(Address, including zip code, and telephone number,
including area code, of registrant’s principal executive office)
(Name, address, including zip code, and telephone
number, including area code, of agent for service)
If the only securities being registered on this
Form are being offered pursuant to dividend or interest reinvestment plans, please check the following box. ☐
If any of the securities being registered on this
Form are to be offered on a delayed or continuous basis pursuant to Rule 415 under the Securities Act of 1933, other than securities offered
only in connection with dividend or interest reinvestment plans, check the following box. ☒
If this Form is filed to register additional securities
for an offering pursuant to Rule 462(b) under the Securities Act, please check the following box and list the Securities Act registration
statement number of the earlier effective registration statement for the same offering. ☐
If this Form is a post-effective amendment filed
pursuant to Rule 462(c) under the Securities Act, check the following box and list the Securities Act registration statement number of
the earlier effective registration statement for the same offering. ☐
If this Form is a registration statement pursuant
to General Instruction I.D. or a post-effective amendment thereto that shall become effective upon filing with the Commission pursuant
to Rule 462(e) under the Securities Act, check the following box. ☐
If this Form is a post-effective amendment to
a registration statement filed pursuant to General Instruction I.D. filed to register additional securities or additional classes of securities
pursuant to Rule 413(b) under the Securities Act, check the following box. ☐
Indicate by check mark whether the registrant
is a large accelerated filer, an accelerated filer, a non-accelerated filer, a smaller reporting company or an emerging growth company.
See the definitions of “large accelerated filer,” “accelerated filer,” “smaller reporting company”
and “emerging growth company” in Rule 12b-2 of the Exchange Act.
If an emerging growth company, indicate by check
mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting
standards provided to Section 7(a)(2)(B) of the Securities Act. ☐
Subject to Completion,
dated July 20, 2021
PROSPECTUS

ADIAL PHARMACEUTICALS, INC.
1,666,668 Shares of Common Stock
This prospectus relates to the resale from time
to time of up to 1,666,668 shares of Adial Pharmaceuticals, Inc. common stock, par value $0.001 per share, by the selling stockholders
identified in this prospectus or in supplements to this prospectus or its transferees. The shares offered for resale consist of an
aggregate of 166,668 shares of common stock that were purchased by the selling stockholders on July 6, 2021 in a private placement transaction
pursuant to the terms of securities purchase agreements, dated July 6, 2021, we entered into with each of the selling stockholders, as
investors in such private placement transaction (collectively, the “Securities Purchase Agreements”), and an aggregate of
1,500,000 shares that the selling stockholders have committed to purchase and that will be issued and sold to them in a second tranche
of purchases under the Securities Purchase Agreements, which second tranche purchase will occur on the second business day following the
effective date of the registration statement of which this prospectus forms a part. We are filing the registration statement on Form S-3,
of which this prospectus forms a part, to fulfill our contractual obligations with the selling stockholders to provide for the resale
by the selling stockholders of all of the shares of common stock that have been and will be purchased by them in both tranches of purchases
in the private placement transaction pursuant to the Securities Purchase Agreements. See “Selling Stockholders” beginning
on page 12 of this prospectus for more information about the selling stockholders. The registration of the shares of common stock to
which this prospectus relates does not require the selling stockholders to sell any of its shares of our common stock.
We are not offering any shares of common stock
under this prospectus and will not receive any proceeds from the sale or other disposition of the shares covered hereby; however, we have
received an aggregate of $500,002 from the sale of an aggregate of 166,668 shares that the selling stockholders have purchased in the
first tranche purchase under the Securities Purchase Agreements, and we expect to receive an additional aggregate amount of $4,500,000
from the sale of an aggregate of 1,500,000 additional shares that the selling stockholders are committed to purchase in the second tranche
purchase under the Securities Purchase Agreements on the second business day following the effective date of the registration statement
of which this prospectus forms a part.
The selling stockholders may from time to time
sell, transfer or otherwise dispose of any or all of their shares of common stock in a number of different ways at fixed prices, at prevailing
market prices at the time of the sale, at varying prices determined at the time of sale or at negotiated prices, as described in more
detail in this prospectus. We will bear all costs, expenses and fees in connection with the registration of the shares of common stock
included in this prospectus. The selling stockholders will pay or assume discounts, commissions, fees of underwriters, selling brokers
or dealer managers and similar expenses, if any, incurred for the sale of shares of our common stock included in this prospectus. For
more information, see “Plan of Distribution” beginning on page 21 of this prospectus for more information about how the selling
stockholders may sell their shares of common stock.
No underwriter or other person has been engaged
to facilitate the sale of the shares of common stock in this offering. The selling stockholders, individually but not severally, and any
broker-dealers or agents that are involved in selling the securities may be deemed to be “underwriters” within the meaning
of the Securities Act of 1933, as amended (the “Securities Act”), and any commission paid, or any discounts or concessions
allowed to, any such broker-dealer may be deemed to be underwriting commissions or discounts under the Securities Act.
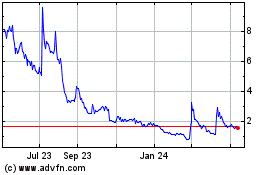
Our common stock is listed on the Nasdaq Capital
Market under the symbol “ADIL.” Our warrants that we issued in our initial public offering are listed on the Nasdaq Capital
Market under the symbol “ADILW.” On July 15, 2021, the last reported sale price of our common stock and our warrants on the
Nasdaq Capital Market was $2.49 per share and $0.77 per warrant, respectively.
We are an “emerging growth company”
as that term is used in the Jumpstart Our Business Startups Act of 2012 (the “JOBS Act”), and, as such, elect to comply with
certain reduced public company reporting requirements for future filings.
An investment in our shares of common stock
involves a high degree of risk. Before deciding whether to invest in our common stock, you should consider carefully the risks and uncertainties
described in the section captioned “Risk Factors” contained in our Annual Report on Form 10-K for the fiscal year ended December
31, 2020 filed with the Securities and Exchange Commission on March 22, 2021 and other filings we make with the Securities and Exchange
Commission from time to time, which are incorporated by reference herein in their entirety, together with other information in this prospectus
and the information incorporated by reference herein.
Neither the Securities and Exchange Commission
nor any state securities commission has approved or disapproved of these securities or determined if this prospectus is truthful or complete.
Any representation to the contrary is a criminal offense.
The date of this prospectus is , 2021
PROSPECTUS SUMMARY
This summary highlights some information
about us and selected information contained elsewhere in this prospectus and in the documents we incorporate by reference. This
summary does not contain all of the information you should consider before investing in our common stock. You should read this
entire prospectus and the documents incorporated by reference carefully, especially the risks of investing in our common stock
discussed under and incorporated by reference in “Risk Factors” on page 8 of this prospectus, along with our
consolidated financial statements and notes to those consolidated financial statements and the other information incorporated by
reference in this prospectus, before making an investment decision.
Overview
We are a clinical-stage biopharmaceutical
company focused on the development of therapeutics for the treatment or prevention of addiction and related disorders. Our lead investigational
new drug product, AD04, is being developed as a therapeutic agent for the treatment of alcohol use disorder (“AUD”). In January
2021, we expanded our portfolio in the field of addiction with the acquisition of Purnovate, LLC, and we continue to explore opportunities
to expand our portfolio in the field of addiction and related disorders, both through internal development and through acquisitions. Our
vision is to create the world’s leading addiction focused pharmaceutical company.
AUD is characterized by an urge to consume
alcohol and an inability to control the levels of consumption. We have commenced the landmark ONWARD™ pivotal Phase 3
clinical trial using AD04 for the potential treatment of AUD in subjects with certain target genotypes. As of this filing, all 25 planned
clinical sites were actively enrolling patients, and the ONWARD trial was 90% enrolled. The trial is expected to be completed by the first
quarter of 2022. We believe our approach is unique in that it targets the serotonin system and individualizes the treatment of AUD, through
the use of genetic screening (i.e., a companion diagnostic genetic biomarker). We have created an investigational companion diagnostic
biomarker test for the genetic screening of patients with certain biomarkers that, as reported in the American Journal of Psychiatry
(Johnson, et. al. 2011 & 2013), we believe will benefit from treatment with AD04. Our strategy is to integrate the pre-treatment genetic
screening into AD04’s label to create a patient-specific treatment in one integrated therapeutic offering. AD04, is dependent on
a successful development, approval, and commercialization of a genetic test, which is expected to be classified as a companion diagnostic
and will require clearance or approval of the FDA prior to commercialization. Our goal is to develop a genetically targeted, effective
and safe product candidate to treat AUD by reducing or eliminating the patients’ consumption of alcohol.
We have a worldwide, exclusive license from
the University of Virginia Patent Foundation (d.b.a. the Licensing & Venture Group) (“UVA LVG”), which is the licensing
arm of the University of Virginia, to commercialize our investigational drug candidate, AD04, subject to Food and Drug Administration
(“FDA”) approval of the product, based upon three separate patent application families, with patents issued in over 40 jurisdictions,
including seven issued patents in the U.S. Our investigational agent has been used in several investigator-sponsored trials and we possess
or have rights to use toxicology, pharmacokinetic and other preclinical and clinical data that supports our landmark ONWARD pivotal Phase
3 clinical trial. Our therapeutic agent was the product candidate used in a University of Virginia investigator sponsored Phase 2b clinical
trial of 283 patients. In this Phase 2b clinical trial, ultra-low dose ondansetron, the active pharmaceutical agent in AD04, showed a
statistically significant difference between ondansetron and placebo for both the primary endpoint and secondary endpoint, which were
reduction in severity of drinking measured in drinks per drinking day (1.71 drinks/drinking day; p=0.0042), and reduction in frequency
of drinking measured in days of abstinence/no drinking (11.56%; p=0.0352), respectively. Additionally, and importantly, the Phase 2b results
showed a significant decrease in the percentage of heavy drinking days (11.08%; p=0.0445) with a “heavy drinking day” defined
as a day with four (4) or more alcoholic drinks for women or five (5) or more alcoholic drinks for men consumed in the same day.
The active pharmaceutical agent in AD04,
our lead investigational new drug product, is ondansetron, which is also the active ingredient in Zofran®, which was granted
FDA approval in 1991 for nausea and vomiting post-operatively and after chemotherapy or radiation treatment and is now commercially available
in generic form. In studies of Zofran®, conducted as part of its FDA review process, ondansetron was given acutely at dosages
up to almost 100 times the dosage expected to be formulated in AD04 with the highest doses of Zofran® given intravenously
(“i.v.”), which results in approximately 160% of the exposure level as oral dosing. Even at high doses given i.v. the studies
found that ondansetron is well-tolerated and results in few adverse side effects at the currently marketed doses, which reach more than
80 times the AD04 dose and are given i.v. The formulation dosage of ondansetron used in our drug candidate (and expected to be used by
us in our Phase 3 clinical trials) has the potential advantage that it contains a much lower concentration of ondansetron than the generic
formulation/dosage that has been used in prior clinical trials, is dosed orally, and is available with use of a companion diagnostic genetic
biomarker. Our development plan for AD04 is designed to demonstrate both the efficacy of AD04 in the genetically targeted population and
the safety of ondansetron when administered chronically at the AD04 dosage. However, to the best of our knowledge, no comprehensive clinical
study has been performed to date that has evaluated the safety profile of ondansetron at any dosage for long-term use as anticipated in
our ongoing and planned clinical trials.
According to the National Institute of Alcohol
Abuse and Alcoholism (the “NIAAA”) and the Journal of the American Medical Association (“JAMA”), in the United
States alone, approximately 35 million people each year have AUD (such number is based upon the 2012 data provided in Grant et. al. the
JAMA 2015 publication and has been adjusted to reflect a compound annual growth rate of 1.13%, which is the growth rate reported by U.S.
