As
filed with the Securities and Exchange Commission on August 28, 2023
Registration
Statement No. 333-
UNITED
STATES
SECURITIES
AND EXCHANGE COMMISSION
Washington,
D.C. 20549
FORM
F-1
REGISTRATION
STATEMENT Under The Securities Act of 1933
TC
BIOPHARM (HOLDINGS) PLC
(Exact
name of Registrant as specified in its charter)
| Scotland |
|
8731 |
|
Not
applicable |
(State
or other jurisdiction of
incorporation
or organization) |
|
(Primary
Standard Industrial
Classification
Code Number) |
|
(IRS
Employer
Identification
Number) |
Maxim
1, 2 Parklands Way
Holytown,
Motherwell, ML1 4WR
Scotland,
United Kingdom
+44
(0) 141 433 7557
(Address,
including zip code, and telephone number, including area code, of registrant’s principal executive offices)
TC
BioPharm (North America) Inc.
c/o
Business Filings, Inc.
108
West 13th Street
Wilmington,
Delaware 19801
(800)
981-7183
(Name,
address, including zip code, and telephone number, including area code, of agent for service)
Copy
of all communications including communications sent to agent for service, should be sent to:
Richard
A. Friedman, Esq.
Stephen
Cohen, Esq.
Sheppard,
Mullin, Richter & Hampton LLP
30
Rockefeller Plaza
New
York, NY 10112
Telephone:
(212) 653-8700
Facsimile:
(212) 653-8701 |
|
|
Approximate
date of commencement of proposed sale to the public: As soon as practicable after this registration statement becomes effective.
If
any of the securities being registered on this Form are to be offered on a delayed or continuous basis pursuant to Rule 415 under the
Securities Act of 1933 (as amended, the “Securities Act”), check the following box. ☒
If
this Form is filed to register additional securities for an offering pursuant to Rule 462(b) under the Securities Act, check the following
box and list the Securities Act registration statement number of the earlier effective registration statement for the same offering.
☐
If
this Form is a post-effective amendment filed pursuant to Rule 462(c) under the Securities Act, check the following box and list the
Securities Act registration statement number of the earlier effective registration statement for the same offering. ☐
If
this Form is a post-effective amendment filed pursuant to Rule 462(d) under the Securities Act, check the following box and list the
Securities Act registration statement number of the earlier effective registration statement for the same offering. ☐
Indicate
by check mark whether the registrant is an emerging growth company as defined in Rule 405 of the Securities Act.
Emerging
growth company ☒
If
an emerging growth company that prepares its financial statements in accordance with U.S. GAAP, indicate by check mark if the registrant
has elected not to use the extended transition period for complying with any new or revised financial accounting standards† provided
pursuant to Section 7(a)(2)(B) of the Securities Act. ☐
| † |
The
term “new or revised financial accounting standard” refers to any update issued by the Financial Accounting Standards
Board to its Accounting Standards Codification after April 5, 2012. |
The
Registrant hereby amends this registration statement on such date or dates as may be necessary to delay its effective date until the
Registrant shall file a further amendment which specifically states that this registration statement shall thereafter become effective
in accordance with Section 8(a) of the Securities Act, or until this registration statement shall become effective on such date as the
Securities and Exchange Commission, acting pursuant to said Section 8(a) may determine.
The
information in this preliminary prospectus is not complete and may be changed. These securities may not be sold until the registration
statement filed with the Securities and Exchange Commission is effective. This preliminary prospectus is not an offer to sell nor does
it seek an offer to buy these securities in any jurisdiction where the offer or sale is not permitted.
| PRELIMINARY
PROSPECTUS |
|
SUBJECT
TO COMPLETION |
|
DATED
AUGUST 28, 2023 |
UP TO 9,900,990 ORDINARY SHARES
AND UP TO 9,900,990 PRE-FUNDED WARRANTS TO PURCHASE
UP TO
9,900,990 AMERICAN DEPOSITARY SHARES
AND PLACEMENT AGENT WARRANTS TO PURCHASE UP
TO 742,574 AMERICAN DEPOSITARY SHARES
(and 9,900,990 American Depositary Shares underlying the Pre-Funded Warrants
and 742,574 American Depositary Shares underlying the Placement Agent Warrants)
TC
BIOPHARM (HOLDINGS) PLC

We
are offering 9,900,990 American depositary shares, or ADSs representing ordinary shares, par value $0.0001 per share. The assumed
public offering price for each ADS is $0.505, which is based upon the closing price of our ADSs on The Nasdaq Capital Market
on August 22, 2023.
We are also offering
to certain purchasers whose purchase of ADSs in this offering would otherwise result in the purchaser, together with its affiliates and
certain related parties, beneficially owning more than 4.99% (or, at the election of the purchaser, 9.99%) of our outstanding ADSs immediately
following the consummation of this offering, the opportunity to purchase, if any such purchaser so chooses, pre-funded warrants, in lieu
of ADSs that would otherwise result in such purchaser’s beneficial ownership exceeding 4.99% (or, at the election of the purchaser,
9.99%) of our ADSs. The public offering price of each pre-funded warrant will be equal to the price at which an ADS is sold to the public
in this offering, minus $0.001, and the exercise price of each pre-funded warrant will be $0.001 per ADS. The pre-funded warrants will
be immediately exercisable and may be exercised at any time until all of the pre-funded warrants are exercised in full. For each pre-funded
warrant we sell, the number of ADSs we are offering will be decreased on a one-for-one basis. The ADSs and pre-funded warrants can only
be purchased together in this offering but will be issued separately and will be immediately separable upon issuance.
There is no established
public trading market for the Warrants and pre-funded warrants, and we do not expect a market to develop. We do not intend to apply for
listing of the Warrants and pre-funded warrants on any securities exchange or other nationally recognized trading system. Without an
active trading market, the liquidity of the warrants will be limited.
This
offering will terminate on [*], 2023, unless we decide to terminate the offering (which we may do at any time in our discretion) prior
to that date. We will have one closing for all the securities purchased in this offering.
Our
ADSs are listed on the Nasdaq Capital Market, or Nasdaq, under the symbol “TCBP”. On August 22, 2023, the closing
trading price for our ADSs, as reported on Nasdaq, was $0.505 per ADS. The recent market price used throughout this prospectus
may not be indicative of the final offering price. The final public offering price will be determined through negotiation between us
and investors based upon a number of factors, including our history and our prospects, the industry in which we operate, our past and
present operating results, the previous experience of our executive officers and the general condition of the securities markets at the
time of this offering.
We
have engaged [*], or the Placement Agent, to act as our exclusive placement agent in connection with this offering. The placement agent
has agreed to use its reasonable best efforts to arrange for the sale of the securities offered by this prospectus. The placement agent
is not purchasing or selling any of the securities we are offering and the placement agent is not required to arrange the purchase or
sale of any specific number of securities or dollar amount. We have agreed to pay to the placement agent the placement agent fees set
forth in the table below, which assumes that we sell all of the securities offered by this prospectus. There is no arrangement for funds
to be received in escrow, trust or similar arrangement. There is no minimum offering requirement as a condition of closing of this offering.
We will bear all costs associated with the offering. See “Plan of Distribution” on page 31 of this prospectus for more information
regarding these arrangements.
We
are a “foreign private issuer,” and an “emerging growth company” each as defined under the federal securities
laws, and, as such, we are subject to reduced public company reporting requirements. See the section entitled “Prospectus Summary—Implications
of Being an Emerging Growth Company and a Foreign Private Issuer” for additional information.
Investing
in our securities involves a high degree of risk. Before buying any ADSs, you should carefully read the discussion of material risks
of investing in the ADSs and the company. See “Risk Factor Summary” beginning on page 11 for a discussion of information
that should be considered in connection with an investment in our securities.
Neither
the Securities and Exchange Commission nor any state securities commission has approved or disapproved of these securities or determined
if this prospectus is truthful or complete. Any representation to the contrary is a criminal offense.
| | |
Per
ADS or Pre-Funded Warrant | | |
Total | |
| Public offering price | |
$ | | | |
$ | | |
| Placement agent fees (1) | |
$ | | | |
$ | | |
| Proceeds to us, before expenses (2) | |
$ | | | |
$ | | |
| 1. |
We have
agreed to pay the Placement Agent cash fee equal to 7.5% of the gross proceeds raised in this offering. We have also
agreed to reimburse the Placement Agent for certain of its offering-related expenses, including a management fee of 1.0%
of the gross proceeds raised in this offering, to reimburse the Placement Agent for its non-accountable expenses in the amount of
$50,000, for its legal fees and expenses and other out-of-pocket expenses in an amount up to $100,000, and for its clearing expenses
in the amount of $15,950. In addition, we have agreed to issue to the Placement Agent, or its designees, warrants to purchase up to
a number of our ADSs and pre-funded warrants equal to 7.5% of the number of ADSs, including ADSs underlying the pre-funded warrants being offered at an exercise price equal to 125% of
the combined public offering price per ADS. See “Plan of Distribution” for additional information and a description
of the compensation payable to the Placement Agent. |
| |
|
| 2. |
We
estimate the total expenses of this offering payable by us, excluding the placement agent fee, will be approximately $0.4 million. |
We
anticipate that delivery of the securities against payment will be made on or about , 2023, subject to satisfaction of customary closing
conditions.
[*]
Prospectus
dated , 2023
TABLE
OF CONTENTS
Neither
we nor the underwriters have authorized anyone to provide information different from that contained in this prospectus, any amendment
or supplement to this prospectus or in any free writing prospectus prepared by us or on our behalf. Neither we nor the underwriters take
any responsibility for, and can provide no assurance as to the reliability of, any information other than the information in this prospectus,
any amendment or supplement to this prospectus, and any free writing prospectus prepared by us or on our behalf. Neither the delivery
of this prospectus nor the sale of the ADSs means that information contained in this prospectus is correct after the date of this prospectus.
This prospectus is not an offer to sell or the solicitation of an offer to buy the ADSs in any circumstances under which such offer or
solicitation is unlawful.
You
should rely only on the information contained in this prospectus and any free writing prospectus prepared by or on behalf of us or to
which we have referred you. We or the underwriters have not authorized anyone to provide you with information that is different. We and
the underwriters are offering to sell the ADSs, and seeking offers to buy the ADSs, only in jurisdictions where offers and sales are
permitted. The information in this prospectus is accurate only as of the date of this prospectus, regardless of the time of delivery
of this prospectus or any sale of the ADSs.
For
investors outside of the United States: Neither we nor the underwriters have done anything that would permit this offering or possession
or distribution of this prospectus in any jurisdiction where action for that purpose is required, other than in the United States. Persons
outside the United States who come into possession of this prospectus must inform themselves about and observe any restrictions relating
to this offering and the distribution of this prospectus outside the United States.
ABOUT
THIS PROSPECTUS
Unless
the context requires otherwise, in this prospectus TC BioPharm (Holdings) plc (formerly TC BioPharm (Holdings) Limited, which was re-registered
as a public limited company on January 10, 2022) and its subsidiaries (“Subsidiar(y/ies)”), and TC BioPharm Limited (our
principal trading subsidiary) shall collectively be referred to as “TCB,” “the Company,” “the Group”,
“we,” “us,” and “our” unless otherwise noted.
On
December 17, 2021, prior to our initial public offering, the Company undertook a corporate reorganization pursuant to which TC BioPharm
(Holdings) plc became the group holding company. The Company in turn effected a forward split of its ordinary shares on a 10 for 1 basis.
On November 18, 2022 the Company undertook a reverse share split such that fifty issued ordinary share were exchanged for one new ordinary
share. As a result of the share splits, all references included in this document to units of ordinary shares or per share amounts are
reflective of the forward and reverse share splits for all periods presented. In addition, the exercise prices and the numbers of ordinary
shares issuable upon the exercise of any outstanding options to purchase ordinary shares were proportionally adjusted pursuant to the
respective anti-dilution terms of the share-based payment plans.
The
consolidated financial statement data as at December 31, 2022 and 2021, and for the years ended December 31, 2022, 2021 and
2020 have been derived from our consolidated financial statements, which have been prepared in accordance with International Financial
Reporting Standards, or IFRS, as issued by the International Accounting Standards Board, or IASB. The December 31, 2022 and 2021 consolidated
financial statements were audited in accordance with the standards of the Public Company Accounting Oversight Board (United States).
Our
financial information is presented in pounds sterling. The figures as at December 31, 2022 and for the year ended December 31, 2022 have
been translated from pounds sterling into U.S. dollars at the rate of £1.00 to $1.2077, which was the noon buying rate of the Federal
Reserve Bank of New York on December 30, 2022. Such U.S. dollar amounts are not necessarily indicative of the amounts of U.S. dollars
that could actually have been purchased upon exchange of pounds sterling at the dates indicated or any other date. All references in
this Prospectus to “$” mean U.S. dollars and all references to “£” and “GBP” mean pounds sterling.
We
have made rounding adjustments to reach some of the figures included in this prospectus. As a result, numerical figures shown as totals
in some tables may not be an arithmetic aggregation of the figures that precede them.
This
prospectus includes statistical, market and industry data and forecasts which we obtained from publicly available information and independent
industry publications and reports that we believe to be reliable sources. These publicly available industry publications and reports
generally state that they obtain their information from sources that they believe to be reliable, but they do not guarantee the accuracy
or completeness of the information. Although we believe that these sources are reliable, we have not independently verified the information
contained in such publications. In addition, assumptions and estimates of our and our industry’s future performance are necessarily
subject to a high degree of uncertainty and risk due to a variety of factors, including those described in the “Risk Factor Summary”.
These and other factors could cause our future performance to differ materially from our assumptions and estimates.
Some
of our trademarks and trade names are used in this prospectus, which are intellectual property owned by the Company. This prospectus
also includes trademarks, trade names, and service marks that are the property of other organizations. Solely for convenience, our trademarks
and trade names referred to in this prospectus appear without the TM symbol, but those references are not intended to indicate,
in any way, that we will not assert, to the fullest extent under applicable law, our rights, or the right of the applicable licensor
to these trademarks and trade names.
ENFORCEABILITY
OF CIVIL LIABILITIES
TCB
is a corporation organized under the laws of Scotland. Substantially all of TCB’s assets and the majority of its directors and
executive officers are located and reside, respectively, outside the United States. Because of the location of TCB’s assets and
board members, it may not be possible for investors to serve process within the United States upon TCB or those persons with respect
to matters arising under the United States federal securities laws or to enforce against TCB or persons located outside the United States
judgments of United States courts asserted under the civil liability provisions of the United States federal securities laws.
TCB
understands that there is doubt as to the enforceability in Scotland and the United Kingdom, in original actions or in actions for enforcement
of judgments of United States courts, of civil liabilities predicated solely upon the federal securities laws of the United States insofar
as they are fines or penalties. In addition, awards of punitive damages in actions brought in the United States or elsewhere may be unenforceable
in Scotland and the United Kingdom by reason of being a penalty.
TC
BioPharm (North America) Inc., a Delaware corporation, with a registered office at Business Filings, Inc. 108 West 13th Street, Wilmington,
Delaware 19801, has been appointed agent to receive service of process in any action against TC BioPharm (Holdings) plc in any state
or federal court in the State of New York.
SPECIAL
NOTE REGARDING FORWARD-LOOKING STATEMENTS
TCB
discusses in this prospectus its business strategy, market opportunity, capital requirements, product introductions and development plans
and the adequacy of the Company’s funding. Other statements contained in this prospectus, which are not historical facts, are also
forward-looking statements. TCB has tried, wherever possible, to identify forward-looking statements by terminology such as “may,”
“will,” “could,” “should,” “expects,” “anticipates,” “intends,”
“plans,” “believes,” “seeks,” “estimates” and other comparable terminology.
TCB
cautions investors that any forward-looking statements presented in this prospectus, or that TCB may make orally or in writing from time
to time, are based on the beliefs of, assumptions made by, and information currently available to, TCB. These statements are based on
assumptions, and the actual outcome will be affected by known and unknown risks, trends, uncertainties and factors that are beyond its
control or ability to predict. Although TCB believes that its assumptions are reasonable, they are not a guarantee of future performance,
and some will inevitably prove to be incorrect. As a result, its actual future results can be expected to differ from its expectations,
and those differences may be material. Accordingly, investors should use caution in relying on forward-looking statements, which are
based only on known results and trends at the time they are made, to anticipate future results or trends. Certain risks are discussed
in this prospectus and also from time to time in TCB’s other filings with the Securities and Exchange Commission (“SEC”).
This
prospectus and all subsequent written and oral forward-looking statements attributable to the Company or any person acting on its behalf
are expressly qualified in their entirety by the cautionary statements contained or referred to in this section. The Company does not
undertake any obligation to release publicly any revisions to its forward-looking statements to reflect events or circumstances after
the date of this prospectus.
In
particular, you should consider the risks provided under “Risk factor summary” in this prospectus and in the Form 20-F for
the fiscal year ended December 31, 2022 as filed with the Securities and Exchange Commission (the “2022 Form 20-F”) incorporated
by reference in this prospectus.
PROSPECTUS
SUMMARY
The
following summary highlights selected information contained elsewhere in this prospectus. This summary does not contain all the information
you should consider before investing in our securities. You should read this entire prospectus carefully, including the information incorporated
by reference in this prospectus and any free writing prospectus prepared by us or on our behalf, including in particular the section
entitled “Risk Factor Summary” in this prospectus, “Item 3. Key Information”, Item 4, “Information on the
Company”; Item 5, “Operating and Financial review and Prospects”; Item 6,”Directors, Senior Management and Employees”;
Item 7, Major Shareholders and Related Party Transactions”; Item 8, “Financial Information” in our 2022 Form 20-F and
incorporated by reference in this prospectus, the other sections of the documents incorporated by reference in this prospectus and the
financial statements and the related notes incorporated by reference in this prospectus, before making an investment in our ADSs.
The
Company
Corporate
Overview
TCB
based in Scotland, is a clinical-stage biopharmaceutical company focused on developing novel immunotherapy products based on our proprietary
allogeneic gamma delta T (GD-T) cell platform. Harnessing the innate ability of GD-Ts has enabled us to develop a range of clinical-stage
cell therapies designed to combat cancer and viral infection.
In-house
clinical studies have demonstrated that our unmodified allogeneic GD-T products are (i) well tolerated and (ii) show preliminary evidence
of disease modification in patients with the late-stage blood cancer, known as acute myeloid leukemia (AML). Based on clinical
data generated by us believe that unmodified GD-Ts have the potential to treat all blood cancers.
TCB
now is embarking on phase 2b-into-pivotal (phase 3) clinical studies with a view to launching its first oncology product for the treatment
of AML. Clinical results generated thus far have enabled us to obtain FDA orphan drug status for treatment of AML.
In
addition to unmodified allogenic GD-Ts for treatment of blood cancers, we are also developing an innovative range of genetically-modified
CAR-T products for treatment of solid cancers. We believe that solid cancers are more difficult to treat than blood cancers and may require
the addition of a CAR “chimeric antigen receptor” (i) to help therapeutic cells to “navigate” into diseased cancerous
tissue and (ii) to retain therapeutic cells in-situ at the lesion for maximal efficacy (increased persistence).
In
order to manufacture our portfolio of allogeneic products, TCB selects the highest quality GD-T cells from healthy donors, activate the
cells and grow them in large numbers at our in-house GMP-compliant manufacturing facility before administration to a patient in order
to target and then destroy malignant or virally-infected tissues. TCB believes that we have introduced a step-change to our manufacturing
platform by implementing a freeze-thaw process that will allow product to be shipped from cleanroom to patient without any shelf-life
issue. Resulting products, TCB believes, will be more cost-effective and straightforward to ship form cleanroom to clinic.
At
this stage, TCB does not have any approved products. Accordingly, TCB has not generated any revenue from the sale of products, and TCB
does not expect to generate any such revenue unless and until it obtains regulatory approvals for, and commercialize any of, our product
candidates. In the future, TCB will seek to generate revenue primarily from product sales and, potentially, regional or global collaborations
with strategic partners, which may produce license fee income.
See
“Business - Overview” in our 2022 Form 20-F incorporated by reference in this prospectus.
Patent
Portfolio and Intellectual Property
We
believe TCB has a strong portfolio of patents and licenses covering the manufacture and commercialization of GD-T cell products and their
modification via CAR-T. We own fourteen granted patents and forty-three patent applications in four patent families and have an exclusive
license to an additional one family of twelve granted patents and ten patent applications. We protect our proprietary position, generally,
by filing an initial priority filing at the United Kingdom Intellectual Property Office, or UKIPO, followed by patent applications under
the Patent Co-operation Treaty claiming priority from the initial application(s) and then progressing to national applications in, for
example, the United States, Europe, Japan, China, Australia, New Zealand, South Korea, Israel and Canada.
