Our Common Shares are listed on Nasdaq under
the symbol “ONCY” and on the Toronto Stock Exchange (“
TSX
”) under the symbol “ONC”.
On August 13, 2019, the closing price of our Common Shares on Nasdaq was US$0.90 per Common Share and on the TSX was C$1.19 per
Common Share. We have applied to list the Unit Shares and the Warrant Shares distributed under this Prospectus Supplement on Nasdaq
and the TSX. Listing will be subject to us fulfilling the listing requirements of Nasdaq and the TSX, respectively. The Warrants
are transferable but will not be listed for trading on any exchange and as a result, there is no market through which the Warrants
may be sold and purchasers may not be able to resell the Warrants purchased under this Prospectus Supplement. This may affect
the pricing of the Warrants in the secondary market, the transparency and availability of trading and the liquidity of the Warrants.
See “
Risk Factors
”.
Unless the context otherwise requires, references
to “Units”, “Unit Shares”, “Warrants” and “Warrant Shares” in this Prospectus Supplement
include the Over-Allotment Unit Shares, and Over-Allotment Warrants issuable upon exercise of the Over-Allotment Option, respectively.
The following table sets out information relating
to the Over-Allotment Option:
Delivery of the securities comprising the
Units is expected to be made on or about August 16, 2019, and in any event not later than September 13, 2019.
If the Underwriter exercises the Over-Allotment
Option in full, the total underwriting discounts and commissions payable by us will be US$301,232.24, and the total proceeds to
us, before expenses, will be US$4,002,085.54. We estimate the total expenses of the Offering, excluding underwriting commissions
and discounts, to be approximately US$300,000.
Messrs. Wayne Pisano, William G. Rice and Bernd
R. Seizinger are directors of the Corporation who reside outside of Canada. Messrs. Pisano, Rice and Seizinger have appointed the
Corporation, at its principal place of business, as agent for service of process. Purchasers are advised that it may not be possible
for investors to enforce judgments obtained in Canada against any person that resides outside of Canada, even if the party has
appointed an agent for service of process.
The Underwriter, as principal, conditionally
offers the Units subject to prior sale if, as and when issued by Oncolytics and accepted by the Underwriter in accordance with
the conditions contained in the Underwriting Agreement described under “
Underwriting
” and subject to the approval
of certain legal matters on behalf of Oncolytics by McCarthy Tétrault LLP, with respect to Canadian legal matters, and by
Dorsey & Whitney LLP, with respect to U.S. legal matters, and on behalf of the Underwriter by Osler, Hoskin & Harcourt
LLP, with respect to Canadian legal matters, and by Ellenoff Grossman & Schole LLP, with respect to U.S. legal matters. Subscriptions
will be received subject to rejection or allotment in whole or in part and the right is reserved to close the subscription books
at any time without notice.
It is anticipated that one or more global
certificates representing the Unit Shares and Warrants will be issued and registered in the name of the Depository Trust Corporation
(“
DTC
”), as directed by the Underwriter, and will be deposited with DTC on August 16, 2019, or such other date
as may be agreed between the Corporation and the Underwriter. No beneficial holder of such Unit Shares or Warrants will receive
definitive certificates representing their interest in the Unit Shares or Warrants, as applicable. Beneficial holders of Unit Shares
or Warrants will receive only a customer confirmation from the Underwriter or other registered dealer who is a DTC participant
and from or through whom a beneficial interest in the Unit Shares or Warrants is acquired.
The financial information of the Corporation
incorporated by reference in the Prospectus is presented in Canadian dollars. Unless otherwise noted herein, all references to
“US$”, “United States dollars” or “US dollars” are to United States dollars and all references
to “C$” or “$”, are to Canadian dollars. See “
Exchange Rate
”.
Our head office and principal place of business
is located at 210, 1167 Kensington Crescent N.W., Calgary, Alberta, T2N 1X7. Our registered office is located at 4000, 421 - 7
th
Avenue S.W., Calgary, Alberta, T2P 4K9.
Base Shelf
Prospectus dated May 4, 2018
IMPORTANT NOTICE ABOUT
INFORMATION IN THIS PROSPECTUS SUPPLEMENT
This document is in two parts. The first part
is this Prospectus Supplement, which describes the specific terms of the Offering and Units and the method of distribution of the
Units and also supplements and updates information regarding Oncolytics Biotech Inc. contained and incorporated by reference in
the Prospectus. The second part is the accompanying Prospectus, which gives more general information, some of which may not apply
to the Units. Both documents contain important information you should consider when making your investment decision. If the description
of the Units varies between this Prospectus Supplement and the accompanying Prospectus, investors should rely on the information
in this Prospectus Supplement. This Prospectus Supplement is deemed to be incorporated by reference into the Prospectus solely
for the purpose of the Offering. If information in this Prospectus Supplement is inconsistent with the Prospectus or the information
incorporated by reference in the Prospectus, you should rely on this Prospectus Supplement. You should read both this Prospectus
Supplement and the accompanying Prospectus, together with the additional information about us to which we refer you in the section
of this Prospectus Supplement entitled “
Where You Can Find Additional Information
.”
You should rely only on the information
contained in this Prospectus Supplement, the Prospectus and the documents incorporated by reference in the Prospectus. The Corporation
and the Underwriter have not authorized anyone to provide you with different information. If anyone provides you with any different
or inconsistent information, you should not rely on it. The Corporation is offering the Units only in jurisdictions where such
offers are permitted by law.
You should assume that the information contained
in this Prospectus Supplement, the Prospectus and the documents incorporated by reference in the Prospectus is accurate only as
of their respective dates, regardless of the time of delivery of this Prospectus Supplement and the accompanying Prospectus.
Our
business, financial condition, results of operations and prospects may have changed since those dates.
Market data and certain industry forecasts
used in this Prospectus Supplement, the Prospectus and the documents incorporated by reference in the Prospectus were obtained
from market research, publicly available information and industry publications. We believe that these sources are generally reliable,
but the accuracy and completeness of this information is not guaranteed. We have not independently verified such information, and
we do not make any representation as to the accuracy of such information.
In
this Prospectus Supplement, “
Oncolytics
,”
the “
Corporation
,” “
we
,”
“
us
,” and “our” refer to Oncolytics Biotech Inc. and its subsidiaries.
FORWARD-LOOKING STATEMENTS
This Prospectus Supplement, the Prospectus
and the documents incorporated by reference in the Prospectus contain certain statements relating to future events or the Corporation’s
future performance which constitute forward-looking statements. Such forward-looking statements involve known and unknown risks,
uncertainties and other factors which may cause the actual results, performance or achievements of the Corporation, or industry
results, to be materially different from any future results, performance or achievements expressed or implied by such forward-looking
statements. Forward-looking statements are statements that are not historical facts, and include, but are not limited to, estimates
and their underlying assumptions; statements regarding plans, objectives and expectations with respect to the efficacy of our technologies;
the timing and results of clinical studies related to our technologies; future operations, products and services; the impact of
regulatory initiatives on our operations; the size of and opportunities related to the markets for our technologies; general industry
and macroeconomic growth rates; expectations related to possible joint and/or strategic ventures and statements regarding future
performance. Forward-looking statements generally, but not always, are identified by the words “expects,” “anticipates,”
“believes,” “intends,” “estimates,” “projects”, “potential”, “possible”
and similar expressions, or that events or conditions “will,” “may,” “could” or “should”
occur.
The forward-looking statements in this Prospectus
Supplement, the Prospectus and the documents incorporated by reference in the Prospectus are subject to various risks and uncertainties,
most of which are difficult to predict and generally beyond the Corporation’s control, including without limitation:
|
|
·
|
risks related to all of our products, including pelareorep, being in the research and development
stage and requiring further development and testing before they can be marketed commercially;
|
|
|
·
|
risks inherent in pharmaceutical research and development;
|
|
|
·
|
risks related to timing and possible delays in our clinical trials;
|
|
|
·
|
risks related to some of our clinical trials being conducted in, and subject to the laws of, foreign
countries;
|
|
|
·
|
risks related to our pharmaceutical products being subject to intense regulatory approval processes
in the United States and other foreign jurisdictions;
|
|
|
·
|
risks related to being subject to government manufacturing and testing regulations;
|
|
|
·
|
risks related to the extremely competitive biotechnology industry and our competition with larger
companies with greater resources;
|
|
|
·
|
risks related to our reliance on patents and proprietary rights to protect our technology;
|
|
|
·
|
risks related to potential product liability claims;
|
|
|
·
|
risks related to our limited manufacturing experience and reliance on third parties to commercially
manufacture our products, if and when developed;
|
|
|
·
|
risks related to our new products not being accepted by the medical community or consumers;
|
|
|
·
|
risks related to our technologies becoming obsolete;
|
|
|
·
|
risks related to our dependence on third party relationships for research and clinical trials;
|
|
|
·
|
risks related to our license, development, supply and distribution agreement with Adlai Nortye
Biopharma Co. Ltd.;
|
|
|
·
|
risks related to our lack of operating revenues and history of losses;
|
|
|
·
|
uncertainty regarding our ability to obtain third-party reimbursement for the costs of our product;
|
|
|
·
|
risks related to other third-party arrangements;
|
|
|
·
|
risks related to our ability to obtain additional financing to fund future research and development
of our products and to meet ongoing capital requirements;
|
|
|
·
|
risks related to potential increases in the cost of director and officer liability insurance;
|
|
|
·
|
risks related to our dependence on key employees and collaborators;
|
|
|
·
|
risks related to Barbados law, including those relating to the enforcement of judgments obtained
in Canada or the United States;
|
|
|
·
|
risks related to the effect of changes in the law on our corporate structure;
|
|
|
·
|
risks related to expenses in foreign currencies and our exposure to foreign currency exchange rate
fluctuations;
|
|
|
·
|
risks related to fluctuations in interest rates;
|
|
|
·
|
risks related to information technology systems; and
|
|
|
·
|
risks related to our Common Shares and Warrants.
|
This list is not exhaustive of the factors
that may affect any of the Corporation’s forward-looking statements. Some of the important risks and uncertainties that could
affect forward-looking statements are described further under the heading “
Risk Factors
” in this Prospectus
Supplement, in the Prospectus and in the Corporation’s Annual Report (as defined below). If one or more of these risks or
uncertainties materializes, or if underlying assumptions prove incorrect, our actual results may vary materially from those expected,
estimated or projected. Forward-looking statements in this document are not a prediction of future events or circumstances, and
those future events or circumstances may not occur. Given these uncertainties, users of the information included herein, including
investors and prospective investors, are cautioned not to place undue reliance on such forward-looking statements. Investors should
consult our quarterly and annual filings with the securities commissions or similar regulatory authorities in Canada and the SEC
for additional information on risks and uncertainties relating to forward-looking statements.
The Corporation cautions that the foregoing
list of factors that may affect future results is not exhaustive. The forward-looking information contained in this Prospectus
Supplement, the Prospectus and the documents incorporated by reference in the Prospectus is made as of the date of such documents.
The forward-looking information contained in this Prospectus Supplement, the Prospectus and in the documents incorporated by reference
in the Prospectus is expressly qualified by this cautionary statement. The Corporation does not undertake any obligation to publicly
update or revise any forward-looking information except as required pursuant to applicable securities laws.
DOCUMENTS INCORPORATED
BY REFERENCE
This Prospectus Supplement is deemed to
be incorporated by reference into the Prospectus solely for the purposes of the Offering.
Information has been incorporated by reference
in the Prospectus from documents filed with securities commissions or similar authorities in Canada.
Copies of the documents
incorporated herein by reference may be obtained on request without charge from our Corporate Secretary at 210, 1167 Kensington
Crescent N.W., Calgary, Alberta, T2N 1X7 telephone (403) 670-7377, and are available electronically under the Corporation’s
profile on SEDAR (www.sedar.com) and on EDGAR (www.sec.gov/edgar.shtml).
The following documents, filed with the securities
commissions or similar regulatory authorities in each of the provinces of Canada and filed with, or furnished to, the SEC are specifically
incorporated by reference into, and form an integral part of, the Prospectus:
|
|
·
|
our annual report on Form 20-F (“
Annual Report
”) dated March 15, 2019, for the
year ended December 31, 2018 (filed in Canada with the Canadian securities regulatory authorities in lieu of an annual information
form);
|
|
|
·
|
our management information circular dated March 7, 2019 relating to the annual general meeting
of shareholders held on May 2, 2019;
|
|
|
·
|
our audited consolidated financial statements, together with the notes thereto, as at
December 31, 2018 and 2017, which comprise the consolidated statements of financial position as at December 31, 2018 and
2017, and the consolidated statements of loss and comprehensive loss, changes in equity, and cash flows for the years ended
December 31, 2018, 2017 and 2016, together with the independent auditors’ report thereon;
|
|
|
·
|
our management’s discussion and analysis of financial condition and results of operations
dated March 7, 2019, for the year ended December 31, 2018;
|
|
|
·
|
our unaudited interim consolidated financial statements, together with the notes thereto, as at
June 30, 2019 and 2018, which comprise the interim consolidated statements of financial position as at June 30, 2019 and 2018,
and the interim consolidated statements of loss and comprehensive loss, changes in equity, and cash flows for the three and six
months ended June 30, 2019 and 2018;
|
|
|
·
|
our management’s discussion and analysis of financial condition and results of operations
dated August 2, 2019, for the three and six months ended June 30, 2019.
|
Any documents of the type required by National
Instrument 44-101 -
Short Form Prospectus Distributions
to be incorporated by reference in a short form prospectus, including
any annual information form, annual report on Form 20-F, comparative annual consolidated financial statements and the auditors’
report thereon, comparative interim consolidated financial statements, management’s discussion and analysis of financial
condition and results of operations, material change report (except a confidential material change report), business acquisition
report and information circular, if filed by us with the securities commissions or similar authorities in Canada after the date
of this Prospectus Supplement and prior to the date on which the Offering under this Prospectus Supplement ends, shall be deemed
to be incorporated by reference in the Prospectus.
In addition, to the extent that any document
or information incorporated by reference in the Prospectus is included in any report filed with or furnished to the SEC pursuant
to the
United States Securities Exchange Act of 1934, as amended
(the “
U.S. Exchange Act
”), after the
date of this Prospectus Supplement and prior to the date on which the Offering under this Prospectus Supplement ends, such document
or information shall be deemed to be incorporated by reference as an exhibit to the registration statement of which this Prospectus
Supplement and the Prospectus forms a part (in the case of documents or information deemed furnished on Form 6-K or Form 8-K, only
to the extent specifically stated therein).
Any statement contained in this Prospectus
Supplement, the Prospectus or in a document incorporated or deemed to be incorporated by reference in the Prospectus shall be deemed
to be modified or superseded for the purposes of this Prospectus Supplement and the Prospectus to the extent that a statement contained
herein or in any other subsequently filed document which also is, or is deemed to be, incorporated by reference in the Prospectus
or therein modifies or supersedes such statement. The modifying or superseding statement need not state that it has modified or
superseded a prior statement or include any other information set forth in the document that it modifies or supersedes. The making
of a modifying or superseding statement shall not be deemed an admission for any purposes that the modified or superseded statement,
when made, constituted a misrepresentation, an untrue statement of a material fact or an omission to state a material fact that
was required to be stated or that was necessary to make a statement not misleading in light of the circumstances in which it was
made. Any statement so modified or superseded shall not be deemed, except as so modified or superseded, to constitute a part of
this Prospectus Supplement or the Prospectus.
DOCUMENTS FILED AS PART
OF THE REGISTRATION STATEMENT
The following documents have been or will be
filed with the SEC as part of the registration statement of which this Prospectus Supplement and the Prospectus forms a part: (i)
the documents set out under the heading “
Documents Incorporated by Reference
” in this Prospectus Supplement
and the Prospectus; (ii) the consents of the Corporation’s auditor and legal counsels; (iii) the powers of attorney from
the directors and certain officers of the Corporation; (iv) the Underwriting Agreement described in this Prospectus Supplement;
and (v) the Warrant Agency Agreement described in this Prospectus Supplement.
CURRENCY AND EXCHANGE
RATE INFORMATION
In this Prospectus Supplement and the accompanying Prospectus, unless
otherwise indicated, all dollar amounts and references to “$” and “US$” are to U.S. dollars and references
to “C$” are to Canadian dollars. This Prospectus Supplement and the accompanying Prospectus and the documents incorporated
by reference in the Prospectus contain translations of some Canadian dollar amounts into U.S. dollars solely for your convenience.
The following table sets forth, for the periods indicated, the high,
low, average and period-end rates of exchange for US$1.00, expressed in Canadian dollars, posted by the Bank of Canada:
|
|
|
Year
Ended December 31
|
|
|
|
|
2018
(1)
|
|
|
2017
(2)
|
|
|
2016
(2)
|
|
|
Highest rate during the
period
|
|
C
|
1.3642
|
|
|
C$
|
1.3743
|
|
|
C$
|
1.4589
|
|
|
Lowest rate during the period
|
|
C
|
1.2288
|
|
|
C$
|
1.2128
|
|
|
C$
|
1.2544
|
|
|
Average rate for the period
|
|
C
|
1.2957
|
|
|
C$
|
1.2986
|
|
|
C$
|
1.3248
|
|
|
Rate at the end of the period
|
|
C
|
1.3642
|
|
|
C$
|
1.2545
|
|
|
C$
|
1.3427
|
|
Notes:
|
|
(1)
|
2018 and 2017 data from the Bank of Canada reflects the
daily average noon rates.
|
|
|
(2)
|
2016 data from the Bank of Canada reflects the noon exchange
rates.
|
On August 13, 2019, the daily average exchange rate posted by the Bank of Canada for conversion of U.S.
dollars into Canadian dollars was US$1.00 = C$1.3236. Unless otherwise indicated, currency translation in this Prospectus Supplement
reflect the August 13, 2019 rate.
SUMMARY OF THE OFFERING
|
Issuer:
|
Oncolytics Biotech Inc.
|
|
|
|
|
Offering:
|
US$3,742,016 aggregate amount of Units.
|
|
|
|
|
Price to the Public:
|
US$0.81 per Unit
|
|
|
|
|
Over-allotment Option:
|
The Underwriter has the option (the “
Over-Allotment Option
”), exercisable in whole or in part to purchase up to an additional 692,965 Unit Shares (each, an “
Over-Allotment Unit Share
”) and/or 692,965 Warrants (each, an “
Over-Allotment Warrant
”) (up to 15% of the number of Unit Shares and Warrants sold in the primary offering) from us at the Price to the Public for a period of 30 days from and including the date of this Prospectus Supplement to cover over-allotments, if any, and for market stabilization purposes.
|
|
|
|
|
Warrants:
|
Each Warrant is exercisable into one Warrant Share at an exercise price of $0.90 per Warrant Share at any time prior to 5:00 p.m. (New York City time) on the date that is five years following the closing of the Offering, subject to adjustment in certain events.
|
|
|
|
|
Use of Proceeds:
|
The estimated net proceeds to be received
by us from the sale of the Units will be US$3,180,075 (approximately C$4,209,150), after deducting the Underwriter Fee of US$261,941.13
and the expenses of the Offering, estimated at US$300,000 (or US$3,702,085.54 (approximately C$4,900,000) if the Over-Allotment
Option is exercised in full).
We intend to allocate approximately US$2.5
million for the advancement of the Corporation’s BRACELET-1 study; approximately US$0.5 million for manufacturing and drug
supply costs; and the remainder for general corporate and administrative expenses. The amounts actually expended for the purposes
described above may vary significantly depending upon a number of factors, including those listed under the heading “
Risk
Factors
” in this Prospectus Supplement. See “
Use of Proceeds
”.
|
|
|
|
|
Nasdaq Symbol:
|
ONCY
|
|
|
|
|
TSX Symbol:
|
ONC
|
|
|
|
|
Risk Factors:
|
You should carefully read and consider the information set forth under the heading “Risk Factors” beginning on page S-10 of this Prospectus Supplement and on page 6 of the Prospectus.
|
RISK FACTORS
Prospective purchasers of Units should consider
carefully the risk factors set out in this Prospectus Supplement, the Prospectus and the documents incorporated by reference in
the Prospectus. Discussions of certain risks affecting Oncolytics in connection with its business are set forth under “
Risk
Factors
” in the Prospectus and in our annual disclosure documents filed with the various securities regulatory authorities
which are incorporated by reference in the Prospectus.
Volatility of market price of the Common
Shares
The market price of the Common Shares may be
volatile. The volatility may affect the ability of holders of Common Shares to sell the Common Shares at an advantageous price.
Market price fluctuations in the Common Shares may be due to the Corporation’s operating results failing to meet the expectations
of securities analysts or investors in any quarter, downward revision in securities analysts’ estimates, governmental regulatory
action, adverse change in general market conditions or economic trends, acquisitions, dispositions or other material public announcements
by the Corporation or its competitors, along with a variety of additional factors, including, without limitation, those set forth
under “
Special Notice Regarding Forward-Looking Statements
” in this Prospectus Supplement. In addition, the
market price for securities in the stock markets, including the Nasdaq and the TSX, recently experienced significant price and
trading fluctuations. These fluctuations have resulted in volatility in the market prices of securities that often has been unrelated
or disproportionate to changes in operating performance. These broad market fluctuations may adversely affect the market price
of the Common Shares.
