CareDx Showcases Digital Solutions and Pipeline in Cellular Transplant and Therapy Monitoring at the 2024 Tandem Meetings
February 21 2024 - 7:00AM
Business Wire
CareDx, Inc. (Nasdaq: CDNA) —The Transplant Company™ — a leading
precision medicine company focused on the discovery, development,
and commercialization of clinically differentiated, high-value
healthcare solutions for transplant patients and caregivers — today
announced that it will be showcasing the breadth of its specialized
digital solutions and the latest advancements in its cellular
therapy and transplant monitoring pipeline at the 2024 Tandem
Meetings, Transplantation & Cellular Therapy Meetings of ASTCT™
and CIBMTR®, being held February 21-25 in San Antonio, Texas.
“Cellular transplant centers are recognizing the value of our
digital and patient monitoring solutions, and we look forward to
sharing our latest advancements as we work to expand our presence
in this dynamic ecosystem,” said Kashif Rathore, Chief of Patient
and Digital Solutions at CareDx.
CareDx Digital Product Theatre
CareDx will be showcasing its broad portfolio of digital health
solutions that address needs in the cellular transplant and therapy
ecosystem including workflow management, regulatory reporting,
medication management, and remote patient monitoring. Product
solution areas include:
- Ottr® Cellular: Designed as a workflow management
solution for hematopoietic cellular transplant and cellular therapy
centers.
- MedActionPlan PRO®: Designed to improve
medication adherence and simplify medication regimens.
- AlloHome® remote patient monitoring: Designed for
patient engagement and timely interventions.
- AlloCare® patient mobile health app: Helps
patients track personal health metrics and test results and manage
their complex medication regimens.
CareDx is advancing its product pipeline with new data for
AlloHeme™ and AlloCell™ that can help monitor treatment response
after allogeneic hematopoietic stem cell transplantation and
allogeneic cell therapy, respectively.
Data being presented:
- Oral Presentation: TScan’s TSC-100 and TSC-101, TCR-T
Cell Therapies That Target Residual Recipient Cells after Reduced
Intensity Conditioning Transplantation, Induce Complete Donor
Chimerism with Favorable Prognosis: Early Results of a Phase 1
Trial. In this TScan study AlloHeme is used as an exploratory
marker in subjects with mismatched HA1 and HA2 minor HLA antigens,
and respective controls.
- Poster 555: Micro-Chimerism Detected By Highly Sensitive
AlloHeme™ Solution Is Associated with Treatment Response and
Relapse after Allogeneic Stem Cell Transplantation. Presenting
interim data from the prospective multi-center ACROBAT study that
supports the clinical utility of ultra-sensitive chimerism
monitoring for detection of early relapse post allogeneic HCT.
- Poster 299: Analytical Validation of AlloCell cfDNA - a
Highly Sensitive, Precise, and Accurate Cell-Free DNA-Based Test
for Allogeneic Cell Therapy Monitoring.
- Poster 645: Regimen Adherence Insights after Allogeneic
Hematopoietic Stem Cell Transplantation from a Central Specialty
Pharmacy.
“We’re thrilled to present interim study findings from the
ACROBAT study, revealing AlloHeme is able to more accurately
measure chimerism levels with much higher sensitivity than the
current methods for patients with AML, ALL, or MDS undergoing
allogeneic hematopoietic cell therapy,” said Dr. Stefan Ciurea of
the University of California, Irvine. “The ACROBAT study aims to
assess the role of AlloHeme for early relapse detection,
potentially paving the way for improved therapeutic interventions
to prevent disease relapse post-transplant and improved treatment
outcomes.”
About CareDx – The Transplant Company
CareDx, Inc., headquartered in Brisbane, California, is a
leading precision medicine solutions company focused on the
discovery, development, and commercialization of clinically
differentiated, high-value healthcare solutions for transplant
patients and caregivers. CareDx offers testing services, products,
and digital healthcare solutions along the pre- and post-transplant
patient journey and is the leading provider of genomics-based
information for transplant patients. For more information, please
visit:www.CareDx.com.
Forward Looking Statements
This press release includes forward-looking statements related
to CareDx, Inc., including statements regarding the potential
benefits and results that may be achieved with AlloHeme™,
AlloCell™, CareDx’s digital health solutions, and CareDx’s
presentation of its latest pipeline advancements in cellular
transplant and therapy monitoring at the 2024 Tandem Meetings (the
“Tandem Presentation”). These forward-looking statements are based
upon information that is currently available to CareDx and its
current expectations, speak only as of the date hereof, and are
subject to risks and uncertainties that could cause actual results
to differ materially from those projected, including risks that
CareDx does not realize the expected benefits of AlloHeme,
AlloCell, its digital health solutions or its Tandem Presentation;
risks that the Tandem Meetings and the data to be presented may not
follow the agenda as stated in this press release; risks that the
findings in the studies supporting the data may be inaccurate,
general economic and market factors; and other risks discussed in
CareDx’s filings with the SEC, including the Annual Report on Form
10-K for the fiscal year ended December 31, 2022 filed by CareDx
with the SEC on February 27, 2023, the quarterly report on Form
10-Q for the quarter ended March 31, 2023 filed by CareDx with the
SEC on May 10, 2023, the quarterly report on Form 10-Q for the
quarter ended June 30, 2023 filed by CareDx with the SEC on August
8, 2023, the quarterly report on Form 10-Q for the quarter ended
September 30, 2023 filed by CareDx with the SEC on November 8,
2023, and other reports that CareDx has filed with the SEC. Any of
these may cause CareDx’s actual results, performance, or
achievements to differ materially and adversely from those
anticipated or implied by CareDx’s forward-looking statements.
CareDx expressly disclaims any obligation, except as required by
law, or undertaking to update or revise any such forward-looking
statements.
View source
version on businesswire.com: https://www.businesswire.com/news/home/20240221575484/en/
CareDx, Inc. Media Relations Anna Czene 818-731-2203
aczene@caredx.com
Investor Relations Greg Chodaczek investor@caredx.com
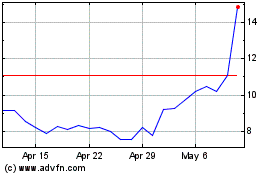
CareDx (NASDAQ:CDNA)
Historical Stock Chart
From Mar 2024 to Apr 2024
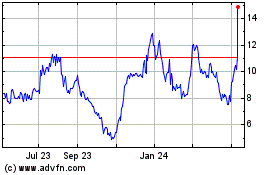
CareDx (NASDAQ:CDNA)
Historical Stock Chart
From Apr 2023 to Apr 2024