As
a platform technology, we believe the co-stimulatory CAR-T GD-T cell system has a wealth of potential options to build added functionality.
We plan to continue to innovate and partner in the field to augment our drug products and introduce next generation attributes. We will
also continue to innovate our manufacturing and supply chains to efficiently scale our processes and simplify the interface with patients
and healthcare professionals, whilst continually seeking to reduce manufacturing costs to improve patient access.
We
intend to continue building on our technology platform, comprised of intellectual property, proprietary methods and know-how in the field
of GD-T cells. These assets form the foundation for our ability, not only to strengthen our product pipeline, but also to successfully
defend and expand our position as a leader in the field of GD-T based immuno-oncology.
See
“Business - Intellectual Property” in our 2022 Form 20-F incorporated by reference in this prospectus.
Our
Product Strategy
Our
strategic objective is to build a global therapeutic business with an extensive portfolio based upon unmodified and modified gamma delta
T cells (GD-T) with the potential to significantly improve the outcomes of patients with cancer and infectious disease.
Our
strategy is to take a step-wise approach to clinical development and commercialization. After our inception, we made clinical transitions
from autologous GD-Ts to allogeneic GD-Ts to CAR-modified allogeneic GD-Ts. Our commercialization strategy is to introduce clinical studies
for products firstly in blood cancers (AML initially) and then solid tumor indications. Complementarily, since GD-T cells are dysfunctional
in patients with severe viral diseases, TCB plans to develop its unmodified GD-T product to treat infectious diseases and believes that
this approach may be particularly relevant in relation to the treatment of viral pandemics.
Since
2015, TCB has built and maintained cell therapy medicinal product manufacturing facilities for Investigational Medicinal Products MIA
(abbreviated IMP), operated under license from the United Kingdom Medicines and Healthcare Products Regulatory Agency (abbreviated MHRA).
In April 2016, the MHRA granted a ‘Specials’ license to TCB, which allows it to treat patients under supervision of a qualified
doctor outside a clinical trial, and approved the company’s facility for ongoing Good Manufacturing Process (“GMP”)
compliance, which permits the manufacture and release of Advanced Therapy Medicinal Products (abbreviated ATMPs) for use in clinical
trials. TCB maintains a rigorous Quality Management System, which is based on the principles of the current GMP of the European and UK
law and regulation and EudraLex Volume 4, as revised. The Company complies with the two directives laying down principles and guidelines
of GMP for medicinal products adopted by the Commission. Directive 2003/94/EC applies to medicinal products for human use and Directive
91/412/EEC for veterinary use. Detailed guidelines in accordance with those principles are published in the Guide to Good Manufacturing
Practice which will be used in assessing applications for manufacturing authorizations and as a basis for inspection of manufacturers
of medicinal products.
Regulatory
approval of all aspects of medicinal therapy development, testing, manufacture and commercialization always is of concern. In the case
of treatment for AML, TCB has developed the novel approach of antibody-based immunotherapy and adoptive cell therapy with the aim to
improve anti-leukemia T cell function. Therefore, TCB is able to take advantage of orphan medicine regulation provided by the European
Medicines Agency (abbreviated EMA) and the United States Federal Drug Administration (abbreviated FDA), which are designed to encourage
medicine development for small numbers of patients where there is little commercial incentive under normal market conditions.
Part
of our strategy is to collaborate with appropriate partners from time to time.
See
“Business - Business Strategy” in our 2022 Form 20-F incorporated by reference in this prospectus.
TCB’s
Strengths
TCB
believes it has certain identified strengths. These include:
| |
● |
Clinical
trials that have provided strong evidence of safety and some preliminary indications of clinical benefit; |
| |
● |
A
proprietary co-stimulatory CAR-T technology platform which we believe allows solid cancers to be treated without toxic side-effects; |
| |
● |
Identification
of a large pool of cancer targets for which we believe we can develop therapeutic candidates; |
| |
● |
Retention
of key business elements, especially in-house ability to manufacture cell-based product and conduct our own clinical research; |
| |
● |
Robust,
and growing intellectual property portfolio protecting our products and proprietary platform; |
| |
● |
Our
policy is to develop strategic collaborations with leading, international companies to work together with us to develop certain GD-T
CAR-T products into clinic. We believe that existing and future collaborations will provide us with experience in scale-up and automation,
and post-authorization sales and marketing; |
| |
● |
A
highly knowledgeable and experienced management team with extensive industry experience and expertise in the United States and in
Europe; and |
| |
● |
Ability
to treat patients under the ‘Specials’ regulatory framework in Europe. |
Corporate
Information
Our
principal executive offices are located in Scotland, United Kingdom, with a mailing address of Maxim 1, 2 Parklands Way, Holytown, Motherwell,
ML1 4WR, United Kingdom and our telephone number at that location is +44 (0) 141 433 7557. Our website address is https://www.tcbiopharm.com.
The information contained on, or that can be accessed through, our website is not part of this prospectus. We have included our website
address in this prospectus solely as an inactive textual reference.
Implications
of Being an “Emerging Growth Company”
We
are an “emerging growth company,” as defined in Section 2(a) of the Securities Act of 1933, as amended, or the
Securities Act. As such, we are eligible to, and intend to, take advantage of certain exemptions from various reporting requirements
applicable to other public companies that are not “emerging growth companies” such as not being required to comply with
the auditor attestation requirements in the assessment of our internal control over financial reporting of Section 404 of the
Sarbanes-Oxley Act of 2002, or the Sarbanes-Oxley Act. We could remain an “emerging growth company” for up to five
years, or until the earliest of (a) the last day of the first fiscal year in which our annual gross revenue exceeds $1.235
billion, (b) the date that we become a “large accelerated filer” as defined in Rule 12b-2 under the Securities Exchange
Act of 1934, as amended, or the Exchange Act, which would occur if the market value of all our ordinary shares, including those
represented by the ADSs, that are held by non-affiliates exceeds $700 million as of the last business day of our most recently
completed second fiscal quarter, or (c) the date on which we have issued more than $1 billion in nonconvertible debt during the
preceding three-year period.
Implications
of being a “Foreign Private Issuer”
We
are subject to the information reporting requirements of the Securities and Exchange Act of 1934, as amended, the Exchange Act, that
are applicable to “foreign private issuers,” and under those requirements we file reports with the SEC. As a foreign private
issuer, we are not subject to the same requirements of the SEC applicable to U.S. domestic issuers. Under the Exchange Act, we are subject
to reporting obligations that, in certain respects, are less detailed and less frequent than those of U.S. domestic reporting companies.
For example, we are not required to issue quarterly reports, proxy statements that comply with the requirements applicable to U.S. domestic
reporting companies, or individual executive compensation information that is as detailed as that required of U.S. domestic reporting
companies. We also have four months after the end of each fiscal year to file our annual report with the SEC and are not required to
file current reports as frequently or promptly as U.S. domestic reporting companies. Our officers, directors and principal shareholders
are exempt from the requirements to report transactions in our equity securities and from the short-swing profit liability provisions
contained in Section 16 of the Exchange Act. As a foreign private issuer, we are not subject to the requirements of Regulation FD (Fair
Disclosure) promulgated under the Exchange Act. In addition, as a foreign private issuer, we are permitted to follow certain home country
corporate governance practices instead of those otherwise required under the Nasdaq Stock Market rules for domestic U.S. issuers and
are not required to be compliant with all Nasdaq Stock Market rules as of the date of our initial listing on Nasdaq as would domestic
U.S. issuers These exemptions and leniencies will reduce the frequency and scope of information and protections available to you in comparison
to those applicable to a U.S. domestic reporting company. We intend to take advantage of the exemptions available to us as a foreign
private issuer during and after the period we qualify as an “emerging growth company.”
Recent
Developments
On
March 27, 2023, we entered into a Securities Purchase Agreement (the “Purchase Agreement”) with certain accredited
investors (the “Investors”), pursuant to which we agreed to issue and sell an aggregate of 215,000 ADSs, pre-funded
warrants to purchase up to 3,222,500 ADS (the “March 2023 Pre-Funded Warrants”), and series C purchase warrants to
purchase up to 3,437,500 ADSs (the “Ordinary Warrants”). The purchase price for each ADS and associated Ordinary Warrants
was $1.60 and the purchase price per each March 2023 Pre-Funded Warrant and associated Ordinary Warrants was $1.599. The Ordinary
Warrants were immediately exercisable, expire five (5) years from the date of issuance and have an exercise price of $1.75 per ADS. The
March 2023 Pre-Funded Warrants may be exercised at any time until all of the March 2023 Pre-Funded Warrants are exercised
in full at an exercise price of $0.001 per ADS. The total net proceeds from this offering were approximately $4.6 million, after deducting
estimated offering expenses of approximately $0.9 million.
In
connection with the March 2023 offering, we agreed that certain existing warrants to purchase up to an aggregate of 2,800,000
ADSs of the Company that were previously issued on November 30, 2022, at an exercise price of $5.00 per ADS and expiration dates of May
30, 2025 and May 30, 2028, were amended effective upon the closing of the March 2023 offering so that the amended warrants will
have a reduced exercise price of $1.75 per ADS.
In
the period from January 1, 2023 to August 28, 2023, the holders of prefunded warrants, exercised prefunded warrants to purchase
4,537,500 ADSs.
On
April 3, 2023, the Company agreed with the loan note holder to extend the Redemption Date (as defined in the Loan Note) to January 15,
2024 and amend the Conversion Price (as defined in the Loan Note) of the outstanding loan notes to be the lesser of $1.00 or the
lowest closing price of the ADSs during the ten (10) day period prior to the date the Noteholder delivers a notice of conversion
to the Company, not to be lower than $0.20. In other respects the terms of the Loan Note remain unaltered. In addition, in consideration
of amending the Loan Note, the Company agreed to issue a 5-year warrant to the loan note holder to subscribe for 200,000 Ordinary Shares
in the share capital of the Company at an exercise price of $5.00 per Ordinary Share.
In
the period from April 3, 2023 to August 21, 2023, the holders of Loan Notes, converted notes with a value of $813,302 into 1,590,130
ADSs. As at the date of this prospectus, there are no
convertible loan notes in issue.
On
July 10, 2023, we entered into a warrant amendment with an existing investor pursuant to which the Company and the investor agreed that
certain existing warrants to purchase 2,800,000 ADSs of the Company that were previously issued on November 30, 2022 (the “November
2022 Warrants”) and certain existing warrants to purchase 3,437,500 ADSs of the Company that were previously issued on March 30,
2023 (the “March 2023 Warrants,” and together with the November 2022 Warrants, the “Existing Warrants”) would
be amended as follows: (i) amend the current exercise price on all Existing Warrants so that it is now equal to £0.35, (ii) extend
the termination date on 50% of the November 2022 Warrants and all of the March 2023 Warrants until May 30, 2028 and (iii) amend to the
definition of “Black Scholes Value” included in Section 3(e) of the Existing Warrants.
In
follow up to a written notification received from the listing qualifications staff of The Nasdaq Stock Market, LLC (“Nasdaq”)
on January 12, 2023 indicating that the Company had not regained compliance with the minimum Market Value of Listed Securities (“MVLS”)
of $35,000,000 required for continued listing on The Nasdaq Capital Market, as set forth in Nasdaq Listing Rule 5550(b)(2) (the “MVLS
Requirement”), the Company announced on July 10, 2023 that the Company believes that, as at July 10, 2023, it has stockholders’
equity well above the $2.5m minimum stockholders’ equity requirement.
The
Offering
| Securities,
offered by us |
|
Up to 9,900,990
ADSs or pre-funded warrants to purchase ADSs. See “Description of Securities.” We are also registering the
ADSs issuable upon exercise of the pre-funded warrants. |
| |
|
|
| ADSs |
|
Each
ADS represents one ordinary share. As a holder of ADSs, we will not treat you as one of our shareholders. The depositary, through
its custodian, will be the holder of the ordinary shares underlying the ADSs, and you will have the rights of a holder of ADSs or
beneficial owner (as applicable) as provided in the deposit agreement among us, the depositary and owners and holders of ADSs from
time to time. To better understand the terms of the ADSs you should read Item 10B (“Memorandum and Articles of Association”)
in our 2022 Form 20-F incorporated by reference in this prospectus. We also encourage you to read the deposit agreement, the form
of which is filed as an exhibit to the registration statement of which this prospectus forms a part. |
| |
|
|
| Pre-Funded Warrants Offered |
|
We are also offering to certain
purchasers whose purchase of ADSs in this offering would otherwise result in the purchaser, together with its affiliates and certain
related parties, beneficially owning more than 4.99% (or, at the election of the purchaser, 9.99%) of our outstanding ADSs immediately
following the closing of this offering, the opportunity to purchase, if such purchasers so choose, pre-funded warrants to purchase
ADSs, in lieu of ADSs that would otherwise result in any such purchaser’s beneficial ownership exceeding 4.99% (or, at the
election of the purchaser, 9.99%) of our outstanding ADSs. Each pre-funded warrant will be exercisable for one ADS. The purchase
price of each pre-funded warrant will be equal to the price at which an ADS and accompanying Ordinary Warrant is being sold to the
public in this offering, minus $0.001, and the exercise price of each pre-funded warrant will be $0.001 ADS. The pre-funded warrants
will be exercisable immediately and may be exercised at any time until all of the pre-funded warrants are exercised in full. This
prospectus also relates to the ADSs issuable upon exercise of any pre-funded warrants sold in this offering. For each pre-funded
warrant we sell, the number of ADSs we are offering will be decreased on a one-for-one basis. |
| |
|
|
| Term
of the offering |
|
This
offering will terminate on [*], 2023, unless we decide to terminate the offering (which we may do at any time in our discretion)
prior to that date. |
| |
|
|
| Ordinary
shares outstanding before this offering |
|
7,292,588
ordinary shares |
| |
|
|
| Warrants
outstanding before this offering |
|
Warrants
to purchase 7,283,037 ADSs
|
| |
|
|
| Ordinary
shares to be outstanding after this offering, including ordinary shares represented by ADSs |
|
17,193,578
ordinary shares, assuming no sale of pre-funded
warrants, which, if sold, would reduce the number of ADSs that we are offering on a one-for-one basis, and no exercise of the warrants
issued to the placement agent (the “Placement Agent Warrants”). |
| |
|
|
| Use
of proceeds |
|
We
estimate that our net proceeds from this offering will be approximately $4.2 million. This is based on a public offering
price of $0.505 per ADS, which is the last reported trading price of our ADSs on The Nasdaq Capital Market on August
22, 2023, after deducting the estimated placement agent fees and commissions and estimated offering expenses payable by us.
We intend to use the net proceeds of this offering to advance our preclinical and clinical
pipeline, and for continuing operating expenses and working capital. |
| |
|
|
| Risk
factors |
|
You
should read the “Risk Factor Summary” section within this prospectus and in Item 3D (“Risk Factors”) in our
2022 Form 20-F included by reference in this prospectus, for a discussion of factors to consider carefully before deciding to invest
in our securities. |
| |
|
|
| Nasdaq
Capital market symbols |
|
ADSs
on the Nasdaq Capital Market under the symbol “TCBP.” |
The
number of our ordinary shares (including shares represented by ADSs) to be outstanding after this offering is based on 7,292,588 ordinary
shares outstanding as of August 28, 2023 and excludes:
| |
● |
106,585
ordinary shares issuable upon the exercise of options outstanding under our 2014 Share Option Scheme as of December 31, 2022, with
a weighted-average exercise price of £23.00 per share; |
| |
|
|
| |
● |
52,305
ordinary shares issuable upon the exercise of options outstanding under our 2021 Share Option Scheme, as of December 31, 2022, with
a weighted-average exercise price of $212.00 per share; and |
| |
|
|
| |
● |
15,891
ordinary shares issuable upon the exercise of options outstanding, at a future date based on the achievement of certain clinical
and commercial milestones with an exercise price of £215.00 per share. |
For
the description of the 2014 Share Option Scheme and 2021 Share Option Scheme see “Item 6.E Share Ownership” in the 2022 Form
20-F, which is incorporated by reference herein.
Unless otherwise stated, all
information in this prospectus assumes no exercise of the outstanding options described above into ordinary shares or ADSs, treats all
restricted shares issued with outstanding restrictions to be vested as issued and outstanding shares, no exercise of the Placement Agent
Warrants issued in this offering and no sale of pre-funded warrants in this offering.
Except
as otherwise indicated all references to our articles of association in this prospectus refer to our articles of association, as amended
as currently in force for TC BioPharm (Holdings) plc at the date of this prospectus.
Summary
Consolidated Financial Data
The
following table summarizes our consolidated financial data as at the dates and for the periods indicated. The consolidated financial
statement data as at December 31, 2022 and 2021, and for the years ended December 31, 2022 and 2021 audited in accordance with the standards
of the Public Company Accounting Oversight Board (United States) have been derived from our consolidated financial statements, which
have been prepared in accordance with International Financial Reporting Standards, or IFRS, as issued by the International Accounting
Standards Board, or IASB.
Our
financial information is presented in pounds sterling. The figures as at December 31, 2022 and for the year ended December 31, 2022 have
been translated from pounds sterling into U.S. dollars at the rate of £1.00 to $1.2077, which was the noon buying rate of the Federal
Reserve Bank of New York on December 30, 2022. Such convenience translation should not be construed as a representation that the pound
sterling amounts have been or could be converted into U.S. dollars at this or at any other rate of exchange, or at all.
Our
historical results are not necessarily indicative of the results that may be expected in the future.
This
information should be read together with, and is qualified in its entirety by, our consolidated financial statements and the notes thereto.
You should read the following summary consolidated financial and other data in conjunction with “Item 5. Operating and Financial
Review and Prospects” and Item 8 (“Financial Information’), our consolidated financial statements and the notes thereto
and the other financial information included in our 2022 Form 20-F annual report and incorporated by reference in this prospectus.
| | |
Year
Ended December 31 | |
| Consolidated
Statement of Comprehensive Loss: | |
2022 | | |
2022 | | |
2021 | |
| | |
$’000 | | |
£’000 | | |
£’000 | |
| | |
| | |
| | |
| |
| Revenue | |
| 4,644 | | |
| 3,845 | | |
| 1,979 | |
| Research and development expenses | |
| (8,995 | ) | |
| (7,448 | ) | |
| (6,157 | ) |
| Administrative expenses | |
| (8,471 | ) | |
| (7,014 | ) | |
| (2,059 | ) |
| Administrative expenses – costs related
to preparing for a listing | |
| (1,576 | ) | |
| (1,305 | ) | |
| (1,050 | ) |
| Foreign exchange losses | |
| (146 | ) | |
| (121 | ) | |
| (83 | ) |
| Other income | |
| - | | |
| - | | |
| 7 | |
| Loss on modification of convertible loan | |
| (169 | ) | |
| (140 | ) | |
| | |
| Change in fair value of convertible loan derivatives | |
| 8,589 | | |
| 7,112 | | |
| (4,182 | ) |
| Change in fair value of warrant derivatives | |
| 15,440 | | |
| 12,785 | | |
| - | |
| Change in fair value of other derivatives | |
| (4,628 | ) | |
| (3,832 | ) | |
| - | |
| Finance income | |
| 2 | | |
| 2 | | |
| - | |
| Finance costs | |
| (8,447 | ) | |
| (6,994 | ) | |
| (3,439 | ) |
| Loss before tax | |
| (3,756 | ) | |
| (3,110 | ) | |
| (14,984 | ) |
| Income tax credit | |
| 2,077 | | |
| 1,720 | | |
| 1,408 | |
| Net
loss for the year and Total comprehensive loss | |
| (1,679 | ) | |
| (1,390 | ) | |
| (13,576 | ) |
| Per share data | |
| | | |
| | | |
| | |
| Basic and diluted loss per
share (1) | |
| (2.44 | ) | |
| (2.02 | ) | |
| (34.76 | ) |
| Weighted average shares outstanding (1) | |
| 687,199 | | |
| 687,199 | | |
| 390,585 | |
| | |
As
at December 31 | |
| | |
2022 | | |
2022 | | |
2021 | |
| | |
$’000 | | |
£’000 | | |
£’000 | |
| | |
| |
| Consolidated Statement of
Financial Position items: | |
| | | |
| | | |
| | |
| Cash and cash equivalents | |
| 5,807 | | |
| 4,808 | | |
| 1,567 | |
| Working capital (2) | |
| (2,072 | ) | |
| (1,716 | ) | |
| (16,724 | ) |
| Total assets | |
| 13,226 | | |
| 10,951 | | |
| 8,024 | |
| Total liabilities | |
| (13,256 | ) | |
| (10,976 | ) | |
| (24,583 | ) |
| Share capital | |
| 20,525 | | |
| 16,995 | | |
| 195 | |
| Other reserves | |
| 20,182 | | |
| 16,711 | | |
| 16,711 | |
| Accumulated deficit | |
| (40,738 | ) | |
| (33,732 | ) | |
| (33,465 | ) |
| Total equity attributable to the equity shareholders
of the parent | |
| (32 | ) | |
| (26 | ) | |
| (16,559 | ) |
| (1) |
Prior
to our initial public offering, the Company undertook a corporate reorganization pursuant to which TC BioPharm (Holdings) plc became
the group holding company. The Company in turn effected a forward split of its ordinary shares on a 10 for 1 basis. On November 18,
2022 the Company undertook a reverse share split such that fifty issued ordinary share were exchanged for one new ordinary share.
