but not quite as well as blood or blood plasma. The company's
shares fell precipitously, and in 2009 it was liquidated under
Chapter 11. Generex is under a similar non-inferiority burden,
though it is not a test of insulin. Insulin is insulin. The body
makes it. No, it was a test of how well the insulin gets into the
blood stream as compared with a needle. And the formula has been
improved since its Phase III of 2011.
As regards the Veneto acquisition, the owners accepted a
promissory note from Generex. In January, that has to be replaced
by cash (or perhaps another note). I put it to Moscato, and he did
not think it was an insurmountable problem, and he has raised
millions for Generex research.
And More Darkness
But the financials are not a pretty sight. There's nothing
there. A paltry $700,000 payment by Shenzhen Bioscien is all the
income the 10K shows. "Stockholder's Deficiency" sheds light on the
losses that have plagued the company. There is, of course, a "Going
Concern" note. This is not a company for the faint of heart.
Next year, assuming Generex makes it past all of its payment
schedule hurdles, (a sampling from the 8-K's are Regentys and Olaregen), which is not a given
considering what's in the kitty, the 10K will look very
different with the recent purchase of Veneto, and these other
acquisitions.
But if I haven't given you pause, I have not succeeded. I should
have given you pause.
Let's just get it out in the open. This is not Merck or Pfizer.
If you want Merck or Pfizer safety buy Merck or Pfizer. This is
riskier.
On the other hand, there is also a downside to Merck and Pfizer.
I have in mind a longish piece about the granddaddy's, the huge
companies. It will begin with a discussion of U.S. Steel. It was,
in effect, the steel trust. In 1901 J.P. Morgan had bought Andrew
Carnegie's steel company for $480 million, plus ten other steel
companies. It had capital of $1.4 billion, the largest company in
the world with 67% percent of the domestic steel market. But get
this: ten years later, they were down to 50%, and a hundred years
later, they have around 10% of the market. It's the safety of slow
death.
Speaking of huge, it turns out that Big Pharma is only so-so at
research by its own scientists. Per the Fortune article, "Inside Pfizer's
Palace Coup," Pfizer spent $60 billion on research 2000-2008, and
that only resulted in nine drugs. It seems to me, the big job of
Big Pharma is to recognize where the good work is being done by the
research start-ups and buy those companies or partner up and fund
their research. It may very well determine the success of those
granddaddies.
To summon up Bob Dylan, if you aren't busy being born, you are
busy dying. So, you may wish to have a few fresh faces in the mix.
But buy no more than you can afford to lose, this is high risk. A
sliver. As my father would say, ten cents worth.
Buy a little now, and watch the announcements, if you see
something really significant, like a partner for the buccal
technology, or positive Phase II results for the cancer vaccines,
or other new partners for the cancer vaccines, increase your
stake.
The Summing Up
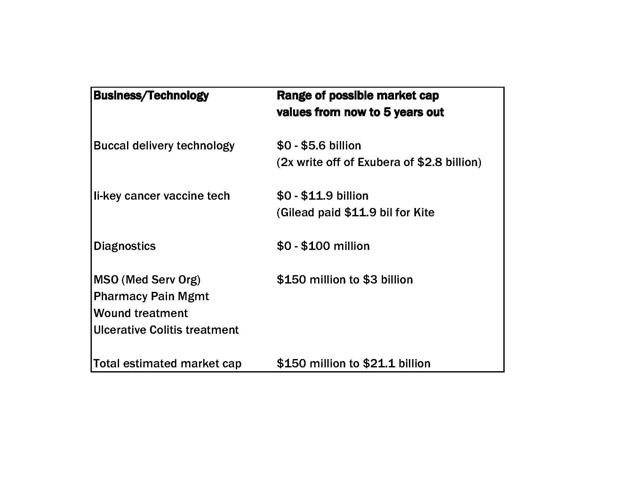
I figure Generex has three (3) chances to achieve a market cap
of $1 billion in five years. One - is the buccal delivery business,
of which insulin is the test case. It would be worth at least a
billion. Two - is the Ii-key peptide vaccines, which are being
tested on breast cancer and prostate cancer, and where Jason showed
me that Juno and Kite achieved billion dollar valuations even
before their cancer vaccines proved out. (Think of the stock market
reaction to a successful Phase II now that Generex is awake). And
three, the MSO/pharmacy business (plus newly purchased treatments)
is being programmed for rapid growth of a kind that may well result
in a billion dollar sales in five years all by itself. I only need
one of these three to succeed to hit my target.
For now, I am assigning a market cap of $150 million to
NuGenerex Distribution Solutions (NDS), which holds Veneto. We were
told in the conference call that Veneto will likely generate
millions of EBITDA, and they paid a little over four times adjusted
EBITDA, or $35 million. If you assign a multiplier of 20, you get
$152 million. But the new entity NDS also includes the new
acquisitions, Regentys and Olaregen, the Puration (OTCPK:PURA) deal, and an as yet unnamed pharmacy which
has been conducting audits prior to inclusion. I have no way to
assign numbers to these things. $150 million is a minimum figure.
Another way to look at it is this: one to two times sales for the
MSO/pharmacy unit (Veneto and including the unnamed pharmacy
conducting audits) plus contributions from the Hema diagnostic
products (if any), the new Regentys ulcerative colitis treatment
(if any) and Olaregen wound treatment (if any), and that still does
not assign any value to buccal and cancer vaccines. The cancer
vaccines look promising, but I cannot put anything into the
equation for that because the market is resistant. We have to wait
for more data. But if insulin users are to avoid the pain of a
needle, then the buccal treatment (since the lung method has been
rejected by consumers twice) will likely become the standard
delivery method for insulin. Given the hundreds of millions that
have been spent on testing for that (even if badly invested in a
poorly designed trial), no one was harmed, and now the formula has
been amped up, if the market changed its viewpoint about that
technology and unmet need, I'm okay with multiple times the $150
million.
Alpha to Spare
All said and done, I don't believe it will ever be possible for
me to deliver more alpha to investors than I will with this piece.
(To refresh - the excess return of an investment relative to
the return of a benchmark index
is an investment's alpha.) In part, this is because the value of
Generex's technologies drifted to ridiculously low values, and even
after the recent run-up, still remains remarkably low. And in part
it is because prospects of these businesses, the two they have had
for years, plus the new businesses, appears to be remarkably good.
As I have stated above, I'm setting a five year market cap target
of $1 billion.
= = = = = =
Breaking News
Serious investors should read the conference call transcript.
Press releases and other pertinent links and information can be
found at Generex's investor site. Also, Generex had
only a little over 1 million shares outstanding after the
1-for-1,000 reverse split. The company decided that was too few
shares, so to increase the number of shares outstanding and they
paid a stock dividend to shareholders in November of twenty (20)
shares for every one (1) share owned.
Disclosure: I am/we are long GNBT. I
wrote this article myself, and it expresses my own opinions. I am
not receiving compensation for it. I have no business relationship
with any company whose stock is mentioned in this article.
Additional disclosure: I have owned shares
of Generex continuously since 2008. I first spoke with CEO Joseph
Moscato in 2010 at which time he was not an employee or board
member of the company, but contracted as a consultant to help raise
funds and seek out opportunities for them. I again contacted him
after he became the CEO in 2017, and met with him in April and
November of 2018. This article results from those meetings and from
telephone interviews with the key officers – James Anderson, Eric
von Hofe, Jason Terrell, Terry Thompson, and Harold Haines.
Orgianl source - https://seekingalpha.com/article/4228973-new-generex-unlocking-value?page=1