ALX Oncology Gets FDA Orphan Drug Designation for Evorpacept
January 27 2022 - 6:43PM
Dow Jones News
By Stephen Nakrosis
Clinical-stage immuno-oncology company ALX Oncology Holdings
Inc. on Thursday said it received an orphan drug designation for
evorpacept from the U.S. Food and Drug Administration.
ALX Oncology said it received the designation for evorpacept to
treat patients with gastric cancer and gastroesophageal junction
cancer.
The FDA grants orphan drug status to treatments for rare
diseases and the designation qualifies the sponsor of the drug for
various development incentives, including tax credits for qualified
clinical testing.
Sophia Randolph, the company's chief medical officer, said,
"Receiving orphan drug designation from the FDA is an important
regulatory milestone for ALX Oncology and reinforces the FDA's
recognition of evorpacept's potential to improve clinical outcomes
in patients with GC."
Write to Stephen Nakrosis at stephen.nakrosis@wsj.com
(END) Dow Jones Newswires
January 27, 2022 18:28 ET (23:28 GMT)
Copyright (c) 2022 Dow Jones & Company, Inc.
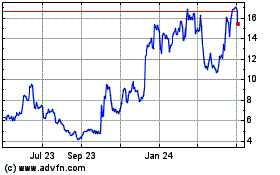
ALX Oncology (NASDAQ:ALXO)
Historical Stock Chart
From Aug 2024 to Sep 2024
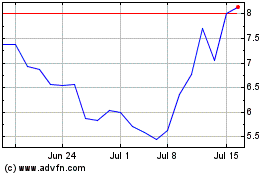
ALX Oncology (NASDAQ:ALXO)
Historical Stock Chart
From Sep 2023 to Sep 2024