Ironwood Pharmaceuticals Announces Adjustment of Conversion Rate for its 2.25% Convertible Senior Notes due 2020
April 16 2019 - 4:02PM
Business Wire
Ironwood Pharmaceuticals, Inc. (NASDAQ: IRWD), a GI-focused
healthcare company, today announced an adjustment to the conversion
rate applicable to its existing 2.25% Convertible Senior Notes due
2022, effective April 15, 2019. The adjustment was effected
pursuant to the indenture, dated as of June 15, 2015, and as
supplemented April 5, 2019, between Ironwood and U.S. Bank National
Association, as trustee, pursuant to which the Convertible Notes
were originally issued.
In connection with the separation of Ironwood and Cyclerion
Therapeutics, Inc. (Cyclerion) into two independent, publicly
traded companies, on April 1, 2019, Ironwood distributed to its
shareholders one share of Cyclerion common stock for every 10
shares of Ironwood common stock held as of the close of business on
March 19, 2019, the record date for the distribution.
As a result of the separation, and pursuant to Section 4.05(c)
of the indenture, effective April 15, 2019, the conversion rate
under the indenture has been adjusted to equal 68.9172 shares of
Ironwood Common Stock per $1,000 principal amount of the
Convertible Notes.
About Ironwood Pharmaceuticals
Ironwood Pharmaceuticals (Nasdaq: IRWD) is a GI-focused
healthcare company focused on creating medicines that make a
difference for patients living with GI diseases. We discovered,
developed and are commercializing linaclotide, the U.S. branded
prescription market leader for adults with irritable bowel syndrome
with constipation (IBS-C) or chronic idiopathic constipation (CIC).
We are currently advancing a Phase IIIb trial evaluating the
efficacy and safety of linaclotide on multiple abdominal symptoms,
including bloating, pain, and discomfort, in adult patients with
IBS-C.
We are also advancing two late-stage, first-in-category GI
product candidates: IW-3718 is a gastric retentive formulation of a
bile acid sequestrant being developed for the potential treatment
of persistent gastroesophageal reflux disease, and MD-7246 is a
delayed-release formulation of linaclotide that is being evaluated
as an oral, intestinal, non-opioid, pain-relieving agent for
patients suffering from abdominal pain associated with IBS with
diarrhea.
Ironwood was founded in 1998 and is headquartered in Cambridge,
Mass. For more information, please visit our newly launched website
at www.ironwoodpharma.com or www.twitter.com/ironwoodpharma;
information that may be important to investors will be routinely
posted in both these locations.
View source
version on businesswire.com: https://www.businesswire.com/news/home/20190416005884/en/
Media and Investors:Meredith Kaya, 617-374-5082Vice
President, Investor Relations and Corporate
Communicationsmkaya@ironwoodpharma.com
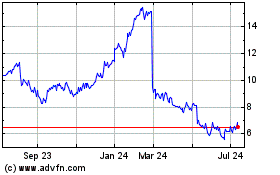
Ironwood Pharmaceuticals (NASDAQ:IRWD)
Historical Stock Chart
From Aug 2024 to Sep 2024
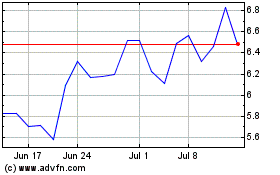
Ironwood Pharmaceuticals (NASDAQ:IRWD)
Historical Stock Chart
From Sep 2023 to Sep 2024