EU Drug Regulator Recommends Bayer Kidney Treatment for Marketing Authorization
December 17 2021 - 8:14AM
Dow Jones News
By Ed Frankl
Bayer AG said Friday that European drug regulators recommended
its finerenone kidney-disease treatment for marketing authorization
in the EU.
The German pharmaceutical company said the Committee for
Medicinal Products for Human Use, a branch of the European
Medicines Agency, gave a positive opinion for finerenone as a new
treatment of chronic kidney disease associated with type 2 diabetes
in adults.
"The positive CHMP opinion for finerenone brings us closer to
providing this new treatment option to patients with chronic kidney
disease associated with type 2 diabetes," said Christian Rommel,
Bayer's head of pharmaceuticals research and development.
The regulatory opinion is based on the results of a Phase 3
study investigating the efficacy and safety of finerenone on kidney
and cardiovascular outcomes in more than 13,000 patients.
Finerenone was approved by the U.S. Food and Drug Administration
in July 2021 under the brand name Kerendia.
Write to Ed Frankl at edward.frankl@dowjones.com
(END) Dow Jones Newswires
December 17, 2021 07:59 ET (12:59 GMT)
Copyright (c) 2021 Dow Jones & Company, Inc.
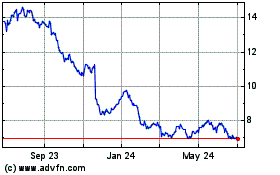
Bayer Aktiengesellschaft (PK) (USOTC:BAYRY)
Historical Stock Chart
From Aug 2024 to Sep 2024
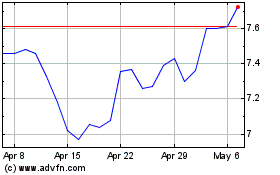
Bayer Aktiengesellschaft (PK) (USOTC:BAYRY)
Historical Stock Chart
From Sep 2023 to Sep 2024