Tenax Therapeutics Schedules Conference Call to Discuss Its Transformative Acquisition of PH Precision Med
January 20 2021 - 7:45AM
Business Wire
-Conference to be held at 4:30 pm EST on
January 21, 2021-
Tenax Therapeutics, Inc. (Nasdaq: TENX), a specialty
pharmaceutical company focused on identifying, developing and
commercializing products for the critical care market, today
announced that it will host an investor conference call for
analysts and investors on Thursday, January 21, 2021 at 4:30 p.m.
EST. On the call, management will review the previously announced
acquisition of PH Precision Med (PHPM), a privately-held clinical
stage biotech company focused on developing imatinib for the
treatment of pulmonary arterial hypertension (PAH), and the
clinical development plan to advance a delayed release oral
formulation of imatinib, designed to avoid the gastric irritation,
into a single pivotal trial pursuant to the 505(b)(2) pathway.
A live webcast of the conference call and accompanying slides
will be available under “Events and Presentations” in the Investors
section of the Company’s website at investors.tenaxthera.com. The
conference call can be accessed by dialing (877) 405-1242 from the
U.S. and (201) 389-0852 internationally, followed by the conference
ID: 13715313. An archived webinar recording of the event will be
available on the website for approximately 30 days.
About Tenax Therapeutics
Tenax Therapeutics, Inc., is a specialty pharmaceutical company
focused on identifying, developing, and commercializing products
that address cardiovascular and pulmonary diseases with high unmet
medical need. The Company has a world-class scientific advisory
team including recognized global experts in pulmonary hypertension.
The Company owns North American rights to develop and commercialize
levosimendan and has recently released topline data regarding their
Phase 2 clinical trial for the use of levosimendan in the treatment
of Pulmonary Hypertension associated with Heart Failure and
preserved Ejection Fraction (PH-HFpEF). Tenax plans to advance a
delayed release oral formulation of imatinib, designed to avoid the
gastric irritation, into a single pivotal trial pursuant to the
505(b)(2) pathway. For more information, visit
www.tenaxthera.com.
About Levosimendan
Levosimendan is a calcium sensitizer that works through a unique
triple mechanism of action. It initially was developed for
intravenous use in hospitalized patients with acutely decompensated
heart failure. It was discovered and developed by Orion Pharma,
Orion Corporation of Espoo Finland, and is currently approved in
over 60 countries for this indication and not available in the
United States. Tenax Therapeutics acquired North American rights to
develop and commercialize levosimendan from Phyxius Pharma,
Inc.
About Imatinib
Imatinib is an antiproliferative agent developed to target the
BCR-ABL tyrosine kinase in patients with chronic myeloid leukemia.
The inhibitory effects of imatinib on PDGF receptors and c-KIT
suggested that it may be efficacious in PAH. Imatinib reversed
experimentally induced pulmonary hypertension and has pulmonary
vasodilatory effects in animal models and proapoptotic effects on
pulmonary artery smooth muscle cells from patients with idiopathic
PAH. In a phase 3 clinical trial imatinib produced significant
improvements in exercise capacity, but a high rate of dropouts
attributed largely to gastric intolerance prevented regulatory
approval.
Caution Regarding Forward-Looking Statements
This news release contains certain forward-looking statements by
the Company that involve risks and uncertainties and reflect the
Company’s judgment as of the date of this release. The
forward-looking statements are subject to a number of risks and
uncertainties, including, but not limited to matters beyond the
Company’s control that could lead to delays in the clinical study,
new product introductions and customer acceptance of these new
products; matters beyond the Company’s control that could impact
the Company’s continued compliance with Nasdaq listing
requirements; the impact of management changes on the Company’s
business and unanticipated charges, costs and expenditures not
currently contemplated that may occur as a result of management
changes; and other risks and uncertainties as described in the
Company’s filings with the Securities and Exchange Commission,
including in its annual report on Form 10-K filed on March 30,
2020, and its quarterly report on Form 10-Q filed on November 16,
2020, as well as its other filings with the SEC. The Company
disclaims any intent or obligation to update these forward-looking
statements beyond the date of this release. Statements in this
press release regarding management’s future expectations, beliefs,
goals, plans or prospects constitute forward-looking statements
within the meaning of the Private Securities Litigation Reform Act
of 1995.
View source
version on businesswire.com: https://www.businesswire.com/news/home/20210120005056/en/
Investor Contact: Westwicke, an ICR Company Stephanie
Carrington, 646-277-1282 stephanie.carrington@westwicke.com
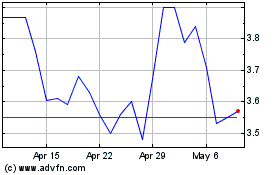
Tenax Therapeutics (NASDAQ:TENX)
Historical Stock Chart
From Aug 2024 to Sep 2024
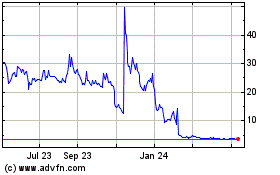
Tenax Therapeutics (NASDAQ:TENX)
Historical Stock Chart
From Sep 2023 to Sep 2024