Census Bureau for the general adult population from 2012-2017), resulting in significant health, social and financial costs with excessive
alcohol use being the third leading cause of preventable death and is responsible for 31% of driving fatalities in the United States (NIAAA
Alcohol Facts & Statistics). AUD contributes to over 200 different diseases and 10% of children live with a person that has an alcohol
problem. According to the American Society of Clinical Oncologists, 5-6% of new cancers and cancer deaths globally are directly attributable
to alcohol. And, The Lancet published that alcohol is the leading cause of death in people ages 15-49 globally, with the World
Health Organization reporting that 3 million deaths a year are due to harmful alcohol use. The Centers for Disease Control (the “CDC”)
has reported that AUD costs the U.S. economy about $250 billion annually, with heavy drinking accounting for greater than 75% of the social
and health related costs. Despite this, according to the article in the JAMA 2015 publication, only 7.7% of patients (i.e., approximately
2.7 million people) with AUD are estimated to have been treated in any way and only 3.6% by a physician (i.e., approximately 1.3 million
people). In addition, according to the JAMA 2017 publication, the problem in the United States appears to be growing with almost a 50%
increase in AUD prevalence between 2002 and 2013.
AUD is characterized by an urge to consume
alcohol and an inability to control the levels of consumption. Until the publication of the fifth revision of the Diagnostic and Statistical
Manual of Mental Disorders in 2013 (the “DSM-5”), AUD was broken into “alcohol dependence” and “alcohol
abuse”. More broadly, overdrinking due to the inability to moderate drinking is called alcohol addiction and is often called “alcoholism”,
sometimes pejoratively.
Since ondansetron is already manufactured
for generic sale, the active ingredient for AD04 is readily available from several manufacturers, and we have contracted with a U.S. manufacturer
to acquire ondansetron at a cost expected to be under $0.01 per dose. Clinical trial material (“CTM”) has already been manufactured
for the ONWARD Phase 3 trial. The CTM has demonstrated good stability after four years with the stability studies to date.
We have also developed the manufacturing
process at a third-party vendor to produce tablets at what we expect will serve for commercial scale production (i.e., greater than 1
million tablets per batch), also at a cost expected to be less than $0.01 per dose. A proprietary packaging process has been developed,
which appears to extend the stability of the drug product. Packaging costs are expected to be less than $0.05 per dose. We do not have
a written commitment for supply of either the tablets or the packaging and believe that alternative suppliers are available to whom we
can transfer the processes that have been developed.
Methods for the companion diagnostic genetic
test have been developed as a blood test, and we established the test with a third-party vendor capable of supporting the ONWARD Phase
3 clinical trial. Additionally, we have built validation and possible approval of the companion diagnostic into the Phase 3 program, including
that we plan to store blood samples for all patients in the event additional genetic testing is required by regulatory authorities.
Recent Developments
Private Placement
On July 6, 2021, we entered into Securities Purchase Agreements
(the “Securities Purchase Agreements”) with each of Bespoke Growth Partners, Inc. (“Bespoke”), a company controlled
by Mark Peikin, the Company’s non-executive Chief Strategy Officer (who is neither an executive officer not director of our company),
Keystone Capital Partners, LLC (“Keystone”) and Richard Gilliam, a private investor (“Gilliam”) (collectively,
the “Investors,” and each an “Investor”), pursuant to which: (i) Bespoke has agreed to purchase an aggregate of
833,334 shares of our common stock at a purchase price of $3.00 per share for aggregate gross proceeds of $2,500,002; (ii) Keystone has
agreed to purchase an aggregate of 500,000 shares of our common stock at a purchase price of $3.00 per share for aggregate gross proceeds
of $1,500,000 and (iii) Gilliam has agreed to purchase an aggregate of 333,334 shares of our common stock at a purchase price of $3.00
per share for aggregate gross proceeds of $1,000,002.
Under the terms of the Securities Purchase
Agreements: (i) Bespoke purchased 83,334 shares of our common stock and agreed to purchase an additional 750,000 shares of our common
stock upon the effectiveness of the Registration Statement; (ii) Keystone purchased 50,000 shares of our common stock and agreed to purchase
an additional 450,000 shares of our common stock upon the effectiveness of the Registration Statement and (iii) Gilliam purchased 33,334
shares of our common stock and agreed to purchase an additional 300,000 shares of our common stock upon the effectiveness of the Registration
Statement. The Securities Purchase Agreement provides that until thirty (30) days
after the effective date of this Registration Statement, if we issue or sell any shares of common stock at a price that is less than $3.00
(a “Dilutive Share Issuance”), subject to certain exceptions, then we are required to pay each Investor as a penalty an amount
in cash equal to the number of shares of common stock purchased by such Investor multiplied by the difference between the greater of the
price per share of common stock paid in the Dilutive Share Issuance and $2.48.
In connection with the Securities Purchase
Agreements, we entered into Registration Rights Agreements (“Registration Rights Agreements”), dated July 6, 2021, with each
of the Investors pursuant to which we were obligated to file a registration statement (the “Registration Statement”) with
the SEC within thirty (30) days following the date of the Registration Rights Agreement, and use all commercially reasonable efforts to
have the Registration Statement declared effective by the SEC within thirty (30) days after the Registration Statement is filed (or, in
the event of a “full review” by the SEC, within sixty (60) days after the Registration Statement is filed).
The Securities Purchase Agreements and the
Registration Rights Agreements contain customary representations, warranties, conditions and indemnification obligations of the parties,
which were made only for purposes of such Securities Purchase Agreements and Registration Rights Agreements as of specific dates and solely
for the benefit of the parties. The Securities Purchase Agreements and Registration Rights Agreements may be subject to limitations agreed
upon by the contracting parties.
Equity Line Sale
On July 14, 2021, under the terms of an equity purchase agreement,
we sold 205,762 shares of common stock to Keystone at a price of $2.43 per share for total proceeds of $500,000.
Share Issuance
One July 14, 2021, we issued 20,000 shares
to consultant under the terms of a consulting agreement at a total cost of $54,600.
Emerging Growth Company
We are an emerging growth company under the
JOBS ACT, which was enacted in April 2012. We shall continue to be deemed an emerging growth company until the earliest of:
|
|
(i)
|
the
last day of the fiscal year in which we have total annual gross revenues of $1.07 billion or more;
|
|
|
(ii)
|
the
last day of the fiscal year of the issuer following the fifth anniversary of the date of the first sale of common equity securities of
the issuer pursuant to an effective registration statement;
|
|
|
(iii)
|
the
date on which we have issued more than $1.0 billion in non-convertible debt, during the previous 3-year period, issued; or
|
|
|
(iv)
|
the
date on which we are deemed to be a large accelerated filer.
|
As an emerging growth company, we are subject
to reduced public company reporting requirements and are exempt from Section 404(b) of Sarbanes Oxley. Section 404(a) requires issuers
to publish information in their annual reports concerning the scope and adequacy of the internal control structure and procedures for
financial reporting. Section 404(b) requires that the registered accounting firm shall, in the same report, attest to and report on the
assessment on the effectiveness of the internal control structure and procedures for financial reporting.
As an emerging growth company, we are also
exempt from Section 14A (a) and (b) of the Exchange Act, which requires the shareholder approval, on an advisory basis, of executive compensation
and golden parachutes.
We have elected to use the extended transition
period for complying with new or revised accounting standards under Section 102(b)(2) of the Jobs Act, that allows us to delay the adoption
of new or revised accounting standards that have different effective dates for public and private companies until those standards apply
to private companies. As a result of this election, our financial statements may not be comparable to companies that comply with public
company effective dates.
Corporate Information
ADial Pharmaceuticals, L.L.C. was formed
as a Virginia limited liability company in November 2010. ADial Pharmaceuticals, L.L.C. converted from a Virginia limited liability company
into a Virginia corporation on October 3, 2017, and then reincorporated in Delaware on October 11, 2017 by merging the Virginia Corporation
with and into Adial Pharmaceuticals, Inc., a Delaware corporation that was incorporated on October 5, 2017 as a wholly owned subsidiary
of the Virginia corporation. We refer to this as the corporate conversion/reincorporation. In connection with the corporate conversion/reincorporation,
each unit of ADial Pharmaceuticals, L.L.C. was converted into shares of common stock of the Virginia corporation and then into shares
of common stock of Adial Pharmaceuticals, Inc., the members of ADial Pharmaceuticals, L.L.C. became stockholders of Adial Pharmaceuticals,
Inc. and Adial Pharmaceuticals, Inc. succeeded to the business of ADial Pharmaceuticals, L.L.C. The Company has one wholly owned subsidiary,
Purnovate, Inc.
Our principal executive offices are located
at 1180 Seminole Trail, Suite 495, Charlottesville Virginia 22901, and our telephone number is (434) 422-9800. Our website address is
www.adialpharma.com. Information contained in our website does not form part of this Annual Report on Form 10-K and is intended
for informational purposes only.
Additional Information
For additional information related to our
business and operations, please refer to the reports incorporated herein by reference, including our 2020 Form 10-K, our Quarterly Report
on Form 10-Q for the quarterly period ended March 31, 2021 as filed with the SEC on May 17, 2021 and our Current Reports on Form 8-K as
filed with the SEC, as described in the section entitled “Incorporation of Certain Documents By Reference” in this prospectus.
This prospectus and the information incorporated
by reference into this prospectus contain references to our trademarks and to trademarks belonging to other entities. Solely for convenience,
trademarks and trade names referred to in this prospectus and the information incorporated by reference into this prospectus, including
logos, artwork, and other visual displays, may appear without the ® or TM symbols, but such references are not intended to indicate,
in any way, that we will not assert, to the fullest extent under applicable law, our rights or the rights of the applicable licensor to
these trademarks and trade names. We do not intend our use or display of other companies’ trade names or trademarks to imply a relationship
with, or endorsement or sponsorship of us by, any other company.
Summary Risk Factors
Our business faces significant risks and
uncertainties of which investors should be aware before making a decision to invest in our common stock. If any of the following risks
are realized, our business, financial condition and results of operations could be materially and adversely affected. The following
is a summary of the more significant risks relating to the Company. A more detailed description of our risk factors set forth under
the caption “Risk Factors” in this prospectus.
Risks Relating to our Company
|
|
●
|
We have a limited operating history with which to compare, have incurred significant losses since our inception, and expect to incur substantial and increasing losses for the foreseeable future.
|
|
|
|
|
|
|
●
|
We currently have no product revenues and may not generate revenue at any time in the near future, if at all.
|
|
|
|
|
|
|
●
|
We and our independent registered accounting firm have expressed substantial doubt about our ability to continue as a going concern.
|
|
|
|
|
|
|
●
|
We will need to secure additional financing, which may not be available to us on favorable terms, if at all.
|
|
|
|
|
|
|
●
|
We have identified weaknesses in our internal controls, and it cannot be assured that these weaknesses will be effectively remediated or that additional material weaknesses will not occur in the future.
|
|
|
|
|
|
|
●
|
We rely on a license to use various technologies that are material to our business and there is no guarantee that such license agreements won’t be terminated, or that other rights necessary to commercialize our products will be available to us on acceptable terms or at all.
|
|
|
|
|
|
|
●
|
Our business is dependent upon the success of our lead product candidate, AD04, which requires significant additional clinical testing before we can seek regulatory approval and potentially launch commercial sales.
|
|
|
|
|
|
|
●
|
The active ingredient of our product candidate, ondansetron, is currently available in generic form and has been shown to have adverse effects on patients.
|
|
|
|
|
|
|
●
|
Coronavirus or other global health crises could adversely impact our business, including our clinical trials.
|
|
|
|
|
|
|
●
|
While there exists a large body of evidence supporting the safety of our primary API, ondansetron, under short-term use, there are currently no long-term use clinical safety data available.
|
|
|
●
|
All of our current data for our lead product candidate are the result of Phase 2 clinical trials conducted by third parties and do not necessarily provide sufficient evidence that our products are viable as potential pharmaceutical products.
|
|
|
|
|
|
|
●
|
The FDA and/or EMA may not accept our planned Phase 3 endpoints for final approval of AD04 and may determine additional clinical trials are required for approval of AD04.
