Filed pursuant to Rule 424(b)(5)
Registration Statement No. 333-262402
PROSPECTUS SUPPLEMENT
(To Prospectus Dated February 4, 2022)

Lexaria Bioscience Corp.
$5,925,000
Common Stock
We have entered into an equity distribution agreement (the “Sales Agreement”) with Maxim Group LLC (“Maxim” or the “Distribution Agent”), dated August 12, 2022, relating to the sale of shares of our common stock offered by this prospectus supplement and the accompanying prospectus. In accordance with the terms of the Sales Agreement, we may offer and sell shares of our common stock, par value $0.001 per share, having an aggregate offering price of up to $5,925,000 from time to time through the Distribution Agent, acting as agent under this prospectus supplement and accompanying prospectus.
Sales of our common stock, if any, under this prospectus supplement and the accompanying prospectus will be made by any method permitted that is deemed an “at the market offering” as defined in Rule 415 under the Securities Act of 1933, as amended (the “Securities Act”), including sales made directly on or through the NASDAQ Capital Market, the existing trading market for our common stock, sales made to or through a market maker other than on an exchange or otherwise, in negotiated transactions at market prices prevailing at the time of sale or at prices related to such prevailing market prices and/or any other method permitted by law, including in privately negotiated transactions. Maxim will use its commercially reasonable efforts to sell on our behalf all the shares of common stock requested to be sold by us, consistent with its normal trading and sales practices, on mutually agreed terms between Maxim and us. There is no arrangement for funds to be received in any escrow, trust or similar arrangement. We provide more information about how the shares of common stock will be sold in the section entitled “Plan of Distribution.”
The Distribution Agent will be entitled to a commission equal to 3.0% of the gross sales price per share sold. In connection with the sale of shares of common stock on our behalf, the Distribution Agent will be deemed to be an “underwriter” within the meaning of the Securities Act and the compensation of the Distribution Agent will be deemed to be underwriting commissions or discounts. We have also agreed to provide indemnification and contribution to the Distribution Agent with respect to certain liabilities, including liabilities under the Securities Act.
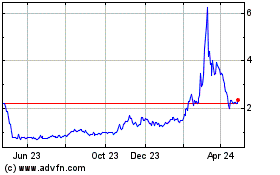
Our common stock and public warrants are listed on the Nasdaq Capital Market under the symbols “LEXX” and “LEXXW”, respectively. The last reported sale prices of our common stock and public warrants on the Nasdaq Capital Market on August 11, 2022 were $3.24 per share and $0.85 per public warrant, respectively.
The aggregate market value of our outstanding common stock held by non-affiliates is $17,776,444.37 based on 5,950,998 shares of outstanding common stock, of which 5,370,527 shares are held by non-affiliates, and a per share price of $3.31, which was the closing sale price of our common stock on the Nasdaq Capital Market on June 28, 2022. During the 12 calendar month period ending August 12, 2022, we have not offered and sold any of our securities pursuant to General Instruction I.B.6 of Form S-3.Pursuant to General Instruction I.B.6 of Form S-3, in no event will we sell securities in public primary offerings on Form S-3 with a value exceeding more than one-third of our public float in any 12 calendar month period so long as our public float remains below $75.0 million.
Investing in our common stock involves risks. See “Risk Factors” beginning on page S-8 of this prospectus supplement and the risks and uncertainties described in the documents we file with the Securities and Exchange Commission that are incorporated in this prospectus supplement by reference for certain risks and uncertainties relating to an investment in our common stock.
Neither the Securities and Exchange Commission, nor any state securities commission, nor any other regulatory body has approved or disapproved of these securities or passed upon the adequacy or accuracy of this prospectus supplement or the accompanying prospectus. Any representation to the contrary is a criminal offense.
Maxim Group LLC
This prospectus supplement is dated August 12, 2022.
TABLE OF CONTENTS
Prospectus Supplement
Prospectus
We have not, and the Distribution Agent has not, authorized anyone to provide you with information different from that contained or incorporated by reference in this prospectus supplement, the accompanying prospectus or any free writing prospectus we prepare or authorize, and neither we nor the Distribution Agent takes any responsibility for any other information that others may give you. This prospectus supplement is not an offer to sell, nor is it a solicitation of an offer to buy, the securities in any jurisdiction where the offer or sale is not permitted. You should not assume that the information contained in this prospectus supplement, the accompanying prospectus or any free writing prospectus is accurate as of any date other than the date on the front cover of those documents, or that the information contained in any document incorporated by reference is accurate as of any date other than the date of the document incorporated by reference, regardless of the time of delivery of this prospectus supplement or any sale of a security. Our business, financial condition, results of operations and prospects may have changed since those dates.
ABOUT THIS PROSPECTUS SUPPLEMENT
This prospectus supplement and the accompanying prospectus relate to the offering of our common stock. Before buying any of the common stock that we are offering, you should carefully read this prospectus supplement, the accompanying prospectus, the information and documents incorporated herein by reference and the additional information under the heading “Where You Can Find Additional Information” and “Incorporation of Certain Information by Reference.” These documents contain important information that you should consider when making your investment decision.
This prospectus supplement and the accompanying prospectus are part of a registration statement on Form S-3 (File No. 333-262402) that we filed with the Securities and Exchange Commission, or SEC, and that was declared effective by the SEC on February 4, 2022. Under this shelf registration process, we may, from time to time, offer common stock, warrants, rights and units, of which this offering is a part.
This document is in two parts. The first part is this prospectus supplement, which describes the terms of this offering of common stock and also adds, updates and changes information contained in the accompanying prospectus and the documents incorporated herein by reference. The second part is the accompanying prospectus, which provides more general information about our common stock and other securities that do not pertain to this offering of common stock. If information in this prospectus supplement is inconsistent with the accompanying prospectus, you should rely on this prospectus supplement. To the extent there is a conflict between the information contained in this prospectus supplement, on the one hand, and the information contained in any document incorporated by reference in this prospectus supplement, on the other hand, you should rely on the information in this prospectus supplement. If any statement in one of these documents is inconsistent with a statement in another document having a later date - for example, a document incorporated by reference into this prospectus supplement - the statement in the document having the later date modifies or supersedes the earlier statement.
You should rely only on the information contained in, or incorporated by reference into this prospectus and in any free writing prospectus that we may authorize for use in connection with this offering. We have not, and the Distribution Agent has not, authorized any other person to provide you with different information. If anyone provides you with different or inconsistent information, you should not rely on it. We are not, and the Distribution Agent is not, making an offer to sell or soliciting an offer to buy our securities in any jurisdiction in which an offer or solicitation is not authorized or in which the person making that offer or solicitation is not qualified to do so or to anyone to whom it is unlawful to make an offer or solicitation. You should assume that the information appearing in this prospectus, the documents incorporated by reference into this prospectus, and in any free writing prospectus that we may authorize for use in connection with this offering, is accurate only as of the date of those respective documents. Our business, financial condition, results of operations and prospects may have changed since those dates. You should read this prospectus, the documents incorporated by reference into this prospectus, and any free writing prospectus that we may authorize for use in connection with this offering, in their entirety before making an investment decision. You should also read and consider the information in the documents to which we have referred you in the sections of this prospectus entitled “Where You Can Find More Information” and “Incorporation of Certain Information by Reference.”
We are offering to sell, and seeking offers to buy, shares of common stock only in jurisdictions where offers and sales are permitted. The distribution of this prospectus and the offering of the common stock in certain jurisdictions may be restricted by law. Persons outside the United States who come into possession of this prospectus must inform themselves about, and observe any restrictions relating to, the offering of the common stock and the distribution of this prospectus outside the United States. This prospectus does not constitute, and may not be used in connection with, an offer to sell, or a solicitation of an offer to buy, any securities offered by this prospectus by any person in any jurisdiction in which it is unlawful for such person to make such an offer or solicitation.
References in this prospectus supplement to “Lexaria,” “we,” “us,” “our,” “our Company,” or “the Company” mean Lexaria Bioscience Corp., a Nevada corporation, and its consolidated subsidiaries, unless we state otherwise or the context indicates otherwise.
CAUTIONARY NOTE REGARDING FORWARD-LOOKING STATEMENTS
This prospectus supplement, the accompanying prospectus and the documents that we incorporate by reference contains forward-looking statements within the meaning of Section 27A of the Securities Act and Section 21E of the Securities Exchange Act of 1934, as amended (the “Exchange Act”). Any statements in this prospectus supplement and the accompanying prospectus about our expectations, beliefs, plans, objectives, assumptions or future events or performance are not historical facts and are forward-looking statements. These statements are often, but not always, made through the use of words or phrases such as “believe,” “will,” “expect,” “anticipate,” “estimate,” “intend,” “plan” and “would.” For example, statements concerning financial condition, possible or assumed future results of operations, growth opportunities, industry ranking, plans and objectives of management, markets for our common stock and future management and organizational structure are all forward-looking statements. Forward-looking statements are not guarantees of performance. They involve known and unknown risks, uncertainties and assumptions that may cause actual results, levels of activity, performance or achievements to differ materially from any results, levels of activity, performance or achievements expressed or implied by any forward-looking statement.
Any forward-looking statements are qualified in their entirety by reference to the risk factors discussed throughout this prospectus supplement and the documents that we incorporate herein by reference. Some of the risks, uncertainties and assumptions that could cause actual results to differ materially from estimates or projections contained in the forward-looking statements include, but are not limited to the following, which may be amplified by the novel coronavirus (COVID-19) pandemic: changes in the market acceptance of our products, increased levels of competition, changes in political, economic or regulatory conditions generally and in the markets in which we operate, our relationships with our key customers, our ability to retain and attract senior management and other key employees, our ability to quickly and effectively respond to new technological developments, our ability to protect our trade secrets or other proprietary rights, operate without infringing upon the proprietary rights of others and prevent others from infringing on our proprietary rights, and other risks and uncertainties discussed in this and our other filings with the SEC.