Purchasers will suffer immediate and substantial
dilution
Because the price of the Common Shares under
the Offering significantly exceeds the net tangible book value per share of the Common Shares, a purchaser of Common Shares in
the Offering will incur immediate and substantial dilution of his, her or its investment. If the Over-Allotment Option is exercised,
you will incur additional dilution. See “
Dilution
” in this Prospectus Supplement.
The Corporation will have broad discretion
over the use of the net proceeds from the Offering and the Corporation may not use these proceeds in a manner desired by the Corporation’s
shareholders
Management will have broad discretion with
respect to the use of the net proceeds from the Offering and investors will be relying on the judgment of management regarding
the application of these proceeds. Management could spend most of the net proceeds from the Offering in ways that the Corporation’s
shareholders may not desire or that do not yield a favorable return. You will not have the opportunity, as part of your investment
in the Units, to influence the manner in which the net proceeds of the Offering are used. At the date of this Prospectus Supplement,
the Corporation intend to use the net proceeds from the Offering as described under the heading “
Use of Proceeds
”.
However, the Corporation’s needs may change as the business and the industry the Corporation addresses evolve. As a result,
the proceeds to be received in the Offering may be used in a manner significantly different from the Corporation’s current
expectations.
The Warrants will not be listed on any exchange
The Warrants are not listed on any exchange
and the Corporation does not intend to list the Warrants on any exchange. Investors may be unable to sell the Warrants at the prices
desired or at all. There is no existing trading market for the Warrants and there can be no assurance that a liquid market will
develop or be maintained for the Warrants, or that an investor will be able to sell any of the Warrants at a particular time (if
at all). The liquidity of the trading market in the Warrants and the sale price, if any, for the Warrants, may be adversely affected
by, among other things:
|
|
·
|
changes in the overall market for the Warrants;
|
|
|
·
|
changes in the Corporation’s financial performance
or prospects;
|
|
|
·
|
changes or perceived changes in the Corporation’s
creditworthiness;
|
|
|
·
|
the prospects for companies in the industry generally;
|
|
|
·
|
the number of holders of the Warrants; and
|
|
|
·
|
the interest of securities dealers in making a market for
the Warrants.
|
Positive return on an investment in the
Units is not guaranteed
There is no guarantee that an investment in
the Units will earn any positive return in the short term or long term. A purchase under the Offering involves a high degree of
risk and should be undertaken only by purchasers whose financial resources are sufficient to enable them to assume such risks and
who have no need for immediate liquidity in their investment. An investment in the Units is appropriate only for purchasers who
have the capacity to absorb a loss of some or all of their investment.
The Corporation does not currently intend
to pay any cash dividends on the Common Shares in the foreseeable future; therefore, the Corporation’s shareholders may not
be able to receive a return on their Common Shares until they sell them
The Corporation has never paid or declared
any cash dividends on its Common Shares. The Corporation does not anticipate paying any cash dividends on its Common Shares in
the foreseeable future because, among other reasons, the Corporation currently intends to retain any future earnings to finance
its business. The future payment of dividends will be dependent on factors such as cash on hand and achieving profitability, the
financial requirements to fund growth, the Corporation’s general financial condition and other factors the board of directors
of the Corporation may consider appropriate in the circumstances. Until the Corporation pays dividends, which it may never do,
its shareholders will not be able to receive a return on their Common Shares unless they sell them.
You may be unable to enforce actions against
us, certain of our directors and officers, or the experts named in this Prospectus Supplement under U.S. federal securities laws.
We are a company continued under the laws of
the Province of Alberta, Canada. Most of our directors and officers as well as the certain of the experts named in this Prospectus
Supplement and the accompanying Prospectus, reside principally in Canada. Because all or a substantial portion of our assets and
the assets of these persons are located outside of the United States, it may not be possible for you to effect service of process
within the United States upon us or those persons. Furthermore, it may not be possible for you to enforce against us or those persons
in the United States, judgments obtained in U.S. courts based upon the civil liability provisions of the U.S. federal securities
laws or other laws of the United States. There is doubt as to the enforceability, in original actions in Canadian courts, of liabilities
based upon U.S. federal securities laws and as to the enforceability in Canadian courts of judgments of U.S. courts obtained in
actions based upon the civil liability provisions of the U.S. federal securities laws. Therefore, it may not be possible to enforce
those actions against us, certain of our directors and officers or certain of the experts named in this Prospectus Supplement.
The Corporation is likely a “passive
foreign investment company” which may have adverse U.S. federal income tax consequences for U.S. shareholders
U.S. holders of Common Shares should be aware
that the Corporation believes it was classified as a passive foreign investment company (“
PFIC
”) during the
tax year ended December 31, 2018 and based on current business plans and financial expectations, the Corporation expects that
it will be a PFIC for the current tax year and may be a PFIC in future tax years. If the Corporation is a PFIC for any year during
a U.S. shareholder’s holding period of the Common Shares, then such U.S. shareholder generally will be required to treat
any gain realized upon a disposition of Common Shares, or any “excess distribution” received on its Common Shares,
as ordinary income, and to pay an interest charge on a portion of such gain or distribution, unless the shareholder makes a timely
and effective “qualified electing fund” election (“
QEF Election
”) or a “mark-to-market”
election with respect to the Common Shares. A U.S. shareholder who makes a QEF Election generally must report on a current basis
its share of the Corporation’s net capital gain and ordinary earnings for any year in which the Corporation is a PFIC, whether
or not the Corporation distributes any amounts to its shareholders. A U.S. shareholder who makes a mark-to-market election generally
must include as ordinary income each year the excess of the fair market value of the Common Shares over the taxpayer’s adjusted
tax basis therein. This paragraph is qualified in its entirety by the discussion below under the heading “
Certain United
States Federal Income Tax Considerations.
” Each U.S. shareholder should consult its own tax advisors regarding the PFIC
rules and the U.S. federal income tax consequences of the acquisition, ownership, and disposition of Common Shares.
THE CORPORATION
Oncolytics Biotech Inc. was incorporated pursuant
to the ABCA on April 2, 1998 as 779738 Alberta Ltd. On April 8, 1998, we amended our articles of incorporation (the “
Articles
”)
and changed our name to Oncolytics Biotech Inc. On July 29, 1999, we amended our Articles by removing the private company
restrictions included therein and subdivided the 2,222,222 Common Shares issued and outstanding into 6,750,000 Common Shares. On
February 9, 2007, we amended our Articles to permit shareholder meetings to be held at any place in Alberta or at any other
location as determined by our board of directors (the “
Board
”). On May 22, 2018, we amended our Articles of
Incorporation to effect a consolidation (the “
Consolidation
”) of the Common Shares on the basis of 9.5 pre-Consolidation
Common Shares for each one post-Consolidation Common Share.
We have two material operating subsidiaries:
Oncolytics Biotech (Barbados) Inc. and Oncolytics Biotech (US) Inc., a Delaware corporation. Oncolytics Biotech (Barbados) Inc.
is incorporated pursuant to the laws of Barbados and is a wholly-owned direct subsidiary of the Corporation. Oncolytics Biotech
(U.S.) Inc. is incorporated pursuant to the laws of Delaware and is a wholly-owned direct subsidiary of Oncolytics Biotech (Barbados)
Inc.
Our head office and principal place of business
is located at 210, 1167 Kensington Crescent N.W., Calgary, Alberta, T2N 1X7. Our registered office is located at 4000, 421 - 7
th
Avenue
S.W., Calgary, Alberta, T2P 4K9.
RECENT DEVELOPMENTS
On June 5, 2019, the Corporation announced
that it has entered into an agreement with Merck KGaA, Darmstadt, Germany, a leading science and technology company, which operates
in the United States and Canada as EMD Serono, and Pfizer Inc. (NYSE:PFE). The agreement is to conduct a clinical study of pelareorep
in combination with paclitaxel and avelumab*, a human anti-PD-L1 antibody, for the treatment of hormone-receptor positive, human
epidermal growth factor 2-negative (HR+ / HER2-) metastatic breast cancer (“
mBC
”). The Corporation and Pfizer
will equally share costs associated with the phase 2 clinical trial.
The study known as BRACELET-1 (
BR
east
c
A
n
CE
r with the Oncolytic Reovirus Pe
L
areor
E
p in Combina
T
ion with anti-PD-L 1 and paclitaxel)
is an open label study that will enroll 45 patients into three cohorts with 15 patients per cohort: paclitaxel alone, paclitaxel
in combination with pelareorep and paclitaxel in combination with both pelareorep and avelumab. The study will examine the expression
of immune-related biomarkers to identify changes in T cell clonality between pre-treatment and on-therapy biopsies to confirm our
previously identified biomarker and is designed to assess efficacy in terms of overall response rate at week 16 per RECIST 1.1
and iRECIST. The safety of the combination will also be evaluated. During the period of the study and for 90 days after a pre-determined
interim analysis, the Corporation may exclusively develop pelareorep in HR+ / HER2- mBC with Merck KGaA, Darmstadt, Germany, and
Pfizer.
*Avelumab is under clinical investigation for
treatment of hormone receptor-positive, human epidermal growth factor 2-negative (HR+ / HER2-) metastatic breast cancer and has
not been demonstrated to be safe and effective for this use. There is no guarantee that avelumab will be approved for HR+ / HER2-
metastatic breast cancer by any health authority worldwide.
BUSINESS OF THE CORPORATION
General
Since our inception in April of 1998, Oncolytics
Biotech Inc. has been a development stage company and we have focused our research and development efforts on the development of
pelareorep, a systemically administered immuno-oncology (“
I-O
”) viral agent with the potential to treat a variety
of cancers. We have not been profitable since our inception and expect to continue to incur substantial losses as we continue research
and development efforts. We do not expect to generate significant revenues until, if and when, pelareorep becomes commercially
viable.
Our potential product for human use, pelareorep,
an unmodified reovirus, is a first in class systemically administered I-O viral agent for the treatment of solid tumors and hematological
malignancies.
Scientific Background
Pelareorep’s anti-tumor activity is based
on three modes of action which are complementary but not interdependent (see Figure 1, below):
|
|
·
|
Selective viral replication in permissive cancer cells which leads to tumor cell lysis.
|
|
|
·
|
Activation of innate immunity in response to the infection which results in a cascade of chemokines/cytokines
causing natural killer (“
NK
”) cells to be activated and attack cancer cells.
|
|
|
·
|
A specific adaptive immune response triggered by tumor- and viral-associated antigens displayed
by antigen-presenting cells (including infected tumor cells and/or dendritic cells, “
APCs
”) to T cells.
|
Summary of Research and Development highlights
Preclinical and Translational Research data
to date indicates the following:
|
|
·
|
Pelareorep has anticancer effects in models of metastatic cancers that can prolong survival in
these models when using immuno-competent rodents.
|
|
|
·
|
The survival benefit in animal models can be enhanced when pelareorep is given in combination with
chemotherapy, immunotherapy or radiotherapy.
|
|
|
·
|
A toxic dose of pelareorep has not been reached/established in animal models and infection presents
with minimal side-effects.
|
Clinical data to date indicate the following:
|
|
·
|
More than 1,400 patients have been enrolled in clinical studies conducted in the United States,
Canada and the European Union. Of these, more than 1,000 patients received pelareorep, with over 930 via intravenous (“
IV
”)
administration and over 90 by intratumoral injections (“
ITu
”). The remaining patients were randomized to control
arms on other therapies.
|
|
|
·
|
Pelareorep has been administered as single or multiple doses (intratumoral or intravenous), either
as a mono-therapy or in combination with chemotherapy, immunotherapy (e.g., checkpoint inhibitors), and radiotherapy.
|
|
|
·
|
No Maximum Tolerated Dose (“
MTD
”) for intravenous pelareorep as mono-therapy
was defined in the two Phase 1 trials (REO 004 and 005). Dose-limiting toxicities (“
DLTs
”) were seen in some
of the combination trials with pelareorep and chemotherapy, which generally enrolled heavily pre-treated patients.
|
|
|
·
|
When combined with chemotherapeutic agents, pelareorep does not appear to enhance either the frequency
or severity of the adverse effects of the chemotherapeutic agents.
|
|
|
·
|
There is emerging evidence that pelareorep may impact overall survival (“
OS
”)
in mBC and metastatic adenocarcinoma of the pancreas (“
MAP
”):
|
|
|
·
|
In a randomized, controlled Phase 2 study of paclitaxel with pelareorep versus paclitaxel alone
in mBC (Canadian Cancer Trials Group IND. 213) median survival time was greater for subjects treated with paclitaxel and pelareorep
(median 17.4 months) than subjects treated with paclitaxel alone (10.4 months, hazard ratio (“
HR
”) 0.65).
|
|
|
·
|
In a single arm study with gemcitabine plus pelareorep in first line MAP (REO 017) the median overall
survival (mOS) was 10 months with a 1-year and 2-year survival of 46% and 24%, respectively.
|
|
|
·
|
In a two-arm Phase 2 randomized study (NCI 8601), patients with MAP were randomized to receive
either carboplatin, paclitaxel and pelareorep (test arm) or carboplatin and paclitaxel alone (control arm). The median OS was similar
for both arms, but the probability of survival at Year 2 was 20% in the test arm versus 9% in the control arm.
|
Mechanism of Action
Figure 1. Proposed mechanism of action for pelareorep.

Direct cell lysis - Reovirus Replication in Permissive Cancer
Cells
Selective viral replication and lysis in cancer
cells and not normal cells is mediated by the host cellular protein dsRNA-activated protein kinase (“
PKR
”).
In non-cancer cells that are infected with reovirus, PKR activates in the presence of the virus which in turn inhibits viral gene
translation. However, in permissive cancer cells, PKR activation is inhibited, allowing for viral gene translation and eventual
cell lysis.
It was originally established that selective
lysis with reovirus was mediated by tumor cells with an activated rat sarcoma virus oncogene (“
RAS
”) pathway,
since active RAS inhibits PKR activation. However, more recent investigations have revealed that reovirus replication is not just
restricted to cells with an active RAS pathway, oncogenic mutations and amplifications in upstream and downstream mediators of
the RAS-pathway also allow for viral replication and oncolysis. Moreover, active RAS is known to stimulate over 18 downstream effector
proteins, many of which have been shown to facilitate viral replication, such as. Cells bearing dysfunctional or deleted tumor
suppressor genes and or chemo- or radiation-induced cell stress also show increased sensitivity to reovirus replication and lysis.
Induction of Innate Immunity
Preclinical and clinical studies provide compelling
lines of evidence that pelareorep functions as an immunogenic agent. Indeed, preclinical studies by Steele and colleagues demonstrated
that melanoma cells infected with pelareorep can produce an innate immune response triggering the release of inflammatory cytokines.
This inflammatory environment promotes a chemotactic response in NK cells, dendritic cells, and cytotoxic T-cells, altering the
tumour microenvironment to support bystander immune-mediated cancer cell death. Intriguingly, preclinical studies have also demonstrated
that the beneficial immunogenic functions of pelareorep can occur independent of viral replication. Pelareorep performs this immunogenic
function, in part, by activating dendritic cells, key regulators of both adaptive and innate immunity. Dendritic cells activated
by reovirus in turn stimulate the innate antitumor activity of natural killer (“
NK
”) cells and aid in the priming
of specific antitumor cytotoxic lyphocyte, demonstrating that dendritic cells’ recognition of reovirus may trigger a beneficial
innate immune response.
A clinical trial with pelareorep (REO 013)
provided an opportunity to study human NK cell activation in a controlled manner. Ten colorectal cancer patients with liver metastases
received between one and five doses of pelareorep prior to surgical resection of their tumor. NK cell activation peaked 24 to 48
hours post-infection, coincident with a peak of pro-inflammatory cytokines. NK cells within reovirus-treated blood mononuclear
cells were stimulated to kill tumor targets, but not normal hepatocytes. Moreover, peripheral blood mononuclear cells were able
to hand-off virus to tumors for direct oncolytic killing. Similarly, NK cells within liver mononuclear cells became selectively
cytotoxic towards tumor cells when activated by reovirus. These results showed that reovirus modulates human NK cell activity in
vivo and suggest that this may contribute to the therapeutic effect of pelareorep.
Induction of Adaptive Immunity
Adaptive anti-tumor immunity allows for elimination
of existing cancer cells and performs constant surveillance, preventing relapse, and increasing patient overall survival. An adaptive
immune response requires two signals: a signal from an APC, as well as a co-stimulation signal in the form of cytokines. In the
absence of both signals, the adaptive immune response fails. Therapy with pelareorep has the potential to activate both signals.
Following its therapeutic administration, pelareorep enhances the expression of ‘foreign’ antigens/markers on tumor
cells. Oncolysis of tumor cells exposes tumor-associated antigens (“
TAAs
”) and viral-associated antigens (“
VAAs
”)
for processing and presentation by APCs, such as dendritic cells. Through the combined actions of these immunological events, pelareorep
facilitates the display of novel ‘foreign’ antigens on the surface of infected tumor cells and APCs. Simultaneously,
pelareorep induces an inflammatory response promoting the expression of co-stimulatory molecules and inflammatory cytokines. Together,
pelareorep mediated immunological events over-rule tumor antigen presentation impairments and initiate adaptive anti-tumor immunity.
By promoting the expression of novel antigens
and the release of inflammatory cytokines, pelareorep, promotes an inflamed tumor phenotype. An inflamed tumor phenotype is characterized
by NK and T-cell infiltration, increased expression of chemokines/ cytokines, and increased expression of checkpoint ligands. This
phenotype correlates with an increase in overall survival and has a positive prognostic value for early stage cancers. In patients
with metastatic cancer, an inflamed tumor phenotype is associated with better clinical outcomes when treated with immunotherapies,
including immune checkpoint blockade inhibitors, cancer vaccines, and adoptive T-cell therapies. By promoting an inflamed tumor
phenotype, pelareorep primes an anti-cancer immune response (see Figure 2, below).
Figure 2. Pelareorep primes an anti-cancer immune response

Clinical Development Plan
The primary objective of our clinical development
plan is to obtain regulatory approval for pelareorep as quickly as possible and is based on the compelling mBC survival data that
was presented at the 2017 American Association for Cancer Research Annual Meeting, in Washington, D.C. The second objective is
to expand pelareorep into commercially valuable new treatment areas. Our clinical development program focuses on the three components
of pelareorep’s mechanism of action and includes the following:
Immunotherapy Combinations
This program focuses on the potential for pelareorep
to cause a specific adaptive immune response in combination with checkpoint inhibitors. In 2017, we announced our first data set
combining a checkpoint inhibitor with pelareorep and pembrolizumab (Keytruda®) in pancreatic cancer, which demonstrated safety
and tolerability and in five efficacy evaluable patients, one had a partial response (six-month duration) and two had stable disease
(lasting 126 and 221 days). In the first half of 2019 we announced the regulatory approval and the start of enrollment in our AWARE-1
study which examines the use of pelareorep and atezolizumab (Tecentriq®) in a breast cancer window-of -opportunity study. Additional
studies are now being planned.
Chemotherapy Combinations
This program focuses on the investigation of
chemotherapy combination clinical trials investigating the use of different chemotherapy agents in various tumor types. In 2017,
we reported additional clinical data from our randomized clinical program which includes clinical trial collaborations with the
Canadian Cancer Trials Group (formerly known as the National Cancer Institute of Canada). Specifically, subgroup analysis in the
IND 213 trial in mBC revealed a significant improvement in the overall survival of patients that are hormone receptor positive
(HR+) / human epidermal growth factor receptor 2 negative (HER2-). In HR+/HER2- patients, pelareorep therapy in combination with
paclitaxel doubled the overall survival from 10.8 month with paclitaxel therapy alone to 21.8 months with pelareorep plus paclitaxel.
This increase in overall survival is consistent with previous survival data reported from our U.S. NCI pancreatic trial which suggests
a long-term survival benefit when comparing test and control arms at 24 months.
Business Strategy
Our business strategy is to develop and market
pelareorep in an effective and timely manner, and access additional technologies at a time and in a manner that we believe is best
for our development. We intend to achieve our business strategy by focusing on these key areas:
|
|
·
|
Develop pelareorep through our clinical development plan assessing the safety and efficacy in human
subjects;
|
|
|
·
|
Establish collaborations with experts to assist us with scientific and clinical developments of
this new potential pharmaceutical product;
|
|
|
·
|
Implement strategic alliances with select biopharmaceutical companies and laboratories, at a time
and in a manner whereby such alliances may complement and expand our own research and development efforts, and which may eventually
expand to include providing additive sales and marketing capabilities;
|
|
|
·
|
Utilize our broadening patent base and collaborator network as a mechanism to meet our strategic
objectives; and
|
|
|
·
|
Develop relationships with companies that could be instrumental in assisting us to access other
innovative therapeutics.
|
Our business strategy is based on attaining
a number of commercial objectives, which, in turn, are supported by a number of product development goals. In this Prospectus Supplement,
statements of our “belief” are based primarily upon our results derived to date from our research and development program
with animals, early stage human trials and our most recent data in HR+/HER2- mBC patients, upon which we believe that we have a
reasonable scientific basis to expect the particular results to occur. It is not possible to predict, based upon studies in animals,
or early stage human trials, whether a new therapeutic will ultimately prove to be safe and effective in humans. There are no assurances
that the particular result expected by us will occur.