|
|
|
●
|
Our lead investigational product, AD04, is dependent on a successful development, approval, and commercialization of a genetic test, which is expected to be classified as a companion diagnostic, which may not attain regulatory approval.
|
|
|
|
|
|
|
●
|
We have limited experience as a company conducting clinical trials, any may experience delays in our clinical trials and may fail to demonstrate adequately the safety and efficacy of AD04 or any future product candidates.
|
|
|
|
|
|
|
●
|
Our product candidate and the product candidates of Purnovate are in the early stages of development and there is uncertainty as to market acceptance of our technology and product candidates.
|
Risks Relating to our Acquisition of Purnovate
|
|
●
|
The combined company may not experience the anticipated strategic benefits of the Acquisition and we may be unable to successfully integrate the Purnovate businesses.
|
|
|
|
|
|
|
●
|
Purnovate has a limited operating history upon which to evaluate its ability to commercialize its products.
|
Risks Relating to our Business and Industry
|
|
●
|
We must obtain and maintain regulatory approvals in every jurisdiction in which we intend to sell our product candidate and the regulatory approval in one jurisdiction does not guarantee the approval in another jurisdiction.
|
|
|
|
|
|
|
●
|
AD04 and any future product candidates may cause undesirable side effects or have other properties that could halt their clinical development, prevent their regulatory approval, limit their commercial potential or result in significant negative consequences such as incurring product liability lawsuits
|
|
|
|
|
|
|
●
|
We will continue to be subject to ongoing and extensive regulatory requirements even after regulatory approval, and compliance with such regulatory requirements cannot be assured.
|
|
|
|
|
|
|
●
|
Our employees, independent contractors, consultants, commercial partners and vendors may engage in misconduct or other improper activities, including noncompliance with regulatory standards and requirements.
|
|
|
|
|
|
|
●
|
We have no experience selling, marketing or distributing products and have no internal capability to do so.
|
|
|
|
|
|
|
●
|
We may not be successful in establishing and maintaining strategic partnerships.
|
|
|
|
|
|
|
●
|
We have limited protection for our intellectual property. Our licensed patents and proprietary rights may not prevent us from infringing on the rights of others or prohibit potential competitors from commercializing products.
|
|
|
|
|
|
|
●
|
We may be involved in lawsuits to protect or enforce the patents of our licensors, which could be expensive, time-consuming and unsuccessful.
|
|
|
|
|
|
|
●
|
We rely on key executive officers and scientific, regulatory and medical advisors, and their knowledge of our business and technical expertise would be difficult to replace.
|
|
|
|
|
|
|
●
|
Certain of our officers may have a conflict of interest.
|
Risks Related to our Securities and Investing
in our Securities
|
|
●
|
Certain of our shareholders have sufficient voting power to make corporate governance decisions that could have a significant influence on us and the other stockholders.
|
|
|
|
|
|
|
●
|
We have never paid dividends and have no plans to pay dividends in the foreseeable future.
|
|
|
|
|
|
|
●
|
Our failure to meet the continued listing requirements of The Nasdaq Capital Market could result in a de-listing of our common stock.
|
|
|
|
|
|
|
●
|
Our stock price has fluctuated in the past, has recently been volatile and may be volatile in the future, and as a result, investors in our common stock could incur substantial losses.
|
|
|
|
|
|
|
●
|
Our need for future financing may result in the issuance of additional securities which will cause investors to experience dilution.
|
|
|
|
|
|
|
●
|
Provisions in our corporate charter documents and under Delaware law could make an acquisition of our company, which may be beneficial to our stockholders, more difficult and may prevent attempts by our stockholders to replace or remove our current management.
|
|
|
|
|
|
|
●
|
Our Certificate of Incorporation and our bylaws provide that the Court of Chancery of the State of Delaware will be the exclusive forum for certain types of state actions that may be initiated by our stockholders, which could limit our stockholders’ ability to obtain a favorable judicial forum for disputes with us or our directors, officers, or employees.
|
|
|
|
|
|
|
●
|
If securities or industry analysts do not publish research or publish inaccurate or unfavorable research about our business, our stock price and trading volume could decline.
|
THE OFFERING
|
Common Stock offered by
us in this offering
|
|
We are not selling any shares of common stock pursuant to this prospectus.
|
|
|
|
|
|
Common Stock
offered by
the selling
stockholders
|
|
Up to 1,666,668 shares of common stock.
|
|
|
|
|
|
Terms of the offering
|
|
The
selling stockholders will determine when and how they will sell the common stock offered in this prospectus, as described
in “Plan of Distribution” on page 21 of this prospectus.
|
|
|
|
|
|
Use of Proceeds
|
|
The
selling stockholders will receive all of the proceeds from the sale of shares of common stock offered from time to time pursuant to
this prospectus. Accordingly, we will not receive any proceeds from the sale of shares of common stock that may be sold from time to
time pursuant to this prospectus; however, we have received aggregate gross proceeds of $500,002 from the sale of an aggregate of
166,668 to the selling stockholders on July 6, 2021 under the Securities Purchase Agreements and expect to receive aggregate gross
proceeds of $4,500,000 from the sale of an aggregate of 1,500,000 additional shares, which the selling stockholders have committed
to purchase in a second tranche purchase that will occur on the second business day following the effective date of the registration
statement of which this prospectus forms a part. We intend to use the proceeds that we receive for working capital purposes,
acquisitions and other strategic purposes. See “Use of Proceeds” on page 10 of this prospectus.
|
|
|
|
|
|
Risk factors
|
|
You should read the “Risk Factors” section of this prospectus on page 8 and the information incorporated by reference therein for a discussion of factors to consider before deciding to purchase shares of our common stock.
|
|
|
|
|
|
Nasdaq Capital Market symbol
|
|
Our common stock is listed on the Nasdaq Capital Market under the symbol
“ADIL.” Our warrants are listed on the Nasdaq Capital Market under the symbol “ADILW.” On July 15, 2021, the last
reported sale price of our common stock and warrants on the Nasdaq Capital Market was $2.49 per share and $0.77 per warrant, respectively.
|
RISK FACTORS
An investment in our common stock involves a high
degree of risk. Before deciding whether to invest in our common stock, you should consider carefully the risks and uncertainties described
in the section captioned “Risk Factors” contained in our Annual Report on Form 10-K for the fiscal year ended December 31,
2020 filed with the SEC on March 22, 2021 and other filings we make with the SEC from time to time, which are incorporated by reference
herein in their entirety, together with other information in this prospectus and the information incorporated by reference herein. For
a description of these reports and documents, and information about where you can find them, see “Where You Can Find More Information”
and “Incorporation of Certain Documents By Reference.” Additional risks not presently known or that we presently consider
to be immaterial could subsequently materially and adversely affect our and the information incorporated by reference herein. If any of
these risks actually occurs, our business, financial condition, results of operations or cash flow could suffer materially. In such event,
the trading price of our common stock could decline, and you might lose all or part of your investment.
CAUTIONARY NOTE REGARDING FORWARD-LOOKING STATEMENTS
This prospectus and the documents incorporated
by reference herein contain forward-looking statements that are based on current management expectations. Statements other than statements
of historical fact included in this prospectus, including statements about us and the future growth and anticipated operating results
and cash expenditures, are forward-looking statements within the meaning of Section 27A of the Securities Act and Section 21E of the Securities
Exchange Act of 1934, as amended (the “Exchange Act”). When used in this prospectus the words “anticipate,” “objective,”
“may,” “might,” “should,” “could,” “can,” “intend,” “expect,”
“believe,” “estimate,” “predict,” “potential,” “plan” or the negative of these
and similar expressions identify forward-looking statements. These statements reflect our current views with respect to uncertain future
events and are based on imprecise estimates and assumptions and subject to risk and uncertainties. Given these uncertainties, you should
not place undue reliance on these forward-looking statements. While we believe our plans, intentions and expectations reflected in those
forward-looking statements are reasonable, these plans, intentions or expectations may not be achieved. Our actual results, performance
or achievements could differ materially from those contemplated, expressed or implied by the forward-looking statements contained in,
or incorporated by reference into, this prospectus for a variety of reasons. Those risks and uncertainties include, among others:
|
|
●
|
our
ability to implement our business plan;
|
|
|
●
|
our ability to raise additional capital to meet our liquidity needs;
|
|
|
●
|
our ability to generate product revenues;
|
|
|
●
|
our ability to achieve profitability;
|
|
|
●
|
our ability to satisfy U.S. (including FDA) and international regulatory requirements;
|
|
|
●
|
our ability to obtain market acceptance of our technology and products;
|
|
|
●
|
our ability to compete in the market;
|
|
|
●
|
our ability to advance our clinical trials;
|
|
|
●
|
our ability to fund, design and implement clinical trials;
|
|
|
●
|
our ability to demonstrate that our lead product candidate is safe for human use and effective for indicated uses;
|
|
|
●
|
our ability to gain acceptance of physicians and patients for use of our lead product;
|
|
|
●
|
our dependency on third-party researchers, manufacturers and payors;
|
|
|
●
|
our ability to establish and maintain strategic partnerships, including for the distribution of our lead product and any future products that we may acquire;
|
|
|
●
|
our ability to attract and retain a sufficient qualified personnel;
|
|
|
●
|
our ability to obtain or maintain patents or other appropriate protection for the intellectual property;
|
|
|
●
|
our dependency on the intellectual property licensed to us or possessed by third parties;
|
|
|
●
|
our ability to adequately support future growth;
|
|
|
●
|
potential product liability or intellectual property infringement claims;
|
|
|
|
|
|
|
●
|
our ability to successfully integrate
the Purnovate business into our business; and
|
|
|
●
|
disruption or delay of our ongoing clinical trial, disruption of our corporate operations or those of our critical vendors, or general significant disruption to the global economy as a resulting of ongoing the COVID-19 pandemic.
|
We urge investors to review carefully risks contained
in the section of this prospectus entitled “Risk Factors” above as well as other risks and factors identified from time to
time in our SEC filings in evaluating the forward-looking statements contained in this prospectus. We caution investors not to place significant
reliance on forward-looking statements contained in this document; such statements need to be evaluated in light of all the information
contained herein.
All forward-looking statements attributable to
us or persons acting on our behalf are expressly qualified in their entirety by the risk factors and other cautionary statements set forth,
or incorporated by reference, in this prospectus. Other than as required by applicable securities laws, we are under no obligation, and
we do not intend, to update any forward-looking statement, whether as result of new information, future events or otherwise.
USE OF PROCEEDS
The selling stockholders will receive all of the
proceeds of the sale of shares of common stock offered from time to time pursuant to this prospectus. Accordingly, we will not receive
any proceeds from the sale of shares of common stock that may be sold from time to time pursuant to this prospectus; however, we have
received aggregate gross proceeds of $500,002 from the sale of an aggregate of 166,668 to the selling stockholders on July 6, 2021 under
the Securities Purchase Agreements and expect to receive aggregate gross proceeds of $4,500,000 from the sale of an aggregate of 1,500,000
additional shares, which the three selling stockholders have committed to purchase in a second tranche purchase that will occur on the
second business day following the effective date of the registration statement of which this prospectus forms a part. We intend to use
the proceeds that we receive for working capital purposes, acquisitions and other strategic purposes.
We will bear the out-of-pocket costs, expenses
and fees incurred in connection with the registration of shares of our common stock to be sold by the selling stockholders pursuant to
this prospectus. Other than registration expenses, the selling stockholders will bear underwriting discounts, commissions, placement agent
fees or other similar expenses payable with respect to sales of shares of our common stock.
DIVIDEND POLICY
We do not anticipate paying dividends on our common
stock. We currently intend to retain all of our future earnings, if any, to finance the growth and development of our business. We are
not subject to any legal restrictions respecting the payment of dividends, except that we may not pay dividends if the payment would render
us insolvent. Any future determination as to the payment of cash dividends on our common stock will be at our board of directors’
discretion and will depend on our financial condition, operating results, capital requirements and other factors that our board of directors
considers to be relevant.