As a result of the share splits, all references included in this document to units of ordinary shares or per share amounts are reflective
of the forward and reverse share splits for all periods presented. In addition, the exercise prices and the numbers of ordinary shares
issuable upon the exercise of any outstanding options to purchase ordinary shares were proportionally adjusted pursuant to the respective
anti-dilution terms of the share-based payment plans. |
| (2) |
Working
capital is defined as current assets less current liabilities. |
RISK
FACTOR SUMMARY
Our
business is subject to a number of risks and uncertainties, including those risks discussed at length in Item 3D (“Risk Factors”)
in our 2022 Form 20-F incorporated into this prospectus by reference. These risks include among others those summarized below. Investing
in our company and its securities involves a high degree of risk. You should carefully consider the risks and uncertainties described
below, together with all of the other information in this prospectus, including the information incorporated by reference to our 2022
Form 20-F, before investing in our company and our securities. If any of these risks materialize, our business, financial condition,
operating results and prospects could be materially and adversely affected. In that event, the price or value of our ADSs in the public
market could decline, and you could lose part or all of your investment.
The
following is a summary of some of the principal risks we face. The list below is not exhaustive, and investors should read the risks
described under the heading “Risk Factors” in our 2022 Form 20-F incorporated by reference herein, as well as the additional
risks set forth in this section, in full.
| |
● |
We
have generated operating losses since inception and expect to continue to generate losses. We may never achieve or maintain profitability.
We will continue to require financing to continue to implement our business plan and sustain operations. |
| |
|
|
| |
● |
We,
as well as our independent registered public accounting firm, in relation to our financial position, have expressed substantial doubt
about our ability to continue as a going concern. The reasons for expressing that doubt in relation to our historical financial
statements remains relevant and applicable to this offering. |
| |
|
|
| |
● |
Our
lack of any approved products and our limited operating history may make it difficult for an investor to evaluate the success of
our business to date and to assess our future viability. |
| |
|
|
| |
● |
GD-T
cell therapies are a novel approach to treating cancers and infectious diseases, which have development risks and will require us
to obtain regulatory approvals for development, testing, commercialization, manufacturing and distribution. We may not achieve all
the required regulatory approvals or approvals may not be obtained as timely as needed. |
| |
|
|
| |
● |
Because
GD-T cell therapies are a novel approach, potential side effects, and long-term efficacy, regulatory approval will require considerable
time for trials, data collection, regulatory submissions and funding for the process. |
| |
|
|
| |
● |
Enrolling
patients in clinical trials may be difficult for many reasons, including high screen failure, GD-T cell proliferation capacity, timing,
proximity and availability of clinical sites, perceived risks, and publicity about the success or lack of success in the methods
of treatment. |
| |
|
|
| |
● |
Because
GD-T cell therapies are novel, our research and development and clinical trial results may not support our products intended purposes
and regulatory approval. We are heavily dependent on the success of our lead product candidate (OmnImmune®), and intend to seek
breakthrough therapy designation for some or all of our other therapeutic candidates in due course. |
| |
|
|
| |
● |
Market
opportunities for certain of our product candidates may be limited to those patients who are ineligible for or have failed prior
treatments. This class of patient may be limited in number, difficult to locate and service, require special governmental approval,
and unable to pay or obtain reimbursement. |
| |
|
|
| |
● |
We
rely on many third parties for aspects of our product development and commercialization, such as raw material supply, clinical trials,
obtaining approvals, aspects of manufacturing, development of additional product candidates and distribution. We may not be able
to control these parties and their business practices, such as compliance with good manufacturing requirements or their ability to
supply or service us timely, which will likely disrupt our business. |
| |
|
|
| |
● |
We
face substantial competition: others may discover, develop and/or commercialize competing products before or more successfully than
TCB. |
| |
● |
Even
if we are able to commercialize any product candidates, such drugs may become subject to unfavorable pricing regulations or third-party
coverage and reimbursement policies. Commercialized products may not be adopted by the medical profession. |
| |
|
|
| |
● |
Because
we operate internationally, we are subject to a wide array of regulation of the United Kingdom, European Union and United States.
In addition to regulation surrounding new drug development and their manufacture, distribution and use, we will be subject, for example
to data protection rules relating to medical records, medical and general privacy laws, environmental laws regarding medical waste,
and bribery and corrupt practices law, in addition to all the drug related approval, manufacturing and distribution rules. |
| |
|
|
| |
● |
Product
liability claims are frequent in drug development of novel therapies and insurance is mandatory and expensive. The inability to obtain
insurance may prevent product development and claims may surpass our ability to pay and call into question the efficacy of a product
with resulting reputational damage. |
| |
|
|
| |
● |
Protecting
our intellectual property is paramount in our ability to be able to commercialize our products and generate revenues and investment
return for our stockholders. We may not be able to obtain the intellectual property protection we seek due to its cost, requirement
to pursue it in many jurisdictions, challenges by others and patent office rejection. |
| |
|
|
| |
● |
Obtaining
and maintaining our patent protection depends on compliance with various procedural, document submission, fee payment and other requirements
imposed by governmental patent agencies acting in multiple jurisdictions, and our patent protection could be reduced or eliminated
for non-compliance with these requirements. |
| |
|
|
| |
● |
As
part of product development, we may need to license aspects of our research and products from third parties or if our IP is challenged,
we may have to seek license accommodation, any of which may be expensive, limited in scope, or unavailable. |
| |
|
|
| |
● |
We
currently have a limited number of employees, and our future success depends on our ability to retain key executives and to attract,
retain and motivate qualified personnel at all levels. |
| |
|
|
| |
● |
We
will need to grow the size and capabilities of our organization, and we may experience difficulties in managing this growth including,
but not limited to, operating as a public company and taking a therapeutic through to market approval and acceptance. |
| |
|
|
| |
● |
We
expect to expand our development and regulatory capabilities and potentially implement sales, marketing and distribution capabilities,
and as a result, we may encounter difficulties in achieving and managing our growth, which could disrupt our operations. We expect
to require further funding for these expansions of activity. |
| |
|
|
| |
● |
We
incur substantial costs as a result of operating as a public company in the United States, and our management is required to devote
substantial time to required SEC compliance and corporate governance practices. |
| |
|
|
| |
● |
If
we fail to maintain proper and effective internal controls, our ability to produce accurate financial statements on a timely basis
could be impaired, which would adversely affect our business and our stock price. |
| |
● |
Certain
of our existing stockholders, members of our board of directors and senior management maintain the ability to exercise significant
control over us. The interests of investors may conflict with the interests of these other stockholders. |
| |
|
|
| |
● |
Our
ADSs provide rights that are different from directly holding our ordinary shares. The outstanding Warrants do not have the rights
of shareholders until exercised. Our Warrants form a substantial part of our capitalization, and they have substantial protective
provisions, which may limit our ability to raise capital. |
| |
|
|
| |
● |
Future
sales, or the possibility of future sales, of a substantial number of our ordinary shares, through the additional deposit of ordinary
shares for ADSs and exercises of our Warrants, could adversely affect the price of our ADSs or Warrants in the market. After any
lock up period, a substantial number of our issued and outstanding ordinary shares will be eligible for trading on the public securities
market by their being deposited with the depositary for ADSs. |
| |
|
|
| |
● |
As
a foreign private issuer, we, and our stockholders, have certain exceptions to disclosure regulation under United States federal
securities regulation, and we will take certain NASDAQ governance exceptions. Consequently, investors may not have the totality of
disclosure about and governance controls in TCB as compared to United States domestic reporting companies. |
| |
|
|
| |
● |
Shareholder
rights and recourse will be governed by and ultimately determined by Scottish and United Kingdom law and judicial process, which
in many ways are more limited than United States law and practice. Most of our directors and officers are not resident in the United
States. Most of our assets are located in the United Kingdom. |
| |
|
|
| |
● |
If
we fail to meet the requirements for continued listing on the Nasdaq Global Market or Nasdaq, our ADSs could be delisted from trading,
which would decrease the liquidity of our ADSs and our ability to raise additional capital. |
Risks
Related to this Offering and Ownership of ADSs
The
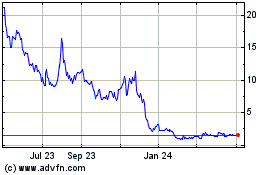
price of the ADSs has been, and is likely to continue to be, highly volatile, which could result in substantial losses for purchases
of ADSs in this offering.
The
price of the ADSs has been, and is likely to continue to be, highly volatile. The stock market in general and the market for smaller
pharmaceutical and biotechnology companies in particular have experienced extreme volatility that has often been unrelated to the operating
performance of particular companies. As a result of this volatility, purchasers of securities sold pursuant to this registration statement
may not be able to sell their ADSs at or above the price paid by such purchasers and, as such, they may lose some or all of their investment.
Additionally, in the past, securities class action litigation has often been brought against a company following a decline in the market
price of its securities. This risk is especially relevant for us in light of the significant stock price volatility we and other pharmaceutical
companies have experienced in recent years. If we face such litigation, it could result in substantial costs and a diversion of management’s
attention and resources, which could harm our business.
We
have broad discretion in the use of the net proceeds from this offering and any exercise of the Warrants and consequently may
not use them effectively.
Our
management will have broad discretion in the application of the net proceeds from this offering and any exercise of any Warrant and could
spend any such proceeds in ways that do not improve our results of operations or enhance the value of our ADSs. The failure by our management
to apply these funds effectively could result in financial losses that could cause the price of our ADSs to decline and delay the development
of our product candidates.
If
we fail to meet the requirements for continued listing on the Nasdaq Global Market or Nasdaq, our ADSs could be delisted from trading,
which would decrease the liquidity of our ADSs and our ability to raise additional capital.
Our
ADSs are currently listed for quotation on The Nasdaq Capital Market. We are required to meet specified financial requirements in order
to maintain our listing on the Nasdaq Capital Market. These requirements include maintaining a minimum bid price of at least $1.00 per
share for our ADSs, which is referred to as the Bid Price Rule, and maintaining a minimum market value of listed securities, or the MVLS,
of $35,000,000. On July 12 and 15, 2022, we received deficiency letters from the Listings Qualifications Department of the Nasdaq Stock
Market notifying that we were not in compliance with the Bid Price Rule and the MVLS, respectively.
On
December 6, 2022, we received written notification from the listing qualifications staff of the Nasdaq Stock Market, LLC (“Nasdaq”)
indicating that the Company regained compliance with the Bid Price Rule. On January 12, 2023, we received written notification from the
listing qualifications staff of the Nasdaq indicating that we have not regained compliance with the MVLS and
that our securities would be subject to delisting unless we timely request a hearing before a Nasdaq Hearings Panel (the “Panel”).
On March 9, 2023 the Company presented a formal plan to regain compliance to the Panel. On March 17, 2023, the Company announced that
the TC BioPharm (Holdings) plc has been granted a formal extension until June 30, 2023, to regain compliance under Nasdaq Listing Rule
5550(b)(2) or its alternative criteria. On July 27, 2023, the Company received a letter, dated July 26, 2023 (the “Letter”)
from Nasdaq notifying the Company that the Panel has concluded that the Company has regained compliance with Nasdaq’s continued
listing requirements. The Letter stated that, pursuant to Listing Rule 5815(d)(4)(A), the Company will be subject to a Panel Monitor
for a period of one year from the date of the Letter. If, within that one-year monitoring period, the Listing Qualifications staff (the
“Staff”) finds the Company again out of compliance with any continued listing requirement, notwithstanding Rule 5810(c)(2),
the Company will not be permitted to provide the Staff with a plan of compliance with respect to any deficiency and the Staff will not
be permitted to grant additional time for the Company to regain compliance with respect to any deficiency, nor will the Company be afforded
an applicable cure or compliance period. Instead, the Staff will issue a Delist Determination Letter and the Company will have an opportunity
to request a new hearing with the initial Panel or a newly convened Hearings Panel if the initial Panel is unavailable.
On
June 22, 2023, we received a deficiency letter from the Staff notifying that we again were not in compliance with the Bid Price
Rule. We have been provided an initial period of 180 calendar days, or until December 19, 2023, to regain compliance with the applicable
listing requirement. If the Company does not regain compliance with the Bid Price Rule by such date, the Company may be afforded a second
180 calendar day period to regain compliance. To qualify, the Company would be required to meet the continued listing requirement for
market value of publicly held shares and all other initial listing standards for The Nasdaq Capital Market, except for the minimum bid
price requirement. In addition, the Company would be required to notify Nasdaq of its intent to cure the deficiency during the second
compliance period. If the Company does not regain compliance with the minimum bid price requirement by the end of the compliance period
(or the second compliance period, if applicable), the Company’s ADSs will become subject to delisting. In the event that the Company
receives notice that its ADSs is being delisted, the Nasdaq listing rules permit the Company to appeal a delisting determination by the
Staff to a hearings panel.
The
Company continues to execute its business plan and is looking into various options available to regain compliance with Nasdaq’s
continued listing standards and maintain its continued listing on the Nasdaq Capital Market. However, there can be no assurance that
the Company will be able to regain compliance with the Bid Price Rule requirement or will otherwise be in compliance with other Nasdaq
listing rules. In addition, there can be no assurance that the Panel will determine to continue the Company’s listing on The Nasdaq
Capital Market or that the Company will timely evidence compliance with the terms of the extension that was granted by the Panel following
the hearing.
There
is no public market for Warrants or pre-funded warrants being offered by us in this offering.
There
is no established public trading market for the Warrants or pre-funded warrants, and we do not expect a market to develop. In addition,
we do not intend to apply to list the Warrants or pre-funded warrants on any national securities exchange or other nationally recognized
trading system. Without an active market, the liquidity of the Warrants or pre-funded warrants will be limited.
The
pre-funded warrants are speculative in nature.
The
pre-funded warrants offered hereby do not confer any rights of ADS ownership on their holders, such as voting rights, but rather merely
represent the right to acquire shares of ADS at a fixed price. Specifically, holders of the pre-funded warrants may acquire the ADSs
issuable upon exercise of such warrants at an exercise price of $0.001 per ADS. Moreover, following this offering, the market value of
the pre-funded warrants is uncertain and there can be no assurance that the market value of the pre-funded warrants will equal or exceed
their public offering prices. There can be no assurance that the market price of the ADSs will ever equal or exceed the exercise price
of the pre-funded warrants, and consequently, whether it will ever be profitable to exercise the pre-funded warrants.
Holders
of the pre-funded warrants offered hereby will have no rights as ADS holders with respect to the ADSs underlying the pre-funded warrants
until such holders exercise their warrants and acquire our ADSs, except as otherwise provided in the pre-funded warrants.
Until
holders of the pre-funded warrants acquire ADSs upon exercise thereof, such holders will have no rights with respect to the ADSs underlying
such pre-funded warrants, except to the extent that certain rights may be granted to warrant holders as set forth in the warrants. Upon
exercise of the pre-funded warrants, the holders will be entitled to exercise the rights of an ADS holder only as to matters for which
the record date occurs after the exercise date.
This
is a best efforts offering, no minimum amount of securities is required to be sold, and we may not raise the amount of capital we believe
is required for our business plans, including our near-term business plans.
The
placement agent has agreed to use its reasonable best efforts to solicit offers to purchase the securities in this offering. The placement
agent has no obligation to buy any of the securities from us or to arrange for the purchase or sale of any specific number or dollar
amount of the securities. There is no required minimum number of securities that must be sold as a condition to completion of this offering.
Because there is no minimum offering amount required as a condition to the closing of this offering, the actual offering amount, placement
agent fees and proceeds to us are not presently determinable and may be substantially less than the maximum amounts set forth above.
We may sell fewer than all of the securities offered hereby, which may significantly reduce the amount of proceeds received by us, and
investors in this offering will not receive a refund in the event that we do not sell an amount of securities sufficient to support our
continued operations, including our near-term continued operations. Thus, we may not raise the amount of capital we believe is required
for our operations in the short-term and may need to raise additional funds, which may not be available or available on terms acceptable
to us.
You
will experience immediate dilution in the book value per ADS purchased in the offering.
Because
the price per share of our ADSs being offered may be higher than the net tangible book value per ADS, you will experience dilution to
the extent of the difference between the offering price per ADS you pay in this offering and the net tangible book value per ADS immediately
after this offering. Our net tangible book value as of December 31, 2022, was approximately $0.7 million, or $0.74
per ADS. Net tangible book value per ADS is equal to our total tangible assets minus total liabilities, all divided by the number of
ADSs outstanding. See the section titled “Dilution” for a more detailed discussion of the dilution you will incur if you
purchase shares in this offering.
If
you purchase our securities in this offering you may experience future dilution as a result of future equity offerings or other equity
issuances.
In
order to raise additional capital, we believe that we will offer and issue additional ADSs or other securities convertible into or exchangeable
for our ADSs in the future. We cannot assure you that we will be able to sell ADSs or other securities in any other offering at a price
per ADS that is equal to or greater than the price per ADS paid by investors in this offering, and investors purchasing other securities
in the future could have rights superior to existing stockholders. The price per ADS at which we sell additional ADSs or other securities
convertible into or exchangeable for our ADSs in future transactions may be higher or lower than the price per ADS in this offering.
In
addition, we have a significant number of share options and warrants outstanding. To the extent that outstanding share options or warrants
have been or may be exercised or other shares issued, you may experience further dilution. Further, we may choose to raise additional
capital due to market conditions or strategic considerations even if we believe we have sufficient funds for our current or future operating
plans.
The
exercise of outstanding ADS purchase warrants and share options will have a dilutive effect on the percentage ownership of our capital
stock by existing stockholders.
As
of August 28, 2023, we had outstanding warrants to acquire 7,283,037 ADSs, and share options to purchase 174,781 shares
of our ordinary shares. A significant number of such warrants have exercise prices above our ADSs’ recent trading prices, but the
holders have the right, in certain circumstances, to effect a cashless exercise of such warrants. If a significant number of such warrants
and share options are exercised by the holders, the percentage of our ADSs owned by our existing ADS holders will be diluted.
We
face risks and uncertainties related to litigation, regulatory actions and government investigations and inquiries.
We
are subject to, and may become a party to, litigation, claims, suits, regulatory actions and government investigations and inquiries.
The
outcome of any litigation, regardless of its merits, is inherently uncertain. Any claims and lawsuits, and the disposition of such
claims and lawsuits, could be time-consuming and expensive to resolve, divert management attention and resources, and lead to attempts
on the part of other parties to pursue similar claims. Negative perceptions of our business may result in additional regulation, enforcement
actions by the government and increased litigation, or harm to our ability to attract or retain customers or strategic partners,
any of which may affect our business. Any damage to our reputation, including from publicity from legal proceedings against us or companies
that work within our industry, governmental proceedings, unfavorable media coverage or class action could adversely affect our business,
financial condition and results of operations.
An
unfavorable outcome or settlement or any other legal, administrative and regulatory proceeding may result in a material adverse impact
on our business, results of operations, financial position and overall trends. In addition, regardless of the outcome, litigation can
be costly, time-consuming, and disruptive to our operations. Any claims or litigation, even if fully indemnified or insured, could
damage our reputation and make it more difficult to compete effectively or to obtain adequate insurance in the future.
The
Company has received a lawsuit asserting, among other things, breach of contract under the terms of certain convertible promissory notes.
The lawsuit is pending before the High Court in England, and the Company
has retained English legal representatives to defend it. The company does not believe that the outcome of the claims is likely to
be material to the balance sheet of TC BioPharm (Holdings) plc.