As of the date hereof, we do not intend to
become a fully integrated pharmaceutical company with substantial in-house research and development, marketing and distribution
or manufacturing capabilities. We are pursuing a strategy of establishing relationships with larger companies as strategic partners.
It is anticipated that future clinical development into large international or pivotal trials would generally occur in conjunction
with a strategic partner or partners, who would contribute expertise and financial assistance. In exchange for certain product
rights and commitments to market our products, the strategic partners would be expected to share in proceeds from the sale of our
product or products.
USE OF PROCEEDS
The estimated net proceeds to be received
by us from the sale of the Units will be approximately US$3,180,075 (approximately C$4,209,150), after deducting the Underwriter
Fee of US$261,941.13 and the expenses of the Offering, estimated at US$300,000 (or approximately US$3,702,085 if the Over-Allotment
Option is exercised in full).
|
|
·
|
US$2.5 million for the advancement of the Corporation’s
BRACELET-1 study;
|
|
|
·
|
US$0.5 million for manufacturing and drug supply costs;
and
|
|
|
·
|
US$180,000 for general corporate and administrative
expenses.
|
The amounts actually expended for the purposes
described above may vary significantly depending upon a number of factors, including those listed under the heading “
Risk
Factors
” in this Prospectus Supplement.
DETAILS OF THE OFFERING
The Offering consists of 4,619,773
Units
(5,312,738 Units if the Underwriter exercises the Over-Allotment Option in full) at a price of US$0.81 per Unit.
Common Shares
We are authorized to issue an unlimited number
of Common Shares. Each Common Share entitles the holder to one vote per share held at meetings of shareholders, to receive such
dividends as declared by us and to receive our remaining property and assets upon dissolution or winding up. Our Common Shares
are not subject to any future call or assessment and there are no pre-emptive, conversion or redemption rights attached to such
shares.
Warrants
The Warrants issued under the Offering will
be governed by a Warrant Agency Agreement (the “
Warrant Agency Agreement
”) to be entered into between the Corporation
and American Stock Transfer & Trust Company, LLC, as agent for the holders of the Warrants (the “
Warrant Agent
”).
The following description is subject to the detailed provisions of the Warrant Agency Agreement. Reference should be made to the
Warrant Agency Agreement for the full text of attributes of the Warrants, which we will file on SEDAR under our profile at www.sedar.com
following the closing of the Offering.
Exercisability
.
The Warrants
are exercisable immediately upon issuance and at any time up to the date that is five years from the date of issuance. The Warrants
will be exercisable, at the option of each holder, in whole or in part by delivering to us a duly executed exercise notice accompanied
by payment in full for the number of Common Shares purchased upon such exercise (except in the case of a cashless exercise as discussed
below). Unless otherwise specified in the Warrant, the holder will not have the right to exercise any portion of the Warrant if
the holder (together with its affiliates) would beneficially own in excess of 4.99% (or, upon election by a holder prior to the
issuance of any Warrants, 9.99%) of the number of Common Shares outstanding immediately after giving effect to the exercise, as
such percentage ownership is determined in accordance with the terms of the Warrants.
For greater certainty, all Warrants,
including the Warrants issued pursuant to, or in connection with, the Over-Allotment Option, will expire on the same expiry
date five years from the Closing Date, subject to acceleration in certain events.
Cashless Exercise
.
In
the event that a registration statement covering Common Shares underlying the Warrants, or an exemption from registration, is
not available for the resale of such Common Shares underlying the Warrants, the holder may, in its sole discretion, exercise the
Warrant in whole or in part and, in lieu of making the cash payment otherwise contemplated to be made to us upon such exercise
in payment of the aggregate exercise price, elect instead to receive upon such exercise the net number of Common Shares determined
according to the formula set forth in the Warrant. In no event shall we be required to make any cash payments or net cash settlement
to the registered holder in lieu of issuance of Warrant Shares underlying the Warrants.
Exercise Price
.
The initial
exercise price per share of Warrants Shares purchasable upon exercise of the Warrants is US$0.90. The exercise price is subject to
appropriate adjustment in the event of certain stock dividends and distributions, stock splits, stock combinations, reclassifications
or similar events affecting our Common Shares and also upon any distributions of assets, including cash, shares or other property
to our shareholders.
Certain Adjustments
.
The
exercise price and the number of Common Shares purchasable upon the exercise of the Warrants are subject to adjustment upon the
occurrence of specific events, including stock dividends, stock splits, combinations and reclassifications of our Common Shares.
Transferability
.
Subject
to applicable laws, the Warrants may be transferred at the option of the holders upon surrender of the Warrants to us together
with the appropriate instruments of transfer.
Warrant Agent and Exchange Listing
.
The Warrants will be issued in registered form under the Warrant Agency Agreement between the Warrant Agent, as warrant agent,
and the Corporation. There will be no market through which the Warrants may be sold and purchasers may not be able to resell the
Warrants purchased in the Offering. This may affect the pricing of the Warrants in the secondary market, the transparency and availability
of trading prices and the liquidity of the Warrants. See “
Risk Factors
”.
Fundamental Transaction
. If,
at any time while the Warrants are outstanding, (1) we consolidate or merge with or into another corporation and we are not the
surviving corporation, (2) we sell, lease, license, assign, transfer, convey or otherwise dispose of all or substantially all of
our assets, (3) any purchase offer, tender offer or exchange offer (whether by us or another individual or entity) is completed
pursuant to which holders of our Common Shares are permitted to sell, tender or exchange their Common Shares for other securities,
cash or property and has been accepted by the holders of 50% or more of our outstanding Common Shares, (4) we effect any reclassification
or recapitalization of our Common Shares or any compulsory share exchange pursuant to which our Common Shares are converted into
or exchanged for other securities, cash or property, or (5) we consummate a stock or share purchase agreement or other business
combination with another person or entity whereby such other person or entity acquires more than 50% of our outstanding Common
Shares, each, a “
Fundamental Transaction
,” then upon any subsequent exercise of the Warrants, the holders thereof
will have the right to receive the same amount and kind of securities, cash or property as it would have been entitled to receive
upon the occurrence of such Fundamental Transaction if it had been, immediately prior to such Fundamental Transaction, the holder
of the number of Warrant Shares then issuable upon exercise of the Warrant, and any additional consideration payable as part of
the Fundamental Transaction. Notwithstanding the foregoing, in the event of a fundamental transaction (other than certain fundamental
transactions where the Company remains the surviving company) as described above, the holder may, subject to certain conditions,
require the Company or a successor entity to purchase the warrant from the holder by paying to the holder an amount in cash equal
to the Black-Scholes value of the remaining unexercised portion of the warrant on the effective date of such change of control;
provided, however, that, if the change of control is not within the Company’s control, including not approved by the Company’s
board of directors, the holder will only be entitled to receive from the Company or any successor entity, as of the date of consummation
of such change of control, the same type or form of consideration (and in the same proportion), at the Black-Scholes value of the
unexercised portion of the warrant, that is being offered and paid to the holders of our common stock in connection with the change
of control, whether that consideration is in the form of cash, stock or any combination thereof, or whether the holders of common
stock are given the choice to receive from among alternative forms of consideration in connection with the change of control.
Rights as a Shareholder
.
Except as otherwise provided in the Warrants or by virtue of such holder’s ownership of Common Shares, the holder of a Warrant
does not have the rights or privileges of a holder of our Common Shares, including any voting rights, until the holder exercises
the Warrant.
No fractional Warrant
Shares will be issuable upon the exercise of any Warrants, and no cash or other consideration will be paid in lieu of fractional
shares.
CONSOLIDATED CAPITALIZATION
The following table sets forth the consolidated
capitalization of the Corporation as at June 30, 2019 before and after giving effect to the Offering. There has not been any material
change in the share and loan capital of the Corporation, on a consolidated basis, since the Corporation’s most recently filed
financial statements for the three and six months ended June 30, 2019. The table below should be read in conjunction with our interim
consolidated financial statements for the three and six months ended June 30, 2019 and the related management’s discussion
and analysis thereof, incorporated in each case by reference in this Prospectus Supplement and the Prospectus.
|
|
|
As
at June 30, 2019
|
|
|
As at June 30, 2019 after
giving effect to the
Offering
(1)(3)
|
|
|
As at June 30, 2019 after
giving effect to the
Offering, including the
Over-Allotment Option
(2)(3)
|
|
|
Share capital
|
|
|
C$292,611,439
(20,359,295
Common Shares)
|
|
|
|
C$296,768,621
(24,979,068 Common
Shares)
|
|
|
|
C$297,451,025
(25,672,033 Common Shares)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Warrants
|
|
C$
|
3,617,570
|
|
|
C
$
|
3,669,535
|
|
|
C$
|
3,730,030
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Contributed surplus
|
|
C$
|
28,752,377
|
|
|
C$
|
28,752,377
|
|
|
C$
|
28,752,377
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Accumulated other comprehensive income
|
|
C$
|
482,549
|
|
|
C$
|
482,549
|
|
|
C$
|
482,549
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Accumulated deficit
|
|
C$
|
(321,675,987)
|
|
|
C$
|
(321,675,987
|
)
|
|
C$
|
(321,675,987
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Total shareholders’ equity
|
|
C$
|
3,787,948
|
|
|
C$
|
7,997,095
|
|
|
C$
|
8,688,028
|
|
Notes:
|
|
(1)
|
Based on the issuance of 4,619,773 Common Shares underlying
Units pursuant to the Offering for aggregate gross proceeds of US$3,742,016.13, less the Underwriter Fee of US$261,941.13 and
the expenses of the Offering, estimated at US$300,000.
|
|
|
(2)
|
Based on the issuance of 5,312,738 Common Shares underlying
Units pursuant to the Offering, assuming the exercise in full of the Over-Allotment Option, for aggregate gross proceeds of US$4,303,317.78,
less the Underwriter Fee of US$301,232.24 and the expenses of the Offering, estimated at US$300,000.
|
|
|
(3)
|
Assumes no exercise of Warrants underlying Units pursuant
to the Offering.
|
DILUTION
If you invest in the Common Shares, your interest
will be diluted to the extent of the difference between the public Offering price per Common Share and the as adjusted tangible
book value per Common Share after the Offering. The Corporation’s net tangible book value as of June 30, 2019 was C$3,787,948,
or C$0.1861 per share. The Corporation’s as adjusted net tangible book value per share
set forth below represents its total tangible assets less total liabilities, divided by the number of Common Shares outstanding.
Dilution results from the fact that the
per Common Share Offering price is substantially in excess of the book value per Common Share attributable to the existing shareholders
for the Corporation’s presently outstanding Common Shares. After giving effect to the Corporation’s issuance and sale
of 4,619,773 Common Shares and 4,619,773 Warrants under the Offering at a public Offering price of US$0.81 per Unit (approximately
C$1.07), and after deducting the Underwriter Fee and estimated offering expenses payable by the Corporation, the as adjusted net
tangible book value as of June 30, 2019 would have been C$7,997,095, or C$0.3202 per share. This represents an immediate increase
in net tangible book value to existing shareholders of C$0.1341 per share. The public Offering price per share will significantly
exceed the net tangible book value per share. Accordingly, new investors who purchase shares in the Offering will suffer an immediate
dilution of their investment of approximately C$0.75 per share. The following table illustrates this per share dilution to the
new investors purchasing shares in the Offering:
|
|
|
Offering
(1)(2)
|
|
|
Offering price per Unit
|
|
C$
|
1.07
|
|
|
Net tangible book value per Common Share as of June 30, 2019
|
|
C$
|
0.1861
|
|
|
Increase per Common Share attributable to the Offering
|
|
C$
|
0.1341
|
|
|
As adjusted net tangible book value per Common Share after the Offering
|
|
C$
|
0.3202
|
|
|
Dilution per Common Share to new investors
|
|
C$
|
0.7498
|
|
Notes:
|
|
(1)
|
Based on the issuance of 4,619,773 Units pursuant to the Offering for aggregate gross proceeds
of US$3,742,016.13, less the Underwriter Fee of US$ 261,941.13 and the expenses of the Offering, estimated at US$300,000.
|
|
|
(2)
|
Assumes no exercise of Warrants underlying Units pursuant to the Offering.
|
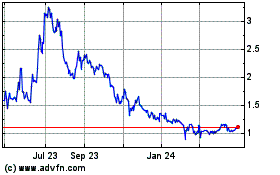
TRADING PRICE AND VOLUME
The Common Shares are listed and posted for trading on the TSX under the trading symbol “ONC”
and on the NASDAQ under the trading symbol “ONCY”. On August 13, 2019, the closing price of our Common Shares on Nasdaq
was US$0.90 per Common Share and on the TSX was C$1.19 per Common Share.
The following table sets forth the market price
ranges and the aggregate volume of trading of the Common Shares on the TSX and NASDAQ for the periods indicated:
|
|
|
TSX
|
|
|
NASDAQ
|
|
|
|
|
High
|
|
|
Low
|
|
|
Close
|
|
|
Volume
|
|
|
High
|
|
|
Low
|
|
|
Close
|
|
|
Volume
|
|
|
Period
|
|
(C$)
|
|
|
(C$)
|
|
|
(C$)
|
|
|
(Shares)
|
|
|
(US$)
|
|
|
(US$)
|
|
|
(US$)
|
|
|
(Shares)
|
|
|
2018
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
August
|
|
|
6.76
|
|
|
|
5.23
|
|
|
|
5.46
|
|
|
|
447,865
|
|
|
|
5.31
|
|
|
|
4.00
|
|
|
|
4.18
|
|
|
|
1,362,577
|
|
|
September
|
|
|
6.52
|
|
|
|
4.94
|
|
|
|
4.96
|
|
|
|
747,789
|
|
|
|
5.12
|
|
|
|
3.77
|
|
|
|
3.86
|
|
|
|
1,412,087
|
|
|
October
|
|
|
5.47
|
|
|
|
3.41
|
|
|
|
4.05
|
|
|
|
1,006,423
|
|
|
|
4.33
|
|
|
|
2.60
|
|
|
|
3.07
|
|
|
|
1,943,334
|
|
|
November
|
|
|
4.12
|
|
|
|
2.82
|
|
|
|
3.50
|
|
|
|
503,826
|
|
|
|
3.20
|
|
|
|
2.13
|
|
|
|
2.61
|
|
|
|
858,710
|
|
|
December
|
|
|
3.44
|
|
|
|
2.26
|
|
|
|
2.51
|
|
|
|
526,966
|
|
|
|
2.61
|
|
|
|
1.6382
|
|
|
|
1.81
|
|
|
|
1,158,436
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
2019
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
January
|
|
|
3.25
|
|
|
|
2.43
|
|
|
|
2.72
|
|
|
|
704,629
|
|
|
|
2.50
|
|
|
|
1.76
|
|
|
|
2.09
|
|
|
|
2,008,478
|
|
|
February
|
|
|
3.13
|
|
|
|
2.55
|
|
|
|
2.70
|
|
|
|
605,259
|
|
|
|
2.37
|
|
|
|
1.95
|
|
|
|
2.035
|
|
|
|
1,490,764
|
|
|
March
|
|
|
2.87
|
|
|
|
2.35
|
|
|
|
2.48
|
|
|
|
711,198
|
|
|
|
2.14
|
|
|
|
1.7501
|
|
|
|
1.86
|
|
|
|
1,935,669
|
|
|
April
|
|
|
2.68
|
|
|
|
2.41
|
|
|
|
2.59
|
|
|
|
639,246
|
|
|
|
2.01
|
|
|
|
1.80
|
|
|
|
1.91
|
|
|
|
2,962,881
|
|
|
May
|
|
|
2.75
|
|
|
|
2.20
|
|
|
|
2.32
|
|
|
|
578,061
|
|
|
|
1.96
|
|
|
|
1.5993
|
|
|
|
1.72
|
|
|
|
935,942
|
|
|
June
|
|
|
2.61
|
|
|
|
2.18
|
|
|
|
2.26
|
|
|
|
546,095
|
|
|
|
1.94
|
|
|
|
1.61
|
|
|
|
1.70
|
|
|
|
1,205,291
|
|
|
July
|
|
|
2.26
|
|
|
|
1.75
|
|
|
|
1.79
|
|
|
|
349,000
|
|
|
|
1.78
|
|
|
|
1.311
|
|
|
|
1.34
|
|
|
|
644,978
|
|
|
August 1 to 13
|
|
|
1.75
|
|
|
|
1.13
|
|
|
|
1.19
|
|
|
|
660,921
|
|
|
|
1.33
|
|
|
|
0.8301
|
|
|
|
0.90
|
|
|
|
2,432,449
|
|
PRIOR SALES
Except as disclosed under this heading, no
other Common Shares or securities exchangeable or convertible into Common Shares have been issued during the twelve month period
preceding the date of this Prospectus Supplement.
Common Shares
Except as disclosed under this heading, no
other Common Shares or securities exchangeable or convertible into Common Shares have been issued during the twelve month period
preceding the date of this Prospectus Supplement.
Common Shares
During the twelve month period prior to the
date of this Prospectus Supplement, the Corporation has issued:
|
|
(a)
|
an aggregate of 1,590,747 Common Shares pursuant to the Equity Distribution Agreement and a prospectus
supplement dated October 24, 2018 to the Prospectus, at prices ranging from US$1.6014 to US$3.1167 per Common Share, with the weighted
average price being US$1.9368 per Common Share;
|
|
|
(b)
|
an aggregate of 2,188,063 Common Shares pursuant to a purchase agreement dated September 28, 2018
between the Corporation and Lincoln Park Capital Fund and a prospectus supplement dated September 28, 2018 to the Prospectus, at
prices ranging from US$1.7358 to US$4.1082 per Common Share, with the weighted average price being US$2.3878 per Common Share;
and
|
|
|
(c)
|
an aggregate of 79,550 Common Shares issued on the exercise
of stock options granted pursuant to its amended and restated stock option plan or on the vesting of restricted share awards granted
pursuant to its amended and restated incentive share award plan, particulars of which are set forth in the following table:
|
|
Date of Issue
|
|
Number of Common Shares Issued
|
|
|
Price per Common Share (US$)
|
|
|
September 12, 2018
|
|
|
6,473
|
|
|
|
2.82
|
|
|
September 17, 2018
|
|
|
3,000
|
|
|
|
2.04
|
|
|
September 18, 2018
|
|
|
5,700
|
|
|
|
2.39
|
|
|
September 27, 2018
|
|
|
4,420
|
|
|
|
2.39
|
|
|
October 2, 2018
|
|
|
4,210
|
|
|
|
2.20
|
|
|
November 30, 2018
|
|
|
5,263
|
|
|
|
2.38
|
|
|
November 30, 2018
|
|
|
21,052
|
|
|
|
2.35
|
|
|
January 4, 2019
|
|
|
1,982
|
|
|
|
1.78
|
|
|
February 22, 2019
|
|
|
2,105
|
|
|
|
2.03
|
|
|
April 3, 2019
|
|
|
2,575
|
|
|
|
1.85
|
|
|
April 16, 2019
|
|
|
8,354
|
|
|
|
1.90
|
|
|
July 4, 2019
|
|
|
12,311
|
|
|
|
1.72
|
|
|
July 5, 2019
|
|
|
2,105
|
|
|
|
1.71
|
|
Stock Options
During the twelve month period preceding the
date of this Prospectus Supplement, the Corporation granted stock options pursuant to its amended and restated stock option plan
exercisable for an aggregate of 444,500 Common Shares. The particulars of such grants are set forth in the following table:
|
Date of Grant
|
|
Number of Options Granted
|
|
|
Exercise Price (C$)
|
|
|
September 20, 2018
|
|
|
1,500
|
|
|
|
5.67
|
|
|
November 19, 2018
|
|
|
203,000
|
|
|
|
3.44
|
|
|
November 29, 2018
|
|
|
25,000
|
|
|
|
3.23
|
|
|
December 14, 2018
|
|
|
195,000
|
|
|
|
2.73
|
|
|
July 16, 2019
|
|
|
20,000
|
|
|
|
2.12
|
|
Share Awards
During the twelve month period preceding the
date of this Prospectus Supplement, the Corporation granted restricted share awards pursuant to its amended and restated incentive
share award plan which, upon vesting, will entitle the holders thereof to receive up to an aggregate of 113,225 Shares. The particulars
of such grants are set forth in the following table:
|
Date of Grant
|
|
Number of Share Awards Granted
|
|
|
November 27, 2018
|
|
|
75,698
|
|
|
December 31, 2018
|
|
|
18,266
|
|
|
March 31, 2019
|
|
|
9,113
|
|
|
June 30, 2019
|
|
|
10,148
|
|
CERTAIN UNITED STATES
FEDERAL INCOME TAX CONSIDERATIONS
The following is a general summary of certain
U.S. federal income tax considerations applicable to a U.S. Holder (as defined below) arising from and relating to the acquisition,
ownership and disposition of Units acquired pursuant to this Prospectus Supplement, the acquisition, ownership, and disposition
of Unit Shares acquired as part of the Units, the exercise, disposition, and lapse of Warrants acquired as part of the Units, and
the acquisition, ownership, and disposition of Warrant Shares received upon exercise of the Warrants.