SELLING STOCKHOLDERS
This prospectus covers the resale or other disposition
by the selling stockholders identified in the table below of up to an aggregate of 1,666,668 shares of our common stock issuable. These
shares have been or will be acquired by the selling stockholders in the private placement transaction described above under the heading
“Prospectus Summary––Private Placement.”
On July 6, 2021, we entered into Securities Purchase
Agreements with each of Bespoke, a company controlled by Mark Peikin, our non-executive Chief Strategy Officer, Keystone, and Gilliam
(collectively, the “Investors,” and each an “Investor”), pursuant to which: (i) Bespoke has agreed to purchase
an aggregate of 833,334 shares of the Company’s common stock at a purchase price of $3.00 per share for aggregate gross proceeds
of $2,500,002; (ii) Keystone has agreed to purchase an aggregate of 500,000 shares of the Company’s common stock at a purchase price
of $3.00 per share for aggregate gross proceeds of $1,500,000; and (iii) Gilliam has agreed to purchase an aggregate of 333,334 shares
of the Company’s common stock at a purchase price of $3.00 per share for gross proceeds of $1,000,002. On July 6, 2021, under
the Securities Purchase Agreements we sold an aggregate of 166,668 shares of our common stock for $3.00 per share to the selling stockholders
for aggregate gross proceeds to us of $500,002, as follows: (i) 83,334 shares were purchased by Bespoke for a total purchase price of
$250,000, (ii) 50,000 shares were purchased by Keystone for a total purchase price of $150,000 and (iii) 33,334 shares were purchased
by Gilliam for a total purchase price of $100,002. Under the terms of the Securities Purchase Agreements, the selling stockholders agreed
to purchase an aggregate of 1,500,000 additional shares of our common stock at a price of $3.00 per share in a second tranche purchase
that will occur on the second business day after the effective date of the registration statement of which this prospectus is a part,
as follows: (i) Bespoke agreed to purchase 750,000 additional shares of our common stock in the second tranche purchase for a purchase
price of $2,250,000, (ii) Keystone agreed to purchase 450,000 additional shares of our common stock in the second tranche purchase for
a purchase price of $1,350,000 and (iii) Gilliam agreed to purchase 300,000 additional shares of our common stock in the second tranche
purchase for a purchase price of $900,000.
In connection with the Securities Purchase Agreements,
the Company entered into Registration Rights Agreements), dated July 6, 2021, with each of the Investors pursuant to which the Company
is obligated to file a registration statement (the “Registration Statement”) with the U.S. Securities and Exchange Commission
(the “SEC”) within thirty (30) days following the date of the Registration Rights Agreement, and use all commercially reasonable
efforts to have the Registration Statement declared effective by the SEC within thirty (30) days after the Registration Statement is filed
(or, in the event of a “full review” by the SEC, within sixty (60) days after the Registration Statement is filed).
Bespoke is a company controlled by Mark Peikin,
our non-executive Chief Strategy Officer. In the last three years, Bespoke has been issued 250,000 shares of our common stock as compensation
for services provided. Bespoke purchased 357,143 shares of our common stock on September 21, 2020 for an aggregate purchase price of $500,000
in a private placement transaction. In addition, on March 11, 2021 we engaged in a private placements of our common stock and entered
into a Securities Purchase Agreement (the “March SPA”) and Registration Rights Agreement (the “March Registration Rights
Agreement”) with certain investors that included Bespoke pursuant to which Bespoke agreed to purchase an aggregate of 336,667 shares
of our common stock at a purchase price of $3.00 per shares for a total purchase price of $1,010,001. Pursuant
to the Registration Rights Agreement (the “April RRA”), we filed a registration statement on Form S-3 with the SEC on April
20, 2020 to register the resale of shares of our common stock that have been or may be issued and sold to Bespoke and the other investors
under the March SPA, which registration statement was declared effective by the SEC on May 26, 2021. Pursuant to the terms
of the March SPA, on March 12, 2021, Bespoke purchased and was issued 33,667 shares of our common stock for a total purchase price of
$101,001 and on May 28, 2021, Bespoke purchased 303,000 shares of our common stock for a total purchase price of $909,000. None of the
shares of common stock that were issued as compensation or purchased by Bespoke on September 21, 2020 or that were issued pursuant to
the March SPA are included in the registration statement of which this prospectus is a part.
On November 18, 2020, we entered into a Common
Stock Purchase Agreement (the “Keystone Purchase Agreement”) and a Registration Rights Agreement (the “Keystone Registration
Rights Agreement”) with Keystone. Pursuant to the Keystone Purchase Agreement, we have the right to sell to Keystone Capital up
to $15,000,000 of our common stock from time to time during the term of the Purchase Agreement. Upon signing the Keystone Purchase Agreement
and the Registration Rights Agreement, we issued to Keystone 175,000 shares of common stock in consideration of its commitment to purchase
our common stock at our direction from time to time under the Keystone Purchase Agreement. As of the date of this prospectus, we have
sold an aggregate of 1,645,907 shares of common stock to Keystone in sales we made pursuant to the Keystone Purchase Agreement and received
aggregate gross proceeds of $3,850,000 from such sales. All of such issuances and sales of our common stock to Keystone under the Keystone
Purchase Agreement are made in reliance upon exemptions from the registration requirements of the Securities Act afforded by Section 4(a)(2)
of the Securities Act and Rule 506(b) of Regulation D thereunder, as well as exemptions from registration or qualification under applicable
state securities laws. Pursuant to the Keystone Registration Rights Agreement, we filed a registration statement on Form S-1 with the
SEC on December 4, 2020 to register the resale of shares of our common stock that have been or may be issued and sold to Keystone under
the Keystone Purchase Agreement, which was declared effective by the SEC on December 15, 2020. In addition, Keystone was one of the investors
in our March private placement and on March 11, 2021 we entered into the March SPA and March RRA with Keystone pursuant to which Keystone
agreed to purchase an aggregate of 333,334 shares of our common stock at a purchase price of $3.00 per shares for a total purchase price
of $1,000,002. The registration statement on Form S-3 that we filed with the SEC on April 20, 2020,
which was declared effective by the SEC on May 26, 2021, included the 333,334 shares issued and at that time to be issued to Keystone
under the March SPA. Pursuant to the terms of the March SPA, on March 12, 2021, Keystone purchased and was issued 33,334 shares
of our common stock for a total purchase price of $100,002 and on June 1, 2021, Keystone purchased and was issued 300,000 shares of our
common stock for a total purchase price of $900,000. None of the shares of common stock that have been or may be issued and sold to Keystone
under the Keystone Purchase Agreement or the March SPA are included in the registration statement of which this prospectus is a part.
Kermit Anderson, one of our Directors, manages
Richard Gilliam’s family office.
Other than as described herein and the documents
incorporated by reference herein, we have not had a material relationship with any of the selling stockholders during the last three years.
The number of shares of common stock beneficially
owned by the selling stockholders has been determined in accordance with Rule 13d-3 under the Exchange Act and includes, for such purpose,
shares of common stock that the selling stockholders has the right to acquire within 60 days of July 15, 2021. The maximum number of shares
of common stock and percentage of shares of common stock beneficially owned by the selling stockholders after the offering shown in the
table below assumes all of the offered shares are sold by the selling stockholders and is based on an aggregate of 20,298,156 shares of
our common stock, which includes an aggregate of 18,798,156 shares of our outstanding on July 15, 2021, and includes the 1,500,000 shares
of common stock that the selling stockholders have committed to purchase in a second tranche purchase pursuant to the terms of the Securities
Purchase Agreements.
All information with respect to the ownership
of the shares of common stock by the selling stockholders has been furnished by or on behalf of the selling stockholders. We believe,
based on information supplied by the selling stockholders, that except as may otherwise be indicated in the footnotes to the table below,
the selling stockholders have sole voting and dispositive power with respect to the shares of common stock reported as beneficially owned
by it. Because the selling stockholders identified in the table may sell some or all of the shares of common stock beneficially owned
by it and covered by this prospectus, and because there are currently no agreements, arrangements or understandings with respect to the
sale of any of the shares of common stock, no estimate can be given as to the number of shares of common stock available for resale hereby
that will be held by the selling stockholders upon termination of this offering. In addition, the selling stockholders may have sold,
transferred or otherwise disposed of, or may sell, transfer or otherwise dispose of, at any time and from time to time, the shares of
common stock they beneficially own in transactions exempt from the registration requirements of the Securities Act after the date on which
they provided the information set forth in the table below. We have, therefore, assumed for the purposes of the following table, that
the selling stockholders will sell all of the shares of common stock that it owns or may own beneficially that are covered by this prospectus,
but will not sell any other shares of our common stock that they presently own that are not covered by this prospectus.
|
|
|
Number of Shares of
Common Stock Beneficially Owned
Prior to
|
|
|
Maximum
Number of
Shares of
Common
Stock that may be
Offered
|
|
|
Number of Shares of
Common
Stock Owned
Beneficially Owned After
|
|
|
Percent
Beneficial Ownership
After
|
|
|
Name of Selling Stockholder
|
|
Offering
|
|
|
for Resale
|
|
|
Offering
|
|
|
Offering
|
|
|
Bespoke Growth Partners, Inc.(1)
|
|
|
741,638
|
(2)
|
|
|
833,334
|
(3)
|
|
|
658,304
|
|
|
|
3.24
|
%
|
|
Keystone Capital Partners, LLC(4)
|
|
|
377,507
|
(5)
|
|
|
500,000
|
(6)
|
|
|
327,507
|
|
|
|
1.61
|
%
|
|
Richard Gilliam(7)
|
|
|
294,233
|
(8)
|
|
|
333,334
|
(9)
|
|
|
260,899
|
|
|
|
1.28
|
%
|
|
*
|
Represents beneficial ownership of less than 1% of the outstanding shares of our common stock.
|
|
(1)
|
The address for Bespoke Growth Partners, Inc. is 1875 N.W. Corporate Boulevard, Suite 290 Boca Raton, Florida 33431.
|
|
(2)
|
The shares beneficially owned by Bespoke Growth Partners consist of 741,638 shares of common stock which include: (i) 83,334 shares of our common stock purchased by Bespoke on July 6, 2021 under the Securities Purchase Agreement for a total purchase price of $250,002, all of which shares are being registered for resale in the registration statement of which this prospectus is a part and (ii) 658,304 shares of our common stock owned by Bespoke, none of which 658,304 shares are being registered for resale in the registration statement of which this prospectus is a part. In accordance with Rule 13d-3(d) under the Exchange Act, we have excluded from the number of shares beneficially owned prior to the offering all of the 750,000 additional shares of common stock that Bespoke has committed to purchase from us in a second tranche purchase under the Securities Purchase Agreement; however, we have included in the maximum number of shares of common stock that may be offered for resale the 750,000 additional shares of common stock that Bespoke has committed to purchase in a second tranche purchase pursuant to the terms of the Securities Purchase Agreement, because Bespoke’s obligation to purchase such 750,000 additional shares of common stock is subject to conditions set forth in the Securities Purchase Agreement, the satisfaction of which are entirely outside of Bespoke’s control, including the registration statement that includes this prospectus becoming and remaining effective. Such 750,000 additional shares that Bespoke has committed to purchase in a second tranche purchase subject to the terms and conditions of the Securities Purchase Agreement are, however, being registered for resale in the registration statement of which this prospectus is a part together with the 83,334 shares of common stock purchased by Bespoke on July 6, 2021 and beneficially owned by Bespoke immediately prior to this offering.
|
|
|
(3)
|
The maximum number of shares that may be offered for resale
by Bespoke through this prospectus consist of: (i) 83,334 shares of our common stock purchased by Bespoke on July 6, 2021 under the Securities
Purchase Agreement for a total purchase price of $250,002 and (ii) the 750,000 additional shares of common stock that Bespoke has committed
to purchase from us in a second tranche purchase under the Securities Purchase Agreement that will occur on the second business day after
the effective date of the registration statement of which this prospectus is a part
|
|
|
(4)
|
The business address of Keystone Capital Partners, LLC is 139
Fulton Street, Suite 412, New York, New York 10038. Keystone Capital Partners, LLC’s principal business is that of a private investor.