The foregoing list sets forth some, but not all, of the factors that could affect our ability to achieve results described in any forward-looking statements. You should read this prospectus and any accompanying prospectus supplement and the documents that we reference herein and therein and have filed as exhibits to the registration statement, of which this prospectus is part, completely and with the understanding that our actual future results may be materially different from what we expect. You should assume that the information appearing in this prospectus and any accompanying prospectus supplement is accurate as of the date on the front cover of this prospectus or such prospectus supplement only. Because the risk factors referred to on page S-8 of this prospectus supplement and incorporated herein by reference, could cause actual results or outcomes to differ materially from those expressed in any forward-looking statements made by us or on our behalf, you should not place undue reliance on any forward-looking statements. Further, any forward-looking statement speaks only as of the date on which it is made, and we undertake no obligation to update any forward-looking statement to reflect events or circumstances after the date on which the statement is made or to reflect the occurrence of unanticipated events. New factors emerge from time to time, and it is not possible for us to predict which factors will arise. In addition, we cannot assess the impact of each factor on our business or the extent to which any factor, or combination of factors, may cause actual results to differ materially from those contained in any forward-looking statements. We qualify all of the information presented in this prospectus and any accompanying prospectus supplement, and particularly our forward-looking statements, by these cautionary statements.
PROSPECTUS SUPPLEMENT SUMMARY
This summary highlights selected information about us, this offering and selected information appearing elsewhere in this prospectus supplement, the accompanying prospectus, and in the documents we incorporate by reference therein. This summary is not complete and does not contain all of the information that you should consider before deciding whether to invest in our common stock. You should read this entire prospectus supplement, and the accompanying prospectus, carefully, including the “Risk Factors” section beginning on page S-8 of this prospectus supplement, the “Risk Factors” section of our most recent Annual Report on Form 10-K, the “Risk Factors” section of our most recent Quarterly Reports on Form 10-Q, and our financial statements and the related notes and the other documents incorporated by reference in this prospectus supplement.
Overview
We are a biotechnology company seeking to enhance the bioavailability of a broad variety of active pharmaceutical ingredients (“APIs”) with our DehydraTECHTM drug delivery technology. DehydraTECH combines lipophilic APIs with specific fatty acid and carrier compounds thereby improving the way APIs enter the bloodstream while increasing the effectiveness of fat-soluble active molecules allowing lowering overall dosing and promoting healthier oral ingestion methods. DehydraTECH can be used with a wide variety of APIs encompassing fat-soluble vitamins, hormones, phosphodiesterase inhibitors, antivirals, nicotine and its analogs, and all cannabinoids including tetrahydrocannabinol (“THC”) for a variety of potential therapeutic indications, including hypertension, SARS-CoV-2/COVID-19 and HIV/AIDS. Our technology applies to a host of different ingestible or topically administered product formats including foods, beverages, oral suspensions, tablets, capsules, creams, lotions, and skin patches.
We began filing patents for DehydraTECH in 2014 with two initial US provisional patent application filings by the original inventors Poppy’s Teas LLC, which we acquired by way of exclusive, worldwide license rights and controlling interest in the founding company. We have since increased the number of patent applications to approximately 50 with 26 patents granted worldwide to date. In addition to the US patent filings, we have also pursued international patent protection through filings under the Patent Cooperation Treaty, followed by national filings in over 40 jurisdictions of highest commercial potential thereunder. Our patent family includes intellectual property addressing the manufacturing, formulations, and processing methods used to combine the long chain fatty acids with active pharmaceutical ingredients.
Our patent applications developed from our Research and Development programs (“R&D”) currently include fat-soluble versions of vitamins, nicotine, cannabinoids, hormones, phosphodiesterase inhibitors, and antivirals. 2018 animal studies demonstrated a propensity for DehydraTECH technology to elevate the quantity of drug delivered across the blood-brain-barrier. This expanded our patent applications and opened possibilities for improved delivery of certain central nervous system-targeted drugs that require additional R&D.
In a human clinical study performed in 2018 and published in 2019 in a peer reviewed medical journal, Advances in Therapy titled “Examination of a New Delivery Approach for Oral Cannabidiol in Healthy Subjects: A Randomized, Double-Blinded, Placebo-Controlled Pharmacokinetics Study” available on the PubMed.gov website with the identification of PMID: 31512143, we demonstrated that our technology delivered higher volumes of cannabidiol into the human circulatory system and did so more quickly than a concentration-matched positive control. This same study also demonstrated a statistically significant reduction in human blood pressure from the DehydraTECH processed cannabidiol, versus no statistical reduction in human blood pressure from the positive control.
We operate a Health Canada-licensed laboratory in Canada to conduct basic research and formulation operations, and typically outsource virtually all analytical work to independent third-party laboratories located in Canada, the USA, and Europe. Such third-party evaluation provides independent confirmation of the effects of our technology and processes.
Our formulation and process-oriented operations are primarily conducted in its own laboratory and validated through third-party testing, in preparation for partnering with industry leaders for adoption into their consumer products and/or drugs. Other than for R&D purposes, we do not produce, manufacture, market or distribute drugs.
Although we have experimented with consumer product development in the past, those activities occupy a declining amount of our corporate time. We first began selling trial amounts of ViPova branded black tea fortified with hemp oil and utilizing our technology, in January 2015 and added additional flavours over time.
Beginning in January 2021 we discontinued sales of all consumer products, but our earlier offering of a variety of products helped us to develop final consumer product formulations and understand consumer needs. We continue to be open to the possibility of generating sales from international consumer markets, in those locations where specific products are permissible by law.
ViPova branded products are owned by our wholly owned PoViva Corp. subsidiary. Lexaria Energy, TurboCBD and ChrgD+ branded products are owned 100% by Lexaria Bioscience Corp.
Through our product development we have communicated to the industry the versatility of our technology in specific CPG formats and we believe this strategy has been successful in assisting us in technology licensing discussions with potential new clients. We believe the range of products available and under development are sufficient to prepare for revenue growth and potentially profitable long-term operations if we are able to generate sufficient business clientele demand.
Our business strategy contains an element that we believe will be more impactful to future corporate growth that involves the further development and out-licensing of our intellectual property of molecule delivery that enhances bioactivity or absorption. We have no plans to offer for sale any products containing THC in quantities higher than 0.3%, or indeed any consumer products at all. We have discontinued all direct business activities related to non-FDA-approved uses of THC, including our former business practice of licensing our technology to businesses that were legally state-licensed to offer THC products. We also plan to license our technology to other companies for the delivery of molecules other than THC or cannabinoids, such as nicotine which we have licensed to Altria Ventures Inc., an indirect wholly owned subsidiary of Altria Group, Inc. Our October 31, 2017, announcement of the USPTO Notice of Allowance for our first patent granted and the subsequent granted patents of our technology in the US and in many other countries around the world related to new molecule groups, along with our ongoing patent filing and grants, may enhance our ability to successfully pursue our licensing initiatives during fiscal 2023.
We continue to communicate the benefits of our technology to potential licensing partners; i.e. with higher absorption levels a manufacturer could perhaps infuse smaller amounts of active molecules into a product, potentially reducing their manufacturing input costs; to provide higher bioavailability with the dosing limits being imposed or contemplated in many jurisdictions; to infuse beverages while masking the flavor and smell of the active molecules; and to reduce delivery times to the bloodstream. We believe these to be meaningful competitive advantages that may lead to the potential to generate licensing revenue, and will pursue these opportunities within the cannabinoids, nicotine, and other bioactive molecular markets both within the USA and also internationally, in those locations where they are legal and regulated by government.
Subject to budgetary availability, we also plan to conduct additional in vitro and in vivo studies testing the absorption of many API’s - CBD, PDE5 inhibitors, antiviral drugs, nicotine, and others- to substantiate the effectiveness of our technology. More than simply satisfying scientific curiosity, successful tests could lead to increased awareness and acceptance of our technology as a meaningful method by which to deliver some or all of the named molecules more effectively than their current delivery methods. Therefore, absorption tests could become an important element leading towards higher rates of acceptance of our technology licensing initiatives.
We will pursue technology licensing opportunities as a method of generating highly profitable revenue streams over long periods of time. In addition, while nine of our US patents and nine of our Australian patents have been granted to date, we now have received granted patents in the European Union, Japan, India and Mexico, and have multiple other applications filed in the US and around the world. It is not possible to forecast with certainty when, or if, our remaining patents pending will become granted patents. But if our remaining patent applications do become granted patents, our ability to generate meaningful license revenue from our intellectual property may increase from multiple jurisdictions outside of the US.
We will continue to pursue our remaining patents pending as vigorously as we are able, since the successful granting of more of those applications could lead to material increases in shareholder value. We are pursuing patent protection in more than 40 countries around the world.
Corporate Information
Our mailing address is #100–740 McCurdy Road, Kelowna, British Columbia, V1X 2P7, Canada. Our telephone number is (250) 765-6424 and our website address is www.lexariabioscience.com. The information on our website is not part of, and should not be construed as being incorporated by reference into, this prospectus supplement and the accompanying prospectus.