This summary is for general information purposes
only and does not purport to be a complete analysis or listing of all potential U.S. federal income tax considerations that may
apply to a U.S. Holder as a result of the acquisition of Units pursuant to this Offering. In addition, this summary does not take
into account the individual facts and circumstances of any particular U.S. Holder that may affect the U.S. federal income tax consequences
to such U.S. Holder, including specific tax consequences to a U.S. Holder under an applicable tax treaty. Accordingly, this summary
is not intended to be, and should not be construed as, legal or U.S. federal income tax advice with respect to any particular U.S.
Holder. This summary does not address the U.S. federal net investment income, U.S. federal alternative minimum, U.S. federal estate
and gift, U.S. state and local, and non-U.S. tax consequences to U.S. Holders of the acquisition, ownership, and disposition of
Units, Unit Shares, Warrants and Warrant Shares. In addition, except as specifically set forth below, this summary does not discuss
applicable tax reporting requirements. Each U.S. Holder should consult its own tax advisor regarding the U.S. federal, U.S. federal
net investment income, U.S. federal alternative minimum, U.S. federal estate and gift, U.S. state and local, and non-U.S. tax consequences
relating to the acquisition, ownership and disposition of Units, Unit Shares, Warrants, and Warrant Shares.
No opinion from legal counsel or ruling from
the Internal Revenue Service (the “
IRS
”) has been requested, or will be obtained, regarding the U.S. federal
income tax considerations applicable to U.S. Holders as discussed in this summary. This summary is not binding on the IRS, and
the IRS is not precluded from taking a position that is different from, and contrary to, the positions taken in this summary. In
addition, because the authorities on which this summary is based are subject to various interpretations, the IRS and the U.S. courts
could disagree with one or more of the positions taken in this summary.
Scope of this Summary
Authorities
This summary is based on the Internal Revenue
Code of 1986, as amended (the “
Code
”), Treasury Regulations (whether final, temporary, or proposed) promulgated
under the Code, published rulings of the IRS, published administrative positions of the IRS and U.S. court decisions, that are
in effect and available, as of the date of this document. Any of the authorities on which this summary is based could be changed
in a material and adverse manner at any time, and any such change could be applied retroactively. This summary does not discuss
the potential effects, whether adverse or beneficial, of any proposed legislation that, if enacted, could be applied on a retroactive
or prospective basis.
U.S. Holders
For purposes of this summary, the term “
U.S.
Holder
” means a beneficial owner of Units, Unit Shares, Warrants or Warrant Shares acquired pursuant to this Prospectus
Supplement that is for U.S. federal income tax purposes:
|
|
·
|
a citizen or individual resident of the United States;
|
|
|
·
|
a corporation (or other entity treated as a corporation
for U.S. federal income tax purposes) organized under the laws of the United States, any state thereof or the District of Columbia;
|
|
|
·
|
an estate whose income is subject to U.S. federal income taxation regardless of its source; or
|
|
|
·
|
a trust that (1) is subject to the primary supervision of a court within the United States and the control
of one or more U.S. persons for all substantial decisions or (2) has a valid election in effect under applicable Treasury Regulations
to be treated as a U.S. person.
|
U.S. Holders Subject to Special U.S. Federal
Income Tax Rules Not Addressed
This summary does not address the U.S. federal
income tax considerations applicable to U.S. Holders that are subject to special provisions under the Code, including U.S. Holders
that: (a) are tax-exempt organizations, qualified retirement plans, individual retirement accounts, or other tax-deferred accounts;
(b) are financial institutions, underwriters, insurance companies, real estate investment trusts, or regulated investment companies;
(c) are brokers or dealers in securities or currencies or U.S. Holders that are traders in securities that elect to apply a mark-to-market
accounting method; (d) have a “functional currency” other than the U.S. dollar; (e) own Units, Unit Shares, Warrants
or Warrant Shares as part of a straddle, hedging transaction, conversion transaction, constructive sale, or other integrated transaction;
(f) acquired Units, Unit Shares, Warrants or Warrant Shares in connection with the exercise of employee stock options or otherwise
as compensation for services; (g) hold Units, Unit Shares, Warrants or Warrant Shares other than as a capital asset within the
meaning of Section 1221 of the Code (generally, property held for investment purposes); (h) are partnerships and other pass-through
entities (and investors in such partnerships and entities); (i) are required to accelerate the recognition of any item of gross
income with respect to Units, Unit Shares, Warrants or Warrant Shares as a result of such income being recognized on an applicable
financial statement; or (i) own, have owned or will own (directly, indirectly, or by attribution) 10% or more of the total combined
voting power or value of the Corporation’s outstanding shares. This summary also does not address the U.S. federal income
tax considerations applicable to U.S. Holders who are (a) U.S. expatriates or former long-term residents of the U.S., or (b) subject
to taxing jurisdictions other than, or in addition to, the United States. U.S. Holders that are subject to special provisions under
the Code, including U.S. Holders described immediately above, should consult their own tax advisors regarding the U.S. federal,
U.S. federal net investment income, U.S. federal alternative minimum, U.S. federal estate and gift, U.S. state and local, and non-U.S.
tax consequences relating to the acquisition, ownership and disposition of Units, Unit Shares, Warrants or Warrant Shares.
If an entity or arrangement that is classified
as a partnership for U.S. federal income tax purposes holds Units, Unit Shares, Warrants or Warrant Shares, the U.S. federal income
tax consequences to such entity or arrangement and the owners of such entity or arrangement generally will depend on the activities
of such entity or arrangement and the status of such owners. This summary does not address the tax consequences to any such entity
or arrangement or owner. Owners of entities or arrangements that are classified as partnerships for U.S. federal income tax purposes
should consult their own tax advisor regarding the U.S. federal income tax consequences arising from and relating to the acquisition,
ownership, and disposition of Units, Unit Shares, Warrants and Warrant Shares.
U.S. Federal Income Tax Consequences of
the Acquisition of Units
For U.S. federal income tax purposes, the
acquisition by a U.S. Holder of a Unit will be treated as the acquisition of one Unit Share and one Warrant. The purchase price
for each Unit will be allocated between these two components in proportion to their relative fair market values at the time the
Unit is purchased by the U.S. Holder. This allocation of the purchase price for each Unit will establish a U.S. Holder’s
initial tax basis for U.S. federal income tax purposes in the Unit Share and one Warrant that comprise each Unit.
For this purpose, the Corporation will
allocate U.S.$0.80 of the purchase price for the Unit to the Unit Share and U.S.$0.01 of the purchase price for each Unit to the
Warrant. However, the IRS will not be bound by such allocation of the purchase price for the Units, and therefore, the IRS or a
U.S. court may not respect the allocation set forth above. Each U.S. Holder should consult its own tax advisor regarding the allocation
of the purchase price for the Units.
Passive Foreign Investment Company Rules
If the Corporation is considered a “passive
foreign investment company” within the meaning of Section 1297 of the Code (a “
PFIC
”) at any time during
a U.S. Holder’s holding period, the following sections will generally describe the potentially adverse U.S. federal income
tax consequences to U.S. Holders of the acquisition, ownership, and disposition of Units, Unit Shares, Warrants or Warrant Shares.
The Corporation believes that it was classified
as a PFIC for the tax year ended December 31, 2018, and based on current business plans and financial expectations, the Corporation
expects that it may be a PFIC for its current tax year ending December 31, 2019 and may be a PFIC in future tax years. No opinion
of legal counsel or ruling from the IRS concerning the status of the Corporation as a PFIC has been obtained or is currently planned
to be requested. The determination of whether any corporation was, or will be, a PFIC for a tax year depends, in part, on the application
of complex U.S. federal income tax rules, which are subject to differing interpretations. In addition, whether any corporation
will be a PFIC for any tax year depends on the assets and income of such corporation over the course of each such tax year and,
as a result, the Corporation’s PFIC status for the current year and future years cannot be predicted with certainty as of
the date of this document. Accordingly, there can be no assurance that the IRS will not challenge any PFIC determination made by
the Corporation (or by one of the Corporation’s subsidiaries). Each U.S. Holder should consult its own tax advisor regarding
the Corporation’s status as a PFIC and the PFIC status of each non-U.S. subsidiary of the Corporation.
In any year in which the Corporation is classified
as a PFIC, a U.S. Holder will be required to file an annual report with the IRS containing such information as Treasury Regulations
and/or other IRS guidance may require. In addition to penalties, a failure to satisfy such reporting requirements may result in
an extension of the time period during which the IRS can assess a tax. U.S. Holders should consult their own tax advisors regarding
the requirements of filing such information returns under these rules, including the requirement to file an IRS Form 8621.
The Corporation generally will be a PFIC for
any tax year in which (a) 75% or more of the gross income of the Corporation for such tax year is passive income (the “
PFIC
income test
”) or (b) 50% or more of the value of the assets of the Corporation either produce passive income or are
held for the production of passive income, based on the quarterly average of the fair market value of such assets (the “
PFIC
asset test
”). “Gross income” generally includes sales revenues less the cost of goods sold, plus income
from investments and from incidental or outside operations or sources, and “passive income” generally includes, for
example, dividends, interest, certain rents and royalties, certain gains from the sale of stock and securities, and certain gains
from commodities transactions.
For purposes of the PFIC income test and PFIC
asset test described above, if the Corporation owns, directly or indirectly, 25% or more of the total value of the outstanding
shares of another corporation, the Corporation will be treated as if it (a) held a proportionate share of the assets of such other
corporation and (b) received directly a proportionate share of the income of such other corporation. In addition, for purposes
of the PFIC income test and PFIC asset test described above, “passive income” does not include any interest, dividends,
rents, or royalties that are received or accrued by the Corporation from a “related person” (as defined in Section
954(d)(3) of the Code), to the extent such items are properly allocable to the income of such related person that is not passive
income.
Under certain attribution rules, if the Corporation
is a PFIC, U.S. Holders will be deemed to own their proportionate share of any of the Corporation’s subsidiaries which is
also a PFIC (a “
Subsidiary PFIC
”), and will generally be subject to U.S. federal income tax under the “Default
PFIC Rules Under Section 1291 of the Code” discussed below on their proportionate share of any (i) distribution on the shares
of a Subsidiary PFIC and (ii) disposition or deemed disposition of shares of a Subsidiary PFIC, both as if such U.S. Holders directly
held the shares of such Subsidiary PFIC. Accordingly, U.S. Holders should be aware that they could be subject to tax under the
PFIC rules even if no distributions are received and no redemptions or other dispositions of Units, Unit Shares, Warrants or Warrant
Shares are made. In addition, U.S. Holders may be subject to U.S. federal income tax on any indirect gain realized on the stock
of a Subsidiary PFIC on the sale or disposition of Units, Unit Shares, Warrants or Warrant Shares.
Default PFIC Rules Under Section 1291 of
the Code
If the Corporation is a PFIC, the U.S. federal
income tax consequences to a U.S. Holder of the purchase of Units and the acquisition, ownership, and disposition of Unit Shares,
Warrants and Warrant Shares will depend on whether such U.S. Holder makes a “qualified electing fund” or “QEF”
election (a “
QEF Election
”) or makes a mark-to-market election under Section 1296 of the Code (a “
Mark-to-Market
Election
”) with respect to Unit Shares or Warrant Shares. A U.S. Holder that does not make either a QEF Election or
a Mark-to-Market Election (a “
Non-Electing U.S. Holder
”) will be taxable as described below.
A Non-Electing U.S. Holder will be subject
to the rules of Section 1291 of the Code with respect to (a) any gain recognized on the sale or other taxable disposition of Unit
Shares, Warrants and Warrant Shares and (b) any excess distribution received on the Unit Shares and Warrant Shares. A distribution
generally will be an “excess distribution” to the extent that such distribution (together with all other distributions
received in the current tax year) exceeds 125% of the average distributions received during the three preceding tax years (or during
a U.S. Holder’s holding period for the Unit Shares and Warrant Shares, if shorter).
Under Section 1291 of the Code, any gain recognized
on the sale or other taxable disposition of Unit Shares, Warrants and Warrant Shares of a PFIC (including an indirect disposition
of shares of a Subsidiary PFIC), and any excess distribution received on such Unit Shares and Warrant Shares (or a distribution
by a Subsidiary PFIC to its shareholder that is deemed to be received by a U.S. Holder) must be ratably allocated to each day in
a Non-Electing U.S. Holder’s holding period for the Unit Shares or Warrant Shares. The amount of any such gain or excess
distribution allocated to the tax year of disposition or distribution of the excess distribution and to years before the entity
became a PFIC, if any, would be taxed as ordinary income (and not eligible for certain preferential tax rates, as discussed below).
The amounts allocated to any other tax year would be subject to U.S. federal income tax at the highest tax rate applicable to ordinary
income in each such year, and an interest charge would be imposed on the tax liability for each such year, calculated as if such
tax liability had been due in each such year. A Non-Electing U.S. Holder that is not a corporation must treat any such interest
paid as “personal interest,” which is not deductible.
If the Corporation is a PFIC for any tax year
during which a Non-Electing U.S. Holder holds Unit Shares, Warrant Shares or Warrants, it will continue to be treated as a PFIC
with respect to such Non-Electing U.S. Holder, regardless of whether it ceases to be a PFIC in one or more subsequent tax years.
If the Corporation ceases to be a PFIC, a Non-Electing U.S. Holder may terminate this deemed PFIC status with respect to Unit Shares
and Warrant Shares by electing to recognize gain (which will be taxed under the rules of Section 1291 of the Code as discussed
above) as if such Unit Shares and Warrant Shares were sold on the last day of the last tax year for which the Corporation was a
PFIC. No such election, however, may be made with respect to the Warrants.
Under proposed Treasury Regulations, if a U.S.
holder has an option, warrant, or other right to acquire stock of a PFIC (such as the Warrants), such option, warrant or right
is considered to be PFIC stock subject to the default rules of Section 1291 of the Code. Under rules described below, the holding
period for the Warrant Shares will begin on the date a U.S. Holder acquires the Units. This will impact the availability of the
QEF Election and Mark-to-Market Election with respect to the Warrant Shares. Thus, a U.S. Holder will have to account for Warrant
Shares and Unit Shares under the PFIC rules and the applicable elections differently.
QEF Election
A U.S. Holder that makes a QEF Election for
the first tax year in which its holding period of its Unit Shares begins generally will not be subject to the rules of Section
1291 of the Code discussed above with respect to its Unit Shares. However, a U.S. Holder that makes a QEF Election will be subject
to U.S. federal income tax on such U.S. Holder’s pro rata share of (a) the Corporation’s net capital gain, which will
be taxed as long-term capital gain to such U.S. Holder, and (b) the Corporation’s ordinary earnings, which will be taxed
as ordinary income to such U.S. Holder. Generally, “net capital gain” is the excess of (a) net long-term capital gain
over (b) net short-term capital loss, and “ordinary earnings” are the excess of (a) “earnings and profits”
over (b) net capital gain. A U.S. Holder that makes a QEF Election will be subject to U.S. federal income tax on such amounts for
each tax year in which the Corporation is a PFIC, regardless of whether such amounts are actually distributed to such U.S. Holder
by the Corporation. However, for any tax year in which the Corporation is a PFIC and has no net income or gain, U.S. Holders that
have made a QEF Election would not have any income inclusions as a result of the QEF Election. If a U.S. Holder that made a QEF
Election has an income inclusion, such a U.S. Holder may, subject to certain limitations, elect to defer payment of current U.S.
federal income tax on such amounts, subject to an interest charge. If such U.S. Holder is not a corporation, any such interest
paid will be treated as “personal interest,” which is not deductible.
A U.S. Holder that makes a timely QEF Election
generally (a) may receive a tax-free distribution from the Corporation to the extent that such distribution represents “earnings
and profits” that were previously included in income by the U.S. Holder because of such QEF Election and (b) will adjust
such U.S. Holder’s tax basis in the Unit Shares to reflect the amount included in income or allowed as a tax-free distribution
because of such QEF Election. In addition, a U.S. Holder that makes a QEF Election generally will recognize capital gain or loss
on the sale or other taxable disposition of Unit Shares.
The procedure for making a QEF Election, and
the U.S. federal income tax consequences of making a QEF Election, will depend on whether such QEF Election is timely. A QEF Election
will be treated as “timely” for purposes of avoiding the default PFIC rules discussed above if such QEF Election is
made for the first year in the U.S. Holder’s holding period for the Unit Shares in which the Corporation was a PFIC. A U.S.
Holder may make a timely QEF Election by filing the appropriate QEF Election documents at the time such U.S. Holder files a U.S.
federal income tax return for such year.
A QEF Election will apply to the tax year for
which such QEF Election is made and to all subsequent tax years, unless such QEF Election is invalidated or terminated or the IRS
consents to revocation of such QEF Election. If a U.S. Holder makes a QEF Election and, in a subsequent tax year, the Corporation
ceases to be a PFIC, the QEF Election will remain in effect (although it will not be applicable) during those tax years in which
the Corporation is not a PFIC. Accordingly, if the Corporation becomes a PFIC in another subsequent tax year, the QEF Election
will be effective and the U.S. Holder will be subject to the QEF rules described above during any subsequent tax year in which
the Corporation qualifies as a PFIC.
As discussed above, under proposed Treasury
Regulations, if a U.S. holder has an option, warrant or other right to acquire stock of a PFIC (such as the Warrants), such option,
warrant or right is considered to be PFIC stock subject to the default rules of Section 1291 of the Code. However, a U.S. Holder
of an option, warrant or other right to acquire stock of a PFIC may not make a QEF Election that will apply to the option, warrant
or other right to acquire PFIC stock. In addition, under proposed Treasury Regulations, if a U.S. Holder holds an option, warrant
or other right to acquire stock of a PFIC, the holding period with respect to shares of stock of the PFIC acquired upon exercise
of such option, warrant or other right will include the period that the option, warrant or other right was held.
Consequently, under the proposed Treasury Regulations,
if a U.S. Holder of Unit Shares makes a QEF Election, such election generally will not be treated as a timely QEF Election with
respect to Warrant Shares and the rules of Section 1291 of the Code discussed above will continue to apply with respect to such
U.S. Holder’s Warrant Shares. However, a U.S. Holder of Warrant Shares should be eligible to make a timely QEF Election if
such U.S. Holder elects in the tax year in which such Warrant Shares are received to recognize gain (which will be taxed under
the rules of Section 1291 of the Code discussed above) as if such Warrant Shares were sold for fair market value on the date such
U.S. Holder acquired them by exercising the corresponding Warrant. In addition, gain recognized on the sale or other taxable disposition
(other than by exercise) of the Warrants by a U.S. Holder will be subject to the rules of Section 1291 of the Code discussed above.
Each U.S. Holder should consult its own tax advisor regarding the application of the PFIC rules to the Units, Unit Shares, Warrants,
and Warrant Shares.
The Corporation: (a) will make available to
U.S. Holders, upon their written request, information as to its status as a PFIC and the PFIC status of any subsidiary in which
the Corporation owns more than 50% of such subsidiary’s total aggregate voting power and (b) for each year in which the Corporation
is a PFIC, provide to a U.S. Holder, upon written request, such information and documentation that a U.S. Holder making a QEF Election
with respect to the Corporation and such more than 50% owned subsidiary which constitutes a PFIC is reasonably required to obtain
for U.S. federal income tax purposes. The Corporation may elect to provide such information on its website. With respect to any
Subsidiary PFIC in which the Corporation owns 50% or less of the aggregate voting power, upon the written request of a U.S. Holder
acquiring Unit Shares, the Corporation will request that such Subsidiary PFIC provide such U.S. Holder with the information that
such U.S. Holder requires to report under the QEF rules; provided, however, the Corporation can provide no assurances that such
Subsidiary PFIC will provide such information.
A U.S. Holder makes a QEF Election by attaching
a completed IRS Form 8621, including a PFIC Annual Information Statement, to a timely filed U.S. federal income tax return. However,
if the Corporation does not provide the required information with regard to the Corporation or any of its Subsidiary PFICs, U.S.
Holders will not be able to make a QEF Election for such entity and will continue to be subject to the rules of Section 1291 of
the Code discussed above that apply to Non-Electing U.S. Holders with respect to the taxation of gains and excess distributions.