As
of January 1, 2024, we may no longer qualify as a foreign private issuer, which would then require us to comply with the Exchange
Act’s domestic reporting regime and cause us to incur significant legal, accounting and other expenses.
In
order to maintain our current status as a foreign private issuer, a majority of our outstanding ordinary shares (including those represented
by ADSs) must continue to be either directly or indirectly owned of record by non-residents of the United States. If a majority of our
outstanding ordinary shares (including those represented by the ADSs) are instead held by U.S. residents, then in order to continue to
maintain our foreign private issuer status, (i) a majority of our executive officers or directors must not be U.S. citizens or residents,
(ii) more than 50% of our assets must not be located in the United States, and (iii) our business must be administered principally outside
the United States.
The
determination of foreign private issuer status is made annually on the last business day of an issuer’s most recently completed
second fiscal quarter. We are assessing whether, as of June 30, 2023, we satisfied the requirements for retaining our foreign private
issuer status as of such date. Assuming we did not satisfy the requirements to remain a foreign private issuer as of June 30, 2023,
we would cease to be a foreign private issuer and cease to be eligible for the foregoing exemptions
and privileges effective January 1, 2024.
Losing
our status as a foreign private issuer would require us to comply with all of the periodic disclosure and current reporting requirements
of the Exchange Act applicable to U.S. domestic issuers. We may also be required to make changes in our corporate governance practices
in accordance with various SEC and Nasdaq rules. The regulatory and compliance costs to us under U.S. securities laws, if we are required
to comply with the reporting requirements applicable to a U.S. domestic issuer, may be significantly higher than the cost we would incur
as a foreign private issuer. As a result, we would expect that a loss of foreign private issuer status will increase our legal and financial
compliance costs and will make some activities highly time consuming and costly. We also expect that if we will be required to comply
with the rules and regulations applicable to U.S. domestic issuers, it will make it more difficult and expensive for us to obtain director
and officer liability insurance; we may therefore be required to accept reduced coverage or incur substantially higher costs to obtain
coverage. These rules and regulations could also make it more difficult for us to attract and retain qualified members of our board of
directors.
If we fail to maintain
proper and effective internal controls, our ability to produce accurate financial statements on a timely basis could be impaired, which
would adversely affect our business and our stock price.
Ensuring that
we have adequate internal financial and accounting controls and procedures in place to produce accurate financial statements on a timely
basis is a costly and time-consuming effort that needs to be re-evaluated frequently. We may discover material weaknesses in our internal
financial and accounting controls and procedures that need improvement from time to time.
Management is
responsible for establishing and maintaining adequate internal control over financial reporting to provide reasonable assurance regarding
the reliability of our financial reporting and the preparation of financial statements for external purposes. Management does not expect
that our internal control over financial reporting will prevent or detect all errors and all fraud. A control system, no matter how well
designed and operated, can provide only reasonable, not absolute, assurance that the control system’s objectives will be met. Because
of the inherent limitations in all control systems, no evaluation of controls can provide absolute assurance that misstatements due to
error or fraud will not occur or that all control issues and instances of fraud, if any, within our company will have been detected.
Pursuant to Section
404 of the Sarbanes-Oxley Act of 2002, or Section 404, we are required to furnish a report by our senior management on our internal control
over financial reporting, commencing with our second annual report. However, while we remain an EGC we are not required to include an
attestation report on internal control over financial reporting issued by our independent registered public accounting firm. To prepare
for eventual compliance with Section 404, once we no longer qualify as an EGC, we are engaged in a process to document and evaluate our
internal control over financial reporting, which is both costly and challenging. In this regard, we will need to continue to dedicate
internal resources, potentially engage outside consultants, adopt a detailed work plan to assess and document the adequacy of internal
control over financial reporting, continue steps to improve control processes as appropriate, validate through testing that controls
are functioning as documented, and implement a continuous reporting and improvement process for internal control over financial reporting.
Despite our efforts, there is a risk that we will not be able to conclude, within the prescribed timeframe or at all, that our internal
control over financial reporting is effective as required by Section 404. If we identify one or more material weaknesses, it could result
in an adverse reaction in the financial markets due to a loss of confidence in the reliability of our financial statements. In addition,
if we are unable to produce accurate financial statements on a timely basis, investors could lose confidence in the reliability of our
financial statements, which could cause the market price of either of our ADSs or Warrants, or both, to decline and make it more difficult
for us to finance our operations and growth.
The Company notes
that the auditors identified that the Company experienced difficulty in the accounting for complex financial instruments and the Company
lacked adequate internal control over the account and assessment of complex financial instruments following control deficiencies which
they believed constituted a material weakness in the Company’s internal control over financial reporting as of December 31, 2022.
The Company recognizes this error as a material weakness and has begun to establish controls to support assessment and review of accounting
for complex financial instruments.
Purchasers
who purchase our securities in this offering pursuant to a securities purchase agreement may have rights not available to purchasers
that purchase without the benefit of a securities purchase agreement.
In
addition to rights and remedies available to all purchasers in this offering under federal securities and state law, the purchasers that
enter into a securities purchase agreement will also be able to bring claims of breach of contract against us. The ability to pursue
a claim for breach of contract provides those investors with the means to enforce the covenants uniquely available to them under the
securities purchase agreement including: (i) timely delivery of shares; (ii) agreement to not enter into variable rate financings for
one year from closing, subject to certain exceptions; (iii) agreement to not enter into any financings for forty-five (45) days
from closing; and (iv) indemnification for breach of contract.
Unstable
market and economic factors could adversely affect our business, financial condition or results of operations.
Uncertain
or unfavorable global economic or market conditions, such as a recession, an economic slowdown, inflation or reduced growth rates, could
significantly impact our operating results or lead to significant reductions in funding sources available to the Company, which could
adversely affect our business, results of operations or financial condition. Our operations have required substantial amounts of cash
since inception. We expect to continue to spend substantial amounts to continue the development of our GD-T cell-based therapeutic candidates,
including for future clinical trials. In the event of unstable markets and unfavorable market conditions, we cannot be certain that additional
funding will be available on acceptable terms, or at all. We have no committed source of additional capital. If we are unable to raise
additional capital in sufficient amounts or on terms acceptable to us, we may have to significantly delay, scale back or discontinue
the development or commercialization of our GD-T cell-based therapeutic candidates or other research and development initiatives. Furthermore,
uncertain or unfavorable global economic or market conditions may cause our manufacturers, suppliers, distributors, contractors, logistics
providers and other external business partners to suffer financial or operational difficulties, which could impact their ability to provide
us with or distribute finished product, raw and packaging materials or services in a timely manner or at all. We could also face difficulty
collecting or recovering accounts receivables from third parties facing financial or operational difficulties.
DIVIDEND
POLICY
Since
inception, we have not declared or paid any dividends on our ordinary shares. We do not have any current plans to pay any dividends on
our ordinary shares, including those represented by ADSs, in the foreseeable future. We intend to retain all our available funds and
any future earnings to operate and expand our business. Because we do not anticipate paying any cash dividends in the foreseeable future.
Capital appreciation, if any, will be your sole source of gains, and you may never receive a return on your investment.
The
determination to pay dividends, if any, will be made at the discretion of our board of directors and may be based on a number of factors,
including our future operations and earnings, capital requirements and surplus, general financial condition, contractual and legal restrictions
and other factors that the board of directors may deem relevant.
Under
current Scottish law, among other things, a company’s accumulated realized profits must exceed its accumulated realized losses
(on a non-consolidated basis) before dividends can be paid. Accordingly, we may only pay dividends if we have sufficient distributable
reserves (on a non-consolidated basis), which are our accumulated realized profits that have not been previously distributed or capitalized
less our accumulated realized losses, so far as such losses have not been previously written off in a reduction or reorganization of
capital.
USE
OF PROCEEDS
We
estimate that our net proceeds from this offering will be approximately $4.2 million, after deducting estimated offering
expenses of approximately $0.8 million (based on an assumed public offering price per ADS of $0.505 per ADS and
accompanying warrant, which was the last reported sales price of our ADSs on The Nasdaq Capital Market on August 22,
2023.). However, because this is a best efforts offering and there is no minimum offering amount required as a condition to the
closing of this offering, the actual offering amount, the placement agent’s fees and net proceeds to us are not presently
determinable and may be substantially less than the maximum amounts set forth on the cover page of this prospectus.
If all of the
Placement Agent warrants to purchase 742,574 ADSs issued in connection with this offering are fully exercised for cash, we would receive
additional aggregate proceeds of approximately $0.47 million.
We
intend to use the net proceeds of this offering to support the submission of our Investigational New Drug (IND) Application in respect
of our upcoming clinical trial, and for continuing operating expenses and working capital.
With existing resources
and the funding received as a result of this offering, we expect to be able to fund current operations to December 2023, through the
period in which the Company is targeting the submission on the IND and subsequent anticipated acceptance. The reaching of this value
inflection point is reasonably expected to provide support for additional funding initiatives.
As described
in our 20-F and in common with many clinical development stage biotechnology companies our future liquidity needs, and ability to address
them, will largely be determined by the availability of capital, both generally and in particular to fund our product candidates and
key development and regulatory projects. As a pre-revenue biotechnology company, we have financed our operations though continuously
raising capital; and we expect to continue having to raise capital routinely on the capital markets, taking advantage of our public listing.
We are constantly formulating and implementing potential funding initiatives to ensure we have adequate working capital . These initiatives
could be in the form of further equity raises, as noted earlier and/or non-dilutive financings arising from collaborations or licensing
arrangements.
The
amounts and timing of our actual expenditures will depend on numerous factors, including the progress of our clinical trials, the potential
for achieving accelerated regulatory approval and the amount of cash used in our operations. We therefore cannot estimate with certainty
the amount of net proceeds to be used for the purposes described above. We may find it necessary or advisable to use the net proceeds
for other purposes, and we will have broad discretion in the application of the net proceeds. Our shareholders may not agree with the
manner in which our management chooses to allocate and spend the net proceeds. Moreover, our management may use the net proceeds for
corporate purposes that may not result in our being profitable or increase our market value.
Pending
these uses, we plan to invest these net proceeds in short-term, interest-bearing obligations; investment-grade instruments; certificates
of deposit; or direct or guaranteed obligations of the United States and the United Kingdom. The goal with respect to the investment
of these net proceeds is capital preservation and liquidity so that such funds are readily available to fund our operations. Our investment
positions will also take into consideration the law and rules under the U.S. Investment Company Act of 1940, so as to avoid being characterized
as an investment company thereunder.
CAPITALIZATION
You
should read this information together with our 2022 audited consolidated financial statements and related notes of TC BioPharm (Holdings)
plc included in our 2022 Form 20-F annual report and incorporated by reference in this prospectus and the information set forth under
the sections titled “Use of Proceeds” and “Item 5. Operating and Financial Review and Prospects” included in
our 2022 Form 20-F annual report and incorporated by reference.
The
following table sets forth our cash and cash equivalents, indebtedness and capitalization as of December 31, 2022, on:
(1)
a pro forma basis to give effect to (i) the accrued interest on the Convertible Loan Notes outstanding at December 31, 2022 up to
the point of conversion to equity; (ii) the issue of 1,590,130 Ordinary Shares on conversion of Convertible Loan Notes (including the
interest accrued to the date of conversion) pursuant to the provisions of such notes; (iii) the sale of 215,000 ADSs and pre-funded Warrants
for 3,222,500 ADSs in March 2023, (vii) the issue of Series C Ordinary Warrants to purchase up to 3,437,500 ADSs in March 2023 and the
repricing of Series A Ordinary Warrants and Series B Ordinary Warrants; (viii) the subsequent exercise of pre-funded Warrants for 4,537,500
ADSs; and (ix) the movement in the fair value of the Warrants outstanding as at August 22, 2023.
(2)
a pro forma basis as adjusted to give effect to the sale of 9,900,990 ADSs (assuming no sale of pre-funded warrants) pursuant
to this prospectus at an assumed public offering price of $0.505 per one ADS in this offering and after deducting the underwriting
discounts and commissions and estimated offering expenses payable by us.
The
pro forma as adjusted calculations assume a public offering price of $0.505 per one ADS after deducting the estimated underwriting
discounts and commissions and estimated offering expenses payable by us.
| | |
As
of December 31, 2022 | |
| | |
Actual
(1) | | |
Pro
Forma (2) | | |
Pro Forma
As Adjusted (3) | |
| | |
(in
thousands, except share and per share data) | |
| | |
£ | | |
$ | | |
$ | | |
$ | |
| Cash
and cash equivalents | |
| 4,808 | | |
| 5,807 | | |
| 10,697 | | |
| 14,886 | |
| | |
| | | |
| | | |
| | | |
| | |
| Convertible
Loan Notes | |
| 656 | | |
| 792 | | |
| - | | |
| - | |
| Warrant
liabilities [1] | |
| 6,021 | | |
| 7,271 | | |
| 1,433 | | |
| 1,433 | |
| Warrant liabilities – re offering | |
| - | | |
| - | | |
| - | | |
| 257 | |
| | |
| | | |
| | | |
| | | |
| | |
| Total
equity attributable to equity holders: | |
| | | |
| | | |
| | | |
| | |
| Ordinary
shares, 949,958 shares authorized, issued and outstanding, actual; £0.0001 par value 7,292,588 shares authorized, issued
and outstanding, pro forma; £0.0001 par value 17,193,578 shares authorized, issued and outstanding, pro forma as adjusted | |
| - | | |
| - | | |
| 1 | | |
| 2 | |
| Deferred
shares, £0.4999; 794,955 shares authorized, issued and outstanding, pro forma. | |
| 397 | | |
| 480 | | |
| 480 | | |
| 480 | |
| Share
premium | |
| 16,598 | | |
| 20,045 | | |
| 21,836 | | |
| 25,767 | |
| Other
reserves | |
| 16,711 | | |
| 20,182 | | |
| 20,182 | | |
| 20,182 | |
| Accumulated
deficit | |
| (33,732 | ) | |
| (40,738 | ) | |
| (31,078 | ) | |
| (31,078 | ) |
| Total
shareholders’ equity | |
| (26 | ) | |
| (31 | ) | |
| 11,421 | | |
| 15,353 | |
| | |
| | | |
| | | |
| | | |
| | |
| Total
capitalization | |
| 6,651 | | |
| 8,032 | | |
| 12,853 | | |
| 17,043 | |
[1]
Represents the fair value as of August 22, 2023 of the Warrants to purchase up to 7,283,037 ADSs.
The
number of our ordinary shares (including shares represented by ADSs) to be outstanding after this offering is based on 7,292,588 ordinary
shares outstanding as of August 28, 2023 and excludes:
| |
● |
106,585
ordinary shares issuable upon the exercise of options outstanding under our 2014 Share Option Scheme as of December 31, 2022, with
a weighted-average exercise price of £23.00 per share; |
| |
|
|
| |
● |
52,305
ordinary shares issuable upon the exercise of options outstanding under our 2021 Share Option Scheme, as of December 31, 2022, with
a weighted-average exercise price of $212.00 per share; and |
| |
|
|
| |
● |
15,891
ordinary shares issuable upon the exercise of options outstanding, at a future date based on the achievement of certain clinical
and commercial milestones with an exercise price of £215.00 per share. |
For
the description of the 2014 Share Option Scheme and 2021 Share Option Scheme see “Item 6.E Share Ownership” in the 2022 Form
20-F, which is incorporated by reference herein.
Unless
otherwise stated, all information in this prospectus assumes no exercise of the outstanding options described above into ordinary shares
or ADSs, treats all restricted shares issued with outstanding restrictions to be vested as issued and outstanding shares, no exercise
of the Placement Agent Warrants issued in this offering and no sale of pre-funded warrants in this offering.
Except
as otherwise indicated all references to our articles of association in this prospectus refer to our articles of association, as amended
as currently in force for TC BioPharm (Holdings) plc at the date of this prospectus.
To
the extent these outstanding options or any newly issued options are exercised, or we issue additional ordinary shares in the future,
there will be further dilution to the new investors purchasing ordinary shares represented by ADSs in this offering. In addition, we
may choose to raise additional capital because of market conditions or strategic considerations, even if we believe that we have sufficient
funds for our current or future operating plans. If we raise additional capital through the sale of equity or convertible debt securities,
the issuance of these securities could result in further dilution to our shareholders.
DILUTION
If
you invest in our ADSs in this offering, your ownership interest of our ordinary shares will be immediately diluted to the extent of
the difference between the public offering price per ADS in this offering and the pro forma as adjusted net tangible book value per ADS
after this offering. For the purposes of calculating the potential impact of dilution, the full value of an assumed offering price of
$0.505 per ADS has been ascribed to the ADSs. Dilution results from the fact that the public offering price per ADS is substantially
in excess of the net tangible book value per ADS.
As
of December 31, 2022, we had a historical net tangible book value of £0.6 million, or £0.61 per ADS (equivalent
to $0.74 per ADS). Our net tangible book value per ADS represents total tangible assets less total liabilities, divided
by the number of ordinary shares outstanding on December 31, 2022.
After
giving further effect to (i) the accrued interest on the Convertible Loan Notes outstanding at December 31, 2022 up to the point of
conversion to equity; (ii) the issue of 1,590,130 Ordinary Shares on conversion of Convertible Loan Notes (including the interest accrued
to the date of conversion) pursuant to the provisions of such notes; (iii) the sale of 215,000 ADSs and pre-funded Warrants for 3,222,500
ADSs in March 2023, (iv) the issue of Series C Ordinary Warrants to purchase up to 3,437,500 ADSs in March 2023 and the repricing of
Series A Ordinary Warrants and Series B Ordinary Warrants; (v) the subsequent exercise of pre-funded Warrants for 4,537,500 ADSs; and
(vi) the movement in the fair value of the Warrants outstanding as at August 22, 2023; and (vii) 9,900,990 ADSs in this offering
(assuming no sale of pre-funded warrants) at an assumed offering price of $0.505 per ADS and after deducting the underwriting
discounts and commissions and estimated offering expenses payable by us, our as adjusted net tangible book value at December 31, 2022
would have been $14.7 million, or £12.2 million and $0.85 per share, or £0.71 per share. This
represents an immediate decrease in pro forma as adjusted net tangible book value of $0.62 per ordinary share to existing investors
and immediate dilution of $0.35 per ADS to new investors attributable to this offering. The following table illustrates this dilution
to new investors purchasing ADSs in this offering:
| Assumed public sale price per ADS | |
| | | |
$ | 0.505 | |
| Historical net tangible book value per ADS
as at December 31, 2022 | |
$ | (0.74 | ) | |
| | |
| Increase
in net tangible book value per ADS attributable to transactions in the period to August 22, 2023, as described above | |
| 2.21 | | |
| | |
| Pro forma net tangible book value per ADS as of December 31, 2022 | |
| 1.47 | | |
| | |
| Decrease in net tangible book value per ADS
attributable to this offering | |
| (0.62 | ) | |
| | |
| Pro forma as adjusted
net tangible book value per ADS after this offering | |
| | | |
| 0.85 | |
| Dilution per ADS to
new investors purchasing ADSs in this offering | |
| | | |
$ | (0.35 | ) |
The dilution information discussed
above is illustrative only and will change based on the actual offering price and other terms of this offering determined at pricing.
A $1.00 increase (decrease) in the assumed offering price of $0.505 per ADS, which is the price set forth on the cover page of
this prospectus, would increase (decrease) our pro forma as adjusted net tangible book value as of December 31, 2022, after this offering
by $0.52 per ADS, and would increase (decrease) dilution to new investors by $0.52 per ADS, assuming that the number
of ADSs offered by us, as set forth on the cover page of this prospectus, remains the same. An increase of 1,000,000 in the number of
ADSs we are offering would increase our pro forma as adjusted net tangible book value as of December 31, 2022, after this offering by
$0.02 per ADS, and would decrease dilution to new investors by $0.02 per ADS, assuming the assumed offering price per
ADS remains the same. A decrease of 1,000,000 in the number of ADSs we are offering would decrease our pro forma as adjusted net tangible
book value as of December 31, 2022, after this offering by $0.03 per ordinary share, and would increase dilution to new investors
by $0.03 per ADS, assuming the assumed offering price per ADS remains the same.