THE OFFERING
| Common stock offered pursuant to this prospectus supplement | | Shares of our common stock having an aggregate offering price of up to $5,925,000. |
| | | |
| Common stock to be outstanding after this offering (1) | | Up to 7,779,701 shares, after giving effect to the assumed sale of 1,828,703 of shares of our common stock at a price of $3.24 per share, which was the closing price of our common stock on the Nasdaq Capital Market on August 11, 2022. The actual number of shares issued will vary depending on the price at which shares may be sold from time to time during this offering. |
| | | |
| Manner of offering | | “At the market offering” that may be made from time to time on the Nasdaq Capital Market or other market for our common stock in the United States through our agent, Maxim Group LLC. See the section entitled “Plan of Distribution” on page S-12 of this prospectus supplement. |
| | | |
| Use of proceeds | | We intend to use the net proceeds from this offering for working capital and general corporate purposes including, but not limited to, research and development studies and the patent and legal costs associated thereto. We will retain broad discretion over the use of the net proceeds from the sale of the securities offered hereby. See “Use of Proceeds” on page S-10 of this prospectus supplement for additional information. |
| | | |
| Nasdaq Capital Market symbol | | “LEXX” |
| | | |
| Risk factors | | See “Risk Factors” beginning on page S-8 of this prospectus supplement and the other information included in, or incorporated by reference into, this prospectus supplement and the accompanying prospectus for a discussion of certain factors you should carefully consider before deciding to invest in shares of our common stock, including the risk factors discussed in the sections entitled “Risk Factors” contained in our most recent Annual Report on Form 10-K, most recent Quarterly Reports on Form 10-Q, and our other filings with the SEC. |
| (1) | Based on 5,950,998 shares of common stock outstanding as of August 12, 2022, and excludes: |
| | ● | 321,336 shares of our common stock issuable upon exercise of outstanding options (of which 293,836 were exercisable as of May 31, 2022) at a weighted average exercise price of $6.80 per share; |
| | |
| | ● | 273,764 shares of our common stock reserved for potential future issuance pursuant to our equity incentive plan; and |
| | | |
| | ● | 2,421,983 shares of our common stock issuable upon exercise of outstanding warrants at a weighted average exercise price of $8.04 per share. |
RISK FACTORS
An investment in our common stock involves a high degree of risk. Prior to making a decision about investing in our common stock, you should carefully consider the risk factors described below and the risk factors discussed in the sections entitled “Risk Factors” contained in our most recent Annual Report on Form 10-K, most recent Quarterly Reports on Form 10-Q, and our other filings with the SEC and incorporated by reference in this prospectus supplement, together with all of the other information contained in this prospectus supplement and the accompanying prospectus. Additional risks and uncertainties not presently known to us, or that we currently view as immaterial, may also impair our business. If any of the risks or uncertainties described in our SEC filings or this prospectus supplement and the accompanying prospectus or any additional risks and uncertainties actually occur, our business, financial condition and results of operations could be materially and adversely affected. In that case, the trading price of our common stock could decline and you might lose all or part of your investment.
Risks Related to the Company and our Business
Unfavorable U.S. or global economic conditions could adversely affect our business, financial condition, or results of operations.
Our results of operations could be adversely affected by general conditions in the global economy and financial markets, including by the current COVID-19 pandemic, recent geopolitical events, unfavorable changes related to interest rates and rising inflation. The most recent global financial crisis caused extreme volatility and disruptions in the capital and credit markets. A severe or prolonged economic downturn, such as the most recent global financial crisis, could result in a variety of risks to our business, including weakened demand for our products and our ability to raise additional capital when needed on favorable terms, if at all. A weak or declining economy could strain our suppliers, possibly resulting in supply disruption, or cause delays in payments for our services by third-party payors or our collaborators. Any of the foregoing could harm our business and we cannot anticipate all of the ways in which the current economic climate and financial market conditions could adversely impact our business.
Risks Related to this Offering
Our management will have broad discretion over the use of the net proceeds from this offering, you may not agree with how we use the proceeds, and the proceeds may not be invested successfully.
Our management will have broad discretion as to the use of the net proceeds from any offering by us and could use them for purposes other than those contemplated at the time of this offering. Accordingly, you will be relying on the judgment of our management with regard to the use of these net proceeds, and you will not have the opportunity, as part of your investment decision, to assess whether the proceeds are being used appropriately. It is possible that the proceeds will be invested in a way that does not yield a favorable, or any, return for us.
You may experience immediate and substantial dilution in the net tangible book value per share of the common stock you purchase in the offering.
The offering price per share in this offering may exceed the net tangible book value per share of our common stock outstanding prior to this offering. Assuming that an aggregate of 1,828,703 shares of our common stock are sold at a price of $3.24 per share, the last reported sale price of our common stock on the Nasdaq Capital Market on August 11, 2022, for aggregate gross proceeds of $5,925,000, and after deducting commissions and estimated aggregate offering expenses payable by us, you will experience immediate dilution of $1.50 per share, representing the difference between our net tangible book value per share, as adjusted, as of May 31, 2022, after giving effect to this offering and the assumed offering price. The exercise of outstanding stock options or warrants could result in further dilution of your investment. See the section below entitled “Dilution” for a more detailed illustration of the dilution you would incur if you participate in this offering.
You may experience future dilution as a result of future equity offerings.
In order to raise additional capital, we may in the future offer additional shares of our common stock or other securities convertible into or exchangeable for our common stock at prices that may not be the same as the price per share in this offering. We may sell shares or other securities in any other offering at a price per share that is less than the price per share paid by any investors in this offering, and investors purchasing shares or other securities in the future could have rights superior to existing stockholders. The price per share at which we sell additional shares of our common stock, or securities convertible or exchangeable into common stock, in future transactions may be higher or lower than the price per share paid by any investors in this offering.
Sales of a substantial number of our shares of common stock in the public markets, or the perception that such sales could occur, could cause our stock price to fall.
We may issue and sell additional shares of commons stock in the public markets, including during this offering. As a result, a substantial number of our shares of common stock may be sold in the public market. Sales of a substantial number of our shares of common stock in the public markets, including during this offering, or the perception that such sales could occur, could depress the market price of our common stock and impair our ability to raise capital through the sale of additional equity securities.
Because we do not currently intend to declare cash dividends on our shares of common stock in the foreseeable future, stockholders must rely on appreciation of the value of our common stock for any return on their investment.
We have never paid cash dividends on our common stock and do not plan to pay any cash dividends in the near future. We currently intend to retain all of our future earnings, if any, to finance the operation, development and growth of our business. Furthermore, any future debt agreements may also preclude us from paying or place restrictions on our ability to pay dividends. As a result, capital appreciation, if any, of our common stock will be your sole source of gain with respect to your investment for the foreseeable future.
The exercise of our outstanding options and warrants will dilute stockholders and could decrease our stock price.
The exercise of our outstanding options and warrants may adversely affect our stock price due to sales of a large number of shares or the perception that such sales could occur. These factors also could make it more difficult to raise funds through future offerings of our securities, and could adversely impact the terms under which we could obtain additional equity capital. Exercise of outstanding options and warrants or any future issuance of additional shares of common stock or other equity securities, including but not limited to options, warrants, restricted stock units or other derivative securities convertible into our common stock, may result in significant dilution to our stockholders and may decrease our stock price.
The common stock offered hereby will be sold in “at-the-market” offerings, and investors who buy shares at different times will likely pay different prices.
Investors who purchase shares in this offering at different times will likely pay different prices, and so may experience different outcomes in their investment results. We will have discretion, subject to market demand, to vary the timing, prices, and numbers of shares sold, and there is no minimum or maximum sales price. Investors may experience a decline in the value of their shares as a result of share sales made at prices lower than the prices they paid.
The actual number of shares we will issue under the Sales Agreement, at any one time or in total, is uncertain.
Subject to certain limitations in the Sales Agreement and compliance with applicable law, we have the discretion to deliver a sales notice to the Distribution Agent at any time throughout the term of the Sales Agreement. The number of shares that are sold by the Distribution Agent after delivering a sales notice will fluctuate based on the market price of the common stock during the sales period and limits we set with Maxim. Because the price per share of each share sold will fluctuate based on the market price of our common stock during the sales period, it is not possible at this stage to predict the number of shares that will be ultimately issued.
USE OF PROCEEDS
We may issue and sell shares of our common stock having aggregate gross sales proceeds of up to $5,925,000 from time to time. The amount of proceeds we receive, if any, will depend on the actual number of shares of our common stock sold and the market price at which such shares are sold. There can be no assurance that we will be able to sell any shares or fully utilize the Sales Agreement as a source of financing. Because there is no minimum offering amount required as a condition to close this offering, the actual total public offering amount, commissions and proceeds to us, if any, are not determinable at this time.
We intend to use the net proceeds from this offering, if any, for working capital and general corporate purposes including, but not limited to, research and development studies and the patent and legal costs associated thereto. The precise amount and timing of the application of such proceeds will depend upon our funding requirements and the availability and cost of other capital. As of the date of this prospectus supplement, we cannot specify with certainty all of the particular uses for the net proceeds to us from this offering. Accordingly, we will retain broad discretion over the use of the net proceeds from the sale of the securities offered hereby. Pending any specific application, we may initially invest funds in short-term marketable securities.
DILUTION
If you invest in this offering, your ownership interest will be diluted to the extent of the difference between the public offering price per share and the as adjusted net tangible book value per share after giving effect to this offering.
We calculate net tangible book value per share by dividing the net tangible book value, which is tangible assets less total liabilities, by the number of outstanding shares of our common stock. Dilution represents the difference between the amount per share paid by purchasers of shares in this offering and the as-adjusted net tangible book value per share of our common stock immediately after giving effect to this offering. Our net tangible book value as of May 31, 2022 was approximately $8,428,079, or $1.42 per share.
After giving effect to the sale of our common stock pursuant to this prospectus supplement and the accompanying prospectus in the aggregate amount of $5,925,000 at an assumed offering price of $3.24 per share, the last reported sale price of our common stock on the Nasdaq Capital Market on August 11, 2022, and after deducting commissions and estimated aggregate offering expenses payable by us, our net tangible book value, as adjusted, as of May 31, 2022 would have been approximately $14,112,079, or $1.81 per share of common stock. This represents an immediate increase in $5,684,000 net tangible book value of $0.40 per share to our existing stockholders and an immediate dilution in net tangible book value of $1.50 per share to new investors.