Mark-to-Market Election
A U.S. Holder may make a Mark-to-Market Election
with respect to Unit Shares and Warrant Shares only if the Unit Shares and Warrant Shares are marketable stock. The Unit Shares
and Warrant Shares generally will be “marketable stock” if the Unit Shares and Warrant Shares are regularly traded
on (a) a national securities exchange that is registered with the SEC, (b) the national market system established pursuant to Section
11A of the U.S. Exchange Act or (c) a foreign securities exchange that is regulated or supervised by a governmental authority of
the country in which the market is located, provided that (i) such foreign exchange has trading volume, listing, financial disclosure,
and other requirements and the laws of the country in which such foreign exchange is located, together with the rules of such foreign
exchange, ensure that such requirements are actually enforced and (ii) the rules of such foreign exchange ensure active trading
of listed stocks. If such stock is traded on such a qualified exchange or other market, such stock generally will be considered
“regularly traded” for any calendar year during which such stock is traded, other than in de minimis quantities, on
at least 15 days during each calendar quarter. Provided that the Unit Shares and Warrant Shares are “regularly traded”
as described in the preceding sentence, the Unit Shares and Warrant Shares are expected to be marketable stock. The Corporation
believes that its Unit Shares were “regularly traded” in the second calendar quarter of 2019 and expects that the Unit
Shares should be “regularly traded” in the third calendar quarter of 2019. However, there can be no assurance that
the Unit Shares will be “regularly traded” in subsequent calendar quarters. U.S. Holders should consult their own tax
advisors regarding the marketable stock rules.
A U.S. Holder that makes a Mark-to-Market Election
with respect to its Unit Shares generally will not be subject to the rules of Section 1291 of the Code discussed above with respect
to such Unit Shares. However, if a U.S. Holder does not make a Mark-to-Market Election beginning in the first tax year of such
U.S. Holder’s holding period for the Unit Shares and such U.S. Holder has not made a timely QEF Election, the rules of Section
1291 of the Code discussed above will apply to certain dispositions of, and distributions on, the Unit Shares.
Any Mark-to-Market Election made by a U.S.
Holder for the Unit Shares will also apply to such U.S. Holder’s Warrant Shares. As a result, if a Mark-to-Market Election
has been made by a U.S. Holder with respect to Unit Shares, any Warrant Shares received will automatically be marked-to-market
in the year of exercise. Because, under the proposed Treasury Regulations, a U.S. Holder’s holding period for Warrant Shares
includes the period during which such U.S. Holder held the Warrants, a U.S. Holder will be treated as making a Mark-to-Market Election
with respect to its Warrant Shares after the beginning of such U.S. Holder’s holding period for the Warrant Shares unless
the Warrant Shares are acquired in the same tax year as the year in which the U.S. Holder acquired its Units. Consequently, the
default rules under Section 1291 described above generally will apply to the mark-to-market gain realized in the tax year in which
Warrant Shares are received. However, the general mark-to-market rules will apply to subsequent tax years.
A U.S. Holder that makes a Mark-to-Market Election
will include in ordinary income, for each tax year in which the Corporation is a PFIC, an amount equal to the excess, if any, of
(a) the fair market value of the Unit Shares and any Warrant Shares, as of the close of such tax year over (b) such U.S. Holder’s
tax basis in the Unit Shares and any Warrant Shares. A U.S. Holder that makes a Mark-to-Market Election will be allowed a deduction
in an amount equal to the excess, if any, of (i) such U.S. Holder’s adjusted tax basis in the Unit Shares and any Warrant
Shares, over (ii) the fair market value of such Unit Shares and any Warrant Shares (but only to the extent of the net amount of
previously included income as a result of the Mark-to-Market Election for prior tax years).
A U.S. Holder that makes a Mark-to-Market Election
generally also will adjust such U.S. Holder’s tax basis in the Unit Shares and Warrant Shares to reflect the amount included
in gross income or allowed as a deduction because of such Mark-to-Market Election. In addition, upon a sale or other taxable disposition
of Unit Shares and Warrant Shares, a U.S. Holder that makes a Mark-to-Market Election will recognize ordinary income or ordinary
loss (not to exceed the excess, if any, of (a) the amount included in ordinary income because of such Mark-to-Market Election for
prior tax years over (b) the amount allowed as a deduction because of such Mark-to-Market Election for prior tax years).
A U.S. Holder makes a Mark-to-Market Election
by attaching a completed IRS Form 8621 to a timely filed U.S. federal income tax return. A timely Mark-to-Market Election applies
to the tax year in which such Mark-to-Market Election is made and to each subsequent tax year, unless the Unit Shares and Warrant
Shares cease to be “marketable stock” or the IRS consents to revocation of such election. Each U.S. Holder should consult
its own tax advisor regarding the availability of, and procedure for making, a Mark-to-Market Election.
Although a U.S. Holder may be eligible to make
a Mark-to-Market Election with respect to the Unit Shares and Warrant Shares, no such election may be made with respect to the
stock of any Subsidiary PFIC that a U.S. Holder is treated as owning because such stock is not marketable. Hence, the Mark-to-Market
Election will not be effective to eliminate the interest charge and other income inclusion rules described above with respect to
deemed dispositions of Subsidiary PFIC stock or distributions from a Subsidiary PFIC to its shareholder.
Other PFIC Rules
Under Section 1291(f) of the Code, the IRS
has issued proposed Treasury Regulations that, subject to certain exceptions, would cause a U.S. Holder that had not made a timely
QEF Election to recognize gain (but not loss) upon certain transfers of Unit Shares and Warrant Shares that would otherwise be
tax-deferred (e.g., gifts and exchanges pursuant to corporate reorganizations). However, the specific U.S. federal income tax consequences
to a U.S. Holder may vary based on the manner in which Unit Shares, Warrants, or Warrant Shares are transferred.
If finalized in their current form, the proposed
Treasury Regulations applicable to PFICs would be effective for transactions occurring on or after April 1, 1992. Because the proposed
Treasury Regulations have not yet been adopted in final form, they are not currently effective, and there is no assurance that
they will be adopted in the form and with the effective date proposed. Nevertheless, the IRS has announced that, in the absence
of final Treasury Regulations, taxpayers may apply reasonable interpretations of the Code provisions applicable to PFICs and that
it considers the rules set forth in the proposed Treasury Regulations to be reasonable interpretations of those Code provisions.
The PFIC rules are complex, and the implementation of certain aspects of the PFIC rules requires the issuance of Treasury Regulations
which in many instances have not been promulgated and which, when promulgated, may have retroactive effect. U.S. Holders should
consult their own tax advisors about the potential applicability of the proposed Treasury Regulations.
Certain additional adverse rules will apply
with respect to a U.S. Holder if the Corporation is a PFIC, regardless of whether such U.S. Holder makes a QEF Election. For example
under Section 1298(b)(6) of the Code, a U.S. Holder that uses Unit Shares, Warrants or Warrant Shares as security for a loan will,
except as may be provided in Treasury Regulations, be treated as having made a taxable disposition of such Unit Shares, Warrants
or Warrant Shares.
In addition, a U.S. Holder who acquires Unit
Shares, Warrants or Warrant Shares from a decedent will not receive a “step up” in tax basis of such Unit Shares, Warrants
or Warrant Shares to fair market value.
Special rules also apply to the amount of foreign
tax credit that a U.S. Holder may claim on a distribution from a PFIC. Subject to such special rules, foreign taxes paid with respect
to any distribution in respect of stock in a PFIC are generally eligible for the foreign tax credit. The rules relating to distributions
by a PFIC and their eligibility for the foreign tax credit are complicated, and a U.S. Holder should consult with their own tax
advisor regarding the availability of the foreign tax credit with respect to distributions by a PFIC.
The PFIC rules are complex, and each U.S. Holder
should consult its own tax advisor regarding the PFIC rules (including the applicability and advisability of a QEF Election and
Mark-to-Market Election) and how the PFIC rules may affect the U.S. federal income tax consequences of the acquisition, ownership,
and disposition of Unit Shares, Warrants and Warrant Shares.
U.S. Federal Income Tax Consequences of
the Ownership, Exercise and Disposition of Warrants
The following discussion describes the general
rules applicable to the ownership, exercise and disposition of the Warrants but is subject in its entirety to the special rules
described above under the heading “Passive Foreign Investment Company Rules.”
Exercise of Warrants
A U.S. Holder should not recognize gain or
loss on the exercise of a Warrant and related receipt of a Warrant Share (unless cash is received in lieu of the issuance of a
fractional Warrant Share). A U.S. Holder’s initial tax basis in the Warrant Share received on the exercise of a Warrant
should be equal to the sum of (a) such U.S. Holder’s tax basis in such Warrant plus (b) the exercise price paid by such
U.S. Holder on the exercise of such Warrant. It is unclear whether a U.S. Holder's holding period for the Warrant Share received
on the exercise of a Warrant would commence on the date of exercise of the Warrant or the day following the date of exercise of
the Warrant. If, as anticipated, the Corporation is a PFIC, a U.S. Holder’s holding period for the Warrant Share for PFIC
purposes only will begin on the date on which such U.S. Holder acquired its Units.
Disposition of Warrants
A U.S. Holder will recognize gain or loss on
the sale or other taxable disposition of a Warrant in an amount equal to the difference, if any, between (a) the amount of cash
plus the fair market value of any property received and (b) such U.S. Holder’s tax basis in the Warrant sold or otherwise
disposed of. Subject to the PFIC rules discussed above, any such gain or loss generally will be a capital gain or loss, which will
be long-term capital gain or loss if the Warrant is held for more than one year. Deductions for capital losses are subject to complex
limitations under the Code.
Expiration of Warrants Without Exercise
Upon the lapse or expiration of a Warrant,
a U.S. Holder will recognize a loss in an amount equal to such U.S. Holder’s tax basis in the Warrant. Any such loss generally
will be a capital loss and will be long-term capital loss if the Warrants are held for more than one year. Deductions for capital
losses are subject to complex limitations under the Code.
Certain Adjustments to the Warrants
Under Section 305 of the Code, an adjustment
to the number of Warrant Shares that will be issued on the exercise of the Warrants, or an adjustment to the exercise price of
the Warrants, may be treated as a constructive distribution to a U.S. Holder of the Warrants if, and to the extent that, such adjustment
has the effect of increasing such U.S. Holder’s proportionate interest in the “earnings and profits” or the Corporation’s
assets, depending on the circumstances of such adjustment (for example, if such adjustment is to compensate for a distribution
of cash or other property to the shareholders). Adjustments to the exercise price of Warrants made pursuant to a bona fide reasonable
adjustment formula that has the effect of preventing dilution of the interest of the holders of the Warrants should generally not
be considered to result in a constructive distribution. Any such constructive distribution would be taxable whether or not there
is an actual distribution of cash or other property. (See more detailed discussion of the rules applicable to distributions made
by the Corporation at “Distributions on Unit Shares and Warrant Shares” below).
General Rules Applicable to U.S. Federal
Income Tax Consequences of the Acquisition, Ownership, and Disposition of Unit Shares and Warrant Shares
The following discussion describes the general
rules applicable to the acquisition, ownership and disposition of the Unit Shares and Warrant Shares but is subject in its entirety
to the special rules described above under the heading “Passive Foreign Investment Company Rules.”
Distributions on Unit Shares and Warrant
Shares
A U.S. Holder that receives a distribution,
including a constructive distribution, with respect to a Unit Share or Warrant Share (as well as any constructive distribution
on a Warrant as described above) will be required to include the amount of such distribution in gross income as a dividend (without
reduction for any Canadian income tax withheld from such distribution) to the extent of the Corporation’s current and accumulated
“earnings and profits”, as computed under U.S. federal income tax principles. A dividend generally will be taxed to
a U.S. Holder at ordinary income tax rates if the Corporation is a PFIC for the tax year of such distribution or the preceding
tax year. To the extent that a distribution exceeds the current and accumulated “earnings and profits” of the Corporation,
such distribution will be treated first as a tax-free return of capital to the extent of a U.S. Holder’s tax basis in the
Unit Shares or Warrant Shares and thereafter as gain from the sale or exchange of such Unit Shares or Warrant Shares (see “Sale
or Other Taxable Disposition of Unit Shares and/or Warrant Shares” below). However, the Corporation may not maintain the
calculations of earnings and profits in accordance with U.S. federal income tax principles, and each U.S. Holder may be required
to assume that any distribution by the Corporation with respect to the Unit Shares or Warrant Shares will constitute ordinary dividend
income. Dividends received on Unit Shares or Warrant Shares generally will not be eligible for the “dividends received deduction”
generally applicable to corporations. Subject to applicable limitations and provided the Corporation is eligible for the benefits
of the Convention Between Canada and the United States of America with Respect to Taxes on Income and on Capital, signed September
26, 1980, as amended, or the Unit Shares are readily tradable on a United States securities market, dividends paid by the Corporation
to non-corporate U.S. Holders, including individuals, generally will be eligible for the preferential tax rates applicable to long-term
capital gains for dividends, provided certain holding period and other conditions are satisfied, including that the Corporation
not be classified as a PFIC in the tax year of distribution or in the preceding tax year. The dividend rules are complex, and each
U.S. Holder should consult its own tax advisor regarding the application of such rules.
Sale or Other Taxable Disposition of Unit
Shares and/or Warrant Shares
Upon the sale or other taxable disposition
of Unit Shares or Warrant Shares, a U.S. Holder generally will recognize capital gain or loss in an amount equal to the difference
between (a) the amount of cash plus the fair market value of any property received and (b) such U.S. Holder’s tax basis in
such Unit Shares or Warrant Shares sold or otherwise disposed of. Gain or loss recognized on such sale or other taxable disposition
generally will be long-term capital gain or loss if, at the time of the sale or other taxable disposition, the Unit Shares or Warrant
Shares have been held for more than one year. Preferential tax rates may apply to long-term capital gain of a U.S. Holder that
is an individual, estate, or trust. There are no preferential tax rates for long-term capital gain of a U.S. Holder that is a corporation.
Deductions for capital losses are subject to significant limitations under the Code.
Additional Tax Considerations
Receipt of Foreign Currency
The amount of any distribution paid to a U.S.
Holder in foreign currency or on the sale, exchange or other taxable disposition of Unit Shares, Warrants or Warrant Shares generally
will be equal to the U.S. dollar value of such foreign currency based on the exchange rate applicable on the date of receipt (regardless
of whether such foreign currency is converted into U.S. dollars at that time). If the foreign currency received is not converted
into U.S. dollars on the date of receipt, a U.S. Holder will have a tax basis in the foreign currency equal to its U.S. dollar
value on the date of receipt. Any U.S. Holder who receives payment in foreign currency and engages in a subsequent conversion or
other disposition of the foreign currency may have a foreign currency exchange gain or loss that would be treated as ordinary income
or loss, and generally will be U.S. source income or loss for foreign tax credit purposes. Different rules apply to U.S. Holders
who use the accrual method of tax accounting. Each U.S. Holder should consult its own U.S. tax advisor regarding the U.S. federal
income tax consequences of receiving, owning, and disposing of foreign currency.
Foreign Tax Credit
Subject to the PFIC rules discussed above,
a U.S. Holder that pays (whether directly or through withholding) Canadian income tax with respect to dividends paid on the Unit
Shares or Warrant Shares (or with respect to any constructive dividend on the Warrants) generally will be entitled, at the election
of such U.S. Holder, to receive either a deduction or a credit for such Canadian income tax paid. Generally, a credit will reduce
a U.S. Holder’s U.S. federal income tax liability on a dollar-for-dollar basis, whereas a deduction will reduce a U.S. Holder’s
income subject to U.S. federal income tax. This election is made on a year-by-year basis and applies to all foreign taxes paid
or accrued (whether directly or through withholding) by a U.S. Holder during a year. The foreign tax credit rules are complex and
involve the application of rules that depend on a U.S. Holder’s particular circumstances. Accordingly, each U.S. Holder should
consult its own tax advisor regarding the foreign tax credit rules.
Information Reporting; Backup Withholding
Tax
Under U.S. federal income tax laws certain
categories of U.S. Holders must file information returns with respect to their investment in, or involvement in, a foreign corporation.
For example, U.S. return disclosure obligations (and related penalties) are imposed on U.S. Holders that hold certain specified
foreign financial assets in excess of certain threshold amounts. The definition of specified foreign financial assets includes
not only financial accounts maintained in foreign financial institutions, but also, unless held in accounts maintained by a financial
institution, any stock or security issued by a non-U.S. person. U. S. Holders may be subject to these reporting requirements unless
their Unit Shares, Warrants, and Warrant Shares are held in an account at certain financial institutions. Penalties for failure
to file certain of these information returns are substantial. U.S. Holders should consult their own tax advisors regarding the
requirements of filing information returns, including the requirement to file IRS Form 8938.
Payments made within the U.S., or by a U.S.
payor or U.S. middleman, of dividends on, and proceeds arising from the sale or other taxable disposition of the Unit Shares, Warrants
and Warrant Shares generally may be subject to information reporting and backup withholding tax, currently at the rate of 24%,
if a U.S. Holder (a) fails to furnish its correct U.S. taxpayer identification number (generally on Form W-9), (b) furnishes an
incorrect U.S. taxpayer identification number, (c) is notified by the IRS that such U.S. Holder has previously failed to properly
report items subject to backup withholding tax, or (d) fails to certify, under penalty of perjury, that it has furnished its correct
U.S. taxpayer identification number and that the IRS has not notified such U.S. Holder that it is subject to backup withholding
tax. However, certain exempt persons, such as U.S. Holders that are corporations, generally are excluded from these information
reporting and backup withholding tax rules. Any amounts withheld under the U.S. backup withholding tax rules will be allowed as
a credit against a U.S. Holder’s U.S. federal income tax liability, if any, or will be refunded, if such U.S. Holder furnishes
required information to the IRS in a timely manner.
The discussion of reporting requirements set
forth above is not intended to constitute a complete description of all reporting requirements that may apply to a U.S. Holder.
A failure to satisfy certain reporting requirements may result in an extension of the time period during which the IRS can assess
a tax and, under certain circumstances, such an extension may apply to assessments of amounts unrelated to any unsatisfied reporting
requirement. Each U.S. Holder should consult its own tax advisors regarding the information reporting and backup withholding rules.
THE ABOVE SUMMARY IS NOT INTENDED TO CONSTITUTE A COMPLETE ANALYSIS OF ALL TAX CONSIDERATIONS APPLICABLE
TO U.S. HOLDERS WITH RESPECT TO THE ACQUISITION, OWNERSHIP, AND DISPOSITION OF UNIT SHARES, WARRANTS AND WARRANT SHARES. U.S. HOLDERS
SHOULD CONSULT THEIR OWN TAX ADVISORS AS TO THE TAX CONSIDERATIONS APPLICABLE TO THEM IN THEIR OWN PARTICULAR CIRCUMSTANCES.
CERTAIN CANADIAN FEDERAL
INCOME TAX CONSIDERATIONS
The following is, as of the date hereof, a
general summary of the principal Canadian federal income tax considerations under the Tax Act and the regulations thereunder (the
“
Regulations
”) generally applicable to a holder who acquires Units as beneficial owner pursuant to this Prospectus
Supplement and who, for purposes of the Tax Act and at all relevant times: (a) deals at arm’s length with the Corporation
and the Underwriter; (b) is not affiliated with the Corporation or the Underwriter; (c) acquires and holds the Unit Shares, the
Warrants and any Warrant Shares acquired on the exercise of Warrants (collectively with the Unit Shares referred to as the “
Shares
”)
as capital property; and (d) is neither resident nor deemed to be resident in Canada and does not use or hold, and will not be
deemed to use or hold, the Shares or the Warrants in carrying on a business in Canada (each, a “
Non-Resident Holder
”).
The Shares and Warrants will generally be considered to be capital property to a Non-Resident Holder unless the Non-Resident Holder
holds or uses the Shares or Warrants, or is deemed to hold or use the Shares or Warrants, in the course of carrying on a business
of trading or dealing in securities or has acquired them, or is deemed to have acquired them, in a transaction or transactions
considered to be an adventure in the nature of trade. The term “
U.S. Holder
,” for the purposes of this summary,
means a Non-Resident Holder who, for purposes of Canada-United States Income Tax Convention (1980) (the “
Canada-U.S. Tax
Convention
”), is at all relevant times a resident of the United States and is a “qualifying person” within
the meaning of the Canada-U.S. Tax Convention. In some circumstances, persons deriving amounts through fiscally transparent entities
(including limited liability companies) may be entitled to benefits under the Canada-U.S. Tax Convention. U.S. Holders are urged
to consult their own tax advisors to determine their entitlement to benefits under the Canada-U.S. Tax Convention based on their
particular circumstances.
Special considerations, which are not discussed
in this summary, may apply to a Non-Resident Holder that is an insurer that carries on an insurance business in Canada and elsewhere
or an authorized foreign bank (as defined in the Tax Act). Such Non-Resident Holders should consult their own tax advisors.
This summary is based upon the current provisions
of the Tax Act and the Regulations in force as of the date hereof, specific proposals to amend the Tax Act and the Regulations
(the “
Tax Proposals
”) which have been publically announced by or on behalf the Minister of Finance (Canada)
prior to the date hereof, the current provisions of the Canada-U.S. Tax Convention and counsel’s understanding of the current
published administrative policies and assessing practices of the Canada Revenue Agency (the “
CRA
”). This summary
assumes that the Tax Proposals will be enacted in the form proposed and does not take into account or anticipate any other changes
in law, whether by way of judicial, legislative or governmental decision or action, or changes in the CRA’s administrative
policies and assessing practices, nor does it take into account provincial, territorial or foreign income tax legislation or considerations,
which may differ from the Canadian federal income tax considerations discussed herein. No assurances can be given that the Tax
Proposals will be enacted as proposed or at all, or that legislative, judicial or administrative changes will not modify or change
the statements expressed herein.