The
number of our ordinary shares (including shares represented by ADSs) to be outstanding after this offering is based on 7,292,588 ordinary
shares outstanding as of August 28, 2023 and excludes:
| |
● |
106,585
ordinary shares issuable upon the exercise of options outstanding under our 2014 Share Option Scheme as of December 31, 2022, with
a weighted-average exercise price of £23.00 per share; |
| |
|
|
| |
● |
52,305
ordinary shares issuable upon the exercise of options outstanding under our 2021 Share Option Scheme, as of December 31, 2022, with
a weighted-average exercise price of $212.00 per share; and |
| |
|
|
| |
● |
15,891
ordinary shares issuable upon the exercise of options outstanding, at a future date based on the achievement of certain clinical
and commercial milestones with an exercise price of £215.00 per share. |
Unless otherwise stated, all
information in this prospectus assumes no exercise of the outstanding options described above into ordinary shares or ADSs, treats all
restricted shares issued with outstanding restrictions to be vested as issued and outstanding shares, no exercise of the Placement Agent
Warrants issued in this offering and no sale of pre-funded warrants in this offering.
MATERIAL
INCOME TAX CONSIDERATIONS
The
following summary contains a description of material U.K. and U.S. federal income tax consequences of the acquisition, ownership and
disposition of our ordinary shares. This summary should not be considered a comprehensive description of all the tax considerations that
may be relevant to the decision to acquire our ordinary shares.
U.S.
Federal Income Taxes
The
following is a summary of the material U.S. federal income tax consequences to U.S. Holders (as defined below) of purchasing, owing and
disposing of the ordinary shares or ADSs. This discussion is included for general informational purposes only, does not purport to consider
all aspects of U.S. federal income taxation that might be relevant to a U.S. Holder, and does not constitute, and is not, a tax opinion
for or tax advice to any particular U.S. Holder of ordinary shares or the ADSs. The summary does not address any U.S. tax matters other
than those specifically discussed. The summary is based on the provisions of the U.S. Internal Revenue Code of 1986, as amended (the
“Code”), existing, temporary and proposed Treasury Regulations issued thereunder, judicial decisions and administrative
rulings and pronouncements and other legal authorities, all as of the date hereof and all of which are subject to change, possibly with
retroactive effect. Any such change could alter the tax consequences described herein.
The
discussion below applies only to U.S Holders as capital assets within the meaning of Section 1221 of the Code (generally, property held
for investment), and does not address the tax consequences that may be relevant to U.S. Holders who, in light of their particular circumstances,
may be subject to special tax rules, including without limitation:
| ● |
insurance
companies, tax-exempt organizations, regulated investment companies, real estate investment trusts, brokers or dealers in securities
or foreign currencies, banks and other financial institutions, mutual funds, retirement plans, traders in securities that elect to
mark to market, certain former U.S. citizens or long-term residents; |
| |
|
| ● |
U.S.
Holders that are classified for U.S. federal income tax purposes as partnerships and other pass-through entities and investors therein; |
| |
|
| ● |
U.S.
Holders who hold ordinary shares or ADSs as part of a hedge, straddle, constructive sale, conversion, or other integrated or risk-reduction
transaction, as “qualified small business stock,” within the meaning of Section 1202 of the Code or as Section 1244 stock
for purposes of the Code; |
| |
|
| ● |
U.S.
Holders who hold ordinary shares or ADSs through individual retirement or other tax-deferred accounts; |
| |
|
| ● |
U.S.
Holders that have a functional currency other than the U.S. dollar; |
| |
|
| ● |
U.S.
Holders who are subject to the alternative minimum tax provisions of the Code or the Medicare surtax of 3.8% on net investment income
imposed by Section 1411 of the Code; |
| |
|
| ● |
U.S.
Holders who acquire their ordinary shares or ADSs pursuant to any employee share option or otherwise as compensation; |
| |
|
| ● |
U.S.
Holders required to accelerate the recognition of any item of gross income with respect to their ordinary shares or ADSs as a result
of such income being recognized on an applicable financial statement; or |
| ● |
U.S.
Holders who hold or held, directly or indirectly, or are treated as holding or having held under applicable constructive attribution
rules, 10% or more of the ordinary shares or ADSs of the company, measured by voting power or value. |
Any
such U.S. Holders should consult their own tax advisors.
For
purposes of this discussion, a “U.S. Holder” means a holder of our ordinary shares or ADSs that is or is treated as, for
U.S. federal income tax purposes,
| (i) |
an
individual citizen or resident of the United States; |
| (ii) |
a
corporation (or other entity taxable as a corporation for U.S. federal income tax purposes) created or organized in or under the
laws of the United States, any State thereof or the District of Columbia or any entity treated as such for U.S. federal income tax
purposes; |
| (iii) |
an
estate the income of which is subject to U.S. federal income taxation regardless of its source, or |
| (iv) |
a
trust (A) the administration over which a U.S. court exercises primary supervision and all of the substantial decisions of which
one or more U.S. persons have the authority to control, or (B) that has a valid election in effect under the applicable Treasury
Regulations to be treated as a U.S. person under the Code. |
If
a partnership or other pass-through entity (including any entity or arrangement treated as such for purposes of U.S. federal income tax
law) holds our ordinary shares or ADSs, the tax treatment of a partner of such partnership or member of such entity will generally depend
upon the status of the partner and the activities of the partnership. Partnerships and other pass-through entities holding our ordinary
shares or ADSs, and any person who is a partner or member of such entities should consult their own tax advisors regarding the tax consequences
of purchasing, owning and disposing of the ordinary shares or ADSs.
Passive
Foreign Investment Company Considerations
A
non-U.S. corporation, such as TCB, will be classified as a passive foreign investment company, or PFIC, for U.S. federal income tax purposes,
if, in the case of any particular taxable year, either (i) 75% or more of its gross income for such taxable year consists of certain
types of “passive” income or (ii) 50% or more of the value of its assets (based on an average of the quarterly values of
the assets) during such taxable year is attributable to assets that produce or are held for the production of passive income. For this
purpose, cash is categorized as a passive asset and the company’s un-booked intangibles associated with active business activities
may generally be classified as active assets. Passive income generally includes, among other things, dividends, interest, rents, royalties,
and gains from the disposition of passive assets. For this purpose, a foreign corporation will be treated as owning its proportionate
share of the assets and earning its proportionate share of the income of any other non-U.S. corporation in which it owns, directly or
indirectly, more than 25% (by value) of the stock.
Based
upon its current income and assets and projections as to the value of the ordinary shares or ADSs, it is not presently expected that
we will be classified as a PFIC for the 2022 taxable year or the foreseeable future.
The
determination of whether we will be or become a PFIC will depend upon the composition of its income (which may differ from our historical
results and current projections) and assets and the value of its assets from time to time, including, in particular the value of its
goodwill and other un-booked intangibles (which may depend upon the market value of the ordinary shares or ADSs from time to time and
may be volatile). Among other matters, if our market capitalization is less than anticipated or subsequently declines, we may be classified
as a PFIC for the taxable year in the 2021 taxable year or future taxable years. It is also possible that the IRS may challenge the classification
or valuation of our assets, including its goodwill and other unbooked intangibles, or the classification of certain amounts received
by us, including interest earnings, which may result in our being, or becoming classified as, a PFIC for the taxable year in 2021 or
future taxable years.
The
determination of whether we will be or become a PFIC may also depend, in part, on how, and how quickly, it uses liquid assets and the
cash proceeds of this offering or otherwise. If we were to retain significant amounts of liquid assets, including cash, the risk of our
being classified as a PFIC may substantially increase. Because there are uncertainties in the application of the relevant rules and PFIC
status is a factual determination made annually after the close of each taxable year, there can be no assurance that we will not be a
PFIC for the 2022 taxable year or any future taxable year, and no opinion of counsel has or will be provided regarding the classification
of us as a PFIC. If we were classified as a PFIC for any year during which a holder held our ordinary shares or ADSs, it generally would
continue to be treated as a PFIC for all succeeding years during which such holder held the ordinary shares or ADSs. The discussion below
under “—Dividends Paid on Ordinary Shares or ADSs” and “—Sale or Other Disposition of Ordinary Shares or
ADS” is written on the basis that we will not be classified as a PFIC for U.S. federal income tax purposes.
Dividends
Paid on Ordinary Shares including ordinary shares represented by ADSs
Subject
to the PFIC rules described below, any cash distributions (including constructive distributions) paid on the ordinary shares including
ordinary shares represented by ADSs out of our current or accumulated earnings and profits, as determined under U.S. federal income tax
principles, will generally be includible in the gross income of a U.S. Holder as dividend income on the day actually or constructively
received by the U.S. Holder, in the case of ordinary shares including ordinary shares represented by ADSs. Because we do not intend to
determine our earnings and profits on the basis of U.S. federal income tax principles, any distribution will generally be treated as
a “dividend” for U.S. federal income tax purposes. Under current law, a non-corporate recipient of a dividend from a “qualified
foreign corporation” will generally be subject to tax on the dividend income at the lower applicable net capital gains rate rather
than the marginal tax rates generally applicable to ordinary income provided that certain holding period and other requirements are met.
A
non-U.S. corporation (other than a corporation that is classified as a PFIC for the taxable year in which the dividend is paid or the
preceding taxable year) will generally be considered to be a qualified foreign corporation (i) if it is eligible for the benefits of
a comprehensive tax treaty with the United States which the Secretary of Treasury of the United States determines is satisfactory for
these purposes and which includes an exchange of information program, or (ii) with respect to any dividend paid by such corporation on
its stock, if such stock is readily tradable on an established securities market in the United States. We believe we are eligible for
the benefits of the Convention Between the Government of the United States of America and the Government of the United Kingdom of Great
Britain and Northern Ireland for the Avoidance of Double Taxation and the Prevention of Fiscal Evasion with Respect to Taxes on Income
and On Capital Gains, or the United States-United Kingdom income tax treaty (which the Secretary of the Treasury of the United States
has determined is satisfactory for this purpose and includes an exchange of information program), in which case it would be treated as
a qualified foreign corporation with respect to dividends paid on the ordinary shares or ADSs. U.S. Holders are urged to consult their
tax advisors regarding the availability of the reduced tax rate on dividends in their particular circumstances. Dividends received on
the ordinary shares will not be eligible for the dividends received deduction allowed to corporations.
Sale
or Other Disposition of Ordinary Shares or ADSs
Subject
to the PFIC rules discussed below, a U.S. Holder of our ordinary shares or ADSs will generally recognize capital gain or loss, if any,
upon the sale or other disposition of ordinary shares or ADSs, respectively, in an amount equal to the difference between the amount
realized upon the disposition and the U.S. Holder’s adjusted tax basis in such ordinary shares or ADSs. Any capital gain or loss
will be long-term capital gain or loss if the ordinary shares or ADSs have been held for more than one year and will generally be United
States source capital gain or loss for United States foreign tax credit purposes. Long-term capital gains of non-corporate taxpayers
are currently eligible for reduced rates of taxation.
Disposition
of Foreign Currency
U.S.
Holders are urged to consult their tax advisors regarding the tax consequences of receiving, converting or disposing of any non-U.S.
currency received as dividends on our ordinary shares or ADSs.
Tax
on Net Investment Income
A
U.S. Holder may be subject to a Medicare surtax of 3.8% on some or all of such U.S. Holder’s “net investment income”
as defined in Section 1411 of the Code. Net investment income generally includes income from the ordinary shares or ADSs unless such
income is derived in the ordinary course of the conduct of a trade or business (other than a trade or business that consists of certain
passive or trading activities). You should consult your tax advisors regarding the effect this Medicare tax may have, if any, on your
acquisition, ownership or disposition of ordinary shares or ADSs.
Passive
Foreign Investment Company Rules
If
we are is classified as a PFIC for any taxable year during which a U.S. Holder holds our ordinary shares or ADSs, unless the holder makes
a mark-to-market election (as described below), the holder will, except as discussed below, be subject to special tax rules that have
a penalizing effect, regardless of whether we remain a PFIC, on (i) any excess distribution that we make to the holder (which generally
means any distribution paid during a taxable year to a holder that is greater than 125% of the average annual distributions paid in the
three preceding taxable years or, if shorter, the holder’s holding period for the ordinary shares or ADSs), and (ii) any gain realized
on the sale or other disposition, including, under certain circumstances, a pledge, of our ordinary shares or ADSs. Under the PFIC rules:
| |
● |
The excess distribution
and/or gain will be allocated ratably over the U.S. Holder’s holding period for the ordinary shares or ADSs; |
| |
|
|
| |
● |
The amount of the excess
distribution or gain allocated to the taxable year of the distribution or disposition and any taxable years in the U.S. Holder’s
holding period prior to the first taxable year in which we are classified as a PFIC, or a pre-PFIC year, will be taxable as ordinary
income; and |
| |
|
|
| |
● |
The amount of the excess
distribution or gain allocated to each taxable year other than the taxable year of the distribution or disposition or a pre-PFIC
year, will be subject to tax at the highest tax rate in effect applicable to the individuals or corporations, and the interest charge
generally applicable to underpayments of tax will be imposed on the resulting tax attributable to each such year. |
If
we are a PFIC for any taxable year during which a U.S. Holder holds our ordinary shares or ADSs and any of its non-U.S. subsidiaries
is also a PFIC, such holder would be treated as owning a proportionate amount (by value) of the shares of the lower-tier PFIC for purposes
of the application of these rules. Each U.S. Holder is advised to consult its tax advisors regarding the application of the PFIC rules
to any of our subsidiaries.
As
an alternative to the foregoing rules, a U.S. Holder of “marketable stock” in a PFIC may make a mark-to-market election with
respect to such ordinary shares or ADSs, provided that they are “regularly traded” (as specially defined under the Code)
on The Nasdaq Stock Market. No assurances may be given regarding whether the ordinary shares or ADSs will qualify, or will continue to
be qualified, as being regularly traded in this regard. If a mark-to-market election is made, the U.S. Holder will generally (i) include
as ordinary income for each taxable year that we are a PFIC the excess, if any, of the fair market value of ordinary shares or ADSs held
at the end of the taxable year over the adjusted tax basis of such securities and (ii) deduct as an ordinary loss the excess, if any,
of the adjusted tax basis of such securities over the fair market value of such securities held at the end of the taxable year, but only
to the extent of the net amount previously included in income as a result of the mark-to-market election. The U.S. Holder’s adjusted
tax basis in the ordinary shares or ADSs would be adjusted to reflect any income or loss resulting from the mark-to-market election.
If a U.S. Holder makes an effective mark-to-market election, in each year that we are a PFIC any gain recognized upon the sale or other
disposition of the ordinary shares or ADSs will be treated as ordinary income and loss will be treated as ordinary loss, but only to
the extent of the net amount previously included in income as a result of the mark-to-market election. U.S. Holders of our ordinary shares
or ADSs should consult their tax advisors regarding the availability of a mark-to-market election with respect to such ordinary shares
or ADSs.
If
a U.S. Holder makes a mark-to-market election in respect of a corporation classified as a PFIC and such corporation ceases to be classified
as a PFIC, the holder will not be required to take into account the mark-to-market gain or loss described above during any period that
such corporation is not classified as a PFIC.
Because
a mark-to-market election cannot be made for any lower-tier PFICs that a PFIC may own, a U.S. Holder who makes a mark-to-market election
with respect to the ordinary shares or ADSs may continue to be subject to the general PFIC rules with respect to such holder’s
indirect interest in any of our non-U.S. subsidiaries that is classified as a PFIC.
We
do not intend to provide information necessary for U.S. Holder’s to make qualified electing fund elections, which, if available,
would result in tax treatment different from the general tax treatment for PFICs described above. However, as described above under “Passive
Foreign Investment Company Considerations-PFIC Classification of TCB,” it is not presently expected that we will be classified
as a PFIC for the 2022 taxable year or the foreseeable future.
As
discussed above under “Dividends Paid on Ordinary Shares or ADSs”, dividends that we pay on the ordinary shares or ADSs will
not be eligible for the reduced tax rate that applies to qualified dividend income if we are classified as a PFIC for the taxable year
in which the dividend is paid or the preceding taxable year. In addition, if a U.S. Holder owns the ordinary shares or ADSs during any
taxable year that we are a PFIC, the holder must file an annual information return with the IRS. Each holder is urged to consult its
tax advisor concerning the U.S. federal income tax consequences of purchasing, holding, and disposing ordinary shares or ADSs if we are
or become a PFIC, including the possibility of making a mark-to-market election and the unavailability of the qualified electing fund
election.
Information
reporting and backup withholding
Certain
U.S. Holders are required to report information to the IRS relating to an interest in “specified foreign financial assets,”
including shares issued by a non-U.S. corporation, for any year in which the aggregate value of all specified foreign financial assets
exceeds $50 thousand (or a higher U.S. dollar amount prescribed by the IRS), subject to certain exceptions (including an exception for
shares held in custodial accounts maintained with a United States financial institution). These rules also impose penalties if a holder
is required to submit such information to the IRS and fails to do so.
In
addition, U.S. Holders may be subject to information reporting to the IRS and backup withholding with respect to dividends on and proceeds
from the sale or other disposition of our ordinary shares or ADSs. Information reporting will apply to payments of dividends on, and
to proceeds from the sale or other disposition of, our ordinary shares or ADSs by a paying agent within the United States to a holder,
other than holders that are exempt from information reporting and properly certify their exemption. A paying agent within the United
States will be required to withhold at the applicable statutory rate, currently 24%, in respect of any payments of dividends on, and
the proceeds from the disposition of, our ordinary shares or ADSs within the U.S. to a U.S. Holder (other than holders that are exempt
from backup withholding and properly certify their exemption) if the holder fails to furnish its correct taxpayer identification number
or otherwise fails to comply with applicable backup withholding requirements. U.S. Holders who are required to establish their exempt
status generally must provide a properly completed IRS Form W-9.
Backup
withholding is not an additional tax. Amounts withheld as backup withholding may be credited against a holder’s U.S. federal income
tax liability. A U.S. Holder generally may obtain a refund of any amounts withheld under the backup withholding rules by filing the appropriate
claim for refund with the IRS in a timely manner and furnishing any required information. Each U.S. Holder is advised to consult with
its tax advisor regarding the application of the United States information reporting rules to their particular circumstances.
Material
United Kingdom Tax Considerations
The
following is a description of the material U.K. tax considerations relating primarily to the ownership and disposal of our ordinary shares
or ADSs by the U.S. Holders described above. The U.K. tax comments set out below are based on current U.K. tax law as applied in Scotland,
and HMRC practice (which may not be binding on HMRC) as at the date of this summary, both of which are subject to change, possibly with
retrospective effect. They are intended as a general guide and, save where otherwise stated, only apply to you if you are not resident
in the U.K. for U.K. tax purposes and do not hold our ordinary shares or ADSs for the purposes of a trade, profession or vocation that
you carry on in the U.K. through a branch, agency or permanent establishment in the U.K. and if you hold our ordinary shares as an investment
for U.K. tax purposes and are not subject to special rules.
This
summary does not address all possible tax consequences relating to an investment in our ordinary shares or ADSs. In particular it does
not cover the U.K. inheritance tax consequences of holding our ordinary shares or ADSs. It assumes that the depositary or DTC has not
made an election under section 97A(1) of the Finance Act 1986. It assumes that we do not (and will not at any time) derive 75% or more
of our qualifying asset value, directly or indirectly, from U.K. land, and that we are and remain solely resident in the U.K. for tax
purposes. It assumes that the holder is not our officer or our employee (or of any related company of ours) and has not (and is not deemed
to have) acquired the ordinary shares or ADSs by virtue of an office or employment. It assumes that a holder of ordinary shares or ADSs
is the beneficial owner of the underlying ordinary shares for U.K. tax purposes. This summary is for general information only and is
not intended to be, nor should it be considered to be, legal or tax advice to any particular holder. Holders of our ordinary shares or
ADSs are strongly urged to consult their tax advisers in connection with the U.K. tax consequences of their investment in our securities.
U.K.
Taxation of Dividends and Distributions
We
will not be required to withhold amounts for or on account of U.K. tax at source when paying a dividend or distribution in respect of
our ordinary shares.
Individual
holders who hold our ordinary shares as an investment, who are not resident in the U.K. for U.K. tax purposes should not be subject to
U.K. income tax in respect of any dividends on our ordinary shares, unless they hold their ordinary shares in connection with any trade,
profession or vocation carried on (whether solely or in partnership) by them in the U.K. through a branch, agency or permanent establishment
in the U.K.. In these circumstances, such holder may, depending on his or her individual circumstances, be chargeable to U.K. income
tax in respect of our dividends.
Corporate
holders which are not resident in the U.K. for U.K. tax purposes should not be subject to U.K. corporation tax in respect of any dividends
on our ordinary shares, unless they carry on a trade in the U.K. through a permanent establishment to which the ordinary shares are attributable.
In these circumstances, such holders may, depending on their individual circumstances and if an exemption from U.K. corporation tax in
respect of dividend payments does not apply, be chargeable to U.K. corporation tax in respect of our dividends.