Ranz Group, LLC, a Delaware limited liability company, is the managing member of Keystone Capital Partners, LLC and the beneficial owner
of 97% of the membership interests in Keystone Capital Partners, LLC. Fredric G. Zaino is the managing member of Ranz Group, LLC and
has sole voting control and investment discretion over securities beneficially owned directly by Keystone Capital, LLC and indirectly
by Ranz Group, LLC. We have been advised that none of Mr. Zaino, Ranz Group, LLC or Keystone Capital Partners, LLC is a member of the
Financial Industry Regulatory Authority, or FINRA, or an independent broker-dealer, or an affiliate or associated person of a FINRA member
or independent broker-dealer. The foregoing should not be construed in and of itself as an admission by Mr. Zaino as to beneficial ownership
of the securities beneficially owned directly by Keystone Capital Partners, LLC and indirectly by Ranz Group, LLC.
|
|
(5)
|
The shares beneficially owned by
Keystone prior to the offering consist of: (i) 50,000 shares of our common stock purchased by Keystone on July 6, 2021 under the
Securities Purchase Agreement for a total purchase price of $150,000, all of which shares are being registered for resale in the
registration statement of which this prospectus is a part, and (ii) 327,507 shares of our common stock owned by Keystone, none of
which 327,507 shares are being registered for resale in the registration statement of which this prospectus is a part. In accordance
with Rule 13d-3(d) under the Exchange Act, we have excluded from the number of shares beneficially owned prior to the offering all
of the 450,000 additional shares of common stock that Keystone has committed to purchase from us in a second tranche purchase under
the Securities Purchase Agreement, because Keystone’s obligation to purchase such 450,000 additional shares of common stock is
subject to conditions set forth in the Securities Purchase Agreement, the satisfaction of which are entirely outside of
Keystone’s control, including the registration statement that includes this prospectus becoming and remaining effective. Such
450,000 additional shares that Keystone has committed to purchase in a second tranche purchase subject to the terms and conditions
of the Securities Purchase Agreement are, however, being registered for resale by Keystone in the registration statement of which
this prospectus is a part together with the 50,000 shares of common stock purchased by Keystone on July 6, 2021 and beneficially
owned by Keystone immediately prior to this offering.
|
|
(6)
|
The maximum number of shares that may
be offered for resale by Keystone through this prospectus consist of: (i) 50,000 shares of our common stock purchased by Keystone on July
6, 2021 under the Securities Purchase Agreement for a total purchase price of $500,000 and (ii) the 450,000 additional shares of common
stock that Keystone has committed to purchase from us in a second tranche purchase under the Securities Purchase Agreement that will occur
on the second business day after the effective date of the registration statement of which this prospectus is a part.
|
|
|
(7)
|
The address for Richard Gilliam is 650 Peter Jefferson Parkway,
Suite 230, Charlottesville, VA 22911.
|
|
(8)
|
The shares beneficially owned by Gilliam
prior to the offering consist of: (i) 33,334 shares of our common stock purchased by Gilliam on July 6, 2021 under the Securities Purchase
Agreement for a total purchase price of $100,002, all of which shares are being registered for resale in the registration statement of
which this prospectus is a part, 146,409 shares of common stock owned by Gilliam, none of which 146,409 shares are being registered for
resale in the registration statement of which this prospectus is a part, and (iii) warrants to purchase 114,490 shares of our common stock
owned by Gilliam, none of which 114,490 shares underlying the warrants are being registered for resale in the registration statement of
which this prospectus is a part. In accordance with Rule 13d-3(d) under the Exchange Act, we have excluded from the number of shares beneficially
owned prior to the offering all of the 300,000 additional shares of common stock that Gilliam has committed to purchase from us in a second
tranche purchase under the Securities Purchase Agreement, because Gilliam’s obligation to purchase such 300,000 additional shares
of common stock is subject to conditions set forth in the Securities Purchase Agreement, the satisfaction of which are entirely outside
of Gilliam’s control, including the registration statement that includes this prospectus becoming and remaining effective. Such
300,000 additional shares that Gilliam has committed to purchase in a second tranche purchase subject to the terms and conditions of the
Securities Purchase Agreement are, however, being registered for resale by Gilliam in the registration statement of which this prospectus
is a part together with the 33,334 shares of common stock purchased by Gilliam on July 6, 2021 and beneficially owned by Gilliam immediately
prior to this offering.
|
|
|
|
|
(9)
|
The maximum number of shares that may
be offered for resale by Gilliam through this prospectus consist of: (i) 33,334 shares of our common stock purchased by Gilliam on July
6, 2021 under the Securities Purchase Agreement for a total purchase price of $100,002 and (ii) the 300,000 additional shares of common
stock that Gilliam has committed to purchase from us in a second tranche purchase under the Securities Purchase Agreement that will occur
on the second business day after the effective date of the registration statement of which this prospectus is a part
|
DETERMINATION OF OFFERING PRICE
The prices at which the shares of common stock
covered by this prospectus may actually be sold by the selling stockholders will be determined by prevailing market prices for shares
of our common stock at the time of sale, by negotiations between the selling stockholders and buyers of our common stock or as otherwise
described in “Plan of Distribution.”
DESCRIPTION OF CAPITAL STOCK
The following description of our capital stock
and the provisions of our certificate of incorporation and our bylaws are summaries and are qualified by reference to the certificate
of incorporation and the bylaws. We have filed copies of these documents with the SEC as exhibits to our registration statement of which
this prospectus forms a part.
General
As of the date of this prospectus, our authorized
capital stock consists of 50,000,000 shares of common stock, par value $0.001 per share, and 5,000,000 shares of preferred stock, par
value $0.001 per share.
Common Stock
For a description of our capital stock, please
see the Description of Securities included as Exhibit 4.19 to our Annual Report on Form 10-K for the year ended December 31, 2020, filed
with the SEC on March 22, 2021, which is incorporated by reference herein. See “Incorporation of Certain Documents by Reference”
and “Where You Can Find More Information.”
Common stock outstanding. As of July 15,
2021, there were 18,798,156 shares of our common stock outstanding.
Voting rights. The holders of common stock
are entitled to one vote per share on all matters to be voted upon by the stockholders, except on matters relating solely to terms of
preferred stock. Stockholders do not have cumulative voting rights.
Dividend rights. Subject to preferences
that may be applicable to any outstanding preferred stock, the holders of common stock are entitled to receive ratably such dividends,
if any, as may be declared from time to time by the board of directors out of funds legally available therefor. See “Dividend Policy.”
Rights upon liquidation. In the event of
our liquidation, dissolution or winding up, the holders of common stock are entitled to share ratably in all assets remaining after payment
of liabilities, subject to prior distribution rights of preferred stock, if any, then outstanding.
Other rights. The holders of our common
stock have no preemptive or conversion rights or other subscription rights. There are no redemption or sinking fund provisions applicable
to our common stock.
Preferred Stock
There are no shares of our preferred stock outstanding
as of the date of this prospectus.
Our board of directors has the authority to issue
preferred stock in one or more classes or series and to fix the designations, powers, preferences and rights, and the qualifications,
limitations or restrictions thereof, including dividend rights, conversion right, voting rights, terms of redemption, liquidation preferences
and the number of shares constituting any class or series, without further vote or action by the stockholders. Although we have no present
plans to issue any shares of preferred stock, the issuance of shares of preferred stock, or the issuance of rights to purchase such shares,
could decrease the amount of earnings and assets available for distribution to the holders of common stock, could adversely affect the
rights and powers, including voting rights, of the common stock, and could have the effect of delaying, deterring or preventing a change
of control of us or an unsolicited acquisition proposal. To date, no preferred stock has been issued.
Warrants
At the date of this prospectus, we had outstanding
warrants to purchase 7,957,225 shares of common stock at exercise prices ranging from $0.005 to $7.634 (with a weighted average exercise
price of $4.83) and expiration dates from July 31, 2023 to December 31, 2031.
Anti-Takeover Effects of Delaware Law
The provisions of Delaware law, our certificate
of incorporation and our bylaws described below may have the effect of delaying, deferring or discouraging another party from acquiring
control of us.
Section 203 of the Delaware General Corporation
Law
We are subject to Section 203 of the Delaware
General Corporation Law, which prohibits a Delaware corporation from engaging in any business combination with any interested stockholder
for a period of three years after the date that such stockholder became an interested stockholder, with the following exceptions:
|
|
●
|
before such date, the board of directors of the corporation approved either the business combination or the transaction that resulted in the stockholder becoming an interested stockholder;
|
|
|
|
|
|
|
|
upon completion of the transaction that resulted in the stockholder becoming an interested stockholder, the interested stockholder owned at least 85% of the voting stock of the corporation outstanding at the time the transaction began, excluding for purposes of determining the voting stock outstanding (but not the outstanding voting stock owned by the interested stockholder) those shares owned (i) by persons who are directors and also officers and (ii) employee stock plans in which employee participants do not have the right to determine confidentially whether shares held subject to the plan will be tendered in a tender or exchange offer; or
|
|
|
|
|
|
|
●
|
on or after such date, the business combination is approved by the board of directors and authorized at an annual or special meeting of the stockholders, and not by written consent, by the affirmative vote of at least sixty-six and two-thirds percent (66 2/3%) of the outstanding voting stock that is not owned by the interested stockholder.
|
In general, Section 203 defines business combination to include the
following:
|
|
●
|
any merger or consolidation involving the corporation and the interested stockholder;
|
|
|
|
|
|
|
●
|
any sale, transfer, pledge or other disposition of 10% or more of the assets of the corporation involving the interested stockholder;
|
|
|
|
|
|
|
●
|
subject to certain exceptions, any transaction that results in the issuance or transfer by the corporation of any stock of the corporation to the interested stockholder;
|
|
|
|
|
|
|
●
|
any transaction involving the corporation that has the effect of increasing the proportionate share of the stock or any class or series of the corporation beneficially owned by the interested stockholder; or
|
|
|
|
|
|
|
●
|
the receipt by the interested stockholder of the benefit of any loss, advances, guarantees, pledges or other financial benefits by or through the corporation.
|
Certificate of Incorporation and Bylaws
Our certificate of incorporation and bylaws provide
that:
|
|
●
|
our board of directors is divided into three classes, one class of which is elected each year by our stockholders with the directors in each class to serve for a three-year term;
|
|
|
|
|
|
|
●
|
the authorized number of directors can be changed only by resolution of our board of directors;
|
|
|
|
|
|
|
●
|
directors may be removed only by the affirmative vote of the holders of at least 60% of our voting stock, whether for cause or without cause;
|
|
|
|
|
|
|
●
|
our bylaws may be amended or repealed by our board of directors or by the affirmative vote of sixty-six and two-thirds percent (66 2/3%) of our stockholders;
|
|
|
|
|
|
|
●
|
stockholders may not call special meetings of the stockholders or fill vacancies on the board of directors;
|
|
|
|
|
|
|
●
|
our board of directors will be authorized to issue, without stockholder approval, preferred stock, the rights of which will be determined at the discretion of the board of directors and that, if issued, could operate as a “poison pill” to dilute the stock ownership of a potential hostile acquirer to prevent an acquisition that our board of directors does not approve;
|
|
|
|
|
|
|
●
|
our stockholders do not have cumulative voting rights, and therefore our stockholders holding a majority of the shares of common stock outstanding will be able to elect all of our directors; and
|
|
|
|
|
|
|
●
|
our stockholders must comply with advance notice provisions to bring business before or nominate directors for election at a stockholder meeting.
|
Board Classification
Our board of directors is divided into three classes,
one class of which is elected each year by our stockholders. The directors in each class will serve for a three-year term. For more information
on the classified board, see “Management—Board of Directors and Executive Officers.” The classification of our board
of directors and the limitations on the ability of our stockholders to remove directors could make it more difficult for a third-party
to acquire, or discourage a third-party from seeking to acquire, control of us.
