Merck Gets FDA Approval for Verquvo
January 20 2021 - 7:48AM
Dow Jones News
By Robb M. Stewart
Merck & Co. said Wednesday it received approval from U.S.
regulators for Verquvo, a treatment to reduce the risk of
cardiovascular death and hospitalization from heart failure.
The Food and Drug Administration's okay for Verquvo
(vericiguat), a soluble guanylate cyclase stimulator that Merck
said is the first treatment for chronic heart failure approved
specifically for patients following a hospitalization for heart
failure or need for outpatient IV diuretics, was based on the
results of a late-stage trial and followed a priority regulatory
review.
Verquvo, in 2.5 milligram, 5 mg and 10 mg tablets, is being
jointly developed with Bayer AG.
The approval was based on data from a randomized,
parallel-group, placebo-controlled, double-blind, event-driven,
multi-center clinical trial comparing Verquvo to placebo in 5,050
adult patients with symptomatic chronic heart failure and left
ventricular ejection fraction less than 45%, following a worsening
heart failure event, the company said. It said Verquvo was superior
to placebo in reducing the risk of cardiovascular death or heart
failure hospitalization based on a time-to-event analysis.
Merck has the commercial rights to vericiguat in the U.S. and
Bayer has the exclusive commercial rights in the rest of world. The
companies share equally the costs of the development of
vericiguat.
Write to Robb M. Stewart at robb.stewart@wsj.com
(END) Dow Jones Newswires
January 20, 2021 07:33 ET (12:33 GMT)
Copyright (c) 2021 Dow Jones & Company, Inc.
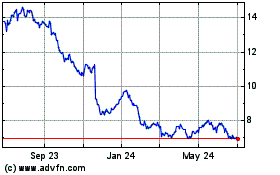
Bayer Aktiengesellschaft (PK) (USOTC:BAYRY)
Historical Stock Chart
From Apr 2024 to May 2024
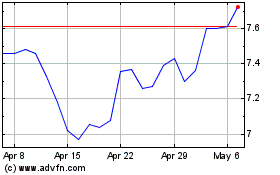
Bayer Aktiengesellschaft (PK) (USOTC:BAYRY)
Historical Stock Chart
From May 2023 to May 2024