U.K.
Taxation of Capital Gains
An
individual holder who is not resident in the U.K. for U.K. tax purposes should not be liable to U.K. capital gains tax on capital gains
realized on the disposal of their ordinary shares unless such holder carries on (whether solely or in partnership) a trade, profession
or vocation in the U.K. through a branch or agency in the U.K. to which our ordinary shares are attributable. In these circumstances,
such holder may, depending on his or her individual circumstances, be chargeable to U.K. capital gains tax on chargeable gains arising
from a disposal of his or her ordinary shares.
Any
such individual holder of our ordinary shares who is temporarily non-resident for U.K. tax purposes will, in certain circumstances, become
liable to U.K. tax on capital gains in respect of gains realized while they were not resident in the U.K.
A
corporate holder of our ordinary shares which is not resident in the U.K. for U.K. tax purposes should not be liable for U.K. corporation
tax on chargeable gains realized on the disposal of our ordinary shares unless it carries on a trade in the U.K. through a permanent
establishment in the U.K. to which our ordinary shares are attributable. In these circumstances, a disposal of ordinary shares by such
holder may give rise to a chargeable gain or an allowable loss for the purposes of U.K. corporation tax.
Stamp
Duty and Stamp Duty Reserve Tax
The
discussion below relates to the holders of our ordinary shares or ADSs wherever resident, however it should be noted that special rules
may apply to certain persons such as market makers, brokers, dealers or intermediaries.
As
a general rule (and except in relation to depositary receipt systems and clearance services (as to which see below)), no UK stamp duty
or stamp duty reserve tax, or SDRT, is payable on the issue of the ordinary shares underlying the ADSs.
An
unconditional agreement to transfer ordinary shares will normally give rise to a charge to SDRT at the rate of 0.5% of the amount or
value of the consideration payable for the transfer. The purchaser of the shares is liable for the SDRT. Transfers of ordinary shares
in certificated form are generally also subject to stamp duty at the rate of 0.5% of the amount or value of the consideration given for
the transfer (rounded up to the next £5.00). Stamp duty is normally paid by the purchaser. The charge to SDRT will be cancelled
or, if already paid, repaid (generally with interest), where a transfer instrument has been duly stamped within six years of the charge
arising, (either by paying the stamp duty or by claiming an appropriate relief) or if the instrument is otherwise exempt from stamp duty.
Under
current UK legislation, an issue or transfer of ordinary shares or an unconditional agreement to transfer ordinary shares to a clearance
service or a depositary receipt system (including to a nominee or agent for, a person whose business is or includes the issue of depositary
receipts or the provision of clearance services) will generally be subject to SDRT (and, in the case of transfers, where the transfer
is effected by a written instrument, stamp duty) at a higher rate of 1.5% of the amount or value of the consideration given for the transfer
or, in certain circumstances, the value of the ordinary shares unless the clearance service has made and maintained an election under
section 97A of the UK Finance Act 1986, or a section 97A election. It is understood that HMRC regards the facilities of DTC as a clearance
service for these purposes and we are not aware of any section 97A election having been made by the DTC.
However,
based on current published HMRC practice following European Union case law in respect of the European Council Directives 69/335/EEC and
2009/7/EC, no SDRT is generally payable in respect of such an issue of ordinary shares and no SDRT or stamp duty is generally payable
in respect of such a transfer of ordinary shares where such transfer is an integral part of an issue of share capital. It is noted that
on January 31, 2020 the United Kingdom ceased to be a Member State of the European Union. Accordingly, the extent to which HMRC’s
position will remain as set out in this paragraph following the end of the transition period on December 31, 2020 is uncertain.
Any
stamp duty or SDRT payable on an issue or transfer of ordinary shares to a depositary receipt system or clearance service (although strictly
accountable by the clearance service or depositary receipt system operator or their nominee) will in practice generally be paid by the
transferors or participants in the clearance service or depositary receipt system. Specific professional advice should be sought before
incurring or reimbursing the costs of a 1.5% stamp duty or SDRT charge in any circumstances.
No
UK SDRT or stamp duty is required to be paid in respect of the issue or transfer of, or an agreement to transfer, ADSs (including by
way of a paperless transfer of ADSs through the facilities of DTC).
DESCRIPTION
OF SECURITIES WE ARE OFFERING
We are
offering up to 9,900,990 ADSs and pre-funded warrants to purchase up to 9,900,990 ADSs. For each pre-funded warrant we sell, the number
of ADSs we are offering will be decreased on a one-for-one basis. We are also registering the ADSs issuable from time to time upon exercise
of the pre-funded warrants offered hereby.
American
Depositary Shares (‘ADSs’)
The
description of our ADSs under the section “Share Capital” in our 2022 Form 20-F is incorporated herein by reference.
Pre-Funded
Warrants
General
The
term “pre-funded” refers to the fact that the purchase price of the pre-funded warrants in this offering includes almost
the entire exercise price that will be paid under the pre-funded warrants, except for a nominal remaining exercise price of $0.001. The
purpose of the pre-funded warrants is to enable investors that may have restrictions on their ability to beneficially own more than 4.99%
(or, at the election of such purchaser, 9.99%) of our outstanding ADSs following the consummation of this offering the opportunity to
invest capital into the Company without triggering their ownership restrictions, by receiving pre-funded warrants in lieu of ADSs which
would result in such ownership of more than 4.99% or 9.99%, as applicable, and receiving the ability to exercise their option to purchase
the ADSs underlying the pre-funded warrants at a nominal price at a later date.
The
following is a brief summary of certain terms and conditions of the pre-funded warrants being offered by us. The following description
is subject in all respects to the provisions contained in the form of pre-funded warrant, the form of which will be filed as an exhibit
to the registration statement of which this prospectus forms a part.
Exercise
Price
Pre-funded
warrants will have an exercise price of $0.001 per ADSs. The exercise price is subject to appropriate adjustment in the event of certain
stock dividends and distributions, stock splits, stock combinations, reclassifications or similar events affecting our ADSs and also
upon any distributions of assets, including cash, stock or other property to our stockholders.
Exercisability
The
pre-funded warrants are exercisable at any time after their original issuance and until exercised in full. The pre-funded warrants will
be exercisable, at the option of each holder, in whole or in part by delivering to us a duly executed exercise notice and by payment
in full of the exercise price in immediately available funds for the number of ADSs purchased upon such exercise. As an alternative to
payment in immediately available funds, the holder may elect to exercise the pre-funded warrant through a cashless exercise, in which
the holder would receive upon such exercise the net number of ADSs determined according to the formula set forth in the pre-funded warrant.
No fractional ADSs will be issued in connection with the exercise of a pre-funded warrant.
Exercise
Limitations
The
pre-funded warrants may not be exercised by the holder to the extent that the holder, together with its affiliates, would beneficially
own, after such exercise more than 4.99% of the ADSs then outstanding (including for such purpose the ADSs issuable upon such exercise).
However, any holder may increase or decrease such beneficial ownership limitation upon notice to us, provided that such limitation cannot
exceed 9.99%, and provided that any increase in the beneficial ownership limitation shall not be effective until 61 days after such notice
is delivered. Purchasers of pre-funded warrants in this offering may also elect prior to the issuance of the pre-funded warrants to have
the initial exercise limitation set at 9.99% of our outstanding ADSs.
Transferability
Subject
to applicable laws, the pre-funded warrants may be offered for sale, sold, transferred or assigned without our consent.
Exchange
Listing
There
is no established trading market for the pre-funded warrants and we do not expect a market to develop. In addition, we do not intend
to apply for the listing of the pre-funded warrants on any national securities exchange or other trading market. Without an active trading
market, the liquidity of the pre-funded warrants will be limited.
Fundamental
Transactions
In
the event of a fundamental transaction, as described in the pre-funded warrants and generally including any reorganization, recapitalization
or reclassification of our ADSs, the sale, transfer or other disposition of all or substantially all of our properties or assets, our
consolidation or merger with or into another person, the acquisition of more than 50% of our outstanding ADSs, or any person or group
becoming the beneficial owner of 50% of the voting power represented by our outstanding ADSs, upon consummation of such a fundamental
transaction, the holders of the pre-funded warrants will be entitled to receive upon exercise of the pre-funded warrants the kind and
amount of securities, cash or other property that the holders would have received had they exercised the pre-funded warrants immediately
prior to such fundamental transaction without regard to any limitations on exercise contained in the pre-funded warrants.
Rights
as a Shareholder
Except
as otherwise provided in the pre-funded warrant or by virtue of such holder’s ownership of shares of our ADSs, the holder of a
pre-funded warrant does not have the rights or privileges of a holder of our ADSs, including any voting rights, until the holder exercises
the pre-funded warrant. The pre-funded warrants will provide that holders have the right to participate in distributions or dividends
paid on our ADSs.
Placement
Agent Warrants
The
following summary of certain terms and provisions of the Placement Agent Warrants that are being issued hereby is not complete and is
subject to, and qualified in its entirety by, the provisions of the Placement Agent Warrants, the form of which will be filed as an exhibit
to the registration statement of which this prospectus forms a part. Prospective investors should carefully review the terms and provisions
of the form of Placement Agent Warrant for a complete description of the terms and conditions of the Placement Agent Warrant.
Duration
and Exercise Price
Each
Placement Agent Warrant offered hereby will have an initial exercise price equal to $0.63 per ADS (125% of the public offering price
per ADS). The Placement Agent Warrants will be immediately exercisable and will expire five years from the commencement of sales in this
offering. The exercise price and number of ADSs issuable upon exercise is subject to appropriate adjustment in the event of stock dividends,
stock splits, reorganizations or similar events affecting our ADSs and the exercise price.
Exercisability
The
Placement Agent Warrants will be exercisable, at the option of each holder, in whole or in part, by delivering to us a duly executed
exercise notice accompanied by payment in full for the number of shares of ADSs purchased upon such exercise (except in the case of a
cashless exercise as discussed below). A holder (together with its affiliates) may not exercise any portion of the Placement Agent Warrant
to the extent that the holder would own more than 4.99% of the outstanding ADSs immediately after exercise, except that upon notice from
the holder to us, the holder may increase or decrease the amount of beneficial ownership of outstanding stock after exercising the holder’s
Placement Agent Warrant up to 4.99% of the number of ADSs outstanding immediately after giving effect to the exercise, as such percentage
ownership is determined in accordance with the terms of the Placement Agent Warrants and in accordance with the rules and regulations
of the SEC, provided that any increase in the beneficial ownership limitation shall not be effective until 61 days following notice to
us.
Cashless
Exercise
If,
at the time a holder exercises its Placement Agent Warrants, a registration statement registering the issuance of the ADSs underlying
the Placement Agent Warrants under the Securities Act is not then effective or available for the issuance of such shares, then in lieu
of making the cash payment otherwise contemplated to be made to us upon such exercise in payment of the aggregate exercise price, the
holder may elect instead to receive upon such exercise (either in whole or in part) the net number of ADSs determined according to a
formula set forth in the Placement Agent Warrants.
Fractional
ADSs
No
fractional ADSs will be issued upon the exercise of the Placement Agent Warrants. Rather, the number of ADSs to be issued will be rounded
up to the next whole share or we will pay a cash adjustment equal to such fraction multiplied by the exercise price to the holder.
Transferability
Subject
to applicable laws, a Placement Agent Warrant may be transferred at the option of the holder upon surrender of the Placement Agent Warrant
to us together with the appropriate instruments of transfer.
Trading
Market
There
is no trading market available for the Placement Agent Warrants on any securities exchange or nationally recognized trading system, and
we do not expect a trading market to develop. We do not intend to list the Placement Agent Warrants on any securities exchange or other
trading market. Without a trading market, the liquidity of the Placement Agent Warrants will be extremely limited. The ADSs issuable
upon exercise of the Placement Agent Warrants is currently listed on the Nasdaq Capital Market.
Right
as a Shareholder
Except
as otherwise provided in the Placement Agent Warrants or by virtue of such holder’s ownership of ADSs, the holders of the Placement
Agent Warrants do not have the rights or privileges of holders of our ADSs, including any voting rights, until they exercise their Placement
Agent Warrants.
Fundamental
Transaction
In
the event of a fundamental transaction, as described in the Placement Agent Warrants and generally including any reorganization, recapitalization
or reclassification of our ADSs, the sale, transfer or other disposition of all or substantially all of our properties or assets, our
consolidation or merger with or into another person, the acquisition of more than 50% of our outstanding ADSs, or any person or group
becoming the beneficial owner of 50% of the voting power represented by our outstanding ADSs, the holders of the Placement Agent Warrants
will be entitled to receive upon exercise of the Placement Agent Warrants the kind and amount of securities, cash or other property that
the holders would have received had they exercised the Placement Agent Warrants immediately prior to such fundamental transaction. In
addition, in the event of a fundamental transaction which is approved by our board of directors, the holders of the Placement Agent Warrants
have the right to require us or a successor entity to redeem the Placement Agent Warrant for cash in the amount of the Black-Scholes
value of the unexercised portion of the Placement Agent Warrant on the date of the consummation of the fundamental transaction. In the
event of a fundamental transaction which is not approved by our board of directors, the holders of the Placement Agent Warrants have
the right to require us or a successor entity to redeem the Placement Agent Warrants for the consideration paid in the fundamental transaction
in the amount of the Black Scholes value of the unexercised portion of the Placement Agent Warrant on the date of the consummation of
the fundamental transaction.
PLAN
OF DISTRIBUTION
We
have engaged [*], or the placement agent, to act as our exclusive placement agent to solicit offers to purchase the securities offered
pursuant to this prospectus on a reasonable best efforts basis. The engagement agreement does not give rise to any commitment by the
placement agent to purchase any of our securities, and the placement agent will have no authority to bind us by virtue of the engagement
agreement. The placement agent is not purchasing or selling any of the securities offered by us under this prospectus, nor is it required
to arrange for the purchase or sale of any specific number or dollar amount of securities, other than to use its “reasonable best
efforts” to arrange for the sale of such securities by us. Therefore, we may not sell all of the securities being offered. The
terms of this offering were subject to market conditions and negotiations between us, the placement agent and prospective investors.
This is a best efforts offering and there is no minimum offering amount required as a condition to the closing of this offering. The
placement agent does not guarantee that it will be able to raise new capital in any prospective offering. The placement agent may engage
sub-agents or selected dealers to assist with the offering.
Investors
purchasing securities offered hereby will have the option to execute a securities purchase agreement with us. In addition to rights and
remedies available to all purchasers in this offering under federal securities and state law, the purchasers which enter into a securities
purchase agreement will also be able to bring claims of breach of contract against us. The ability to pursue a claim for breach of contract
is material to larger purchasers in this offering as a means to enforce the following covenants uniquely available to them under the
securities purchase agreement: (i) a covenant to not enter into variable rate financings for a period of one (1) year following the closing
of the offering, subject to certain exceptions; and (ii) a covenant to not enter into any equity financings for forty-five (45)
days from closing of the offering, subject to certain exceptions.
The
nature of the representations, warranties and covenants in the securities purchase agreements shall include:
| |
● |
standard issuer representations
and warranties on matters such as organization, qualification, authorization, no conflict, no governmental filings required, current
in SEC filings, no litigation, labor or other compliance issues, environmental, intellectual property and title matters and compliance
with various laws such as the Foreign Corrupt Practices Act; and |
| |
|
|
| |
● |
covenants
regarding matters such as registration of warrant shares, no integration with other offerings, filing of a 6-K to disclose entering
into these securities purchase agreements, no shareholder rights plans, no material nonpublic information, use of proceeds, indemnification
of purchasers, reservation and listing of ADSs, and no subsequent equity sales for forty-five (45) days from closing
of the offering, subject to certain exceptions. |
We
expect to deliver the securities being offered pursuant to this prospectus on or about [*], 2023, subject to satisfaction of certain
customary closing conditions.
Fees
and Expenses
The
following table shows the per ADS and per pre-funded warrant and total placement agent fees we will pay in connection with the
sale of the securities in this offering.
| | |
Per ADS | | |
Per
Pre-Funded Warrant | | |
Total | |
| Public offering price | |
$ | 0.5050 | | |
$ | 0.5050 | | |
$ | 5,000,000 | |
| Placement agent fees | |
$ | 0.0379 | | |
$ | 0.0379 | | |
$ | 375,000 | |
| Proceeds to us, before expenses | |
$ | 0.4671 | | |
$ | 0.4671 | | |
$ | 4,625,000 | |
We have agreed to pay the Placement Agent cash fee equal to 7.5% of the gross
proceeds raised in this offering. We have also agreed to reimburse the Placement Agent for certain of its offering-related
expenses, including a management fee of 1.0% of the gross proceeds raised in this offering, to reimburse the Placement Agent for its non-accountable
expenses in the amount of $50,000, for its legal fees and expenses and other out-of-pocket expenses in an amount up to $100,000, and for
its clearing expenses in the amount of $15,950.
Placement
Agent Warrants
In
addition, we have agreed to issue to the placement agent or its designees warrants to purchase up to 742,574 ADSs (which represents 7.5%
of the aggregate number of ADSs issued in this offering and issuable upon the exercise of the pre-funded warrants issued in this offering)
with an exercise price of $0.63 per ADS (representing 125% of the public offering price per ADS) and exercisable for five years from
the date of the commencement of sales in this offering. The placement agent warrants and underlying ADSs are registered on the registration
statement of which this prospectus is a part.
We estimate the
total expenses of this offering payable by us, excluding the placement agent fee, will be approximately $0.4 million.
Tail
We
have also agreed to pay the placement agent a tail fee equal to the cash and warrant compensation in this offering, if any investor,
who was contacted or introduced to us by the placement agent during the term of its engagement, provides us with capital in any public
or private offering or other financing or capital raising transaction during the one (1) year period following expiration or termination
of our engagement of the placement agent.
Lock-up
Agreements
We
and each of our officers and directors have agreed with the placement agent to be subject to a lock-up period of forty-five (45)
days following the date of closing of the offering pursuant to this prospectus. This means that, during the applicable lock-up
period, we and such persons may not offer for sale, contract to sell, sell, distribute, grant any option, right or warrant to purchase,
pledge, hypothecate or otherwise dispose of, directly or indirectly, any of our ADSs or any securities convertible into, or exercisable
or exchangeable for, ADSs, subject to customary exceptions. The placement agent may waive the terms of these lock-up agreements in its
sole discretion and without notice. In addition, we have agreed to not issue any securities that are subject to a price reset based on
the trading prices of our ADS or upon a specified or contingent event in the future, or enter into any agreement to issue securities
at a future determined price for a period of one (1) year following the closing date of this offering, subject to an exception.
Regulation
M
The
placement agent may be deemed to be an underwriter within the meaning of Section 2(a)(11) of the Securities Act, and any commissions
received by it and any profit realized on the resale of the securities sold by it while acting as principal might be deemed to be underwriting
discounts or commissions under the Securities Act. As an underwriter, the placement agent would be required to comply with the requirements
of the Securities Act and the Exchange Act, including, without limitation, Rule 10b-5 and Regulation M under the Exchange Act. These
rules and regulations may limit the timing of purchases and sales of our securities by the placement agent acting as principal. Under
these rules and regulations, the placement agent (i) may not engage in any stabilization activity in connection with our securities and
(ii) may not bid for or purchase any of our securities or attempt to induce any person to purchase any of our securities, other than
as permitted under the Exchange Act, until it has completed its participation in the distribution.
Indemnification
We
have agreed to indemnify the placement agent against certain liabilities, including certain liabilities arising under the Securities
Act and to contribute to payments that the placement agent may be required to make for these liabilities.
Determination
of Offering Price
The
actual offering price of the securities we are offering has been negotiated between us and the investors in the offering based on the
trading of our ADSs prior to the offering, among other things. Other factors considered in determining the public offering price of the
securities we are offering include our history and prospects, the stage of development of our business, our business plans for the future
and the extent to which they have been implemented, an assessment of our management, the general conditions of the securities markets
at the time of the offering and such other factors as were deemed relevant.
Electronic
Offer, Sale and Distribution of Securities
A
prospectus in electronic format may be made available on the websites maintained by the placement agent, if any, participating in this
offering and the placement agent may distribute prospectuses electronically. Other than the prospectus in electronic format, the information
on these websites is not part of this prospectus or the registration statement of which this prospectus forms a part, has not been approved
or endorsed by us or the placement agent, and should not be relied upon by investors.