Potential Effects of Authorized but Unissued
Stock
We have shares of common stock and preferred stock
available for future issuance without stockholder approval. We may utilize these additional shares for a variety of corporate purposes,
including future public offerings to raise additional capital, to facilitate corporate acquisitions or payment as a dividend on the capital
stock.
The existence of unissued and unreserved common
stock and preferred stock may enable our board of directors to issue shares to persons friendly to current management or to issue preferred
stock with terms that could render more difficult or discourage a third-party attempt to obtain control of us by means of a merger, tender
offer, proxy contest or otherwise, thereby protecting the continuity of our management. In addition, the board of directors has the discretion
to determine designations, rights, preferences, privileges and restrictions, including voting rights, dividend rights, conversion rights,
redemption privileges and liquidation preferences of each series of preferred stock, all to the fullest extent permissible under the Delaware
General Corporation Law and subject to any limitations set forth in our certificate of incorporation. The purpose of authorizing the board
of directors to issue preferred stock and to determine the rights and preferences applicable to such preferred stock is to eliminate delays
associated with a stockholder vote on specific issuances. The issuance of preferred stock, while providing desirable flexibility in connection
with possible financings, acquisitions and other corporate purposes, could have the effect of making it more difficult for a third party
to acquire, or could discourage a third party from acquiring, a majority of our outstanding voting stock.
Limitations of Director Liability and Indemnification
of Directors, Officers and Employees
Our certificate of incorporation limits the liability
of directors to the maximum extent permitted by Delaware law. Delaware law provides that directors of a corporation will not be personally
liable for monetary damages for breach of their fiduciary duties as directors, except for liability for any:
|
|
●
|
breach of their duty of loyalty to us or our stockholders;
|
|
|
|
|
|
|
●
|
act or omission not in good faith or that involves intentional misconduct or a knowing violation of law;
|
|
|
|
|
|
|
●
|
unlawful payments of dividends or unlawful
stock repurchases or redemptions as provided in Section 174 of the Delaware
General Corporation Law; or
|
|
|
|
|
|
|
●
|
transaction from which the directors derived an improper personal benefit.
|
These limitations of liability do not apply to
liabilities arising under the federal or state securities laws and do not affect the availability of equitable remedies such as injunctive
relief or rescission.
Our bylaws provide that we will indemnify our
directors and officers to the fullest extent permitted by law, and may indemnify employees and other agents. Our bylaws also provide that
we are obligated to advance expenses incurred by a director or officer in advance of the final disposition of any action or proceeding.
We have obtained a policy of directors’
and officers’ liability insurance.
We have entered into separate indemnification
agreements with our directors and officers. These agreements, among other things, require us to indemnify our directors and officers for
any and all expenses (including reasonable attorneys’ fees, retainers, court costs, transcript costs, fees of experts, witness fees,
travel expenses, duplicating costs, printing and binding costs, telephone charges, postage, delivery service fees) judgments, fines and
amounts paid in settlement actually and reasonably incurred by such directors or officers or on his or her behalf in connection with any
action or proceeding arising out of their services as one of our directors or officers, or any of our subsidiaries or any other company
or enterprise to which the person provides services at our request provided that such person follows the procedures for determining entitlement
to indemnification and advancement of expenses set forth in the indemnification agreement. We believe that these bylaw provisions and
indemnification agreements are necessary to attract and retain qualified persons as directors and officers.
The limitation of liability and indemnification
provisions in our certificate of incorporation and bylaws may discourage stockholders from bringing a lawsuit against directors for breach
of their fiduciary duties. They may also reduce the likelihood of derivative litigation against directors and officers, even though an
action, if successful, might provide a benefit to us and our stockholders. Our results of operations and financial condition may be harmed
to the extent we pay the costs of settlement and damage awards against directors and officers pursuant to these indemnification provisions.
Insofar as indemnification for liabilities arising
under the Securities Act may be permitted to directors, officers or persons controlling us, we have been informed that, in the opinion
of the SEC, such indemnification is against public policy as expressed in the Securities Act and is therefore unenforceable.
At present, there is no pending litigation or
proceeding involving any of our directors or officers as to which indemnification is required or permitted, and we are not aware of any
threatened litigation or proceeding that may result in a claim for indemnification.
Requirements for Advance Notification of Stockholder
Nominations and Proposals
Our Bylaws establish advance notice procedures
with respect to stockholder proposals and nomination of candidates for election as directors.
Limits on Special Meetings
Special meetings of the stockholders may be called
at any time only by the board of directors, Chairman or our Chief Executive Officer, subject to the rights of the holders of any series
of preferred stock.
Election and Removal of Directors
Directors are elected by a plurality of the votes
of shares present in person or represented by proxy at a meeting and entitled to vote generally on the election of directors. Our stockholders
may remove directors only with the vote of sixty percent (60%) of the stockholders, whether for cause or without cause. Our board of directors
may appoint a director to fill a vacancy, including vacancies created by the expansion of the board of directors. This system of electing
and removing directors may discourage a third party from making a tender offer or otherwise attempting to obtain control of us, because
it generally makes it more difficult for stockholders to replace a majority of our directors. Our certificate of incorporation and bylaws
do not provide for cumulative voting in the election of directors.
Amendments to Our Governing Documents
Generally, the amendment of our certificate of
incorporation requires approval by our board of directors and a majority vote of stockholders. Any amendment to our bylaws requires the
approval of either a majority of our board of directors or approval of at least sixty-six and two-thirds (66 2/3%) of the votes entitled
to be cast by the holders of our outstanding capital stock in elections of our board of directors.
Choice of Forum
Our certificate of incorporation provides
that the Court of Chancery of the State of Delaware will be the exclusive forum for:
|
|
●
|
any derivative action or proceeding brought on our behalf;
|
|
|
|
|
|
|
●
|
any action asserting a breach of
fiduciary duty; any action asserting a claim against us arising pursuant to the Delaware General Corporation Law, our certificate of
incorporation or our bylaws; or
|
|
|
|
|
|
|
●
|
any action asserting a claim against us that is governed by the internal affairs doctrine.
|
The exclusive forum provision does not apply to
suits brought to enforce any liability or duty created by the Securities Act or the Exchange Act or any other claim for which the federal
courts have exclusive jurisdiction. To the extent that any such claims may be based upon federal law claims, Section 27 of the Exchange
Act creates exclusive federal jurisdiction over all suits brought to enforce any duty or liability created by the Exchange Act or the
rules and regulations thereunder. Furthermore, Section 22 of the Securities Act creates concurrent jurisdiction for federal and state
courts over all suits brought to enforce any duty or liability created by the Securities Act or the rules and regulations thereunder.
These exclusive-forum provisions could result in increased costs for
our stockholders’ to bring a claim and may limit a stockholder’s ability to bring a claim in a judicial forum that it finds
favorable for disputes with us or our directors, employees, control persons, underwriters, or agents, which may discourage lawsuits against
us and our directors, employees, control persons, underwriters, or agents. Additionally, a court could determine that the exclusive forum
provision is unenforceable, and our stockholders will not be deemed to have waived our compliance with the federal securities laws and
the rules and regulations thereunder. If a court were to find these provisions of our bylaws inapplicable to, or unenforceable in respect
of, one or more of the specified types of actions or proceedings, we may incur additional costs associated with resolving such matters
in other jurisdictions, which could adversely affect our business, financial condition, or results of operations.
Transfer Agent and
Registrar
The transfer agent and
registrar for our common stock is VStock Transfer, LLC. The transfer agent’s address is 18 Lafayette Place, Woodmere, New York 11598.
Warrant Agent
The warrant agent for the warrants issued in connection
with our initial public offering in July 2018 is VStock Transfer, LLC.
Listing on the Nasdaq
Capital Market
Our common stock is listed
on the Nasdaq Capital Market under the symbol “ADIL.” Our warrants issued in connection with our initial public offering in
July 2018 are currently listed on the Nasdaq Capital Market under the symbol “ADILW.”
Registration Rights
We have filed this registration statement with the SEC that includes
this prospectus to register for resale under the Securities Act the shares of common stock issued and to be issued pursuant to the terms
of the Securities Purchase Agreements to satisfy our obligations in connection with Registration Rights Agreement that we entered into
with the selling stockholders.
PLAN OF DISTRIBUTION
Each selling stockholder of the securities and
any of their pledgees, assignees and successors-in-interest may, from time to time, sell any or all of their securities covered hereby
on the Nasdaq Capital Market or any other stock exchange, market or trading facility on which the securities are traded or in private
transactions. The shares of common stock may be sold in one or more transactions at fixed prices, at prevailing market prices at the time
of the sale, at varying prices determined at the time of sale or at negotiated prices. A selling stockholder may use any one or more of
the following methods when selling securities:
|
|
●
|
ordinary brokerage transactions and transactions in which the broker-dealer solicits purchasers;
|
|
|
●
|
block trades in which the broker-dealer will attempt to sell
the securities as agent but may position and resell a portion of the block as principal to facilitate the transaction;
|
|
|
●
|
purchases by a broker-dealer as principal and resale by the broker-dealer for its account;
|
|
|
●
|
an exchange distribution in accordance with the rules of the applicable exchange;
|
|
|
●
|
privately negotiated transactions;
|
|
|
●
|
settlement of short sales;
|
|
|
●
|
in transactions through
broker-dealers that agree with the selling stockholders to sell a specified number of such securities at a stipulated price per
security;
|
|
|
●
|
through the writing or settlement of options or other hedging transactions, whether through an options exchange or otherwise;
|
|
|
●
|
a combination of any such methods of sale; or
|
|
|
●
|
any other method permitted pursuant to applicable law.
|
The selling stockholders may also sell securities
under Rule 144 or any other exemption from registration under the Securities Act, if available, rather than under this prospectus.
Broker-dealers engaged by the selling stockholders
may arrange for other brokers-dealers to participate in sales. Broker-dealers may receive commissions or discounts from the selling stockholders
(or, if any broker-dealer acts as agent for the purchaser of securities, from the purchaser) in amounts to be negotiated, but except as
set forth in a supplement to this Prospectus, in the case of an agency transaction not in excess of a customary brokerage commission in
compliance with FINRA Rule 2440; and in the case of a principal transaction a markup or markdown in compliance with FINRA IM-2440.
In connection with the sale of the securities
or interests therein, the selling stockholders may enter into hedging transactions with broker-dealers or other financial institutions,
which may in turn engage in short sales of the securities in the course of hedging the positions they assume. The selling stockholders
may also sell securities short and deliver these securities to close out their short positions, or loan or pledge the securities to broker-dealers
that in turn may sell these securities. The selling stockholders may also enter into option or other transactions with broker-dealers
or other financial institutions or create one or more derivative securities which require the delivery to such broker-dealer or other
financial institution of securities offered by this prospectus, which securities such broker-dealer or other financial institution may
resell pursuant to this prospectus (as supplemented or amended to reflect such transaction).
The selling stockholders and any broker-dealers
or agents that are involved in selling the securities may be deemed to be “underwriters” within the meaning of the Securities
Act in connection with such sales. In such event, any commissions received by such broker-dealers or agents and any profit on the resale
of the securities purchased by them may be deemed to be underwriting commissions or discounts under the Securities Act. Each selling stockholder
has informed the Company that it does not have any written or oral agreement or understanding, directly or indirectly, with any person
to distribute the securities.
We are required to pay certain fees and expenses
incurred by us incident to the registration of the shares of common stock. We have agreed to indemnify the selling stockholders against
certain losses, claims, damages and liabilities, including liabilities under the Securities Act.