The following table illustrates this per share dilution:
| Assumed public offering price per share | | $ | 3.24 | |
| Net tangible book value per share as of May 31, 2022 | | $ | 1.42 | |
| Increase in net tangible book value per share attributable to new investors purchasing our common stock in this offering | | $ | 0.40 | |
| As adjusted net tangible book value per share after giving effect to this offering | | $ | 1.81 | |
| Dilution per share to investors purchasing our common stock in this offering | | $ | 1.50 | |
The table above assumes for illustrative purposes that an aggregate of 1,828,703 shares of our common stock are sold pursuant to this prospectus supplement and the accompanying prospectus at a price of $3.24 per share, the last reported sale price of our common stock on the Nasdaq Capital Market on August 11, 2022, for aggregate gross proceeds of $5,925,000. The shares sold in this offering, if any, will be sold from time to time at various prices. An increase of $1.00 per share in the price at which the shares are sold from the assumed offering price of $3.24 per share shown in the table above, assuming all of our common stock in the aggregate amount of $5,925,000 is sold at that price, would result in a net tangible book value per share, as adjusted, after the offering of $2.08 per share and would increase the dilution in net tangible book value per share to new investors in this offering to $2.16 per share, after deducting commissions and estimated aggregate offering expenses payable by us. A decrease of $1.00 per share in the price at which the shares are sold from the assumed offering price of $3.24 per share shown in the table above, assuming all of our common stock in the aggregate amount of $5,925,000 is sold at that price, would result in a net tangible book value per share, as adjusted, after the offering of $1.61 per share and would decrease the dilution in net tangible book value per share to new investors in this offering to $0.63 per share, after deducting commissions and estimated aggregate offering expenses payable by us.
The above discussion and table are based on 5,950,998 shares of our common stock outstanding as of May 31, 2022 and excludes the following, all as of May 31, 2022:
| | ● | 321,336 shares of our common stock issuable upon exercise of outstanding options (of which 293,836 were exercisable as of May 31, 2022) at a weighted average exercise price of $6.80 per share; |
| | | |
| | ● | 273,764 shares of our common stock reserved for potential future issuance pursuant to our equity incentive plan; and |
| | | |
| | ● | 2,421,983 shares of our common stock issuable upon exercise of outstanding warrants at a weighted average exercise price of $8.04 per share. |
To the extent that options or warrants are exercised, there may be further dilution to new investors.
PLAN OF DISTRIBUTION
We have entered into the Sales Agreement with Maxim as the Distribution Agent, dated August 12, 2022, under which we may issue and sell shares of our common stock from time to time through the Distribution Agent, subject to certain limitations, pursuant to this prospectus supplement and the accompanying prospectus. Upon delivery of a placement notice and subject to the terms and conditions of the Sales Agreement, Maxim may sell shares of our common stock by any method permitted by law deemed to be an “at-the-market” equity offering as defined in Rule 415 promulgated under the Securities Act, including sales made directly on or through the Nasdaq Capital Market, the existing trading market for our common stock, sales made to or through a market maker other than on an exchange or otherwise, in negotiated transactions at market prices prevailing at the time of sale or at prices related to such prevailing market prices, and/or any other method permitted by law, including in privately negotiated transactions.
Under the terms of the Sales Agreement, in no event will the Company issue or sell through the Distribution Agent such number or dollar amount of shares of common stock that would (i) exceed the number or dollar amount of shares of common stock registered and available on the Registration Statement, (ii) exceed the number of authorized but unissued shares of common stock, (iii) exceed the number or dollar amount of shares of Common Stock permitted to be sold under Form S-3 (including General Instruction I.B.6 thereof, if applicable), or (iv) exceed the number or dollar amount of common stock for which the Company has filed a prospectus supplement to the Registration Statement.
Each time we wish to issue and sell common stock under the Sales Agreement, we will notify the Distribution Agent of the number of shares to be issued, the dates on which such sales are anticipated to be made, any minimum price below which sales may not be made and other sales parameters as we deem appropriate. Once we have so instructed the Distribution Agent, unless the Distribution Agent declines to accept the terms of the notice, the Distribution Agent has agreed to use its commercially reasonable efforts consistent with its normal trading and sales practices to sell such shares up to the amount specified on such terms. The obligations of the Distribution Agent under the Sales Agreement to sell our common stock are subject to a number of conditions that we must satisfy.
We will pay the Distribution Agent commissions for its services in acting as agent in the sale of our common stock. The Distribution Agent will be entitled to a commission equal to 3.0% of the gross sales price per share sold. Because there is no minimum offering amount required as a condition to closing this offering, the actual total public offering amount, commissions and proceeds to us, if any, are not determinable at this time. In addition, we have agreed to reimburse the Distribution Agent for fees and disbursements related to its legal counsel in an amount not to exceed $43,000. We have also agreed to reimburse the Distribution Agents for fees and disbursements related to its legal counsel for each quarter for which a bringdown is required an amount not to exceed $3,500. We estimate that the total expenses for the offering, excluding compensation payable to the Distribution Agent under the terms of the Sales Agreement, will be approximately $241,000.
Settlement for sales of our common stock will occur on the second business day following the date on which any sales are made, or on such earlier day as is then industry practice for regular-way trading, or on some other date that is agreed upon by us and the Distribution Agent in connection with a particular transaction, in return for payment of the net proceeds to us. There is no arrangement for funds to be received in an escrow, trust or similar arrangement.
In connection with the sale of the common stock on our behalf, the Distribution Agent will be deemed to be an “underwriter” within the meaning of the Securities Act, and the compensation of the Distribution Agent will be deemed to be underwriting commissions or discounts. We have agreed to provide indemnification and contribution to the Distribution Agent against certain civil liabilities, including liabilities under the Securities Act.
The offering pursuant to the Sales Agreement will terminate upon the earlier of (i) the issuance and sale of all shares of our common stock subject to the Sales Agreement, or (ii) the termination of the Sales Agreement as permitted therein.
This summary of the material provisions of the Sales Agreement does not purport to be a complete statement of its terms and conditions. A copy of the Sales Agreement is filed with the SEC as an exhibit to a Current Report on Form 8-K filed on August 12, 2022. See “Where You Can Find Additional Information” below.
The Distribution Agent and its respective affiliates may in the future provide various investment banking and other financial services for us and our affiliates, for which services they may in the future receive customary fees. To the extent required by Regulation M, the Distribution Agent will not engage in any market making or stabilizing activities involving our common stock while the offering is ongoing under this prospectus supplement and the accompanying prospectus.
LEGAL MATTERS
Sichenzia Ross Ference LLP, New York, New York, has passed upon the validity of the common stock offered hereby. Ellenoff Grossman & Schole LLP is counsel for the Distribution Agent in connection with this offering.
EXPERTS
The audited consolidated financial statements of the Company and its subsidiaries, as of and for the years ended August 31, 2021 and 2020, included in this prospectus have been so included in reliance upon the report of Davidson & Company LLP, independent registered public accountants, upon the authority of said firm as experts in accounting and auditing.
WHERE YOU CAN FIND ADDITIONAL INFORMATION
We are subject to the reporting requirements of the Exchange Act and file annual, quarterly and current reports, proxy statements and other information with the SEC. You may read and copy these reports, proxy statements and other information at the SEC’s public reference facilities at 100 F Street, N.E., Room 1580, Washington, D.C. 20549. You can request copies of these documents by writing to the SEC and paying a fee for the copying cost. Please call the SEC at 1-800-SEC-0330 for more information about the operation of the public reference facilities. SEC filings are also available at the SEC’s website at www.sec.gov.
This prospectus supplement and the accompanying prospectus form part of a registration statement on Form S-3 filed by us with the SEC under the Securities Act. As permitted by the SEC, this prospectus supplement and the accompanying prospectus do not contain all the information in the registration statement filed with the SEC. For a more complete understanding of this offering, you should refer to the complete registration statement, including the exhibits thereto, on Form S-3 that may be obtained as described above. Statements contained or incorporated by reference in this prospectus supplement and the accompanying prospectus about the contents of any contract or other document are not necessarily complete. If we have filed any contract or other document as an exhibit to the registration statement or any other document incorporated by reference in the registration statement of which this prospectus supplement forms a part, you should read the exhibit for a more complete understanding of the document or matter involved. Each statement regarding a contract or other document is qualified in its entirety by reference to the actual document.
We also maintain a web site at www.lexariabioscience.com through which you can access our SEC filings. The information set forth on our web site is not part of this prospectus supplement or the accompanying prospectus.
INCORPORATION OF CERTAIN INFORMATION BY REFERENCE
We have filed a registration statement on Form S-3 with the SEC under the Securities Act. This prospectus is part of the registration statement but the registration statement includes and incorporates by reference additional information and exhibits. The SEC permits us to “incorporate by reference” the information contained in documents we file with the SEC, which means that we can disclose important information to you by referring you to those documents rather than by including them in this prospectus. Information that is incorporated by reference is considered to be part of this prospectus and you should read it with the same care that you read this prospectus. Information that we file later with the SEC will automatically update and supersede the information that is either contained, or incorporated by reference, in this prospectus, and will be considered to be a part of this prospectus from the date those documents are filed. We have filed with the SEC, and incorporate by reference in this prospectus:
1. The Company’s Annual Report on Form 10-K for the year ended August 31, 2021;
2. The Company’s Quarterly Reports for the quarters ended November 30, 2021, February 28, 2022, and May 31, 2022;
3. The Company’s Current Reports on Form 8-K filed on September 8, 2021, November 10, 2021, December 16, 2021, January 4, 2022, June 1, 2022, June 3, 2022, June 9, 2022, June 22, 2022 and August 12, 2022;
4. The Company’s definitive Proxy Statement on Schedule 14A, filed on April 13, 2022;
5. The Company’s Form 8-A12B filed on January 11, 2021; and
6. The Company’s Form 8-A12G filed on July 14, 2006.
We also incorporate by reference all additional documents that we file with the SEC under the terms of Sections 13(a), 13(c), 14 or 15(d) of the Exchange Act that are made after the date of the initial registration statement but prior to effectiveness of the registration statement and after the date of this prospectus but prior to the termination of the offering of the securities covered by this prospectus. We are not, however, incorporating, in each case, any documents or information that we are deemed to furnish and not file in accordance with SEC rules.