Other
Relationships
From
time to time, the placement agent or its affiliates have in the past or may in the future provide in the future, various advisory, investment
and commercial banking and other services to us in the ordinary course of business, for which they have received and may continue to
receive customary fees and commissions. However, except as disclosed in this prospectus, we have no present arrangements with the placement
agent for any further services.
Listing
Our
ADSs are listed on The Nasdaq Capital Market under the symbol “TCBP.”
Depositary
The
depositary for our ADSs is The Bank of New York Mellon.
EXPENSES
OF THE OFFERING
Set
forth below is an itemization of the total anticipated expenses, excluding underwriting discounts, expected to be incurred in
connection with the offer and sale of the ADSs by us. With the exception of the SEC registration fee and the FINRA filing fee, all amounts
are estimates, in United States dollars:
| SEC registration fee |
|
$ | 2,307 | |
| FINRA filing fee |
|
$ | 650 | |
| Printer fees and expenses |
|
$ | 2,000 | |
| Transfer agent, depositary fees and expenses |
|
$ | 5,000 | |
| Management fees |
|
$ | 50,000 | |
| Legal fees and expenses |
|
$ | 288,600 | |
| Accounting fees and expenses |
|
$ | 75,000 | |
| Miscellaneous |
|
$ | 12,366 | |
| Total |
|
$ | 435,923 | |
In addition to the expenses noted in the table
above, the estimated fair value of Placement Agent Warrants to be issued as part of this offering is $257,135.
LEGAL
MATTERS
We
are being represented by Sheppard, Mullin, Richter & Hampton LLP, New York, New York with respect to certain legal matters of United
States federal securities and New York state law. We are being represented by Addleshaw Goddard LLP, Glasgow, Scotland with respect to
certain legal matters of the law of Scotland and other applicable law of the United Kingdom and as to certain patent law matters by Murgitroyd
& Company Limited.
EXPERTS
The
consolidated financial statements of TC BioPharm (Holdings) plc incorporated by reference in TC BioPharm (Holdings) plc’s Annual
Report (Form 20-F) for the year ended December 31, 2022, have been audited by Marcum LLP, independent registered public accounting
firm, as set forth in their report thereon, (which contains an explanatory paragraph describing conditions that raise substantial doubt
about the Company’s ability to continue as a going concern as described in Note 1 to the consolidated financial statements) included
therein, and incorporated herein by reference. Such consolidated financial statements are incorporated herein by reference in reliance
upon such report given on the authority of such firm as experts in accounting and auditing.
The
registered business address of Marcum LLP is 730 3rd Avenue, 11th Floor, New York, NY 10017, United States of America.
On
November 11, 2022, the Company appointed Marcum LLP (“Marcum”) as the Company’s independent registered public accounting
firm for the year ending December 31, 2022. Marcum succeeds Ernst & Young LLP who tendered their resignation on October 20, 2022.
The
consolidated financial statements of TC BioPharm (Holdings) plc incorporated by reference in TC BioPharm (Holdings) plc’s Annual
Report (Form 20-F) for the year ended December 31, 2021 and for the fiscal years ended December 31, 2021 and 2020, have been audited
by Ernst & Young LLP, independent registered public accounting firm, as set forth in their report thereon, (which contains an explanatory
paragraph describing conditions that raise substantial doubt about the Company’s ability to continue as a going concern as described
in Note 1 to the consolidated financial statements) included therein, and incorporated herein by reference. Such consolidated financial
statements are incorporated herein by reference in reliance upon such report given on the authority of such firm as experts in accounting
and auditing.
The
registered business address of Ernst & Young LLP is 144 Morrison Street, Edinburgh, EH3 8EX, United Kingdom.
WHERE
YOU CAN FIND MORE INFORMATION
We
have filed with the SEC a registration statement on Form F-1 under the Securities Act relating to this offering. This prospectus does
not contain all of the information contained in the registration statement. The rules and regulations of the SEC allow us to omit certain
information from this prospectus that is included in the registration statement. Statements made in this prospectus concerning the contents
of any contract, agreement or other document are summaries of all material information about the documents summarized, but are not complete
descriptions of all terms of these documents. If we filed any of these documents as an exhibit to the registration statement, you may
read the document itself for a complete description of its terms.
You
may read and copy the registration statement, including the related exhibits and schedules, and any document we file with the SEC without
charge at the SEC’s public reference room at 100 F Street, N.E., Room 1580, Washington, DC 20549. You may also obtain copies of
the documents at prescribed rates by writing to the Public Reference Section of the SEC at 100 F Street, N.E., Room 1580, Washington,
DC 20549. Please call the SEC at 1-800-SEC-0330 for further information on the public reference room. The SEC also maintains an Internet
website that contains reports and other information regarding issuers that file electronically with the SEC. Our filings with the SEC
are also available to the public through the SEC’s website at http://www.sec.gov.
We
are subject to the information reporting requirements of the Exchange Act that are applicable to foreign private issuers, and under those
requirements are filing reports with the SEC. Those other reports or other information may be inspected without charge at the locations
described above. As a foreign private issuer, we are exempt from the rules under the Exchange Act related to the furnishing and content
of proxy statements, and our officers, directors and principal shareholders are exempt from the reporting and short-swing profit recovery
provisions contained in Section 16 of the Exchange Act. In addition, we are not required under the Exchange Act to file annual, quarterly
and current reports and financial statements with the SEC as frequently or as promptly as U.S. registrants whose securities are registered
under the Exchange Act. However, we are required to file with the SEC, within 120 days after the end of each fiscal year, or such applicable
time as required by the SEC, an annual report on Form 20-F containing financial statements audited by an independent registered public
accounting firm, and will furnish to the SEC, on Form 6-K, unaudited interim financial information.
We
maintain a corporate website at https://tcbiopharm.com/. Information contained on, or that can be accessed through, our website does
not constitute a part of this prospectus. We have included our website address in this prospectus solely as an inactive textual reference.
We will post on our website any materials required to be so posted on such website under applicable corporate or securities laws and
regulations, including, posting any XBRL interactive financial data required to be filed with the SEC and any notices of general meetings
of our shareholders.
INFORMATION
INCORPORATED BY REFERENCE
The
rules of the SEC allow us to incorporate information into this prospectus by reference. The information incorporated by reference is
considered to be a part of this prospectus. This prospectus incorporates by reference the documents listed below (including any exhibits,
except where otherwise noted):
| |
● |
our Annual Report on Form 20-F for the fiscal year ended December 31, 2022 filed on May 1, 2023; and |
| |
● |
our Reports of Foreign
Private Issuer on Form 6-K filed with the SEC on January 11, 2023, January 19, 2023, March 30, 2023, June 7, 2023, June 27, 2023,
June 29, 2023, July 10, 2023, July 24, 2023, July 31, 2023 and August 22, 2023; |
| |
● |
the description of our
securities contained in our registration statement on Form 8-A (File No. 001-41231) filed with the SEC on January 14, 2022, including
all amendments and reports filed for the purpose of updating such description. |
Any
statement made in a document incorporated by reference into this prospectus will be deemed to be modified or superseded for purposes
of this prospectus to the extent that a statement contained in this prospectus modifies or supersedes that statement. Any statement so
modified or superseded will not be deemed, except as so modified or superseded, to constitute a part of this prospectus.
You
can obtain any of the filings incorporated by reference into this prospectus through us or from the SEC through the SEC’s website
at http://www.sec.gov. We will provide, without charge, to each person, including any beneficial owner, to whom a copy of this prospectus
is delivered, upon written or oral request of such person, a copy of any or all of the reports and documents referred to above which
have been or may be incorporated by reference into this prospectus. You should direct requests for those documents to:
TC
BioPharm (Holdings) plc
Maxim
1, 2 Parklands Way
Holytown,
Motherwell, ML1 4WR
Scotland,
United Kingdom
+44
(0) 141 433 7557
We
maintain an internet site at http://www.tcbiopharm.com. Our website and the information contained on or connected to it shall not be
deemed to be incorporated into this prospectus or the registration statement of which it forms a part.

UP TO 9,900,990 ORDINARY SHARES
AND UP TO 9,900,990 PRE-FUNDED WARRANTS TO PURCHASE
UP TO
9,900,990 AMERICAN DEPOSITARY SHARES
AND PLACEMENT AGENT WARRANTS TO PURCHASE UP
TO 742,574 AMERICAN DEPOSITARY SHARES
TC
BIOPHARM (HOLDINGS) PLC
PRELIMINARY
PROSPECTUS
[*]
,
2023
PART
II
INFORMATION
NOT REQUIRED IN PROSPECTUS
Item
6. Indemnification of Directors and Officers
Scottish
law does not limit the extent to which a company’s articles of association may provide indemnification of officers and directors,
except to the extent that it may be held by the Scottish and United Kingdom courts to be contrary to public policy, such as providing
indemnification against civil fraud or the consequences of committing a crime.
Our
Memorandum and Articles of Association provide that, to the maximum extent permitted by law, every current and former director and officer
(excluding an auditor) is entitled to be indemnified out of our assets against any liability, action, proceeding, claim, demand, costs,
damages or expenses, including legal expenses, which such indemnified person may incur in that capacity unless such liability arose as
a result of the actual fraud or willful default.
A
company formed under the laws of Scotland may also purchase insurance for directors and certain other officers against liability incurred
as a result of any negligence, default, breach of duty or breach of trust in relation to the company. We expect to maintain director’s
and officer’s liability insurance covering our directors and officers with respect to general civil liability, including liabilities
under the Securities Act of 1933, as amended (or the “Securities Act”), which he or she may incur in his or her capacity
as such. We have entered into a deed of indemnity with each of our directors and members of our senior management, each of which provides
the office holder with indemnification permitted under applicable law and to the extent that these liabilities are not covered by directors’
and officers’ insurance.
Insofar
as indemnification for liabilities arising under the Securities Act may be permitted to directors, officers or persons controlling us
under the foregoing provisions, we have been informed that in the opinion of the SEC such indemnification is against public policy as
expressed in the Securities Act and is therefore unenforceable.
Item
7. Recent Sales of Unregistered Securities
Set
forth below is information regarding share capital issued by TC BioPharm (Holdings) plc and TC BioPharm Limited (prior to completing
a corporate reorganization) since January 1, 2018. Some of the transactions described below involved directors, officers and 5% shareholders
and are more fully described under the section titled “Related Party Transactions”.
| |
● |
On December 17, 2018, the
Company issued an aggregate of 3,499 ordinary shares to 19 accredited investors and insiders at a purchase price of £178.48
per share for aggregate consideration totaling £624,500 in respect of satisfying a convertible loan note. |
| |
|
|
| |
● |
In November 2019, the Company
issued an aggregate of 14,688 A ordinary shares to 25 accredited investors and insiders at a purchase price of £215.00 per
share for aggregate cash consideration totaling £3,157,877. |
| |
|
|
| |
● |
From December 2019 until
July 2020, the Company issued an aggregate of 2,338 A ordinary shares to 8 accredited investors and insiders at a purchase price
of £215.00 per share for aggregate cash consideration totaling £499,187. |
| |
|
|
| |
● |
On August 25, 2020, the
Company issued an aggregate of 15,891 A ordinary shares to 14 accredited investors and insiders at a purchase price of £215.00
per share for aggregate cash consideration totaling £3,416,522. |
| |
|
|
| |
● |
On January 18, 2021, the
Company issued an aggregate of 27 A ordinary shares to one accredited investor and insider at a purchase price of £215.00 per
share for aggregate cash consideration totaling £5,719. |
| |
|
|
| |
● |
On January 19, 2021, the
Company issued an aggregate of 930 A ordinary shares to one accredited investor and insider at a purchase price of £215.00
per share for aggregate cash consideration totaling £199,993. |
| |
● |
On April 30, 2021, the
Company issued an aggregate of 465 A ordinary shares to one accredited investor and insider at a purchase price of £215 per
share for aggregate consideration totaling £100,018. |
| |
|
|
| |
● |
On June 16, 2021, the Company
issued an aggregate of 369 A ordinary shares to one accredited investor and insider at a purchase price of £215.00 per share
for aggregate consideration totaling £79,378. |
From
April 2021 to January 28, 2022, the Company issued Convertible Loan Notes with a face value amount of $17.7 million. The loan note was
issued with a 50% discount. Upon listing, 50% of the face value of the outstanding Convertible Loan Notes (including interest accrued
to date), and any further balance as elected by the noteholders, will convert into ADSs and Warrants at a conversion price for a unit
comprised of one ADS and 1.25 Warrants, which is the lower of (a) the price per share calculated on a fully diluted basis (based on the
number of shares in issue and vested share options immediately prior to the IPO being approved by the shareholders) on an assumed entity
valuation of $120,000,000 and (b) the listing price. The remaining balance of the loan notes are repayable or convertible (at the same
value) at the loan note holders’ option in two equal tranches at 90 days and 180 days after the listing date. In the event of an
act of default (including if the Company does not list despite its and its bankers’ efforts before February 15, 2022) the outstanding
notes become repayable at their face value.
Immediately
prior to completion of the Initial Public Offering, TC BioPharm (Holdings) plc re-organized its share capital whereby all of the outstanding
series A ordinary shares were re-designated as ordinary shares of TC BioPharm (Holdings) plc on a one for one basis. Immediately prior
to the completion of the offering, a further 24,693 ordinary shares were issued, under the terms of our Articles of Association to certain
shareholders who, prior to the IPO, owned A ordinary shares which carried the right, to subscribe at nominal value for a certain number
of additional shares, calculated by reference to the pre-money valuation of the IPO.
On
February 10, 2022, the Company completed an IPO on Nasdaq, issuing 82,353 American Depositary Shares (“ADSs”) representing
82,353 ordinary shares with nominal value of £41,176 and warrants to buy 189,412 ADSs at a combined issue price of $212.50 for
proceeds before expenses of $17.5 million.
At
the date of the IPO loan notes totaling $13,447,012, converted into 63,280 ADSs and 125,560 Warrants at a combined issue price of $212.50.
On
June 8, 2022, the Company completed a secondary public offering, issuing 230,000 ADSs representing 230,000 ordinary shares for aggregate
gross proceeds before expenses of $4.6 million.
On
August 9, 2022, TC BioPharm (Holdings) plc issued 3,676 American Depositary Shares (“ADSs”) representing 3,676 ordinary shares
and warrants to buy 7,352 ADSs on conversion of loan notes totaling $0.8 million.
On
November 15, 2022, TC BioPharm (Holdings) plc issued 21 Ordinary shares for a consideration of $7.565 (£6.362) per share.
On
November 24, 2022, TC BioPharm (Holdings) plc issued 3 Ordinary shares for a consideration of $6.51 per share.
On
November 27, 2022, the Company entered into a Securities Purchase Agreement (the “Purchase Agreement”) with certain accredited
investors (the “Investors”) as purchasers. Pursuant to the Purchase Agreement, the Company sold, and the Investors purchased
in a private placement an aggregate of 155,000 American Depositary Shares (the “ADSs”), pre-funded warrants to purchase up
to 1,315,000 ADS (the “Pre-Funded Warrants”), series A purchase warrants to purchase up to 1,470,000 ADSs (the “Series
A Ordinary Warrants”) and series B purchase warrants to purchase up to 1,470,000 ADSs (the “Series B Ordinary Warrants”
and together with the Series A Ordinary Warrants, the “Ordinary Warrants”) for aggregate gross proceeds of $7,350,000, excluding
any proceeds that may be received upon exercise of the Ordinary Warrants. The purchase price for each ADS and associated Ordinary Warrants
is $5.00 and the purchase price per each Pre-Funded Warrant and associated Ordinary Warrants is $4.999. The offering closed on November
30, 2022.The Series A Ordinary Warrants will be immediately exercisable, will expire five and one-half (5.5) years from the date of issuance
and have an exercise price of $5.00 per ADS, subject to adjustment as set forth therein. The Series B Ordinary Warrants will be immediately
exercisable, will expire thirty (30) months from the date of issuance and have an exercise price of $5.00 per ADS, subject to adjustment
as set forth therein. The placement agent for such offering received warrants to purchase up to 110,250 ADSs (the “Placement Agent
Warrants”) on substantially the same terms as the Series A Ordinary Warrants except that the Placement Agent Warrants have an exercise
price equal to 125% of the offering price, or $6.25 per ADS.
On
March 27, 2023, the Company , entered into a Securities Purchase Agreement (the “Purchase Agreement”) with certain accredited
investors (the “Investors”), pursuant to which the Company agreed to issue and sell an aggregate of 215,000 ADSs, pre-funded
warrants to purchase up to 3,222,500 ADS (the “March 2023 Pre-Funded Warrants”), and series C purchase warrants to purchase
up to 3,437,500 ADSs (the “Ordinary Warrants” and together with the March 2023 Pre-Funded Warrants and the ADSs, the “Securities”).
The purchase price for each ADS and associated Ordinary Warrants was $1.60 and the purchase price per each March 2023 Pre-Funded Warrant
and associated Ordinary Warrants was $1.599. The Ordinary Warrants were immediately exercisable, expire five (5) years from the date
of issuance and have an exercise price of $1.75 per ADS. The March 2023 Pre-Funded Warrants may be exercised at any time until all of
the Pre-Funded Warrants are exercised in full at an exercise price of $0.001 per ADS. The total net proceeds from this offering were
approximately $4.6 million, after deducting estimated offering expenses of approximately $0.9 million.
In
connection with the March 2023 offering, the Company agreed that certain existing warrants to purchase up to an aggregate of 2,800,000
ADSs of the Company that were previously issued on November 30, 2022, at an exercise price of $5.00 per ADS and expiration dates of May
30, 2025 and May 30, 2028, were amended effective upon the closing of the March 2023 offering so that the amended warrants will have
a reduced exercise price of $1.75 per ADS.
In
the period from January 1, 2023 to August 28, 2023, the holders of prefunded warrants, exercised prefunded warrants to purchase
4,537,500 ADSs.
On
April 3, 2023, the Company agreed with the loan note holder to extend the Redemption Date (as defined in the Loan Note) to January 15,
2024 and amend the Conversion Price (as defined in the Loan Note) of the outstanding loan notes to be the lesser of $1.00 or the
lowest closing price of the Ordinary Shares during the ten (10) day period prior to the date the Noteholder delivers a notice of conversion
to the Company, not to be lower than $0.20. In other respects the terms of the Loan Note remain unaltered. In addition, in consideration
of amending the Loan Note, the Company agreed to issue a 5-year warrant to the loan note holder to subscribe for 200,000 Ordinary Shares
in the share capital of the Company at an exercise price of $5.00.
In
the period from April 3, 2023 to August 28, 2023, the holders of Loan Notes, converted notes with a value of $813,302 into 1,590,130
ADSs.
The
offers, sales and issuances of the securities and loan notes described above were exempt from registration either (i) under Section 4(a)(2)
of the Securities Act in that the transactions did not involve any public offering, (ii) under Rule 701 promulgated under the Securities
Act in that the transactions were under compensatory benefit plans and contracts relating to compensation or (iii) under Regulation S
promulgated under the Securities Act in that offers, sales and issuances were not made to persons in the United States and no directed
selling efforts were made in the United States.