This summary is not exhaustive of all possible
Canadian federal income tax considerations applicable to an investment in Units. This summary is of a general nature only and is
not intended to be, nor should it be construed to be, legal or income tax advice to any particular Non-Resident Holder. Non-Resident
Holders should consult their own income tax advisors with respect to the tax consequences applicable to them based on their own
particular circumstances.
Allocation of Purchase Price of Units
The total purchase price of a Unit to a Non-Resident
Holder must be allocated on a reasonable basis between the Unit Share and the Warrant which comprise that Unit to determine the
cost of each to the Non-Resident Holder for purposes of the Tax Act.
For its purposes, the Corporation intends
to allocate approximately US$0.80 of the issue price of each Unit as consideration for the issue of each Unit Share and US$0.01
of the issue price of each Unit for the issue of the Warrant. Although the Corporation believes that its allocation is reasonable,
it is not binding on the CRA or the Non-Resident Holder. The Non-Resident Holder’s adjusted cost base of the Unit Share will
be determined by averaging the cost allocated to the Unit Share with the adjusted cost base to the Non-Resident Holder of all Common
Shares of the Corporation owned by the Non-Resident Holder as capital property immediately prior to such acquisition.
Exercise of Warrants
No gain or loss will be realized by a Non-Resident
Holder upon the exercise of a Warrant to acquire a Warrant Share. When a Warrant is exercised, the Non-Resident Holder’s
cost of the Warrant Share acquired thereby will be the aggregate of the Non-Resident Holder’s adjusted cost base of such
Warrant and the exercise price paid for the Warrant Share. The Non-Resident Holder’s adjusted cost base of the Warrant Share
so acquired will be determined by averaging such cost with the adjusted cost base to the Non-Resident Holder of all Common Shares
of the Corporation owned by the Non-Resident Holder as capital property immediately prior to such acquisition.
Taxation of Dividends
Dividends paid or credited, or deemed to be
paid or credited, to a Non-Resident Holder on the Shares will be subject to Canadian withholding tax under the Tax Act at the rate
of 25% of the gross amount of the dividend. Such rate is generally reduced under the Canada-U.S. Tax Convention to 15% if the beneficial
owner of such dividend is a U.S. Holder. The rate of withholding tax is further reduced to 5% if the beneficial owner of such dividend
is a U.S. Holder that is a company that owns, directly or indirectly, at least 10% of the voting stock of the Corporation. In addition,
under the Canada-U.S. Tax Convention, dividends may be exempt from such Canadian withholding tax if paid to certain U.S. Holders
that are qualifying religious, scientific, literary, educational or charitable tax-exempt organizations or qualifying trusts, companies,
organizations or arrangements operated exclusively to administer or provide pension, retirement or employee benefits or benefits
for the self-employed under one or more funds or plans established to provide pension or retirement benefits or other employee
benefits that are exempt from tax in the United States and that have complied with specific administrative procedures.
Disposition of Shares and Warrants
A Non-Resident Holder will not be subject to
tax under the Tax Act in respect of any capital gain realized by such Non-Resident Holder on a disposition of Shares or Warrants,
unless the Shares or Warrants constitute “taxable Canadian property” (as defined in the Tax Act) of the Non-Resident
Holder at the time of the disposition and are not “treaty-protected property” (as defined in the Tax Act) of the Non-Resident
Holder at the time of the disposition.
Generally, as long as the Shares are then listed
on a designated stock exchange (which currently includes the TSX), the Shares and Warrants will not constitute taxable Canadian
property of a Non-Resident Holder, unless at any time during the 60-month period immediately preceding the disposition the following
two conditions are met concurrently: (a) the Non-Resident Holder, persons with which the Non-Resident Holder does not deal at arm’s
length, partnerships whose members include, either directly or indirectly through one or more partnerships, the Non-Resident Holder
or persons which do not deal at arm’s length with the Non-Resident Holder, or any combination of them, owned 25% or more
of the issued shares of any class or series of shares of the capital stock of the Corporation, and (b) more than 50% of the fair
market value of the Shares was derived directly or indirectly, from one or any combination of real or immovable property situated
in Canada, “Canadian resource properties”, “timber resource properties” (each as defined in the Tax Act)
and options in respect of or interests in, or for civil law rights in, any such property (whether or not such property exists).
In the case of a US Holder, the Shares of such
US Holder will generally constitute “treaty-protected property” for purposes of the Tax Act unless the value of the
Shares is derived principally from real property situated in Canada. For this purpose, “real property” has the meaning
that term has under the laws of Canada and includes any option or similar right in respect thereof and usufruct of real property,
rights to explore for or to exploit mineral deposits, sources and other natural resources and rights to amounts computed by reference
to the amount or value of production from such resources.
Non-Resident Holders whose Common Shares
may be taxable Canadian property should consult their own tax advisors.
UNDERWRITING
Under the terms and subject to the conditions in the Underwriting Agreement, Ladenburg Thalmann &
Co. Inc. (the “
Underwriter
”) has agreed to purchase, and the Corporation has agreed to sell to the Underwriter,
4,619,773 Units.
The obligations of the Underwriter under
the Underwriting Agreement may be terminated at its discretion if (i) the Corporation shall have failed, refused or been
unable, at or prior to such closing date, to perform any agreement on its part to be performed hereunder, (ii) any other
condition of the Underwriters’ obligations hereunder is not fulfilled, (iii) trading on the Nasdaq or the TSX of the
Common Shares shall have been suspended, (iv) minimum or maximum prices for trading shall have been fixed, or maximum ranges
for prices for securities shall have been required, on the Nasdaq or the TSX, by such stock exchange or by order of the SEC
or any other governmental authority having jurisdiction, (v) a banking moratorium shall have been declared by U.S. federal,
Canadian federal or state authorities, or (vi) there shall have occurred any outbreak or escalation of hostilities or any
calamity or crisis that, in the Underwriter’s judgment, is material and adverse and makes it impractical or inadvisable
to proceed with the completion of the sale of and payment for the Units offered under this Prospectus Supplement. The
Underwriter is, however, obligated to take up and pay for all of the Units pursuant to the Offering if any of the Units are
purchased under the Underwriting Agreement. However, the Underwriter is not required to take or pay for the Over-Allotment
Unit Shares or Over-Allotment Warrants covered by the Over-allotment Option.
The Offering is being made in the United States
pursuant to the multijurisdictional disclosure system implemented by the SEC and the securities regulatory authorities in Canada.
The Units will be offered in the United States by the Underwriter. The Units will be offered in the United States by the Underwriter
directly.
No Units offered pursuant to the Offering will be sold to Canadian purchasers.
Subject
to applicable law, the Underwriter may offer the Units outside of the United States and Canada.
The Underwriter is offering the Units, subject
to prior sale, when, as and if issued to and accepted by them, subject to approval of legal matters by their counsel, including
the validity of the Units, and other conditions contained in the Underwriting Agreement, such as the receipt by the Underwriter
of officers’ certificates and legal opinions. The Underwriter reserves the right to withdraw, cancel or modify offers to
the public and to reject orders in whole or in part.
The Underwriter proposes to offer the Units directly to the public at the price set forth on the cover
page of this Prospectus Supplement and to certain dealers at that price less a concession not in excess of US$0.03402 per Unit.
After the Offering, these figures may be changed by the Underwriter. The offering price of the Units was determined by arm’s
length negotiation between the Corporation and the Underwriter.
The Underwriter Fee is equal to the public
offering price per Unit less the amount paid by the Underwriter to the Corporation per Common Share. The following table shows
the per Unit and total underwriting discount to be paid to the Underwriter in the Offering.
|
|
|
Per Unit
(1)
|
|
|
Without Over-Allotment
Option Exercise
|
|
|
With Full Over-Allotment
Option Exercise
|
|
|
Public Offering price
|
|
US$
|
0.81
|
|
|
US$
|
3,742,016.13
|
|
|
US$
|
4,303,317.78
|
|
|
Underwriter Fee
(2)
|
|
US$
|
0.0567
|
|
|
US$
|
261,941.13
|
|
|
US$
|
301,232.24
|
|
|
Proceeds, before expenses, to us
|
|
US$
|
0.7533
|
|
|
US$
|
3,480,075.00
|
|
|
US$
|
4,002,085.54
|
|
Notes:
|
|
(1)
|
The Price to the Public and Underwriter Fee corresponds
to (i) a Price to the Public per Unit Share of US$0.80 (or US$0.7433 per Unit Share after deduction of the Underwriter
Fee), and (ii) a Price to the Public per Warrant of US$0.01 (no Underwriter Fee is being deducted per Warrant).
|
|
|
(2)
|
The Corporation has granted to the Underwriter an option
(the “
Over-Allotment Option
”), exercisable in whole or in part to purchase up to an additional 692,965 Unit
Shares (each, an “
Over-Allotment Unit Share
”) and/or 692,965 Warrants (each, an “
Over-Allotment Warrant
”)
(up to 15% of the number of Unit Shares and Warrants sold in the primary offering) from us at the Public Offering Price for a
period of 30 days from and including the date of this Prospectus Supplement to cover over-allotments, if any, and for market stabilization
purposes. If the Over-Allotment Option is exercised in full, the total Public Offering Price, the Underwriter Fee and net proceeds
to the Corporation (prior to the deduction of the expenses of the Offering) will be US$4,303,317.78, US$301,232.24 and US$4,002,085.54,
respectively.
|
The Corporation estimates that its total
expenses for the Offering will be approximately US$300,000. Pursuant to the terms of the Underwriting Agreement, the Corporation
has also agreed to reimburse the Underwriter for expenses relating to the Offering up to US$150,000.
The Underwriter has advised us that it does
not intend to confirm sales to any account over which it exercises discretionary authority.
The Corporation has agreed to indemnify the
Underwriter against certain liabilities, including civil liabilities under the
Securities Act of 1933, as amended
, and applicable
Canadian securities legislation, or to contribute to payments that the Underwriter may be required to make in respect of those
liabilities.
The Corporation and its executive officers
and directors, are subject to lock-up agreements that prohibit, subject to certain exceptions, the Corporation and such persons
from, for a period of 90 days following the date of this Prospectus Supplement, offering, pledging, announcing the intention to
sell, selling, contracting to sell, selling any option or contract to purchase, purchasing any option or contract to sell, granting
any option, right or warrant to purchase or otherwise transferring or disposing of, directly or indirectly, any Common Shares or
any securities convertible into or exercisable or exchangeable for Common Shares or (B) entering into any swap or other agreement
that transfers, in whole or in part, any of the economic consequences of ownership of the Common Shares, whether any such transaction
described in clause (A) or (B) above is to be settled by delivery of Common Shares or such other securities, in cash or otherwise,
except for (1) sales of the Shares to the Underwriters pursuant to this Agreement, (2) grants of options, share awards or the issuance
of Common Shares by the Corporation pursuant to equity incentive plans described in the Time of Sale Disclosure Package, or (3)
issuance of shares upon exercise or conversion of securities outstanding as of the date hereof. The lock-up agreements do not prohibit
the Corporation’s directors and executive officers from transferring Common Shares for bona fide estate or tax planning purposes
and for certain other purposes, subject to certain requirements, including that the transferee be subject to the same lock-up terms.
The Common Shares of the Corporation are listed
for trading on the Nasdaq under the symbol “ONCY” and on the TSX under the symbol “ONC”. The Corporation
has applied to list the Common Shares qualified for distribution by this Prospectus Supplement on the Nasdaq and the TSX. Listing
will be subject to the Corporation fulfilling all of the listing requirements of the Nasdaq and the TSX, respectively.
The Underwriter may engage in syndicate covering transactions stabilizing
transactions and penalty bids or purchases for the purpose of pegging, fixing or maintaining the price of our Common Shares:
|
|
·
|
Syndicate covering transactions involve purchases of securities in the open market after the distribution has been completed
in order to cover syndicate short positions. Such a naked short position would be closed out by buying securities in the open market.
A naked short position is more likely to be created if the underwriters are concerned that there could be downward pressure on
the price of the securities in the open market after pricing that could adversely affect investors who purchase in the offering.
|
|
|
·
|
Stabilizing transactions permit bids to purchase the underlying security so long as the stabilizing bids do not exceed a specific
maximum and are engaged in for the purpose of preventing or retarding a decline in the market price of the shares of common stock
while this offering is in progress.
|
|
|
·
|
Penalty bids permit the underwriters to reclaim a selling concession from a syndicate member when the securities originally
sold by the syndicate member are purchased in a stabilizing or syndicate covering transaction to cover syndicate short positions.
|
These syndicate covering transactions, stabilizing transactions,
and penalty bids may have the effect of raising or maintaining the market prices of our securities or preventing or retarding a
decline in the market prices of our securities. As a result the price of our common stock may be higher than the price that might
otherwise exist in the open market. Neither we nor the underwriters make any representation or prediction as to the effect that
the transactions described above may have on the price of our common stock. These transactions may be effected on The Nasdaq Capital
Market, TSX, in the over-the-counter market or on any other trading market and, if commenced, may be discontinued at any time.
In connection with this offering, the Underwriter also may engage
in passive market making transactions in our Common Shares in accordance with Regulation M during a period before the commencement
of offers or sales of shares of our Common Shares in this Offering and extending through the completion of the distribution. In
general, a passive market maker must display its bid at a price not in excess of the highest independent bid for that security.
However, if all independent bids are lowered below the passive market maker’s bid that bid must then be lowered when specific
purchase limits are exceeded. Passive market making may stabilize the market price of the securities at a level above that which
might otherwise prevail in the open market and, if commenced, may be discontinued at any time.
Neither we, nor the Underwriter make any representation or prediction
as to the direction or magnitude of any effect that the transactions described above may have on the prices of our securities.
In addition, neither we nor the Underwriter make any representation that the underwriters will engage in these transactions or
that any transactions, once commenced will not be discontinued without notice.
This Prospectus Supplement and the accompanying
Prospectus in electronic format may be made available on the web site maintained by the Underwriter and the Underwriter may distribute
prospectus supplements electronically.
WHERE YOU CAN FIND ADDITIONAL
INFORMATION
The Corporation has filed with the SEC
a registration statement on Form F-10 relating to, among other securities, the Common Shares. This Prospectus Supplement and the
Prospectus, both of which constitute a part of the registration statement, do not contain all of the information contained in
the registration statement, certain items of which are contained in the exhibits to the registration statement as permitted by
the rules and regulations of the SEC. See “
Documents Filed as Part of the Registration Statement
” in this Prospectus
Supplement and the Prospectus. Statements contained in this Prospectus Supplement, the Prospectus or a document incorporated by
reference in the Prospectus about the contents of any contract, agreement or other documents referred to are not necessarily complete,
and in each instance you should refer to the exhibits to the registration statement for a more complete description of the matter
involved. The registration statement, and the items of information omitted from this Prospectus Supplement and the Prospectus
but contained in the registration statement, will be available on EDGAR (
www.sec.gov/edgar.shtml
).
The Corporation is subject to the information
requirements of the U.S. Exchange Act and applicable Canadian securities legislation and, in accordance therewith, files and
furnishes annual and quarterly financial information and material change reports, business acquisition reports and other material
with the securities commission or similar regulatory authority in each of the provinces of Canada and with the SEC. Under MJDS
adopted by the United States and Canada, documents and other information that the Corporation files with the SEC may be prepared
in accordance with the disclosure requirements of Canada, which are different from those of the United States. As a foreign private
issuer within the meaning of rules made under the U.S. Exchange Act, the Corporation is exempt from the rules under the U.S. Exchange
Act prescribing the furnishing and content of proxy statements, and the Corporation’s officers, directors and principal shareholders
are exempt from the reporting and shortswing profit recovery provisions contained in Section 16 of the U.S. Exchange
Act. In addition, the Corporation is not required to publish financial statements as promptly as United States companies.
You may read any document that the Corporation
has filed with the SEC on EDGAR at
www.sec.gov/edgar.shtml
and such information can also be inspected and copies ordered
at the SEC’s public reference room in Washington, D.C. You may also obtain copies of those documents from the public reference
room of the SEC at 100 F Street, N.E., Washington, D.C. 20549 by paying a fee. You should call the SEC at
1-800-SEC-0330 or access its website at www.sec.gov for further information about the public reference rooms. You may read and
download any public document that the Corporation has filed with the Canadian securities regulatory authorities under the Corporation’s
profile on SEDAR (
www.sedar.com
).
LEGAL MATTERS AND INTEREST
OF EXPERTS
The auditors of the Corporation are Ernst &
Young LLP, Chartered Accountants, 1000, 440 – 2
nd
Avenue S.W., Calgary, Alberta, T2P 5E9. Ernst & Young LLP
is independent of the Corporation in accordance with the Rules of Professional Conduct as outlined by the Institute of Chartered
Accountants of Alberta. Ernst & Young LLP is registered with the U.S. Public Corporation Accounting Oversight Board.
Certain legal matters relating to the Offering
will be passed upon on our behalf by McCarthy Tétrault LLP with respect to certain Canadian legal matters and by Dorsey
& Whitney LLP with respect to certain U.S. legal matters and on behalf of the Underwriter by Osler, Hoskin & Harcourt LLP
with respect to certain Canadian legal matters and by Ellenoff Grossman & Schole LLP with respect to certain U.S. legal matters.
As at the date hereof, the partners and associates of each of McCarthy Tetrault LLP and Osler, Hoskin & Harcourt LLP beneficially
own, directly or indirectly, less than 1% of the Common Shares.
AGENT FOR SERVICE OF
PROCESS
Messrs. Wayne Pisano, William G. Rice and Bernd
R. Seizinger are directors of the Corporation who reside outside of Canada. Messrs. Pisano, Rice and Seizinger have appointed the
Corporation, at its principal place of business, as agent for service of process. Purchasers are advised that it may not be possible
for investors to enforce judgments obtained in Canada against any person that resides outside of Canada, even if the party has
appointed an agent for service of process.
BASE SHELF PROSPECTUS
May 4, 2018

Cdn.$150,000,000
Common Shares
Subscription Receipts
Warrants
Units
______________
Oncolytics Biotech Inc. (the “
Corporation
”,
“
Oncolytics
”, “
we
”, “
our
” or “
us
”) may from time to
time offer and issue the following securities: (i) common shares in the capital of the Corporation (“
Common Shares
”);
(ii) subscription receipts of the Corporation exchangeable for Common Shares and/or other securities of the Corporation (“
Subscription
Receipts
”); (iii) warrants exercisable to acquire Common Shares and/or other securities of the Corporation (“
Warrants
”);
and (iv) securities comprised of more than one of Common Shares, Subscription Receipts and/or Warrants offered together as a unit
(“
Units
”), or any combination thereof, up to an aggregate offering price of $150,000,000 (or the equivalent
thereof, at the date of issue, in any other currency or currencies, as the case may be) at any time during the 25-month period
that this short form base shelf prospectus (including any amendments hereto, the “
Prospectus
”) remains valid.
The Common Shares, Subscription Receipts, Warrants and Units (collectively, the “
Securities
”) offered hereby
may be offered separately or together, in separate series, in amounts, at prices and on terms to be set forth in one or more prospectus
supplements (collectively or individually, as the case may be, “
Prospectus Supplements
”).
The specific terms of any offering of Securities
will be set forth in the applicable Prospectus Supplement and may include, without limitation, where applicable: (i) in the case
of Common Shares, the number of Common Shares being offered, the offering price (in the event the offering is a fixed price distribution),
the manner of determining the offering price(s) (in the event the offering is not a fixed price distribution) and any other specific
terms; (ii) in the case of Subscription Receipts, the number of Subscription Receipts being offered, the offering price, the terms,
conditions and procedures for the exchange of the Subscription Receipts into or for Common Shares and/or other securities of the
Corporation and any other specific terms; (iii) in the case of Warrants, the number of such Warrants offered, the offering price,
the terms, conditions and procedures for the exercise of such Warrants into or for Common Shares and/or other securities of the
Corporation and any other specific terms; and (iv) in the case of Units, the number of Units being offered, the offering price,
the terms of the Common Shares, Subscription Receipts and/or Warrants, as the case may be, underlying the Units, and any other
specific terms.
This offering is made by a Canadian issuer
that is permitted, under a multijurisdictional disclosure system adopted by the United States and Canada (“
MJDS
”),
to prepare this Prospectus in accordance with Canadian disclosure requirements. Prospective investors in the United States should
be aware that such requirements are different from those of the United States. Financial statements included or incorporated by
reference herein have been prepared in accordance with International Financial Reporting Standards (“
IFRS
”)
as issued by the International Accounting Standards Board (“
IASB
”) and may not be comparable to financial statements
of United States companies. Such financial statements are subject to Canadian generally accepted auditing standards and auditor
independence standards, in addition to the standards of the Public Company Accounting Oversight Board (United States) and the United
States Securities and Exchange Commission (“
SEC
”) independence standards.