You may request, and we will provide you with, a copy of these filings, at no cost, by calling us at (250) 765-6424 or by writing to us at the following address:
Lexaria Bioscience Corp.
#100 – 740 McCurdy Road
Kelowna, British Columbia V1X 2P7
Canada
Attn: Chief Executive Officer
PROSPECTUS

Lexaria Bioscience Corp.
$50,000,000
Common Stock
Warrants
Rights
Units
From time to time, we may offer and sell up to $50,000,000 in aggregate of the securities described in this prospectus separately or together in any combination, in one or more classes or series, in amounts, at prices and on terms that we will determine at the time of the offering.
This prospectus provides a general description of the securities we may offer. We may provide specific terms of securities to be offered in one or more supplements to this prospectus. We may also provide a specific plan of distribution for any securities to be offered in a prospectus supplement. Prospectus supplements may also add, update or change information in this prospectus. You should carefully read this prospectus and the applicable prospectus supplement, together with any documents incorporated by reference herein, before you invest in our securities.
Our common stock and public warrants are listed on the Nasdaq Capital Market under the symbols “LEXX” and “LEXXW”, respectively. The last reported sale prices of our common stock and public warrants on the Nasdaq Capital Market on January 27, 2022 were $4.17 per share and $1.26 per public warrant, respectively. The aggregate market value of our outstanding common stock held by non-affiliates is $22,436,589.09 based on 5,950,998 shares of outstanding common stock, of which 570,521 shares are held by affiliates, and a per share price of $4.17, which was the closing sale price of our common stock as quoted on the NASDAQ Capital Market on January 27, 2022. During the 12 calendar month period that ends on, and includes, the date of this prospectus, we have not offered and sold any of our securities pursuant to General Instruction I.B.6 of Form S-3.
Investing in any of our securities involves a high degree of risk. Please read carefully the section entitled “Risk Factors” on page 8 of this prospectus, the “Risk Factors” section contained in the applicable prospectus supplement and the information included and incorporated by reference in this prospectus.
Neither the Securities and Exchange Commission nor any state securities commission has approved or disapproved of these securities or determined if this prospectus is truthful or complete. Any representation to the contrary is a criminal offense.
The date of this prospectus is February 4, 2022.
TABLE OF CONTENTS
ABOUT THIS PROSPECTUS
This prospectus is part of a registration statement on Form S-3 that we filed with the Securities and Exchange Commission, or the SEC, using a “shelf” registration or continuous offering process. Under this shelf registration process, we may, from time to time, sell any combination of the securities described in this prospectus in one or more offerings up to a total aggregate offering price of $50,000,000.
This prospectus provides a general description of the securities we may offer. We may provide specific terms of securities to be offered in one or more supplements to this prospectus. We may also provide a specific plan of distribution for any securities to be offered in a prospectus supplement. Prospectus supplements may also add, update or change information in this prospectus. If the information varies between this prospectus and the accompanying prospectus supplement, you should rely on the information in the accompanying prospectus supplement.
Before purchasing any securities, you should carefully read both this prospectus and any prospectus supplement, together with the additional information described under the heading “Information We Incorporate by Reference.” You should rely only on the information contained or incorporated by reference in this prospectus, any prospectus supplement and any free writing prospectus prepared by or on behalf of us or to which we have referred you. Neither we nor any underwriters have authorized any other person to provide you with different information. If anyone provides you with different or inconsistent information, you should not rely on it. We take no responsibility for, and can provide no assurance as to the reliability of, any other information that others may give you. You should assume that the information contained in this prospectus, any prospectus supplement or any free writing prospectus is accurate only as of the date on its respective cover, and that any information incorporated by reference is accurate only as of the date of the document incorporated by reference, unless we indicate otherwise. Our business, financial condition, results of operations and prospects may have changed since those dates. This prospectus contains summaries of certain provisions contained in some of the documents described herein, but reference is made to the actual documents for complete information. All of the summaries are qualified in their entirety by the actual documents. Copies of some of the documents referred to herein have been filed, will be filed or will be incorporated by reference as exhibits to the registration statement of which this prospectus is a part, and you may obtain copies of those documents as described below under the heading “Where You Can Find More Information.”
This prospectus and any applicable prospectus supplement do not constitute an offer to sell or the solicitation of an offer to buy any securities other than the registered securities to which they relate. We are not making offers to sell common stock or any other securities described in this prospectus in any jurisdiction in which an offer or solicitation is not authorized or in which we are not qualified to do so or to anyone to whom it is unlawful to make an offer or solicitation.
Unless otherwise expressly indicated or the context otherwise requires, we use the terms “Lexaria,” the “Company,” “we,” “us,” “our” or similar references to refer to Lexaria Bioscience Corp. and its subsidiaries.
WHERE YOU CAN FIND MORE INFORMATION
We have filed our registration statement on Form S-3 with the SEC under the Securities Act of 1933, as amended, or the Securities Act. We also file annual, quarterly and current reports, proxy statements and other information with the SEC. You may read and copy any document that we file with the SEC, including the registration statement and the exhibits to the registration statement, at the SEC’s Public Reference Room located at 100 F Street, N.E., Washington D.C. 20549. You may obtain further information on the operation of the Public Reference Room by calling the SEC at 1-800-SEC-0330. Our SEC filings are also available to the public at the SEC’s web site at www.sec.gov. These documents may also be accessed on our web site at www.lexariabioscience.com. Information contained on our web site is not incorporated by reference into this prospectus and you should not consider information contained on our web site to be part of this prospectus.
This prospectus and any prospectus supplement are part of a registration statement filed with the SEC and do not contain all of the information in the registration statement. The full registration statement may be obtained from the SEC or us as indicated above. Other documents establishing the terms of the offered securities are filed as exhibits to the registration statement or will be filed through an amendment to our registration statement on Form S-3 or under cover of a Current Report on Form 8-K and incorporated into this prospectus by reference.
INFORMATION WE INCORPORATE BY REFERENCE
The SEC allows us to “incorporate by reference” into this prospectus the information we file with it, which means that we can disclose important information to you by referring you to those documents. The information incorporated by reference is considered to be part of this prospectus. Any statement contained herein or in a document incorporated or deemed to be incorporated by reference into this document will be deemed to be modified or superseded for purposes of the document to the extent that a statement contained in this document or any other subsequently filed document that is deemed to be incorporated by reference into this document modifies or supersedes the statement. We incorporate by reference in this prospectus the following information (other than, in each case, documents or information deemed to have been furnished and not filed in accordance with SEC rules):
| | • | our Annual Report on Form 10-K for the fiscal year ended August 31, 2021 filed with the SEC on November 29, 2021; |
| | • | our Quarterly Report on Form 10-Q for the fiscal quarter ended November 30, 2021 filed with the SEC on January 14, 2022; |
| | • | our Current Reports on Form 8-K filed with the SEC on November 10, 2021, December 16, 2021, and January 4, 2022. |
| | • | our Form 8-A12B, filed with the SEC on January 11, 2021; and |
| | • | our Form 8-A12G, filed with the SEC on July 14, 2006. |
We also incorporate by reference each of the documents that we file with the SEC under Sections 13(a), 13(c), 14 or 15(d) of the Securities Exchange Act of 1934, as amended, or the Exchange Act, (i) after the date of this prospectus and prior to effectiveness of this registration statement on Form S-3 and (ii) on or after the date of this prospectus and prior to the termination of the offerings under this prospectus and any prospectus supplement. These documents include periodic reports, such as Annual Reports on Form 10-K, Quarterly Reports on Form 10-Q and Current Reports on Form 8-K, as well as proxy statements. We will not, however, incorporate by reference in this prospectus any documents or portions thereof that are not deemed “filed” with the SEC, including any information furnished pursuant to Item 2.02 or Item 7.01 of our Current Reports on Form 8-K after the date of this prospectus unless, and except to the extent, specified in such Current Reports.
We will provide to each person, including any beneficial owner, to whom a prospectus (or a notice of registration in lieu thereof) is delivered a copy of any of these filings (other than an exhibit to these filings, unless the exhibit is specifically incorporated by reference as an exhibit to this prospectus) at no cost, upon a request to us by writing or telephoning us at the following address and telephone number:
Lexaria Bioscience Corp.
#100–740 McCurdy Road, Kelowna
British Columbia, Canada V1X 2P7
1-250-765-6424
SPECIAL NOTE REGARDING FORWARD-LOOKING STATEMENTS
This prospectus, including the documents incorporated by reference herein, may contain or incorporate “forward-looking statements” within the meaning of Section 21E of the Securities Exchange Act of 1934. In this context, forward-looking statements mean statements related to future events, may address our expected future business and financial performance, and often contain words such as “expects”, “anticipates”, “intends”, “plans”, “believes”, “will”, “should”, “could”, “would” or “may” and other words of similar meaning. Forward-looking statements by their nature address matters that are, to different degrees, uncertain. For us, particular risks and uncertainties that could cause our actual future results to differ materially from those expressed in our forward-looking statements include but are not limited to the following, which may be amplified by the novel coronavirus (COVID-19) pandemic: changes in the market acceptance of our products, increased levels of competition, changes in political, economic or regulatory conditions generally and in the markets in which we operate, our relationships with our key customers, our ability to retain and attract senior management and other key employees, our ability to quickly and effectively respond to new technological developments, our ability to protect our trade secrets or other proprietary rights, operate without infringing upon the proprietary rights of others and prevent others from infringing on our proprietary rights, and other risks and uncertainties discussed in this and our other filings with the SEC.