Item
8. Exhibits and Financial Statement Schedules
| Exhibit |
|
Description |
|
Schedule/
Form |
|
File
Number |
|
Exhibit |
|
File
Date |
| 2.1 |
|
Deposit Agreement – Bank of New York Mellon for American Depositary Shares |
|
F-1 |
|
333-260492 |
|
4.1 |
|
03/08/2022 |
| 2.2 |
|
Form of American Depositary Share (included in Exhibit 2.1) |
|
F-1 |
|
333-260492 |
|
4.2 |
|
03/08/2022 |
| 2.3 |
|
Warrant Agent Agreement with Computershare Inc. |
|
F-1 |
|
333-260492 |
|
4.4 |
|
03/08/2022 |
| 2.4 |
|
Form of Warrant Certificate (included in Exhibit 2.3) |
|
F-1 |
|
333-260492 |
|
4.5 |
|
03/08/2022 |
| 2.5 |
|
Form of Ordinary Share Certificate |
|
F-1 |
|
333-260492 |
|
4.6 |
|
03/08/2022 |
| 3.1 |
|
Articles of Association of TC BioPharm (Holdings) plc |
|
F-1 |
|
333-260492 |
|
3.2 |
|
03/08/2022 |
| 4.1 |
|
Form of Representative Warrant |
|
F-1 |
|
333-260492 |
|
4.3 |
|
03/08/2022 |
| 4.2 |
|
Form of 2014 Share Option Scheme of Registrant |
|
F-1 |
|
333-260492 |
|
10.1 |
|
03/08/2022 |
| 4.3 |
|
Form of 2021 Share Option Scheme (including sub-plan for U.S. based persons) of Registrant |
|
F-1 |
|
333-260492 |
|
10.2 |
|
03/08/2022 |
| 4.4 |
|
Form of 2021 Company Share Option Plan (CSOP) of Registrant |
|
F-1 |
|
333-260492 |
|
10.3 |
|
03/08/2022 |
| 4.5 |
|
Convertible Loan Note, up to $20,000,000 in principal amount |
|
F-1 |
|
333-260492 |
|
10.6 |
|
03/08/2022 |
| 4.6 |
|
Form of Lock Up Agreement of Pre-IPO Smaller Shareholders |
|
F-1 |
|
333-260492 |
|
10.8 |
|
03/08/2022 |
| 4.7 |
|
Form of Lock Up Agreement of Pre-IPO Management and Larger Shareholders |
|
F-1 |
|
333-260492 |
|
10.9 |
|
03/08/2022 |
| 4.8 |
|
Form of Lock Up Agreement of Holders of Convertible Loan Notes |
|
F-1 |
|
333-260492 |
|
10.10 |
|
03/08/2022 |
| 4.9 |
|
Form of Deed of Indemnity for directors and officer |
|
20-F |
|
001-41231 |
|
4.10 |
|
05/13/2022 |
| 4,10 |
|
Description of Securities of Registrant |
|
20-F |
|
001-41231 |
|
4.11 |
|
05/13/2022 |
| 4.11 |
|
Code of Ethics of the Registrant |
|
F-1 |
|
333-260492 |
|
11.1 |
|
03/08/2022 |
| 4.12 |
|
Form of Pre-Funded Warrant |
|
6-K |
|
001-41231 |
|
10.1 |
|
11/30/2022 |
| 4.13 |
|
Form of Series A and Series B Ordinary Warrant |
|
6-K |
|
001-41231 |
|
10.2 |
|
11/30/2022 |
| 4.14 |
|
Form of Placement Agent Warrant |
|
6-K |
|
001-41231 |
|
10.3 |
|
11/30/2022 |
| 4.15 |
|
Form of Pre-Funded Warrant |
|
6-K |
|
001-41231 |
|
10.1 |
|
03/30/2023 |
| 4.16 |
|
Form of Placement Agent Warrant |
|
6-K |
|
001-41231 |
|
10.3 |
|
03/30/2023 |
| 4.17 |
|
Form of Series C Ordinary Warrant |
|
6-K |
|
001-41231 |
|
10.2 |
|
03/30/2023 |
| 5.1* |
|
Opinion of Addleshaw Goddard LLP |
|
|
|
|
|
|
|
|
| 5.2* |
|
Opinion of Sheppard, Mullin, Richter & Hampton LLP ⸶ |
|
|
|
|
|
|
|
|
| 10.1 |
|
Form of Securities Purchase Agreement for Nov 2022 Private Placement |
|
6-K |
|
001-41231 |
|
10.4 |
|
11/30/2022 |
| 10.2 |
|
Form of Registration Rights Agreement for Nov 2022 Private Placement |
|
6-K |
|
001-41231 |
|
10.5 |
|
03/30/2023 |
| 10.3 |
|
Form of Securities Purchase Agreement |
|
6-K |
|
001-41231 |
|
10.4 |
|
03/30/2023 |
| 10.4 |
|
Warrant Amendment Agreement, dated March 27, 2023 |
|
6-K |
|
001-41231 |
|
10.5 |
|
03/30/2023 |
| 10.5 |
|
Warrant Amendment Agreement, dated July 10, 2023 |
|
6-K |
|
001-41231 |
|
10.1 |
|
07/24/2023 |
| 16.1 |
|
Letter of Ernst & Young LLP, dated November 18, 2022 |
|
6-K |
|
001-41231 |
|
16.1 |
|
11/18/2022 |
| 21.1 |
|
List of Subsidiaries of Registrant |
|
F-1 |
|
333-260492 |
|
21.1 |
|
03/08/2022 |
| 23.1 |
|
Consent of Marcum LLP, independent registered public accounting firm |
|
|
|
|
|
|
|
|
| 23.2 |
|
Consent of Ernst & Young LLP, independent registered public accounting firm |
|
|
|
|
|
|
|
|
| 23.3* |
|
Consent of Addleshaw Goddard LLP (included in Exhibit
5.1) |
|
|
|
|
|
|
|
|
| 23.4* |
|
Consent of Sheppard, Mullin, Richter & Hampton
LLP (included in Exhibit 5.2) |
|
|
|
|
|
|
|
|
| 24.1 |
|
Power of Attorney (included as part of the signature page of original filed Registration Statement) |
|
|
|
|
|
|
|
|
| 107 |
|
Filing Fee Table |
|
|
|
|
|
|
|
|
* To be filed by amendment
Item
9. Undertakings
| (A) |
The undersigned registrant
hereby undertakes: |
| |
(1) |
To
file, during any period in which offers or sales are being made, a post-effective amendment to this registration statement: |
| |
(i) |
To
include any prospectus required by Section 10(a)(3) of the Securities Act of 1933, as amended, or the Securities Act; |
| |
|
|
| |
(ii) |
To
reflect in the prospectus any facts or events arising after the effective date of the registration statement (or the most recent
post-effective amendment thereof) which, individually or in the aggregate, represent a fundamental change in the information set
forth in the registration statement. Notwithstanding the foregoing, any increase or any decrease in volume of securities offered
(if the total dollar value of securities offered would not exceed that which was registered) and any deviation from the low or high
end of the estimated maximum offering range may be reflected in the form of prospectus filed with the SEC pursuant to Rule 424(b)
if, in the aggregate, the changes in volume and price represent no more than a 20% change in the maximum aggregate offering price
set forth in the “Calculation of Registration Fee” table in the effective registration statement; and |
| |
|
|
| |
(iii) |
To
include any material information with respect to the plan of distribution not previously disclosed in the registration statement
or any material change to such information in the registration statement; |
| |
provided,
however, that paragraphs (a)(1)(i), (a)(1)(ii) and (a)(1)(iii) of this section do not apply if the registration statement is
on Form F-3 and the information required to be included in a post-effective amendment by those paragraphs is contained in periodic
reports filed with or furnished to the SEC by the registrant pursuant to Section 13 or Section 15(d) of the Securities Exchange Act
of 1934, as amended, or Exchange Act, that are incorporated by reference in the registration statement, or is contained in a form
of prospectus filed pursuant to Rule 424(b) that is part of the registration statement. |
| |
(2) |
That,
for the purpose of determining any liability under the Securities Act, each such post-effective amendment shall be deemed to be a
new registration statement relating to the securities offered therein, and the offering of such securities at that time shall be
deemed to be the initial bona fide offering thereof. |
| |
|
|
| |
(3) |
To
remove from registration by means of a post-effective amendment any of the securities being registered which remain unsold at the
termination of the offering. |
| |
|
|
| |
(4) |
To
file a post-effective amendment to the registration statement to include any financial statements required by Item 8.A of Form 20-F
at the start of any delayed offering or throughout a continuous offering. Financial statements and information otherwise required
by Section 10(a)(3) of the Exchange Act need not be furnished, provided that the registrant includes in the prospectus, by means
of a post-effective amendment, financial statements required pursuant to this paragraph (a)(4) and other information necessary to
ensure that all other information in the prospectus is at least as current as the date of those financial statements. Notwithstanding
the foregoing, with respect to registration statements on Form F-3, a post-effective amendment need not be filed to include financial
statements and information required by Section 10(a)(3) of the Exchange Act or Rule 3-19 of Regulation S-K if such financial statements
and information are contained in periodic reports filed with or furnished to the SEC by the registrant pursuant to Section 13 or
Section 15(d) of the Exchange Act that are incorporated by reference in this Form F-3. |
| |
(5) |
That,
for the purpose of determining liability under the Securities Act to any purchaser: |
| |
(i) |
Each
prospectus filed by the registrant pursuant to Rule 424(b)(3) shall be deemed to be part of the registration statement as of the
date the filed prospectus was deemed part of and included in the registration statement; and |
| |
(ii) |
Each
prospectus required to be filed pursuant to Rule 424(b)(2), (b)(5), or (b)(7) as part of a registration statement in reliance on
Rule 430B relating to an offering made pursuant to Rule 415(a)(1)(i), (vii), or (x) for the purpose of providing the information
required by section 10(a) of the Securities Act shall be deemed to be part of and included in the registration statement as of the
earlier of the date such form of prospectus is first used after effectiveness or the date of the first contract of sale of securities
in the offering described in the prospectus. As provided in Rule 430B, for liability purposes of the issuer and any person that is
at that date an underwriter, such date shall be deemed to be a new effective date of the registration statement relating to the securities
in the registration statement to which that prospectus relates, and the offering of such securities at that time shall be deemed
to be the initial bona fide offering thereof. Provided, however, that no statement made in a registration statement or prospectus
that is part of the registration statement or made in a document incorporated or deemed incorporated by reference into the registration
statement or prospectus that is part of the registration statement will, as to a purchaser with a time of contract of sale prior
to such effective date, supersede or modify any statement that was made in the registration statement or prospectus that was part
of the registration statement or made in any such document immediately prior to such effective date. |
| |
(6) |
That,
for the purpose of determining liability of the registrant under the Securities Act to any purchaser in the initial distribution
of the securities:
The
undersigned registrant undertakes that in a primary offering of securities of the undersigned registrant pursuant to this registration
statement, regardless of the underwriting method used to sell the securities to the purchaser, if the securities are offered or sold
to such purchaser by means of any of the following communications, the undersigned registrant will be a seller to the purchaser and
will be considered to offer or sell such securities to such purchaser: |
| |
(i) |
Any
preliminary prospectus or prospectus of the undersigned registrant relating to the offering required to be filed pursuant to Rule
424; |
| |
|
|
| |
(ii) |
Any
free writing prospectus relating to the offering prepared by or on behalf of the undersigned registrant or used or referred to by
the undersigned registrant; |
| |
|
|
| |
(iii) |
The
portion of any other free writing prospectus relating to the offering containing material information about the undersigned registrant
or its securities provided by or on behalf of the undersigned registrant; and |
| |
|
|
| |
(iv) |
Any
other communication that is an offer in the offering made by the undersigned registrant to the purchaser. |
| (B) |
Insofar as indemnification
for liabilities arising under the Securities Act may be permitted to directors, officers and controlling persons of the registrant
pursuant to the foregoing provisions, or otherwise, the registrant has been advised that in the opinion of the Securities and Exchange
Commission such indemnification is against public policy as expressed in the Securities Act and is, therefore, unenforceable. In
the event that a claim for indemnification against such liabilities (other than the payment by the registrant of expenses incurred
or paid by a director, officer or controlling person of the registrant in the successful defense of any action, suit or proceeding)
is asserted by such director, officer or controlling person in connection with the securities being registered, the registrant will,
unless in the opinion of its counsel the matter has been settled by controlling precedent, submit to a court of appropriate jurisdiction
the question whether such indemnification by it is against public policy as expressed in the Securities Act and will be governed
by the final adjudication of such issue. |
| (C) |
The undersigned registrant
hereby undertakes that, for purposes of determining any liability under the Securities Act, each filing of the registrant’s
annual report pursuant to Section 13(a) or Section 15(d) of the Exchange Act that is incorporated by reference in the registration
statement shall be deemed to be a new registration statement relating to the securities offered therein, and the offering of such
securities at that time shall be deemed to be the initial bona fide offering thereof. |
SIGNATURES
Pursuant
to the requirements of the Securities Act of 1933, the Registrant certifies it has reasonable grounds to believe that it meets all of
the requirements for filing this amended registration statement on Form F-1 with the Securities and Exchange Commission and has duly
caused this registration statement to be signed on its behalf by the undersigned, thereunto duly authorized, in Glasgow, Scotland, United
Kingdom, on August 28, 2023.
| |
TC BIOPHARM (HOLDINGS) PLC |
| |
|
| |
By: |
/s/
Bryan Kobel |
| |
Name: |
Bryan
Kobel |
| |
Title: |
Chief Executive Officer |
POWER
OF ATTORNEY
Each
of the undersigned officers and directors of TC BioPharm (Holdings) PLC hereby constitutes and appoints Bryan Kobel and Martin Thorp,
and each of them any of whom may act without joinder of the other, the individual’s true and lawful attorneys-in-fact and agents,
each with full power of substitution and resubstitution, for the person and in his or her name, place and stead, in any and all capacities,
to sign this registration statement of TC BioPharm (Holdings) PLC on Form F-1, and any other registration statement relating to the same
offering (including any registration statement, or amendment thereto, that is to become effective upon filing pursuant to Rule 462(b)
under the Securities Act of 1933, as amended), and any and all amendments thereto (including post-effective amendments to the registration
statement), and to file the same, with all exhibits thereto, and all other documents in connection therewith, with the Securities and
Exchange Commission, granting unto said attorneys-in-fact and agents, and each of them, full power and authority to do and perform each
and every act and thing requisite and necessary to be done in connection therewith, as fully to all intents and purposes as he or she
might or could do in person, hereby ratifying and confirming all that said attorneys-in-fact and agents or any of them, or their substitute
or substitutes, may lawfully do or cause to be done by virtue hereof.
Pursuant
to the requirements of the Securities Act of 1933, this registration statement has been signed by the following persons in the capacities
and on the dates indicated.
| Signature |
|
Title |
|
Date |
| |
|
|
|
|
| /s/
Bryan Kobel |
|
Chief Executive Officer
and Director (Principal Executive Officer) |
|
August
28, 2023 |
| Bryan Kobel |
|
|
|
|
| |
|
|
|
|
| /s/
Martin Thorp |
|
Chief Financial Officer
and Director (Principal Financial and |
|
August
28, 2023 |
| Martin Thorp |
|
Accounting Officer) |
|
|
| |
|
|
|
|
| /s/
Dr Mark Bonyhadi |
|
Non-Executive Director |
|
August
28, 2023 |
| Dr Mark Bonyhadi |
|
|
|
|
| |
|
|
|
|
| /s/
James Culverwell |
|
Non-Executive Director |
|
August
28, 2023 |
| James Culverwell |
|
|
|
|
| |
|
|
|
|
| /s/
Arlene Morris |
|
Chair of the Board and Non-Executive Director |
|
August
28, 2023 |
| Arlene Morris |
|
|
|
|
| |
|
|
|
|
| /s/
Edward Niemczyk |
|
Non-Executive Director |
|
August
28, 2023 |
| Edward Niemczyk |
|
|
|
|
SIGNATURE
OF AUTHORIZED U.S. REPRESENTATIVE OF THE REGISTRANT
Pursuant
to the Securities Act of 1933, the undersigned, the duly authorized representative in the United States of the registrant has signed
this registration statement or amendment thereto on August 28, 2023.
| |
TC BioPharm
(North America) Inc. |
| |
|
|
| |
By: |
/s/
Bryan Kobel |
| |
Name: |
Bryan Kobel |
| |
Title: |
Chief Executive Officer
and Director |
| |
|
|
| |
|
Authorized Representative
in the United States |
Exhibit
23.1
Independent
Registered Public Accounting Firm’s Consent
We
consent to the incorporation by reference in this Registration Statement of TC BioPharm (Holdings) PLC on Form F-1 of our report dated
May 1, 2023, which includes an explanatory paragraph as to the Company’s ability to continue as a going concern, with respect to
our audit of the consolidated financial statements of TC BioPharm (Holdings) PLC as of December 31, 2022 and for the year ended December
31, 2022 appearing in the Annual Report on Form 20-F of TC BioPharm (Holdings) PLC for the year ended December 31, 2022. Our report on
the consolidated financial statements refers to a retrospective adjustment to the 2021 and 2020 consolidated financial statements for
the November 2022 reverse share split, which we have audited. We also consent to the reference to our firm under the heading “Experts”
in the Prospectus, which is part of this Registration Statement.
| /s/
Marcum llp |
|
| |
|
| Marcum
llp |
|
| New
York, NY |
|
| August
28, 2023 |
|
Exhibit
23.2
Consent
OF Independent Registered Public Accounting Firm
We
consent to the reference to our firm under the caption “Experts” in the Registration Statement (Form F-1) and related Prospectus
of TC BioPharm (Holdings) plc for the registration of American Depository Shares representing ordinary shares and warrants and to the
incorporation by reference therein of our report dated May 13, 2022, with respect to the consolidated financial statements of TC BioPharm
(Holdings) plc included in its Annual Report (Form 20-F) for the year ended December 31, 2021, filed with the Securities and Exchange
Commission.
| /s/
Ernst & Young LLP |
|
| |
|
| Edinburgh,
United Kingdom |
|
| August
28, 2023 |
|
Exhibit 107
Calculation
of Filing Fee Tables
Form
F-1
(Form
Type)
TC
Biopharm (Holdings) PLC
(Exact
Name of Registrant as Specified in its Charter)
Table
1: Newly Registered Securities
| | |
Security
Type | |
Security
Class
Title | |
Fee
Calculation
or Carry
Forward Rule | | |
Maximum
Aggregate
Offering
Price | | |
Fee
Rate | | |
Amount of
Registration
Fee | |
Fees
to be paid | |
Equity | |
Ordinary Shares, par value £0.0001 per share (1)(2)(3) | |
| 457(o) | | |
$ | 5,000,000.00 | | |
$ | .00011020 | | |
$ | 551.00 | |
| | |
Other | |
Pre-Funded Warrants to purchase American Depositary Shares (2)(3) | |
| 457(g) | | |
| — | | |
| — | | |
$ | 0.00 | |
| | |
Equity | |
Ordinary Shares issuable upon exercise of Pre-Funded Warrants (2)(3) | |
| 457(o) | | |
| — | | |
| — | | |
$ | 0.00 | |
| | |
Other | |
Placement Agent Warrants to purchase American Depositary Shares (4) | |
| 457(g) | | |
| — | | |
| — | | |
$ | 0.00 | |
| | |
Equity | |
Ordinary Shares issuable upon exercise of Placement Agent Warrants (2)(5) | |
| 457(o) | | |
$ | 468,750.00 | | |
$ | .00011020 | | |
$ | 51.66 | |
| | |
Total Offering Amounts |
| | | |
$ | 5,468,750.00 | |
| | |
Total Fees Previously Paid |
| | | |
$ | 0 | |
| | |
Total Fee Offsets |
| | | |
$ | 0 | |
| | |
Net Fee Due |
| | | |
$ | 602.66 | |
| (1) |
Represents
the maximum number of ordinary shares, represented by American Depositary Shares (“ADSs”), each representing one ordinary
share, offered in this Registration Statement. |
| (2) |
This Registration Statement
includes an indeterminate number of additional ordinary shares issuable for no additional consideration pursuant to any stock dividend,
stock split, recapitalization or other similar transaction effected without the receipt of consideration, which results in an increase
in the number of outstanding ordinary shares. In the event of a stock split, stock dividend or similar transaction involving our
common stock, in order to prevent dilution, the number of shares registered shall be automatically increased to cover the additional
shares in accordance with Rule 416(a) under the Securities Act of 1933, as amended (the “Securities Act”). |
| (3) |
The proposed maximum aggregate
offering price of the ADSs will be reduced on a dollar-for-dollar basis based on the offering price of any pre-funded warrants issued
in the offering, and the proposed maximum aggregate offering price of the pre-funded warrants to be issued in the offering will be
reduced on a dollar-for-dollar basis based on the offering price of any ADS issued in the offering. Accordingly, the proposed maximum
aggregate offering price of the ADSs and pre-funded warrants (including the ordinary shares issuable upon exercise of the pre-funded
warrants), if any, is $5,000,000.00. |
| (4) |
No separate registration
fee required pursuant to Rule 457(g) under the Securities Act |
| (5) |
Estimated solely for the
purpose of calculating the registration fee pursuant to Rule 457(g) under the Securities Act. The placement agent warrants are exercisable
for up to the number of ADSs or pre-funded warrants equal to 7.5% of the aggregate number of securities sold in this offering at
a per ADS exercise price equal to 125% of the public offering price of the ADSs. As estimated solely for the purpose of calculating
the registration fee pursuant to Rule 457(g) under the Securities Act, the proposed maximum aggregate offering price of the representative’s
warrants is $468,750, which is equal to 125% of $375,000 (7.5% of the proposed maximum aggregate offering price of $5,000,000). |
TC BioPharm (NASDAQ:TCBP)
Historical Stock Chart
From Oct 2024 to Nov 2024
TC BioPharm (NASDAQ:TCBP)
Historical Stock Chart
From Nov 2023 to Nov 2024