Prospective investors should be aware that
the acquisition of the securities described herein may have tax consequences both in the United States and in Canada. Such consequences
for investors who are resident in, or citizens of, the United States may not be described fully herein. Prospective investors should
read the tax discussion contained in the applicable Prospectus Supplement with respect to a particular offering of Securities.
The enforcement by investors of civil
liabilities under the United States federal securities laws may be affected adversely by the fact that the Corporation is incorporated
under the laws of Alberta, Canada, that the majority of its officers and directors are residents of Canada, that many of the experts
named in the registration statement are not residents of the United States, and that a substantial portion of the assets of the
Corporation and said persons are located outside the United States.
NEITHER THE SEC NOR ANY STATE OR CANADIAN
SECURITIES COMMISSION HAS APPROVED OR DISAPPROVED THE SECURITIES OFFERED HEREBY OR DETERMINED IF THIS PROSPECTUS IS TRUTHFUL OR
COMPLETE. ANY REPRESENTATION TO THE CONTRARY IS A CRIMINAL OFFENCE.
Any investment in Securities involves
significant risks that should be carefully considered by prospective investors before purchasing Securities. The risks outlined
in this Prospectus and in the documents incorporated by reference herein, including the applicable Prospectus Supplement, should
be carefully reviewed and considered by prospective investors in connection with any investment in Securities. See “
Risk
Factors
”.
All shelf information permitted under applicable
securities legislation to be omitted from this Prospectus including, without limitation, the information disclosed in the specific
terms of any offering of Securities, as discussed above, will be contained in one or more Prospectus Supplements that will be delivered
to purchasers together with this Prospectus, except in cases where an exemption from such delivery requirements has been obtained.
Each Prospectus Supplement will be incorporated by reference into this Prospectus for the purposes of securities legislation as
of the date of such Prospectus Supplement and only for the purposes of the distribution of the Securities to which that Prospectus
Supplement pertains.
We may sell the Securities to or through
one or more underwriters or dealers purchasing as principals and may also sell the Securities to one or more purchasers directly,
through applicable statutory exemptions, or through one or more agents designated by us from time to time. The Securities may be
sold from time to time in one or more transactions at fixed prices or not at fixed prices, such as market prices prevailing at
the time of sale, prices related to such prevailing market prices or prices to be negotiated with purchasers, which prices may
vary as between purchasers and during the period of distribution of the Securities. The Prospectus Supplement relating to a particular
offering of Securities will identify each underwriter, dealer or agent engaged in connection with the offering and sale of such
Securities, as well as the method of distribution and the terms of the offering of such Securities, including the initial offering
price (in the event the offering is a fixed price distribution), the manner of determining the offering price(s) (in the event
the offering is not a fixed price distribution), the net proceeds to us and, to the extent applicable, any fees, discounts or any
other compensation payable to underwriters, dealers or agents and any other material terms. See “
Plan of Distribution
”.
In connection with any offering of the
Securities other than an “at-the-market distribution” (as defined under applicable Canadian legislation) (unless otherwise
specified in the relevant Prospectus Supplement), the underwriters or agents may over-allot or effect transactions that stabilize
or maintain the market price of the offered Securities at a level above that which might otherwise prevail on the open market.
Such transactions, if commenced, may be interrupted or discontinued at any time. See “
Plan of Distribution
”.
No underwriter or dealer involved in an
“at-the-market distribution” under this Prospectus, no affiliate of such an underwriter or dealer and no person or
company acting jointly or in concert with such underwriter or dealer will over-allot securities in connection with such distribution
or effect any other transactions that are intended to stabilize or maintain the market price of the Securities.
Owning the Securities may subject you to
tax consequences. This Prospectus and any applicable Prospectus Supplement may not describe the tax consequences fully. You should
read the tax discussion in any applicable Prospectus Supplement and consult with your own tax advisor with respect to your own
particular circumstances.
Unless otherwise specified in the applicable
Prospectus Supplement, the Subscription Receipts, Warrants and Units will not be listed on any securities exchange. There is no
market through which these securities may be sold and purchasers may not be able to resell such securities purchased under this
Prospectus. This may affect the pricing of such securities in the secondary market, the transparency and availability of trading
prices, the liquidity of such securities, and the extent of issuer regulation. See “
Forward-Looking Statements
”
and “
Risk Factors
”.
Our outstanding securities are listed
for trading on the Toronto Stock Exchange (“
TSX
”) under the trading symbol “
ONC
” and are
quoted for trading on the OTCQX Best Market (“
OTCQX
”) under the trading symbol “
ONCYF
”.
On May 3, 2018, the closing price of our Common Shares on the TSX was $0.63 per Common Share.
Messrs. Wayne Pisano, William G. Rice and
Bernd R. Seizinger are directors of the Corporation who reside outside of Canada. Messrs. Pisano, Rice and Seizinger have appointed
the Corporation, at its principal place of business, as agent for service of process. Purchasers are advised that it may not be
possible for investors to enforce judgments obtained in Canada against any person that resides outside of Canada, even if the party
has appointed an agent for service of process.
No underwriter, agent or dealer has
been involved in the preparation of this Prospectus or performed any review of the contents of this Prospectus.
Our head office and principal place of
business is located at 210, 1167 Kensington Crescent N.W., Calgary, Alberta, T2N 1X7. Our registered office is located at
4000, 421 - 7
th
Avenue S.W., Calgary, Alberta, T2P 4K9.
TABLE
OF CONTENTS
Page
ABOUT THIS PROSPECTUS
AND OTHER MATTERS
In this Prospectus and any Prospectus Supplement,
unless otherwise indicated, references to “
we
”, “
us
”, “
our
”, “
issuer
”
“
Oncolytics
” or the “
Corporation
” are to Oncolytics Biotech Inc., including, where the context
requires, its subsidiaries and affiliates.
In this Prospectus and in any Prospectus
Supplement, unless otherwise specified or the context otherwise requires, all references to “
dollars
” or “
$
”
are to Canadian dollars and all references to “
US$
” are to United States dollars.
Unless otherwise indicated, all financial
information included and incorporated by reference in this Prospectus and any Prospectus Supplement is determined using IFRS as
issued by IASB and adopted by the Accounting Standards Board of Canada.
This Prospectus provides you with a general
description of the Securities that the Corporation may offer. Each time the Corporation sells Securities under this Prospectus,
the Corporation will file and deliver, except in cases where an exemption from such delivery requirement has been obtained, a Prospectus
Supplement that will contain specific information about the terms of that offering of Securities. The Prospectus Supplement also
may add, update or change information contained in this Prospectus. Before investing, investors should read both this Prospectus
and any applicable Prospectus Supplement together with additional information described under the heading “
Documents Incorporated
by Reference
”.
You should rely only on the information
contained in or incorporated by reference in this Prospectus or any applicable Prospectus Supplement. The Corporation has not authorized
anyone to provide you with different or additional information. The Corporation is not making an offer of these Securities in any
jurisdiction where the offer is not permitted by law.
FORWARD-LOOKING STATEMENTS
This Prospectus and the documents incorporated
by reference herein contain certain statements relating to future events or the Corporation’s future performance which constitute
forward-looking statements within the meaning of applicable Canadian securities laws and within the meaning of the United States
Private Securities Litigation Reform Act of 1995. Such forward-looking statements involve known and unknown risks, uncertainties
and other factors which may cause the actual results, performance or achievements of the Corporation, or industry results, to be
materially different from any future results, performance or achievements expressed or implied by such forward-looking statements.
Forward-looking statements are statements that are not historical facts, and include, but are not limited to, estimates and their
underlying assumptions; statements regarding plans, objectives and expectations with respect to the efficacy of our technologies;
the timing and results of clinical studies related to our technologies; future operations, products and services; the impact of
regulatory initiatives on our operations; the size of and opportunities related to the markets for our technologies; general industry
and macroeconomic growth rates; expectations related to possible joint and/or strategic ventures and statements regarding future
performance. Forward-looking statements generally, but not always, are identified by the words “expects,” “anticipates,”
“believes,” “intends,” “estimates,” “projects”, “potential”, “possible”
and similar expressions, or that events or conditions “will,” “may,” “could” or “should”
occur.
The forward-looking statements in this
Prospectus are subject to various risks and uncertainties, most of which are difficult to predict and generally beyond our control,
including without limitation:
|
|
·
|
risks related to all of our products, including REOLYSIN®, being in the research and development
stage and requiring further development and testing before they can be marketed commercially;
|
|
|
·
|
risks inherent in pharmaceutical research and development;
|
|
|
·
|
risks related to timing and possible delays in our clinical trials;
|
|
|
·
|
risks related to some of our clinical trials being conducted in, and subject to the laws of foreign
countries;
|
|
|
·
|
risks related to our pharmaceutical products being subject to intense regulatory approval processes
in the United States and other foreign jurisdictions;
|
|
|
·
|
risks related to being subject to government manufacturing and testing regulations;
|
|
|
·
|
risks related to the extremely competitive biotechnology industry and our competition with larger
companies with greater resources;
|
|
|
·
|
risks related to our reliance on patents and proprietary rights to protect our technology;
|
|
|
·
|
risks related to potential product liability claims;
|
|
|
·
|
risks related to our limited manufacturing experience and reliance on third parties to commercially
manufacture our products, if and when developed;
|
|
|
·
|
risks related to our new products not being accepted by the medical community or consumers;
|
|
|
·
|
risks related to our technologies becoming obsolete;
|
|
|
·
|
risks related to our dependence on third party relationships for research and clinical trials;
|
|
|
·
|
risks related to our license, development, supply and distribution agreement with Adlai Nortye
Biopharma Co. Ltd.;
|
|
|
·
|
risks related to our lack of operating revenues and history of losses;
|
|
|
·
|
uncertainty regarding our ability to obtain third-party reimbursement for the costs of our product;
|
|
|
·
|
risks related to other third-party arrangements;
|
|
|
·
|
risks related to our ability to obtain additional financing to fund future research and development
of our products and to meet ongoing capital requirements;
|
|
|
·
|
risks related to potential increases in the cost of director and officer liability insurance;
|
|
|
·
|
risks related to our dependence on key employees and collaborators;
|
|
|
·
|
risks related to Barbados law, including those relating to the enforcement of judgments obtained
in Canada or the United States;
|
|
|
·
|
risks related to the effect of changes in the law on our corporate structure;
|
|
|
·
|
risks related to expenses in foreign currencies and our exposure to foreign currency exchange rate
fluctuations;
|
|
|
·
|
risks related to fluctuations in interest rates;
|
|
|
·
|
risks related to information technology systems; and
|
|
|
·
|
risks related to our Common Shares.
|
This list is not exhaustive of the factors
that may affect any of the Corporation’s forward-looking statements. Some of the important risks and uncertainties that could
affect forward-looking statements are described further under the heading “
Risk Factors
” in our Annual Report.
If one or more of these risks or uncertainties materializes, or if underlying assumptions prove incorrect, our actual results may
vary materially from those expected, estimated or projected. Forward-looking statements in this document are not a prediction of
future events or circumstances, and those future events or circumstances may not occur. Given these uncertainties, users of the
information included herein, including investors and prospective investors, are cautioned not to place undue reliance on such forward-looking
statements. Investors should consult our quarterly and annual filings with the Canadian and U.S. securities commissions for additional
information on risks and uncertainties relating to forward-looking statements. The Corporation does not undertake any obligation
to publicly update or revise any forward-looking statements other than as required under applicable securities laws.
Prospective investors should carefully
consider the information contained under the heading “
Risk Factors
” in our Annual Report and all other information
included in or incorporated by reference in this Prospectus before making investment decisions with regard to the Securities.
RISK FACTORS
An investment in the Securities involves
a high degree of risk. Prospective investors should note that there is no market through which the Subscription Receipts, Warrants
or Units may be sold and purchasers may not be able to resell the Subscription Receipts, Warrants or Units purchased under this
Prospectus. This may affect the pricing of these securities in the secondary market, the transparency and availability of trading
prices, the liquidity of the securities, and the extent of issuer regulation.
Prospective investors should consider carefully
the risks described in the documents incorporated by reference in this Prospectus (including in subsequently filed documents incorporated
by reference) and those described in any Prospectus Supplement before purchasing the Securities offered hereby. Discussions of
certain risks affecting the Corporation in connection with its business are provided under the heading “
Risk Factors
”
in our Annual Report filed with the various securities regulatory authorities, which is incorporated by reference in this Prospectus.
DOCUMENTS INCORPORATED
BY REFERENCE
Information has been incorporated
by reference in this Prospectus from documents filed with securities commissions or similar authorities in Canada.
Copies
of the documents incorporated herein by reference may be obtained on request without charge from our Corporate Secretary at 210,
1167 Kensington Crescent N.W., Calgary, Alberta, T2N 1X7 telephone (403) 670-7377, and are available electronically
under the Corporation’s profile on SEDAR (www.sedar.com) and on EDGAR (www.sec.gov/edgar.shtml).
We have filed the following documents with
the securities commissions or similar regulatory authorities in certain of the provinces of Canada and such documents are specifically
incorporated by reference in, and form an integral part of, this Prospectus:
|
|
(a)
|
our annual report on Form 20-F (“
Annual Report
”) dated March 19, 2018, for the
year ended December 31, 2017 (filed in Canada with the Canadian securities regulatory authorities in lieu of an annual information
form);
|
|
|
(b)
|
our management information circular dated March 27, 2018 relating to the annual general meeting
of shareholders to be held on May 3, 2018;
|
|
|
(c)
|
our audited consolidated financial statements, together with the notes thereto, as at December 31,
2017 and 2016, which comprise the consolidated statements of financial position as at December 31, 2017 and 2016, and the
consolidated statements of loss and comprehensive loss, changes in equity, and cash flows for the years ended December 31,
2017 and 2016, together with the independent auditors’ report thereon; and
|
|
|
(d)
|
our management’s discussion and analysis of financial condition and results of operations
dated March 8, 2018, for the year ended December 31, 2017.
|
Any documents of the type required by National
Instrument 44-101 -
Short Form Prospectus Distributions
to be incorporated by reference in a short form prospectus, including
any annual information form, annual report on Form 20-F, comparative annual consolidated financial statements and the auditors’
report thereon, comparative interim consolidated financial statements, management’s discussion and analysis of financial
condition and results of operations, material change report (except a confidential material change report), business acquisition
report and information circular, if filed by us with the securities commissions or similar authorities in Canada after the date
of this Prospectus and prior to the date which is 25 months from the date of this Prospectus, shall be deemed to be incorporated
by reference in this Prospectus.
In addition, to the extent that any document
or information incorporated by reference into this Prospectus is included in any report filed with or furnished to the SEC pursuant
to the United States Securities Exchange Act of 1934, as amended (the “
U.S. Exchange Act
”), after the date of
this Prospectus, such document or information shall be deemed to be incorporated by reference as an exhibit to the registration
statement of which this Prospectus forms a part (in the case of documents or information deemed furnished on Form 6-K or Form 8-K,
only to the extent specifically stated therein)
Any statement contained in this Prospectus
or in a document incorporated or deemed to be incorporated by reference herein will be deemed to be modified or superseded for
the purposes of this Prospectus to the extent that a statement contained in this Prospectus or in any other subsequently filed
document which also is, or is deemed to be, incorporated by reference into this Prospectus modifies or supersedes that statement.
The modifying or superseding statement need not state that it has modified or superseded a prior statement or include any other
information set forth in the document that it modifies or supersedes. The making of a modifying or superseding statement shall
not be deemed an admission for any purposes that the modified or superseded statement, when made, constituted a misrepresentation,
an untrue statement of a material fact or an omission to state a material fact that is required to be stated or that is necessary
to make a statement not misleading in light of the circumstances in which it was made. Any statement so modified or superseded
shall not be deemed, except as so modified or superseded, to constitute part of this Prospectus.
Upon a new annual information form and
the related audited annual financial statements and management’s discussion and analysis being filed by us with the applicable
securities regulatory authorities during the term of this Prospectus, the previous annual information form, the previous audited
annual financial statements and related management’s discussion and analysis, all unaudited interim financial statements
and related management’s discussion and analysis, material change reports and business acquisition reports filed prior to
the commencement of our financial year in which the new annual information form and the related audited annual financial statements
and management’s discussion and analysis are filed shall be deemed no longer to be incorporated into this Prospectus for
purposes of future offers and sales of Securities hereunder. Upon new interim financial statements and related management’s
discussion and analysis being filed by us with the applicable securities regulatory authorities during the term of this Prospectus,
all interim financial statements and related management’s discussion and analysis filed prior to the new interim consolidated
financial statements and related management’s discussion and analysis shall be deemed no longer to be incorporated into this
Prospectus for purposes of future offers and sales of Securities hereunder. Upon a new information circular relating to an annual
general meeting of holders of Common Shares being filed by us with the applicable securities regulatory authorities during the
term of this Prospectus, the information circular for the preceding annual general meeting of holders of Common Shares shall be
deemed no longer to be incorporated into this Prospectus for purposes of future offers and sales of Securities hereunder.
Any “template version”
of any “marketing materials” (as such terms are defined in National Instrument 41-101) pertaining to a distribution
of Securities will be filed under the Corporation’s profile on SEDAR (www.sedar.com). In the event that such marketing materials
are filed subsequent to the date of filing of the applicable prospectus supplement pertaining to the distribution of the Securities
to which such marketing materials relates and prior to the termination of such distribution, such filed versions of the marketing
materials will be deemed to be incorporated by reference into the Prospectus for purposes of future offers and sales of Securities
hereunder.
One or more Prospectus Supplements containing
the specific variable terms for an issue of the Securities and other information in relation to such Securities will be delivered
to purchasers of such Securities together with this Prospectus, except in cases where an exemption from such delivery requirement
has been obtained, and will be deemed to be incorporated by reference into this Prospectus as of the date of the Prospectus Supplement
solely for the purposes of the offering of the Securities covered by any such Prospectus Supplement.
DOCUMENTS FILED AS
PART OF THE REGISTRATION STATEMENT
The following documents have been or
will be filed with the SEC as part of the registration statement of which this Prospectus forms a part: (i) the documents
set out under the heading “
Documents Incorporated by Reference
”; (ii) the consents of the Corporation’s
auditor and legal counsel; and (iii) the powers of attorney from the directors and certain officers of the Corporation. A
copy of the form of warrant indenture, unit indenture or subscription receipt agreement, as applicable, will be filed by post-effective
amendment or by incorporation by reference to documents filed or furnished with the SEC under the U.S. Exchange Act.
ADDITIONAL INFORMATION
The Corporation has filed with the
SEC a registration statement on Form F-10 relating to the Securities. This Prospectus, which constitutes a part of the registration
statement, does not contain all of the information contained in the registration statement, certain items of which are contained
in the exhibits to the registration statement as permitted by the rules and regulations of the SEC. See “
Documents Filed
as Part of the Registration Statement
”. Statements included or incorporated by reference in this Prospectus about the
contents of any contract, agreement or other documents referred to are not necessarily complete, and in each instance you should
refer to the exhibits to the registration statement for a more complete description of the matter involved. The registration statement,
and the items of information omitted from this Prospectus but contained in the registration statement, will be available on EDGAR
(www.sec.gov/edgar.shtml). Each time we sell Securities under the registration statement, we will provide a Prospectus Supplement
that will contain specific information about the terms of that offering. The Prospectus Supplement may also add to, update or
change information contained in this Prospectus.
The Corporation is subject to the information
requirements of the U.S. Exchange Act and applicable Canadian securities legislation and, in accordance therewith, files
and furnishes annual and quarterly financial information and material change reports, business acquisition reports and other material
with the securities commission or similar regulatory authority in each of the provinces of Canada and with the SEC. Under MJDS
adopted by the United States and Canada, documents and other information that the Corporation files with the SEC may be prepared
in accordance with the disclosure requirements of Canada, which are different from those of the United States. As a foreign private
issuer within the meaning of rules made under the U.S. Exchange Act, the Corporation is exempt from the rules under the U.S. Exchange
Act prescribing the furnishing and content of proxy statements, and the Corporation’s officers, directors and principal
shareholders are exempt from the reporting and shortswing profit recovery provisions contained in Section 16 of the U.S. Exchange
Act. In addition, the Corporation is not required to publish financial statements as promptly as United States companies.
You may read any
document that the Corporation has filed with the SEC on EDGAR at www.sec.gov/edgar.shtml and such information can also be inspected
and copies ordered at the SEC’s public reference room in Washington, D.C. You may also obtain copies of those documents
from the public reference room of the SEC at 100 F Street, N.E., Washington, D.C. 20549 by paying a fee. You
should call the SEC at 1-800-SEC-0330 or access its website at www.sec.gov for further information about the public reference
rooms. You may read and download any public document that the Corporation has filed with the Canadian securities regulatory authorities
under the Corporation’s profile on SEDAR (www.sedar.com).