These factors may not constitute all factors that could cause actual results to differ from those discussed in any forward-looking statement. All written and oral forward-looking statements attributable to us, or persons acting on our behalf, are expressly qualified in their entirety by the cautionary statements disclosed under “Item 1A. Risk Factors,” in our Annual Report on Form 10-K for the year ended August 31, 2021, as such risk factors may be amended, supplemented or superseded from time to time by other reports we file with the SEC, including subsequent Annual Reports on Form 10-K and Quarterly Reports on Form 10-Q, and in any prospectus supplement. Accordingly, forward-looking statements should be not be relied upon as a predictor of actual results. Readers are urged to carefully review and consider the various disclosures made in this prospectus and in our other filings with the SEC that attempt to advise interested parties of the risks and factors that may affect our business. We do not undertake to update our forward-looking statements to reflect events or circumstances that may arise after the date of this prospectus, except as required by law.
Lexaria Bioscience Corp.
We are a biotechnology company seeking to enhance the bioavailability of a broad variety of active pharmaceutical ingredients (“APIs”) with our DehydraTECHTM drug delivery technology. DehydraTECH combines lipophilic APIs with specific fatty acid and carrier compounds thereby improving the way APIs enter the bloodstream while increasing the effectiveness of fat-soluble active molecules allowing lowering overall dosing and promoting healthier oral ingestion methods. DehydraTECH can be used with a wide variety of APIs encompassing fat-soluble vitamins, non-steroidal anti-inflammatory drugs (“NSAIDs”) pain medications, hormones, phosphodiesterase inhibitors, antivirals, nicotine and its analogs, and all cannabinoids including tetrahydrocannabinol (“THC”) for a variety of potential therapeutic indications, including hypertension, SARS-CoV-2/COVID-19 and HIV/AIDS. Our technology applies to a host of different ingestible or topically administered product formats including foods, beverages, oral suspensions, tablets, capsules, creams, lotions, and skin patches.
We began filing patents for DehydraTECH in 2014 with two initial US provisional patent application filings by the original inventors Poppy’s Teas LLC, which we acquired by way of exclusive, worldwide license rights and controlling interest in the founding company. We have since increased the number of patent applications to approximately 60 with 23 patents granted worldwide to date. In addition to the US patent filings, we have also pursued international patent protection through filings under the Patent Cooperation Treaty, followed by national filings in over 40 jurisdictions of highest commercial potential thereunder. Our patent family includes intellectual property addressing the manufacturing, formulations, and processing methods used to combine the long chain fatty acids with active pharmaceutical ingredients.
Our patent applications developed from our Research and Development programs (“R&D”) currently include fat-soluble versions of vitamins, nicotine, cannabinoids, hormones, phosphodiesterase inhibitors, and antivirals. 2018 animal studies demonstrated a propensity for DehydraTECH technology to elevate the quantity of drug delivered across the blood-brain-barrier. This expanded our patent applications and opened possibilities for improved delivery of certain central nervous system-targeted drugs that require additional R&D.
In a human clinical study performed in 2018 and published in 2019 in a peer reviewed medical journal, Advances in Therapy titled “Examination of a New Delivery Approach for Oral Cannabidiol in Healthy Subjects: A Randomized, Double-Blinded, Placebo-Controlled Pharmacokinetics Study” available on the PubMed.gov website with the identification of PMID: 31512143, we demonstrated that our technology delivered higher volumes of cannabidiol into the human circulatory system and did so more quickly than a concentration-matched positive control. This same study also demonstrated a statistically significant reduction in human blood pressure from the DehydraTECH processed cannabidiol, versus no statistical reduction in human blood pressure from the positive control.
We operate a Health Canada-licensed laboratory in Canada to conduct basic research and formulation operations, and typically outsource virtually all analytical work to independent third-party laboratories located in Canada, the USA, and Europe. Such third-party evaluation provides independent confirmation of the effects of our technology and processes.
Our formulation and process-oriented operations are primarily conducted in its own laboratory and validated through third-party testing, in preparation for partnering with industry leaders for adoption into their consumer products and/or drugs. Other than for R&D purposes, we do not produce, manufacture, market or distribute drugs.
Although we have experimented with consumer product development in the past, those activities occupy a declining amount of our corporate time. We first began selling trial amounts of ViPova branded black tea fortified with hemp oil and utilizing our technology, in January 2015 and added additional flavours over time.
We also began offering our first coffee and hot chocolate also fortified with full spectrum hemp oil, and also under the ViPova brand. Beginning in January 2021 we discontinued sales of all consumer products, but our earlier offering of a variety of products helped us to develop final consumer product formulations and understand consumer needs.
Generating meaningful revenue from consumer product sales was challenging and we were unable to achieve widespread retail distribution. We continue to be open to the possibility of generating sales from international markets, in those locations where hemp oil fortified foods are permissible by law.
ViPova branded products are owned by our wholly owned PoViva Corp. subsidiary. Lexaria Energy, TurboCBD and ChrgD+ branded products are owned 100% by Lexaria Bioscience Corp.
Through our product development we have communicated to the industry the versatility of our technology in specific CPG formats and we believe this strategy has been successful in assisting us in technology licensing discussions with potential new clients. We believe the range of products available and under development are sufficient to prepare for revenue growth and potentially profitable long-term operations if we are able to generate sufficient business clientele demand.
Our business strategy contains an element that we believe will be more impactful to future corporate growth that involves the further development and out-licensing of our intellectual property of molecule delivery that enhances bioactivity or absorption. We have no plans to offer for sale any products containing THC in quantities higher than 0.3%. We have discontinued all direct business activities related to non-FDA-approved uses of THC, including our former business practice of licensing our technology to businesses that were legally state-licensed to offer THC products. We also plan to license our technology to other companies for the delivery of molecules other than THC or cannabinoids, such as nicotine which we have licensed to Altria Ventures Inc., an indirect wholly owned subsidiary of Altria Group, Inc. Our October 31, 2017, announcement of the USPTO Notice of Allowance for our first patent granted and the subsequent granted patents of our technology in the US and in many other countries around the world related to new molecule groups, along with our ongoing patent filing and grants, may enhance our ability to successfully pursue our licensing initiatives during fiscal 2022.
We continue to communicate the benefits of our technology to potential licensing partners; i.e. with higher absorption levels a manufacturer could perhaps infuse smaller amounts of active molecules into a product, potentially reducing their manufacturing input costs; to provide higher bioavailability with the dosing limits being imposed or contemplated in many jurisdictions; to infuse beverages while masking the flavor and smell of the active molecules; and to reduce delivery times to the bloodstream. We believe these to be meaningful competitive advantages that may lead to the potential to generate licensing revenue, and will pursue these opportunities within the cannabinoids, nicotine, and other bioactive molecular markets both within the USA and also internationally, in those locations where they are legal and regulated by government.
Subject to budgetary availability, we also plan to conduct additional in vitro and in vivo studies testing the absorption of many API’s - CBD, PDE5 inhibitors, antiviral drugs, nicotine, and others- to substantiate the effectiveness of our technology. More than simply satisfying scientific curiosity, successful tests could lead to increased awareness and acceptance of our technology as a meaningful method by which to deliver some or all of the named molecules more effectively than their current delivery methods. Therefore, absorption tests could become an important element leading towards higher rates of acceptance of our technology licensing initiatives.
We will pursue technology licensing opportunities as a method of generating highly profitable revenue streams over long periods of time. In addition, while nine of our US patents and eight of our Australian patents have been granted to date, we now have received granted patents in the European Union, Japan, India and Mexico, and have multiple other applications filed in the US and around the world. It is not possible to forecast with certainty when, or if, our remaining patents pending will become granted patents. But if our remaining patent applications do become granted patents, our ability to generate meaningful license revenue from our intellectual property may increase from multiple jurisdictions outside of the US.
We will continue to pursue our remaining patents pending as vigorously as we are able, since the successful granting of more of those applications could lead to material increases in shareholder value. We are pursuing patent protection in more than 40 countries around the world.
RISK FACTORS
Investing in our securities involves a high degree of risk. Before making an investment decision, you should carefully consider any risk factors set forth in the applicable prospectus supplement and the documents incorporated by reference in this prospectus, including the factors discussed under the heading “Risk Factors” in our most recent Annual Report on Form 10-K and each subsequently filed Quarterly Report on Form 10-Q and any risk factors set forth in our other filings with the SEC pursuant to Sections 13(a), 13(c), 14 or 15(d) of the Securities Exchange. See “Where You Can Find More Information” and “Information We Incorporate By Reference.” Each of the risks described in these documents could materially and adversely affect our business, financial condition, results of operations and prospects, and could result in a partial or complete loss of your investment. Additional risks and uncertainties not presently known to us, or that we currently deem immaterial, may also adversely affect our business. In addition, past financial performance may not be a reliable indicator of future performance and historical trends should not be used to anticipate results or trends in future periods.
USE OF PROCEEDS
We will retain broad discretion over the use of the net proceeds from the sale of the securities offered hereby. Unless otherwise specified in any prospectus supplement, we currently intend to use the net proceeds from the sale of our securities offered under this prospectus for working capital and general corporate purposes including, but not limited to, research and development studies and the patent and legal costs associated thereto, potential repurchase of certain of our issued shares and for general working capital purposes. Pending any specific application, we may initially invest funds in short-term marketable securities or apply them to the reduction of indebtedness.
DESCRIPTION OF CAPITAL STOCK
The following information describes the common stock, par value $0.001 per share, of the Company, as well as certain provisions of our articles of incorporation, as amended (the “Articles of Incorporation”) and our amended and restated bylaws (“Bylaws”). This description is only a summary. You should also refer to our Articles of Incorporation and Bylaws, which have been filed with the SEC as exhibits to the registration statement of which this prospectus forms a part.
Authorized and Outstanding Capital Stock
Our authorized capital stock consists of 220,000,000 shares of common stock, par value $0.001 per share. As of January 27, 2022, there were 5,950,998 shares of our common stock outstanding.