We agreed to keep this prospectus effective until
the earlier of (i) the date on which the securities may be resold by the selling stockholders without registration and without regard
to any volume or manner-of-sale limitations by reason of Rule 144, without the requirement for the Company to be in compliance with the
current public information under Rule 144 under the Securities Act or any other rule of similar effect or (ii) all of the securities have
been sold pursuant to this prospectus or Rule 144 under the Securities Act or any other rule of similar effect. The resale securities
will be sold only through registered or licensed brokers or dealers if required under applicable state securities laws. In addition, in
certain states, the resale securities covered hereby may not be sold unless they have been registered or qualified for sale in the applicable
state or an exemption from the registration or qualification requirement is available and is complied with.
Under applicable rules and regulations under the
Exchange Act, any person engaged in the distribution of the resale securities may not simultaneously engage in market making activities
with respect to the common stock for the applicable restricted period, as defined in Regulation M, prior to the commencement of the distribution.
In addition, the selling stockholders will be subject to applicable provisions of the Exchange Act and the rules and regulations thereunder,
including Regulation M, which may limit the timing of purchases and sales of the common stock by the selling stockholders or any other
person. We will make copies of this prospectus available to the selling stockholders and have informed them of the need to deliver a copy
of this prospectus to each purchaser at or prior to the time of the sale (including by compliance with Rule 172 under the Securities Act).
LEGAL MATTERS
Unless otherwise indicated in the applicable prospectus
supplement, the validity of the shares of common stock offered hereby will be passed upon for us by Gracin & Marlow, LLP, New York,
New York. If the validity of the securities offered hereby in connection with offerings made pursuant to this prospectus are passed upon
by counsel for the underwriters, dealers or agents, if any, such counsel will be named in the prospectus supplement relating to such offering.
EXPERTS
The financial statements of Adial Pharmaceuticals,
Inc. as of December 31, 2020 and 2019 and for each of the years in the two year period ended December 31, 2020 incorporated by reference
in this Registration Statement have been so included in reliance on the report of Friedman LLP, an independent registered public accounting
firm, (such report includes an explanatory paragraph regarding our ability to continue as a going concern), given on the authority of
said firm as experts in auditing and accounting.
WHERE YOU CAN FIND MORE INFORMATION
We file annual, quarterly and special reports,
proxy statements and other information with the SEC. Our public filings are available to the public at the SEC’s website at www.sec.gov.
Our website address is www.adialpharma.com.
Through our website, we make available, free of charge, the following documents as soon as reasonably practicable after they are electronically
filed with, or furnished to, the SEC: our Annual Reports on Form 10-K; our proxy statements for our annual and special stockholder meetings;
our Quarterly Reports on Form 10-Q; our Current Reports on Form 8-K; Forms 3, 4 and 5 and Schedules 13G and 13D filed on behalf of our
directors and our executive officers; and amendments to those documents. The information contained on, or that may be accessed through,
our website is not part of, and is not incorporated into, this prospectus.
This prospectus is part of a registration statement
on Form S-3 that we have filed with the SEC under the Securities Act. This prospectus does not contain all of the information in the registration
statement. We have omitted certain parts of the registration statement, as permitted by the rules and regulations of the SEC. You may
inspect and copy the registration statement, including exhibits, at the SEC’s website or our website.
INCORPORATION OF CERTAIN DOCUMENTS BY REFERENCE
The SEC allows us to “incorporate by reference”
the information we file with it which means that we can disclose important information to you by referring you to those documents instead
of having to repeat the information in this prospectus. The information incorporated by reference is considered to be part of this prospectus,
and later information that we file with the SEC will automatically update and supersede this information. We incorporate by reference
the documents listed below and any future filings made with the SEC (other than any portions of any such documents that are not deemed
“filed” under the Exchange Act in accordance with the Exchange Act and applicable SEC rules) under Sections 13(a), 13(c),
14 or 15(d) of the Exchange Act including those made after (i) the date of the initial filing of the registration statement of which this
prospectus is a part and prior to the termination of this offering and (ii) the date of this prospectus and before the completion of the
offerings of the shares of our common stock included in this prospectus:
|
|
●
|
Our Annual Report on Form 10-K for the fiscal year ended December 31, 2020 filed with the SEC on March 22, 2021 (File No. 001-38323);
|
|
|
●
|
Our Quarterly report on Form 10-Q for the quarter ended March 31, 2021 filed with the SEC on May 17, 2021 (File No. 001-38323);
|
|
|
●
|
Our Current Reports on Form 8-K filed with the SEC on February 1, 2021, February 12, 2021, February 26, 2021 (other than as indicated therein), March 15, 2021, April 9, 2021, June 4, 2021, June 23, 2021 (other than as indicated therein), June 25, 2021, July 6, 2021 and July 9, 2021 (File No. 001-38323); and
|
|
|
●
|
The
description of our common stock set forth in (i) our registration statements on Form 8-A12B, filed with the SEC on December 11, 2017
and Form 8-A12B/A filed with the SEC on July 23, 2018 (File No. 001-38323) and (ii) Exhibit 4.19—Description of Securities
to our Annual Report on Form 10-K for the fiscal year ended December 31, 2020.
|
Any statement contained in this prospectus or
any prospectus supplement, or in a document incorporated or deemed to be incorporated by reference herein or therein, shall be deemed
to be modified or superseded to the extent that a statement contained herein, or in any subsequent prospectus supplement or in any subsequently
filed document that also is incorporated or deemed to be incorporated by reference herein or therein, modifies or supersedes such statement.
Any statement so modified or superseded shall not be deemed, except as so modified or superseded, to constitute a part of this prospectus
or any prospectus supplement.
You may obtain, free of charge, a copy of any
of these documents (other than exhibits to these documents unless the exhibits are specifically incorporated by reference into these documents
or referred to in this prospectus) from our website (www.adiapharma.com) or by writing or calling us at the following address and
telephone number:
1180 Seminole Trail, Suite 495
Charlottesville VA 22901
Telephone (434) 422-9800
PART II
INFORMATION NOT REQUIRED IN PROSPECTUS
Item 14. Other Expenses of Issuance and Distribution.
The following table sets forth all expenses, other
than the underwriting discounts and commissions, payable by the registrant in connection with the sale of the common stock being registered.
All the amounts shown are estimates except the SEC registration fee.
|
SEC registration fee
|
|
$
|
500
|
|
|
Accounting fees and expenses
|
|
$
|
10,000
|
|
|
Legal fees and expenses
|
|
$
|
25,000
|
|
|
Printing and engraving expenses
|
|
$
|
5,000
|
|
|
Miscellaneous
|
|
$
|
500
|
|
|
Total
|
|
$
|
41,000
|
|
Item 15. Indemnification of Directors and Officers.
The Registrant is incorporated under the laws
of the State of Delaware. Section 145 of the Delaware General Corporation Law provides that a Delaware corporation may indemnify any persons
who were, are, or are threatened to be made, parties to any threatened, pending or completed action, suit or proceeding, whether civil,
criminal, administrative or investigative (other than an action by or in the right of such corporation), by reason of the fact that such
person is or was an officer, director, employee or agent of such corporation, or is or was serving at the request of such corporation
as an officer, director, employee or agent of another corporation or enterprise. The indemnity may include expenses (including attorneys’
fees), judgments, fines and amounts paid in settlement actually and reasonably incurred by such person in connection with such action,
suit or proceeding, provided that such person acted in good faith and in a manner he or she reasonably believed to be in or not opposed
to the corporation’s best interests and, with respect to any criminal action or proceeding, had no reasonable cause to believe that
his or her conduct was illegal. A Delaware corporation may indemnify any persons who were, are, or are threatened to be made, a party
to any threatened, pending or completed action or suit by or in the right of the corporation by reason of the fact that such person is
or was a director, officer, employee or agent of such corporation, or is or was serving at the request of such corporation as a director,
officer, employee or agent of another corporation or enterprise. The indemnity may include expenses (including attorneys’ fees)
actually and reasonably incurred by such person in connection with the defense or settlement of such action or suit provided such person
acted in good faith and in a manner he or she reasonably believed to be in or not opposed to the corporation’s best interests except
that no indemnification is permitted without judicial approval if the officer or director is adjudged to be liable to the corporation.
Where an officer or director is successful on the merits or otherwise in the defense of any action referred to above, the corporation
must indemnify him or her against the expenses (including attorneys’ fees) actually and reasonably incurred.
The Registrant’s certificate of incorporation
and amended and restated bylaws, each of which will become effective immediately prior to the closing of this offering, provide for the
indemnification of its directors and officers to the fullest extent permitted under the Delaware General Corporation Law.
Section 102(b)(7) of the Delaware General Corporation
Law permits a corporation to provide in its certificate of incorporation that a director of the corporation shall not be personally liable
to the corporation or its stockholders for monetary damages for breach of fiduciary duties as a director, except for liability for any:
|
|
●
|
transaction from which the director derives an improper personal benefit;
|
|
|
●
|
act or omission not in good faith or that involves intentional misconduct or a knowing violation of law;
|
|
|
●
|
unlawful payment of dividends or redemption of shares; or
|
|
|
●
|
breach of a director’s duty of loyalty to the corporation or its stockholders.
|
The Registrant’s certificate of incorporation
includes such a provision. Expenses incurred by any officer or director in defending any such action, suit or proceeding in advance of
its final disposition shall be paid by the Registrant upon delivery to it of an undertaking, by or on behalf of such director or officer,
to repay all amounts so advanced if it shall ultimately be determined that such director or officer is not entitled to be indemnified
by the Registrant.
Section 174 of the Delaware General Corporation
Law provides, among other things, that a director who willfully or negligently approves of an unlawful payment of dividends or an unlawful
stock purchase or redemption, may be held liable for such actions. A director who was either absent when the unlawful actions were approved
or dissented at the time may avoid liability by causing his or her dissent to such actions to be entered in the books containing minutes
of the meetings of the board of directors at the time such action occurred or immediately after such absent director receives notice of
the unlawful acts.
As permitted by the Delaware General Corporation
Law, the Registrant has entered into indemnity agreements with each of its directors and executive officers, that require the Registrant
to indemnify such persons against any and all costs and expenses (including attorneys’, witness or other professional fees) actually
and reasonably incurred by such persons in connection with any action, suit or proceeding (including derivative actions), whether actual
or threatened, to which any such person may be made a party by reason of the fact that such person is or was a director or officer or
is or was acting or serving as an officer, director, employee or agent of the Registrant or any of its affiliated enterprises. Under these
agreements, the Registrant is not required to provide indemnification for certain matters, including:
|
|
●
|
indemnification beyond that permitted by the Delaware General Corporation Law;
|
|
|
●
|
indemnification for any proceeding with respect to the unlawful payment of remuneration to the director or officer;
|
|
|
●
|
indemnification for certain proceedings involving a final judgment that the director or officer is required to disgorge profits from the purchase or sale of the Registrant’s stock;
|
|
|
●
|
indemnification for proceedings involving a final judgment that the director’s or officer’s conduct was in bad faith, knowingly fraudulent or deliberately dishonest or constituted willful misconduct or a breach of his or her duty of loyalty, but only to the extent of such specific determination;
|
|
|
●
|
indemnification
for proceedings or claims brought by an officer or director against us or any of the Registrant’s directors, officers, employees
or agents, except for claims to establish a right of indemnification or proceedings or claims approved by the Registrant’s board
of directors or required by law;
|
|
|
●
|
indemnification
for settlements the director or officer enters into without the Registrant’s consent; or
|
|
|
●
|
indemnification
in violation of any undertaking required by the Securities Act or in any registration statement filed by the Registrant.
|
The indemnification agreements also set forth
certain procedures that will apply in the event of a claim for indemnification thereunder.
Except as otherwise disclosed under the heading
“Legal Proceedings” in the “Business” section of this registration statement, there is at present no pending litigation
or proceeding involving any of the Registrant’s directors or executive officers as to which indemnification is required or permitted,
and the Registrant is not aware of any threatened litigation or proceeding that may result in a claim for indemnification.
