FSD Pharma Announces Dismissal of Derivative Complaint
May 06 2022 - 10:41AM
Business Wire
FSD Pharma Inc. (NASDAQ: HUGE) (CSE: HUGE) (FRA: 0K9) (“FSD
Pharma” or the “Company”), a life sciences holding
company dedicated to building a portfolio of assets and biotech
solutions, announced today that the Company was successful in its
motion to dismiss the derivative claim that was filed by Mr. Maheep
Goyal, a shareholder (the “Plaintiff”) on July 20, 2021 in
Delaware against the Company and its directors and officers (the
“Claim”). In a decision dated May 5, 2022, the Delaware
court dismissed the Claim without prejudice on the grounds that Mr.
Maheep Goyal, the Plaintiff, lacked standing to bring his
claims.
“We have always believed that the lawsuits against the Company
are frivolous in nature. We are pleased to hear that the courts of
Delaware have dealt with this particular claim appropriately and
are looking forward to the resolution of the Company’s remaining
litigation,” said Anthony Durkacz, Interim CEO and Co-Executive
Chairman of the Company.
The dismissal of the Claim marks another claim against the
Company that has been successfully dismissed, following the
dismissal of the joint claim filed by Mr. Edward Brennan Jr. and
Huma Qamar two former FSD BioSciences employees in Pennsylvania on
December 13, 2021.
About FSD Pharma
FSD Pharma Inc. is a biotechnology company with three drug
candidates in different stages of development. FSD BioSciences,
Inc. (“FSD BioSciences”), a wholly owned subsidiary, is
focused on pharmaceutical research and development of its lead
compound, ultra-micronized palmitoyl ethylamine (“PEA”) or
FSD-PEA (formerly called FSD-201). Lucid Psycheceuticals Inc.
(“Lucid”), a wholly owned subsidiary, is focused on the
research and development of its lead compounds, Lucid-PSYCH
(formerly Lucid-201) and Lucid-MS (formerly Lucid-21-302). Lucid
PSYCH is a molecular compound identified for the potential
treatment of mental health disorders. Lucid-MS is a molecular
compound identified for the potential treatment of
neurodegenerative disorders.
Forward Looking Information
Certain statements contained herein are “forward-looking
statements.” Often, but not always, forward-looking statement can
be identified by the use of words such as “plans,” “expects,”
“expected,” “scheduled,” “estimates,” “intends,” “anticipates,”
“hopes,” “planned” or “believes,” or variations of such words and
phrases, or states that certain actions, events or results “may,”
“could,” “would,” “might,” “potentially” or “will” be taken, occur
or be achieved. Forward-looking statements contained in this press
release include the comments made with respect to the resolution of
the Company’s remaining litigation. FSD cannot give any assurance
that such forward-looking statements will prove to have been
correct. The reader is cautioned not to place undue reliance on
these forward-looking statements, which speak only as of the date
of this press release.
Since forward-looking statements relate to future events and
conditions, by their very nature they require making assumptions
and involve inherent risks and uncertainties. The Company cautions
that although it is believed that the assumptions are reasonable in
the circumstances, these risks and uncertainties give rise to the
possibility that actual results may differ materially from the
expectations set out in the forward-looking statements. Factors
that may cause such material differences include without
limitation: the fact that the drug development efforts of both
Lucid and FSD BioSciences are at a very early stage; the fact that
preclinical drug development is uncertain, and the drug product
candidates of Lucid and FSD BioSciences may never advance to
clinical trials; the fact that results of preclinical studies and
early-stage clinical trials may not be predictive of the results of
later stage clinical trials; the uncertain outcome, cost, and
timing of product development activities, preclinical studies and
clinical trials of Lucid and FSD BioSciences; the uncertain
clinical development process, including the risk that clinical
trials may not have an effective design or generate positive
results; the potential inability to obtain or maintain regulatory
approval of the drug product candidates of Lucid and FSD
BioSciences; the introduction of competing drugs that are safer,
more effective or less expensive than, or otherwise superior to,
the drug product candidates of Lucid and FSD BioSciences; the
initiation, conduct, and completion of preclinical studies and
clinical trials may be delayed, adversely affected, or impacted by
COVID-19 related issues; the potential inability to obtain adequate
financing; the potential inability to obtain or maintain
intellectual property protection for the drug product candidates of
Lucid and FSD BioSciences; and other risks. Further information
regarding factors that may cause actual results to differ
materially are included in the Company’s annual and other reports
filed from time to time with the Canadian Securities Administrators
on SEDAR (www.sedar.com) and with the U.S. Securities and Exchange
Commission on EDGAR (www.sec.gov) under the heading “Risk Factors.”
Any forward-looking statement contained in this release speaks only
as of its date. The Company does not undertake to update any
forward-looking statements, except to the extent required by
applicable securities laws.
View source
version on businesswire.com: https://www.businesswire.com/news/home/20220506005361/en/
Zeeshan Saeed, Founder, President and Executive Co-Chairman of
the Board, FSD Pharma Inc. Email: Zsaeed@fsdpharma.com Telephone:
(416) 854-8884
Investor Relations: Email: ir@fsdpharma.com, info@fsdpharma.com
Website: www.fsdpharma.com
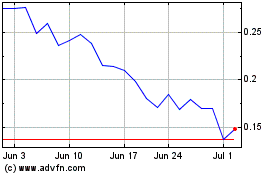
FSD Pharma (NASDAQ:HUGE)
Historical Stock Chart
From Aug 2024 to Sep 2024
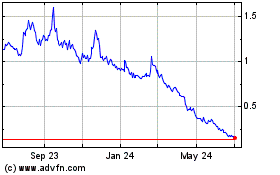
FSD Pharma (NASDAQ:HUGE)
Historical Stock Chart
From Sep 2023 to Sep 2024