THE CORPORATION
Oncolytics Biotech Inc. was incorporated
pursuant to the ABCA on April 2, 1998 as 779738 Alberta Ltd. On April 8, 1998, we amended our articles of incorporation
(the “
Articles
”) and changed our name to Oncolytics Biotech Inc. On July 29, 1999, we further amended our
Articles by removing the private company restrictions included therein and subdivided the 2,222,222 Common Shares issued and outstanding
into 6,750,000 Common Shares. On February 9, 2007, we further amended our Articles to permit shareholder meetings to be held
at any place in Alberta or at any other location as determined by our board of directors (the “
Board
”).
We have two material operating subsidiaries:
Oncolytics Biotech (Barbados) Inc. and Oncolytics Biotech (US) Inc., a Delaware corporation. Oncolytics Biotech (Barbados) Inc.
is incorporated pursuant to the laws of Barbados and is a wholly-owned direct subsidiary of the Corporation. Oncolytics Biotech
(U.S.) Inc. is incorporated pursuant to the laws of Delaware and is a wholly-owned direct subsidiary or Oncolytics Biotech (Barbados)
Inc.
Our head office and principal place of
business is located at 210, 1167 Kensington Crescent N.W., Calgary, Alberta, T2N 1X7. Our registered office is located at
4000, 421 - 7
th
Avenue S.W., Calgary, Alberta, T2P 4K9.
BUSINESS OF THE CORPORATION
General
Since our inception in April of 1998, Oncolytics
Biotech Inc. has been a development stage company and we have focused our research and development efforts on the development of
REOLYSIN (pelareorep), a systemically administered immuno-oncology (“
I-O
”) viral agent with the potential to
treat a variety of cancers. We have not been profitable since our inception and expect to continue to incur substantial losses
as we continue research and development efforts. We do not expect to generate significant revenues until, if and when, pelareorep
becomes commercially viable.
Our potential product for human use, pelareorep,
an unmodified reovirus, is a first in class systemically administered I-O viral agent for the treatment of solid tumors and hematological
malignancies.
Further information regarding the business
of the Corporation is contained in the Annual Report under the heading “
Item 4 – Information on the Company
”,
which document is incorporated by reference in this Prospectus. See “Documents Incorporated by Reference.”
Recent Developments
On February 23, 2018, the Corporation received
approval from the holders of Common Shares to amend the Articles to effect the consolidation of the issued and outstanding Common
Shares on the basis of a consolidation ratio to be selected by the Board, in its sole discretion, provided that the ratio may be
not less than two (2), and not more than fifteen (15), pre-consolidation Common Shares for each one post-consolidation Common Share,
such amendment to become effective at a date in the future to be determined by the Board when the Board considers it to be in the
best interests of the Corporation, but in any event no later than February 22, 2019, subject to approval of the Toronto Stock Exchange.
CONSOLIDATED CAPITALIZATION
There has been no material change in the
share and loan capital of the Corporation on a consolidated basis since December 31, 2017.
USE OF PROCEEDS
The use of proceeds from the issue and
sale of specific Securities pursuant to this Prospectus will be described in the Prospectus Supplement relating to the issuance
and sale of such Securities.
DESCRIPTION OF SHARE
CAPITAL
Authorized Capital
Our authorized capital consists of an unlimited
number of Common Shares. The following is a summary of the provisions attached to our Common Shares.
Common Shares
The holders of our Common Shares are entitled
to one vote per share at meetings of shareholders, to receive such dividends as declared by the Board and to receive our remaining
property and assets upon dissolution or wind up. Our Common Shares are not subject to any future call or assessment and there are
no pre-emptive, conversion or redemption rights attached to such shares.
As at the date hereof, we have 142,325,222
Common Shares issued and outstanding. After giving effect to the exercise of all outstanding options to acquire Common Shares and
all outstanding share awards granted under the Corporation’s Incentive Share Award Plan, we would have 154,164,330 Common
Shares issued and outstanding.
Common Share Purchase
Warrants
As of the date hereof, we have 16,445,000
Common Share purchase warrants (the “
2017 Warrants
”) issued and outstanding. Each 2017 Warrant entitles the
holder to purchase one Common Share until June 1, 2022, at an exercise price of $0.95. The 2017 Warrants are subject to acceleration
if the volume weighted average price of the Common Shares equals or exceeds $2.50 for a period of 15 consecutive trading dates.
In addition, as of the date hereof, the
Corporation has outstanding Common Share purchase warrants as follows:
|
|
·
|
a Common Share purchase warrant (the “
First Adlai Warrant
”) exercisable by the
holder thereof until May 14, 2018 to purchase such number of Common Shares as is calculated by dividing US$2,000,000 by the Exercise
Price (as defined below); and
|
|
|
·
|
a Common Share purchase warrant (the “
Second Adlai Warrant
”) exercisable by
the holder thereof until November 14, 2020 to purchase such number of Common Shares as is calculated by dividing US$6,000,000 by
the Exercise Price.
|
For purposes of the First Adlai Warrant
and the Second Adlai Warrant, the term “
Exercise Price
” means an amount equal to 120% of the volume weighted
average trading price of the Common Shares on the TSX (or, if the Common Shares begin trading on The NASDAQ Capital Market, on
The NASDAQ Capital Market as of the date such trading commences) for the five trading days immediately preceding the exercise date.
The First Adlai Warrant is subject to a
right to call by the Corporation upon the later of: (i) May 14, 2018; and (ii) the date of the enrollment of the first patient
in a Phase III Study related to pelareorep. The Second Adlai Warrant is subject to a right to call by the Corporation upon the
date of the enrollment of the fiftieth (50
th
) patient in a Phase III Study related to pelareorep.
DESCRIPTION OF SUBSCRIPTION
RECEIPTS
The following description of the terms
of Subscription Receipts sets forth certain general terms and provisions of Subscription Receipts in respect of which a Prospectus
Supplement may be filed. The particular terms and provisions of Subscription Receipts offered by any Prospectus Supplement, and
the extent to which the general terms and provisions described below may apply thereto, will be described in the Prospectus Supplement
filed in respect of such Subscription Receipts.
Subscription Receipts may be offered
separately or in combination with one or more other Securities. The Subscription Receipts will be issued under a subscription
receipt agreement (the ”
Subscription Receipt Agreement
”). A copy of the Subscription Receipt Agreement
will be filed by us with the applicable securities regulatory authorities after it has been entered into by us and will be available
electronically under the Corporation’s profile on SEDAR (www.sedar.com) and, if applicable, we will file with the SEC via
EDGAR (www.sec.gov/edgar.shtml) as exhibits to the registration statement of which this Prospectus is a part, or will incorporate
by reference from a Report of Foreign Private Issuer on Form 6-K that we file with the SEC, any Subscription Agreement describing
the terms and conditions of such Subscription Receipts that we are offering before the issuance of such Subscription Receipts.
Pursuant to the Subscription Receipt Agreement,
original purchasers of Subscription Receipts will have a contractual right of rescission against the Corporation, following the
issuance of the underlying Common Share or other securities to such purchasers upon the surrender or deemed surrender of the Subscription
Receipts, to receive the amount paid for the Subscription Receipts in the event that this Prospectus or a Prospectus Supplement,
and any amendment thereto, contains a misrepresentation or is not delivered to such purchaser, provided such remedy for rescission
is exercised within 180 days from the closing date of the offering of Subscription Receipts.
The description of general terms and provisions
of Subscription Receipts described in any Prospectus Supplement will include, where applicable:
|
|
·
|
the number of Subscription Receipts offered;
|
|
|
·
|
the price at which the Subscription Receipts will be offered;
|
|
|
·
|
if other than Canadian dollars, the currency or currency unit in which the Subscription Receipts
are denominated;
|
|
|
·
|
the procedures for the exchange of the Subscription Receipts into Common Shares or other securities;
|
|
|
·
|
the number of Common Shares or other securities that may be obtained upon exercise of each Subscription
Receipt;
|
|
|
·
|
the designation and terms of any other Securities with which the Subscription Receipts will be
offered, if any, and the number of Subscription Receipts that will be offered with each Security;
|
|
|
·
|
the terms applicable to the gross proceeds from the sale of the Subscription Receipts plus any
interest earned thereon;
|
|
|
·
|
the material Canadian tax consequences of owning such Subscription Receipts; and
|
|
|
·
|
any other material terms, conditions and rights (or limitations on such rights) of the Subscription
Receipts.
|
We reserve the right to set forth in a
Prospectus Supplement specific terms of the Subscription Receipts that are not within the options and parameters set forth in this
Prospectus. In addition, to the extent that any particular terms of the Subscription Receipts described in a Prospectus Supplement
differ from any of the terms described in this Prospectus, the description of such terms set forth in this Prospectus shall be
deemed to have been superseded by the description of such differing terms set forth in such Prospectus Supplement.
DESCRIPTION OF WARRANTS
The following description of the terms
of Warrants sets forth certain general terms and provisions of Warrants in respect of which a Prospectus Supplement may be filed.
The particular terms and provisions of Warrants offered by any Prospectus Supplement, and the extent to which the general terms
and provisions described below may apply thereto, will be described in the Prospectus Supplement filed in respect of such Warrants.
Warrants may be offered separately or in combination with one or more other Securities. If applicable, we will file with the SEC
as exhibits to the registration statement of which this Prospectus is a part, or will incorporate by reference from a current report
on Form 6-K that we file with the SEC, any warrant indenture or form of warrant describing the terms and conditions of such Warrants
that we are offering before the issuance of such Warrants.
The description of general terms and provisions
of Warrants described in any Prospectus Supplement will include, where applicable:
|
|
·
|
the designation and aggregate number of Warrants offered;
|
|
|
·
|
the price at which the Warrants will be offered;
|
|
|
·
|
if other than Canadian dollars, the currency or currency unit in which the Warrants are denominated;
|
|
|
·
|
the designation and terms of the Common Shares that may be acquired upon exercise of the Warrants;
|
|
|
·
|
the date on which the right to exercise the Warrants will commence and the date on which the right
will expire;
|
|
|
·
|
the number of Common Shares that may be purchased upon exercise of each Warrant and the price at
which and currency or currencies in which that amount of securities may be purchased upon exercise of each Warrant;
|
|
|
·
|
the designation and terms of any Securities with which the Warrants will be offered, if any, and
the number of the Warrants that will be offered with each Security;
|
|
|
·
|
the date or dates, if any, on or after which the Warrants and the related Securities will be transferable
separately;
|
|
|
·
|
the minimum or maximum amount, if any, of Warrants that may be exercised at any one time;
|
|
|
·
|
whether the Warrants will be subject to redemption or call, and, if so, the terms of such redemption
or call provisions; and
|
|
|
·
|
any other material terms, conditions and rights (or limitations on such rights) of the Warrants.
|
We reserve the right to set forth in a
Prospectus Supplement specific terms of the Warrants that are not within the options and parameters set forth in this Prospectus.
In addition, to the extent that any particular terms of the Warrants described in a Prospectus Supplement differ from any of the
terms described in this Prospectus, the description of such terms set forth in this Prospectus shall be deemed to have been superseded
by the description of such differing terms set forth in such Prospectus Supplement.
DESCRIPTION OF UNITS
We may issue Units comprised of one or
more of the other Securities described in this Prospectus in any combination. Each Unit will be issued so that the holder of the
Unit is also the holder of each Security included in the Unit. Thus, the holder of a Unit will have the rights and obligations
of a holder of each included Security. The unit agreement, if any, under which a Unit is issued may provide that the Securities
comprising the Unit may not be held or transferred separately, at any time or at any time before a specified date. If applicable,
we will file with the SEC as exhibits to the registration statement of which this Prospectus is a part, or will incorporate by
reference from a current report on Form 6-K that we file with the SEC, any unit agreement describing the terms and conditions of
such Units that we are offering before the issuance of such Units.
The particular terms and provisions of
Units offered by any Prospectus Supplement, and the extent to which the general terms and provisions described below may apply
to them, will be described in the Prospectus Supplement filed in respect of such Units.
The particular terms of each issue of Units
will be described in the related Prospectus Supplement. This description will include, where applicable:
|
|
·
|
the designation and aggregate number of Units offered;
|
|
|
·
|
the price at which the Units will be offered;
|
|
|
·
|
if other than Canadian dollars, the currency or currency unit in which the Units are denominated;
|
|
|
·
|
the terms of the Units and of the Securities comprising the Units, including whether and under
what circumstances those securities may be held or transferred separately;
|
|
|
·
|
any provisions for the issuance, payment, settlement, transfer or exchange of the Units or of the
Securities comprising the Units; and
|
|
|
·
|
any other material terms, conditions and rights (or limitations on such rights) of the Units.
|
We reserve the right to set forth in a
Prospectus Supplement specific terms of the Units that are not within the options and parameters set forth in this Prospectus.
In addition, to the extent that any particular terms of the Units described in a Prospectus Supplement differ from any of the terms
described in this Prospectus, the description of such terms set forth in this Prospectus shall be deemed to have been superseded
by the description of such differing terms set forth in such Prospectus Supplement with respect to such Units.
PLAN OF DISTRIBUTION
We may sell the Securities to or through
one or more underwriters or dealers purchasing as principals and we may also sell the Securities to one or more purchasers directly,
through applicable statutory exemptions, or through one or more agents designated from time to time. The Securities may be sold
from time to time in one or more transactions at fixed prices or not at fixed prices, such as market prices prevailing at the time
of sale, prices related to such prevailing market prices or prices to be negotiated with purchasers, which prices may vary as between
purchasers and during the period of distribution of the Securities. The Prospectus Supplement relating to a particular offering
and sale of Securities will identify each underwriter, dealer or agent engaged in connection with the offering and sale of such
Securities, as well as the method of distribution and the terms of the offering and sale of such Securities, including the initial
offering price (in the event the offering is a fixed price distribution), the manner of determining the offering price(s) (in the
event the offering is not a fixed price distribution), the net proceeds to us and, to the extent applicable, any fees, discounts
or any other compensation payable to underwriters, dealers or agents and any other material terms. Only underwriters so named in
the Prospectus Supplement are deemed to be underwriters in connection with the Securities offered and sold thereby.
If the underwriters purchase Securities
from us as principal, the Securities will be acquired by the underwriters for their own account and may be resold from time to
time in one or more transactions, including negotiated transactions, at a fixed public offering price or at varying prices determined
at the time of sale, at market prices prevailing at the time of sale or at prices related to such prevailing market prices. The
obligations of the underwriters to purchase such Securities as principal will be subject to certain conditions precedent, and the
underwriters will be obligated to purchase all the Securities offered and sold by the Prospectus Supplement if any of such Securities
are purchased. Any public offering price and any discounts or concessions allowed or re-allowed or paid to underwriters, dealers
or agents may be changed from time to time.
The Securities may be sold from time to
time in one or more transactions at a fixed price or prices which may be changed or at market prices prevailing at the time of
sale, at prices related to such prevailing market prices or at negotiated prices, including sales in transactions that are deemed
to be “at-the-market distributions” as defined in National Instrument 44-102 –
Shelf Distributions
, including
sales made directly on the TSX or other existing trading markets for the Common Shares. In the event that we elect to pursue an
“at-the-market distribution” in Canada, we will apply for the required exemptive relief from the applicable securities
commissions or similar regulatory authorities in Canada. The prices at which the Securities may be offered may vary as between
purchasers and during the period of distribution. If, in connection with the offering of Securities at a fixed price or prices,
the underwriters have made a bona fide effort to sell all of the Securities at the initial offering price fixed in the applicable
Prospectus Supplement, the public offering price may be decreased and thereafter further changed, from time to time, to an amount
not greater than the initial public offering price fixed in such Prospectus Supplement, in which case the compensation realized
by the underwriters will be decreased by the amount that the aggregate price paid by purchasers for the Securities is less than
the gross proceeds paid to us by the underwriters. Any such reduction to the public offering price will not affect the net proceeds
received by the Corporation.
The Securities may also be sold directly
by us, pursuant to applicable statutory exemptions, at such prices and upon such terms as agreed to by us and the purchaser or
through one or more agents designated by us from time to time. Any agent involved in the offering and sale of the Securities in
respect of which this Prospectus is delivered will be named, and any commissions payable by us to such agent will be set forth,
in the Prospectus Supplement. Unless otherwise indicated in the Prospectus Supplement, any agent would be acting on a best efforts
basis for the period of its appointment.
We may agree to pay the underwriters a
commission for various services relating to the issue and sale of any Securities offered hereby. Any such commission will be paid
out of our general funds. Underwriters, dealers and agents who participate in the distribution of the Securities may be entitled
under agreements to be entered into with us to indemnification by us against certain liabilities under securities legislation,
or to contribution with respect to payments which such underwriters, dealers or agents may be required to make in respect thereof.
Any offering of Subscription Receipts,
Warrants or Units will be a new issue of securities with no established trading market. Unless otherwise specified in the applicable
Prospectus Supplement, the Subscription Receipts, Warrants or Units will not be listed on any securities exchange. Certain dealers
may make a market in these Securities, but will not be obligated to do so and may discontinue any market making at any time without
notice. No assurance can be given that any dealer will make a market in these Securities or as to the liquidity of the trading
market, if any, for these Securities. See “
Risk Factors
”.
Unless otherwise specified in a Prospectus
Supplement, in connection with any offering of the Securities, the underwriters or agents may over-allot or effect transactions
which stabilize or maintain the market price of the Securities offered at a higher level than that which might exist in the open
market. Such transactions, if commenced, may be interrupted or discontinued at any time.
PRIOR SALES
Information regarding prior sales of Securities
will be provided as required in a Prospectus Supplement with respect to the issuance of Securities pursuant to such Prospectus
Supplement.
TRADING PRICE AND
VOLUME
Information regarding trading price and
volume of the Securities will be provided as required for all of the Corporation’s issued and outstanding Securities that
are listed on any securities exchange, as applicable, in each Prospectus Supplement.
CERTAIN INCOME TAX
CONSIDERATIONS
The applicable Prospectus Supplement may
describe certain Canadian federal income tax consequences which may be applicable to a purchaser of Securities offered thereunder,
and may also include a discussion of certain United States federal income tax consequences to the extent applicable.
LEGAL MATTERS AND
INTEREST OF EXPERTS
Unless otherwise specified in the Prospectus
Supplement relating to an offering and sale of Securities, certain legal matters relating to such offering and sale of Securities
will be passed upon on behalf of the Corporation by McCarthy Tétrault LLP with respect to matters of Canadian law and Dorsey
& Whitney LLP, with respect to matters of U.S. law. In addition, certain legal matters in connection with an offering and sale
of Securities will be passed upon for any underwriters, dealers or agents by counsel to be designated at the time of such offering
and sale by such underwriters, dealers or agents with respect to matters of Canadian and, if applicable, United States or other
foreign law. As at the date hereof, the partners and associates of McCarthy Tétrault LLP, as a group, own less than 1% of
the outstanding securities of the Corporation.
AUDITORS, TRANSFER
AGENT AND REGISTRAR
The auditor of the Corporation is Ernst
& Young LLP, Chartered Professional Accountants, Calgary, Alberta. Ernst & Young LLP has confirmed that it is independent
of the Corporation within the meaning of the relevant rules and related interpretations prescribed by the relevant professional
bodies in Canada and applicable legislation or regulations.
The transfer agent and registrar for the
Common Shares is Computershare Trust Company of Canada at its principal offices located in Calgary, Alberta and Toronto, Ontario.
AGENT FOR SERVICE
OF PROCESS
Messrs. Wayne Pisano, William G. Rice and
Bernd R. Seizinger are directors of the Corporation who reside outside of Canada. Messrs. Pisano, Rice and Seizinger have appointed
the Corporation, at its principal place of business, as agent for service of process. Purchasers are advised that it may not be
possible for investors to enforce judgments obtained in Canada against any person that resides outside of Canada, even if the party
has appointed an agent for service of process.
ENFORCEABILITY OF
CIVIL LIABILITIES AGAINST NON-U.S. PERSONS
The Corporation is a corporation existing
under the
Business Corporations Act
(Alberta). Most of the Corporation’s directors and officers, and some or all
of the experts named in this Prospectus, are residents of Canada or otherwise reside outside the United States, and all or a substantial
portion of their assets, and substantially all of the Corporation’s assets, are located outside the United States. The Corporation
has appointed an agent for service of process in the United States, but it may be difficult for holders of Securities who reside
in the United States to effect service within the United States upon those directors, officers and experts who are not residents
of the United States. It may also be difficult for holders of Securities who reside in the United States to realize in the United
States upon judgments of courts of the United States predicated upon the Corporation’s civil liability and the civil liability
of its directors, officers and experts under the United States federal securities laws.
The Corporation filed with the SEC,
concurrently with its registration statement on Form F-10 of which this Prospectus is a part, an appointment of agent for
service of process on Form F-X. Under the Form F-X, the Corporation appointed DL Services Inc. as its agent for
service of process in the United States in connection with any investigation or administrative proceeding conducted by the SEC,
and any civil suit or action brought against or involving the Corporation in a United States court arising out of or related to
or concerning the offering of the Securities under this Prospectus.
Oncolytics Biotech (NASDAQ:ONCY)
Historical Stock Chart
From Aug 2024 to Sep 2024
Oncolytics Biotech (NASDAQ:ONCY)
Historical Stock Chart
From Sep 2023 to Sep 2024