The Registrant has an insurance policy in place
that covers its officers and directors with respect to certain liabilities, including liabilities arising under the Securities Act or
otherwise.
Item 16. Exhibits.
EXHIBIT INDEX
Exhibit
Number
|
|
Description of Exhibit
|
|
1.1#
|
|
Form of Underwriting Agreement
|
|
3.1
|
|
Certificate of Incorporation of Adial Pharmaceuticals, Inc. (Incorporated by reference to the Company’s Registration Statement on Form S-1, File No. 333-220368, filed with the Securities and Exchange Commission on September 7, 2017)
|
|
3.2
|
|
Bylaws of Adial Pharmaceuticals, Inc. (Incorporated by reference to the Company’s Registration Statement on Form S-1, File No. 333-220368, filed with the Securities and Exchange Commission on September 7, 2017)
|
|
4.1
|
|
Specimen Common Stock Certificate (Incorporated by reference to the Company’s Registration Statement on Form S-1, File No. 333-220368, filed with the Securities and Exchange Commission on October 25, 2017)
|
|
4.2
|
|
Description of Securities (Incorporated by reference to the Company’s Annual Report on Form 10-K, File No. 001-38323 filed with the Securities and Exchange Commission on March 22, 2021)
|
|
10.1
|
|
Form of Stock Purchase Agreement dated July 6, 2021 by and between Adial Pharmaceuticals, Inc. and the selling stockholders (Incorporated by reference to Exhibit 10.1 to the Current Report on Form 8-K File No. 001-38323 filed with the Securities and Exchange Commission on July 9, 2021)
|
|
10.2
|
|
Form of Registration Rights Agreement dated July 6, 2021 by and between Adial Pharmaceuticals, Inc. and the selling stockholders (Incorporated by reference to Exhibit 10.2 to the Current Report on Form 8-K File No. 001-38323 filed with the Securities and Exchange Commission on July 9, 2021)
|
|
5.1*
|
|
Opinion of Gracin & Marlow, LLP
|
|
23.1*
|
|
Consent of Friedman LLP
|
|
23.2*
|
|
Consent of Gracin & Marlow, LLP (See Exhibit 5.1 above)
|
|
24.1*
|
|
Power of Attorney (included on signature page hereto)
|
|
*
|
Filed herewith
|
|
+
|
Management contract or compensatory plan or arrangement required to be identified pursuant to Item 15(a)(3) of this report.
|
|
#
|
To be filed, if necessary, by amendment to this Registration Statement or incorporated by reference from documents filed with the SEC under the Exchange Act in connection with the offering of securities.
|
Item 17. Undertakings.
The undersigned Registrant hereby undertakes:
|
(1)
|
To file, during any period in which offers or sales are being made, a post-effective amendment to this Registration Statement:
|
|
|
(a)
|
to
include any prospectus required by Section 10(a)(3) of the Securities Act;
|
|
|
(b)
|
to
reflect in the prospectus any facts or events arising after the effective date of the registration statement (or the most recent post-effective
amendment thereof) which, individually or in the aggregate, represent a fundamental change in the information set forth in the registration
statement. Notwithstanding the foregoing, any increase or decrease in volume of securities offered and any deviation from the low or
high end of the estimated maximum offering range may be reflected in the form of prospectus filed with the Commission pursuant to
Rule 424(b) if, in the aggregate, the changes in volume and price represent no more than 20 percent change in the maximum aggregate
offering price set forth in the “Calculation of Registration Fee” table in the effective registration statement; and
|
|
|
(c)
|
to
include any material information with respect to the plan of distribution not previously disclosed in the registration statement or any
material change to such information in the registration statement;
|
provided, however, that paragraphs
(1)(a), (1)(b) and (1)(c) do not apply if the information required to be included in a post-effective amendment by those paragraphs
is contained in reports filed with or furnished to the Commission by the Registrant pursuant to Section 13 or Section 15(d)
of the Exchange Act that are incorporated by reference in the registration statement, or is contained in a form of prospectus filed pursuant
to Rule 424(b) that is part of the registration statement.
|
|
(2)
|
That,
for the purpose of determining any liability under the Securities Act, each such post-effective amendment shall be deemed to be a new
registration statement relating to the securities offered therein, and the offering of such securities at that time shall be deemed to
be the initial bona fide offering thereof.
|
|
|
(3)
|
To
remove from registration by means of a post-effective amendment any of the securities being registered which remain unsold at the termination
of the offering.
|
|
|
(4)
|
That,
for the purpose of determining liability under the Securities Act to any purchaser:
|
|
|
(a)
|
Each prospectus filed by a Registrant
pursuant to Rule 424(b)(3) shall be deemed to be part of the registration statement as of the date the filed prospectus was deemed
part of and included in the registration statement; and
|
|
|
(b)
|
Each prospectus required to be filed
pursuant to Rule 424(b)(2), (b)(5) or (b)(7) as part of a registration statement in reliance on Rule 430B relating to an offering
made pursuant to Rule 415(a)(1)(i), (vii) or (x) for the purpose of providing the information required by Section 10(a)
of the Securities Act shall be deemed to be part of and included in the registration statement as of the earlier of the date such
form of prospectus is first used after effectiveness or the date of the first contract of sale of securities in the offering described
in the prospectus. As provided in Rule 430B, for liability purposes of the issuer and any person that is at that date an underwriter,
such date shall be deemed to be a new effective date of the registration statement relating to the securities in the registration statement
to which that prospectus relates, and the offering of such securities at that time shall be deemed to be the initial bona fide offering
thereof. Provided, however, that no statement made in a registration statement or prospectus that is part of the
registration statement or made in a document incorporated or deemed incorporated by reference into the registration statement or prospectus
that is part of the registration statement will, as to a purchaser with a time of contract of sale prior to such effective date, supersede
or modify any statement that was made in the registration statement or prospectus that was part of the registration statement or made
in any such document immediately prior to such effective date.
|
|
|
|
|
|
|
(c)
|
That, for purposes of determining any
liability under the Securities Act, each filing of Registrant’s annual report pursuant to Section 13(a) or 15(d) of the Exchange
Act (and, where applicable, each filing of an employee benefit plan’s annual report pursuant to Section 15(d) of the Exchange
Act) that is incorporated by reference in the registration statement shall be deemed to be a new registration statement relating to the
securities offered therein, and the offering of such securities at that time shall be deemed to be the initial bona fide offering
thereof.
|
|
|
|
|
|
|
(d)
|
Insofar as indemnification for liabilities arising under the Securities Act may be permitted to directors, officers and controlling persons of the Registrant pursuant to the foregoing provisions, or otherwise, the Registrant has been advised that in the opinion of the SEC such indemnification is against public policy as expressed in the Securities Act and is, therefore, unenforceable. In the event that a claim for indemnification against such liabilities (other than the payment by a Registrant of expenses incurred or paid by a director, officer or controlling person of a Registrant in the successful defense of any action, suit or proceeding) is asserted by such director, officer or controlling person in connection with the securities being registered, that Registrant will, unless in the opinion of its counsel the matter has been settled by controlling precedent, submit to a court of appropriate jurisdiction the question whether such indemnification by it is against public policy as expressed in the Securities Act and will be governed by the final adjudication of such issue.
|
|
|
(5)
|
That,
for the purpose of determining liability of the registrant under the Securities Act of 1933 to any purchaser in the initial distribution
of the securities, the registrant undertakes that in a primary offering of securities of the registrant pursuant to this registration
statement, regardless of the underwriting method used to sell the securities to the purchaser, if the securities are offered or sold
to such purchaser by means of any of the following communications, the registrant will be a seller to the purchaser and will be considered
to offer or sell such securities to such purchaser:
|
|
|
(a)
|
Any preliminary prospectus or prospectus of the registrant relating to the offering required to be filed pursuant to Rule 424;
|
|
|
|
|
|
|
(b)
|
Any free writing prospectus relating to the offering prepared by or on behalf of the registrant or used or referred to by the registrant;
|
|
|
|
|
|
|
(c)
|
The portion of any other free writing prospectus relating to the offering containing material information about registrant or its securities provided by or on behalf of the registrant; and
|
|
|
|
|
|
|
(d)
|
Any other communication that is an offer in the offering made by a registrant to the purchaser.
|
SIGNATURES
Pursuant to the requirements of the Securities Act of 1933, as amended,
the Registrant certifies that it has reasonable grounds to believe that it meets all of the requirements for filing on Form S-3 and has
duly caused this registration statement to be signed on its behalf by the undersigned, thereunto duly authorized, in the City of Charlottesville,
State of Virginia, on the 20th day of July, 2021.
|
|
ADIAL PHARMACEUTICALS, INC.
|
|
|
|
|
|
|
By:
|
/s/ William B. Stilley
|
|
|
Name:
|
William B. Stilley
|
|
|
Title:
|
President and Chief Executive Officer
|
POWER OF ATTORNEY
Each of the undersigned, whose signature appears
below, hereby constitutes and appoints William B. Stilley and Joseph M. Truluck, and each of them, his true and lawful attorney-in-fact
and agent, with full power of substitution and resubstitution, for him and in his name, place and stead, in any and all capacities, to
sign any and all amendments to this Registration Statement, whether pre-effective or post-effective, and any and all additional registration
statements pursuant to Rule 462(b) under the Securities Act of 1933, and to file the same with all exhibits thereto and other documents
in connection therewith, with the Securities and Exchange Commission, granting unto said attorneys-in-fact and agents, or his or their
substitute or substitutes, full power and authority to do and perform each and every act and thing necessary or appropriate to be done
with respect to this Registration Statement or any amendments hereto in the premises, as fully to all intents and purposes as he might
or could do in person, hereby ratifying and confirming all that said attorneys-in-fact and agents, or his or their substitute or substitutes,
or any of them, may lawfully do or cause to be done by virtue thereof.
Pursuant to the requirements of the Securities
Act of 1933, as amended, this registration statement has been signed by the following persons in the capacities and on the dates indicated.
|
Signature
|
|
Title
|
|
Date
|
|
|
|
|
|
|
|
/s/
William B. Stilley
|
|
Chief Executive Officer
and President
|
|
|
|
William B. Stilley
|
|
(Principal Executive Officer)
|
|
July
20, 2021
|
|
|
|
|
|
|
|
/s/
Joseph M. Truluck
|
|
Chief Operating Officer
and Chief Financial Officer
|
|
|
|
Joseph M. Truluck
|
|
(Principal Financial and
Accounting Officer)
|
|
July
20, 2021
|
|
|
|
|
|
|
|
/s/
J. Kermit Anderson
|
|
|
|
|
|
J. Kermit Anderson
|
|
Member of the Board of
Directors
|
|
July
20, 2021
|
|
|
|
|
|
|
|
/s/
Robertson H. Gilliland
|
|
|
|
|
|
Robertson H. Gilliland
|
|
Member of the Board of
Directors
|
|
July
20, 2021
|
|
|
|
|
|
|
|
/s/
Tony Goodman
|
|
|
|
|
|
Tony Goodman
|
|
Member of the Board of
Directors
|
|
July
20, 2021
|
|
|
|
|
|
|
|
/s/
James W. Newman, Jr.
|
|
|
|
|
|
James W. Newman, Jr.
|
|
Member of the Board of
Directors
|
|
July
20, 2021
|
|
|
|
|
|
|
|
/s/
Kevin Schuyler, CFA
|
|
|
|
|
|
Kevin Schuyler, CFA
|
|
Member of the Board of
Directors
|
|
July
20, 2021
|
II-6
Adial Pharmaceuticals (NASDAQ:ADIL)
Historical Stock Chart
From Aug 2024 to Sep 2024
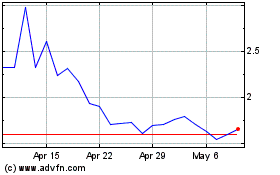
Adial Pharmaceuticals (NASDAQ:ADIL)
Historical Stock Chart
From Sep 2023 to Sep 2024