Common Stock
We are authorized to issue up to a total of 220,000,000 shares of common stock, par value $0.001 per share. Holders of our common stock are entitled to one vote for each share held on all matters submitted to a vote of our stockholders. Holders of our common stock have no cumulative voting rights. Further, holders of our common stock have no pre-emptive or conversion rights or other subscription rights. Upon our liquidation, dissolution or winding-up, holders of our common stock are entitled to share in all assets remaining after payment of all liabilities. As well, holders of our common stock are entitled to receive dividends, if any, as may be declared from time to time by our Board out of our assets which are legally available. Such dividends, if any, are payable in cash, in property or in shares of capital stock.
Holders of shares of our common stock are required to cast at least 33.33% of the total votes entitled to be cast by the holders of all of our outstanding capital stock, present in person or by proxy, in order to constitute a quorum at any meeting. If a quorum is present, an action by stockholders entitled to vote on a matter is approved if the number of votes cast in favor of the action exceeds the number of votes cast in opposition to the action. The vote of 33.33% of our stock held by shareholders present in person or represented by proxy and entitled to vote at the Meeting will be sufficient to elect Directors or to approve a proposal.
Our common stock is traded on the Nasdaq Capital Market under the symbol “LEXX”. The transfer agent and registrar for our common stock is Computershare Trust Company of Canada.
Anti-Takeover Provisions of Nevada State Law
Certain anti-takeover provisions of Nevada law could have the effect of delaying or preventing a third-party from acquiring us, even if the acquisition arguably could benefit our stockholders.
Nevada’s “combinations with interested stockholders” statutes, NRS 78.411 through 78.444, inclusive, prohibit specified types of business “combinations” between certain Nevada corporations and any person deemed to be an “interested stockholder” for two years after such person first becomes an “interested stockholder” unless the corporation’s board of directors approves the combination, or the transaction by which such person becomes an “interested stockholder”, in advance, or unless the combination is approved by the board of directors and sixty percent of the corporation’s voting power not beneficially owned by the interested stockholder, its affiliates and associates. Further, in the absence of prior approval certain restrictions may apply even after such two year period. However, these statutes do not apply to any combination of a corporation and an interested stockholder after the expiration of four years after the person first became an interested stockholder. For purposes of these statutes, an “interested stockholder” is any person who is (1) the beneficial owner, directly or indirectly, of ten percent or more of the voting power of the outstanding voting shares of the corporation, or (2) an affiliate or associate of the corporation and at any time within the two previous years was the beneficial owner, directly or indirectly, of ten percent or more of the voting power of the then outstanding shares of the corporation. The definition of the term “combination” is sufficiently broad to cover most significant transactions between a corporation and an “interested stockholder.” These statutes generally apply to Nevada corporations with 200 or more stockholders of record. However, a Nevada corporation may elect in its articles of incorporation not to be governed by these particular laws, but if such election is not made in the corporation’s original articles of incorporation, the amendment (1) must be approved by the affirmative vote of the holders of stock representing a majority of the outstanding voting power of the corporation not beneficially owned by interested stockholders or their affiliates and associates, and (2) is not effective until 18 months after the vote approving the amendment and does not apply to any combination with a person who first became an interested stockholder on or before the effective date of the amendment. We have made such an election in our original articles of incorporation.
Nevada’s “acquisition of controlling interest” statutes, NRS 78.378 through 78.379, inclusive, contain provisions governing the acquisition of a controlling interest in certain Nevada corporations. These “control share” laws provide generally that any person that acquires a “controlling interest” in certain Nevada corporations may be denied voting rights, unless a majority of the disinterested stockholders of the corporation elects to restore such voting rights. Absent such provision in our bylaws, these laws would apply to us as of a particular date if we were to have 200 or more stockholders of record (at least 100 of whom have addresses in Nevada appearing on our stock ledger at all times during the 90 days immediately preceding that date) and do business in the State of Nevada directly or through an affiliated corporation, unless our articles of incorporation or bylaws in effect on the tenth day after the acquisition of a controlling interest provide otherwise. These laws provide that a person acquires a “controlling interest” whenever a person acquires shares of a subject corporation that, but for the application of these provisions of the NRS, would enable that person to exercise (1) one fifth or more, but less than one third, (2) one third or more, but less than a majority or (3) a majority or more, of all of the voting power of the corporation in the election of directors. Once an acquirer crosses one of these thresholds, shares which it acquired in the transaction taking it over the threshold and within the 90 days immediately preceding the date when the acquiring person acquired or offered to acquire a controlling interest become “control shares” to which the voting restrictions described above apply.
Nevada law also provides that directors may resist a change or potential change in control if the directors determine that the change is opposed to, or not in the best interests of, the corporation. The existence of the foregoing provisions and other potential anti-takeover measures could limit the price that investors might be willing to pay in the future for shares of our common stock. They could also deter potential acquirers of our Company, thereby reducing the likelihood that you could receive a premium for your common stock in an acquisition.
Anti-Takeover Effects of Our Articles of Incorporation and Bylaws
The following provisions of our Articles of Incorporation and Bylaws could have the effect of delaying or discouraging another party from acquiring control of us and could encourage persons seeking to acquire control of us to first negotiate with our Board:
| | • | no cumulative voting in the election of directors, which limits the ability of minority stockholders to elect director candidates; |
| | | |
| | • | the right of our Board to elect a director to fill a vacancy created by the expansion of the Board or the resignation, death or removal of a director, with our stockholders only allowed to fill such a vacancy if not filled by the Board; |
| | | |
| | • | the ability of our Board to alter our Bylaws without obtaining shareholder approval; and |
| | | |
| | • | the requirement that a special meeting of stockholders may be called only by either the President or the Secretary by resolution of the Board of Directors or at the request of stockholders owning a majority of the issued and outstanding capital stock of the Company entitled to vote, which may delay the ability of our stockholders to force consideration of a proposal or to take action, including the removal of directors |
DESCRIPTION OF WARRANTS
General
The following description, together with the additional information we include in any applicable prospectus supplement, summarizes the material terms and provisions of the warrants that we may offer under this prospectus, which consist of warrants to purchase shares of common stock. Warrants may be offered independently or together with shares of common stock by any prospectus supplement and may be attached to or separate from those securities.
While the terms we have summarized below will generally apply to any future warrants we may offer under this prospectus, we will describe the particular terms of any warrants that we may offer in more detail in the applicable prospectus supplement. The specific terms of any warrants may differ from the description provided below as a result of negotiations with third parties in connection with the issuance of those warrants, as well as for other reasons. Because the terms of any warrants we offer under a prospectus supplement may differ from the terms we describe below, you should rely solely on information in the applicable prospectus supplement if that summary is different from the summary in this prospectus.
We will issue the warrants under a warrant agreement, which we will enter into with a warrant agent to be selected by us. We use the term “warrant agreement” to refer to any of these warrant agreements. We use the term “warrant agent” to refer to the warrant agent under any of these warrant agreements. The warrant agent will act solely as an agent of ours in connection with the warrants and will not act as an agent for the holders or beneficial owners of the warrants.
We will incorporate by reference into the registration statement of which this prospectus is a part the form of warrant agreement, including a form of warrant certificate, that describes the terms of the series of warrants we are offering before the issuance of the related series of warrants. The following summaries of material provisions of the warrants and the warrant agreements are subject to, and qualified in their entirety by reference to, all the provisions of the warrant agreement applicable to a particular series of warrants. We urge you to read any applicable prospectus supplement related to the warrants that we sell under this prospectus, as well as the complete warrant agreement that contain the terms of the warrants and defines your rights as a warrant holder.
We will describe in the applicable prospectus supplement the terms relating to a series of warrants. If warrants for the purchase of shares of common stock are offered, the prospectus supplement will describe the following terms, to the extent applicable:
| | · | the offering price and the aggregate number of warrants offered; |
| | | |
| | · | the total number of shares that can be purchased if a holder of the warrants exercises them; |
| | | |
| | · | the number of warrants being offered with each share of common stock; |
| | | |
| | · | the date on and after which the holder of the warrants can transfer them separately from the related shares of common stock; |
| | | |
| | · | the number of shares of common stock that can be purchased if a holder exercises the warrant and the price at which those shares may be purchased upon exercise, including, if applicable, any provisions for changes to or adjustments in the exercise price and in the securities or other property receivable upon exercise; |
| | | |
| | · | the terms of any rights to redeem or call, or accelerate the expiration of, the warrants; |
| | | |
| | · | the date on which the right to exercise the warrants begins and the date on which that right expires; |
| | | |
| | · | federal income tax consequences of holding or exercising the warrants; and |
| | | |
| | · | any other specific terms, preferences, rights or limitations of, or restrictions on, the warrants. |
Warrants for the purchase of shares of common stock will be in registered form only.
A holder of warrant certificates may exchange them for new certificates of different denominations, present them for registration of transfer and exercise them at the corporate trust office of the warrant agent or any other office indicated in the applicable prospectus supplement. Until any warrants to purchase shares of common stock are exercised, holders of the warrants will not have any rights of holders of the underlying shares of common stock, including any rights to receive dividends or to exercise any voting rights, except to the extent set forth under “Warrant Adjustments” below.
Exercise of Warrants
Each holder of a warrant is entitled to purchase the number of shares of common stock, as the case may be, at the exercise price described in the applicable prospectus supplement. After the close of business on the day when the right to exercise terminates (or a later date if we extend the time for exercise), unexercised warrants will become void.
A holder of warrants may exercise them by following the general procedure outlined below:
| | · | deliver to the warrant agent the payment required by the applicable prospectus supplement to purchase the underlying security; |
| | | |
| | · | properly complete and sign the reverse side of the warrant certificate representing the warrants; and |
| | | |
| | · | deliver the warrant certificate representing the warrants to the warrant agent within five business days of the warrant agent receiving payment of the exercise price. |
If you comply with the procedures described above, your warrants will be considered to have been exercised when the warrant agent receives payment of the exercise price, subject to the transfer books for the securities issuable upon exercise of the warrant not being closed on such date. After you have completed those procedures and subject to the foregoing, we will, as soon as practicable, issue and deliver to you the shares of common stock that you purchased upon exercise. If you exercise fewer than all of the warrants represented by a warrant certificate, a new warrant certificate will be issued to you for the unexercised amount of warrants. Holders of warrants will be required to pay any tax or governmental charge that may be imposed in connection with transferring the underlying securities in connection with the exercise of the warrants.
