Ocugen Announces OCU400 Receives Regenerative Medicine Advanced Therapy (RMAT) Designation for Treatment of Retinitis Pigmentosa Associated with RHO Mutations
December 19 2023 - 7:30AM

Ocugen, Inc. (Ocugen or the Company) (NASDAQ: OCGN), a
biotechnology company focused on discovering, developing, and
commercializing novel gene and cell therapies and vaccines, today
announced that the FDA has granted RMAT designation to Ocugen’s
investigational product OCU400 for the treatment of retinitis
pigmentosa (RP) associated with RHO mutations.
“RMAT designation is a significant accomplishment for the OCU400
clinical development program, as it validates the potential for our
game-changing gene therapy approach to fulfill an unmet medical
need for people who are facing blindness due to RP,” said Arun
Upadhyay, PhD, Chief Scientific Officer and Head of R&D at
Ocugen. “FDA’s decision also reinforces the sense of urgency to
bring a therapeutic option to these patients.”
RMAT designation for OCU400 was based on preliminary clinical
data supporting the maintenance and improvement of visual acuity
and function in RP patients in the OCU400 -101 Phase 1/2 clinical
trial as measured by Best Corrected Visual Acuity (BCVA), Low
Luminance Visual Acuity (LLVA), and Multi-Luminance Mobility Test
(MLMT).
RMAT designation is part of the 21st Century Cures Act. The
program was created to expedite the development and review of
regenerative medicine therapies intended to treat, modify, reverse,
or cure a serious condition. Receiving RMAT designation offers
sponsor companies all the benefits of the fast track and
breakthrough therapy designation programs, including early
interactions with the FDA. Ocugen is working with the FDA to
finalize the Phase 3 protocol necessary to advance the clinical
development for OCU400 to support an application for marketing
authorization.
Current data support the gene-agnostic mechanism of action for
OCU400, which suggests that it may be able to provide treatment
benefit to a broader group of RP and Leber congenital amaurosis
(LCA) patients. Ocugen intends to submit additional efficacy and
safety data for OCU400 in RP and LCA patients to the FDA in the
future to potentially expand this RMAT designation to broader RP
and LCA patient populations.
RHO mutations affect more than 10,000 of the 110,000 people in
the United States diagnosed with RP. In the latest clinical study
update from the Phase 1/2 trial of OCU400, 86% (6/7) of RHO
mutation subjects experienced either stabilization or improvement
in MLMT scores from baseline, among which 29% (2/7) demonstrated 3
Lux luminance level improvement. There are currently no treatment
options available for RP patients with RHO gene mutations.
OCU400 represents Ocugen’s modifier gene therapy approach, which
is based on Nuclear Hormone Receptors (NHRs) that regulate diverse
physiological functions, such as homeostasis, reproduction,
development, and metabolism to potentially improve retinal health
and function.
About Ocugen, Inc.Ocugen, Inc. is a
biotechnology company focused on discovering, developing, and
commercializing novel gene and cell therapies and vaccines that
improve health and offer hope for patients across the globe. We are
making an impact on patient’s lives through courageous
innovation—forging new scientific paths that harness our unique
intellectual and human capital. Our breakthrough modifier gene
therapy platform has the potential to treat multiple retinal
diseases with a single product, and we are advancing research in
infectious diseases to support public health and orthopedic
diseases to address unmet medical needs. Discover more
at www.ocugen.com and follow us
on X and LinkedIn.
Cautionary Note on Forward-Looking
StatementsThis press release contains forward-looking
statements within the meaning of The Private Securities Litigation
Reform Act of 1995, including, but not limited to, statements
regarding qualitative assessments of available data, potential
benefits, expectations for ongoing clinical trials, the expected
benefits of RMAT designation, and anticipated regulatory
interactions, which are subject to risks and uncertainties. We may,
in some cases, use terms such as “predicts,” “believes,”
“potential,” “proposed,” “continue,” “estimates,” “anticipates,”
“expects,” “plans,” “intends,” “may,” “could,” “might,” “will,”
“should,” or other words that convey uncertainty of future events
or outcomes to identify these forward-looking statements. Such
statements are subject to numerous important factors, risks, and
uncertainties that may cause actual events or results to differ
materially from our current expectations, including, but not
limited to, the risks that preliminary, interim and top-line
clinical trial results may not be indicative of, and may differ
from, final clinical data; that unfavorable new clinical trial data
may emerge in ongoing clinical trials or through further analyses
of existing clinical trial data; that earlier non-clinical and
clinical data and testing of may not be predictive of the results
or success of later clinical trials; and that that clinical trial
data are subject to differing interpretations and assessments,
including by regulatory authorities. These and other risks and
uncertainties are more fully described in our periodic filings with
the Securities and Exchange Commission (SEC), including the risk
factors described in the section entitled “Risk Factors” in the
quarterly and annual reports that we file with the SEC. Any
forward-looking statements that we make in this press release speak
only as of the date of this press release. Except as required by
law, we assume no obligation to update forward-looking statements
contained in this press release whether as a result of new
information, future events, or otherwise, after the date of this
press release.
Contact:Tiffany HamiltonHead of
CommunicationsIR@ocugen.com
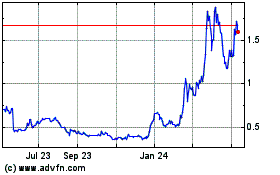
Ocugen (NASDAQ:OCGN)
Historical Stock Chart
From Apr 2024 to May 2024
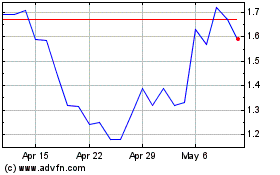
Ocugen (NASDAQ:OCGN)
Historical Stock Chart
From May 2023 to May 2024