Amendments and Supplements to the Warrant Agreements
We may amend or supplement a warrant agreement without the consent of the holders of the applicable warrants to cure ambiguities in the warrant agreement, to cure or correct a defective provision in the warrant agreement, or to provide for other matters under the warrant agreement that we and the warrant agent deem necessary or desirable, so long as, in each case, such amendments or supplements do not materially adversely affect the interests of the holders of the warrants.
Warrant Adjustments
Unless the applicable prospectus supplement states otherwise, the exercise price of, and the number of securities covered by, a warrant for shares of common stock will be adjusted proportionately if we subdivide or combine our common stock, as applicable. In addition, unless the prospectus supplement states otherwise, if we, without payment:
| | · | pay any cash to all or substantially all holders of our common stock, other than a cash dividend paid out of our current or retained earnings; |
| | | |
| | · | issue any evidence of our indebtedness or rights to subscribe for or purchase our indebtedness to all or substantially all holders of our common stock; or |
| | | |
| | · | issue common stock or additional shares or other securities or property to all or substantially all holders of our common stock by way of spinoff, split-up, reclassification, combination of shares or similar corporate rearrangement; |
then the holders of common stock warrants will be entitled to receive upon exercise of the warrants, in addition to the securities otherwise receivable upon exercise of the warrants and without paying any additional consideration, the amount of shares and other securities and property such holders would have been entitled to receive had they held the common stock issuable under the warrants on the dates on which holders of those securities received or became entitled to receive such additional shares and other securities and property.
Except as stated above, the exercise price and number of securities covered by a warrant for shares of common stock, and the amounts of other securities or property to be received, if any, upon exercise of those warrants, will not be adjusted or provided for if we issue those securities or any securities convertible into or exchangeable for those securities, or securities carrying the right to purchase those securities or securities convertible into or exchangeable for those securities.
Holders of common stock warrants may have additional rights under the following circumstances:
| | · | certain reclassifications, capital reorganizations or changes of the common stock; |
| | | |
| | · | certain share exchanges, mergers, or similar transactions involving us that result in changes of the common stock; or |
| | | |
| | · | certain sales or dispositions to another entity of all or substantially all of our property and assets. |
If one of the above transactions occurs and holders of our common stock are entitled to receive shares, securities or other property with respect to or in exchange for their securities, the holders of the common stock warrants then-outstanding, as applicable, will be entitled to receive upon exercise of their warrants the kind and amount of shares and other securities or property that they would have received upon the applicable transaction if they had exercised their warrants immediately before the transaction.
DESCRIPTION OF RIGHTS
The following description, together with the additional information we include in any applicable prospectus supplement, summarizes the general features of the rights that we may offer under this prospectus. We may issue rights to our stockholders to purchase shares of our common stock and/or any of the other securities offered hereby. Each series of rights will be issued under a separate rights agreement to be entered into between us and a bank or trust company, as rights agent. When we issue rights, we will provide the specific terms of the rights and the applicable rights agreement in a prospectus supplement. Because the terms of any rights we offer under a prospectus supplement may differ from the terms we describe below, you should rely solely on information in the applicable prospectus supplement if that summary is different from the summary in this prospectus. We will incorporate by reference into the registration statement of which this prospectus is a part, the form of rights agreement that describes the terms of the series of rights we are offering before the issuance of the related series of rights. The applicable prospectus supplement relating to any rights will describe the terms of the offered rights, including, where applicable, the following:
| | · | the date for determining the persons entitled to participate in the rights distribution; |
| | | |
| | · | the exercise price for the rights; |
| | | |
| | · | the aggregate number or amount of underlying securities purchasable upon exercise of the rights; |
| | | |
| | · | the number of rights issued to each stockholder and the number of rights outstanding, if any; |
| | | |
| | · | the extent to which the rights are transferable; |
| | | |
| | · | the date on which the right to exercise the rights will commence and the date on which the right will expire; |
| | | |
| | · | the extent to which the rights include an over-subscription privilege with respect to unsubscribed securities; |
| | | |
| | · | anti-dilution provisions of the rights, if any; and |
| | | |
| | · | any other terms of the rights, including terms, procedures and limitations relating to the distribution, exchange and exercise of the rights. |
Holders may exercise rights as described in the applicable prospectus supplement. Upon receipt of payment and the rights certificate properly completed and duly executed at the corporate trust office of the rights agent or any other office indicated in the prospectus supplement, we will, as soon as practicable, forward the securities purchasable upon exercise of the rights. If less than all of the rights issued in any rights offering are exercised, we may offer any unsubscribed securities directly to persons other than stockholders, to or through agents, underwriters or dealers or through a combination of such methods, including pursuant to standby underwriting arrangements, as described in the applicable prospectus supplement.
DESCRIPTION OF UNITS
We may issue units comprising two or more securities described in this prospectus in any combination. For example, we might issue units consisting of a combination of common stock and warrants to purchase common stock. The following description sets forth certain general terms and provisions of the units that we may offer pursuant to this prospectus. The particular terms of the units and the extent, if any, to which the general terms and provisions may apply to the units so offered will be described in the applicable prospectus supplement.
Each unit will be issued so that the holder of the unit also is the holder of each security included in the unit. Thus, the unit will have the rights and obligations of a holder of each included security. Units will be issued pursuant to the terms of a unit agreement, which may provide that the securities included in the unit may not be held or transferred separately at any time or at any time before a specified date. A copy of the forms of the unit agreement and the unit certificate relating to any particular issue of units will be filed with the SEC each time we issue units, and you should read those documents for provisions that may be important to you. For more information on how you can obtain copies of the forms of the unit agreement and the related unit certificate, see “Where You Can Find More Information.”
The prospectus supplement relating to any particular issuance of units will describe the terms of those units, including, to the extent applicable, the following:
| | · | the designation and terms of the units and the securities comprising the units, including whether and under what circumstances those securities may be held or transferred separately; |
| | | |
| | · | any provision for the issuance, payment, settlement, transfer or exchange of the units or of the securities comprising the units; and |
| | | |
| | · | whether the units will be issued in fully registered or global form. |
PLAN OF DISTRIBUTION
We may sell the securities from time to time, by a variety of methods, including the following:
| | · | on any national securities exchange or quotation service on which our securities may be listed at the time of sale, including the Nasdaq Capital Market; |
| | | |
| | · | in the over-the-counter market; |
| | | |
| | · | in transactions otherwise than on such exchange or in the over-the-counter market, which may include privately negotiated transactions and sales directly to one or more purchasers; |
| | | |
| | · | through ordinary brokerage transactions and transactions in which the broker-dealer solicits purchasers; |
| | | |
| | · | through purchases by a broker-dealer as principal and resale by the broker-dealer for its account; |
| | | |
| | · | through underwriters, broker-dealers, agents, in privately negotiated transactions, or any combination of these methods; |
| | | |
| | · | through the writing or settlement of options or other hedging transactions, whether through an options exchange or otherwise; |
| | | |
| | · | a combination of any of these methods; or |
| | | |
| | · | by any other method permitted pursuant to applicable law. |
The securities may be distributed from time to time in one or more transactions:
| | · | at a fixed price or prices, which may be changed; |
| | | |
| | · | at market prices prevailing at the time of sale; |
| | | |
| | · | at prices related to such prevailing market prices; or |
| | | |
| | · | at negotiated prices. |
Offers to purchase the securities being offered by this prospectus may be solicited directly. Agents may also be designated to solicit offers to purchase the securities from time to time. Any agent involved in the offer or sale of our securities will be identified in a prospectus supplement.
If a dealer is utilized in the sale of the securities being offered by this prospectus, the securities will be sold to the dealer, as principal. The dealer may then resell the securities to the public at varying prices to be determined by the dealer at the time of resale.
If an underwriter is utilized in the sale of the securities being offered by this prospectus, an underwriting agreement will be executed with the underwriter at the time of sale and the name of any underwriter will be provided in the prospectus supplement that the underwriter will use to make resales of the securities to the public. In connection with the sale of the securities, we, or the purchasers of securities for whom the underwriter may act as agent, may compensate the underwriter in the form of underwriting discounts or commissions. The underwriter may sell the securities to or through dealers, and those dealers may receive compensation in the form of discounts, concessions or commissions from the underwriters and/or commissions from the purchasers for which they may act as agent. Unless otherwise indicated in a prospectus supplement, an agent will be acting on a best efforts basis and a dealer will purchase securities as a principal, and may then resell the securities at varying prices to be determined by the dealer.
Any compensation paid to underwriters, dealers or agents in connection with the offering of the securities, and any discounts, concessions or commissions allowed by underwriters to participating dealers will be provided in the applicable prospectus supplement. Underwriters, dealers and agents participating in the distribution of the securities may be deemed to be underwriters within the meaning of the Securities Act, and any discounts and commissions received by them and any profit realized by them on resale of the securities may be deemed to be underwriting discounts and commissions. In compliance with the guidelines of the Financial Industry Regulatory Authority, Inc., or FINRA, the maximum amount of underwriting compensation, including underwriting discounts and commissions, to be paid in connection with any offering of securities pursuant to this prospectus may not exceed 8% of the aggregate principal amount of securities offered. We may enter into agreements to indemnify underwriters, dealers and agents against civil liabilities, including liabilities under the Securities Act, or to contribute to payments they may be required to make in respect thereof and to reimburse those persons for certain expenses. The securities may or may not be listed on a national securities exchange. To facilitate the offering of securities, certain persons participating in the offering may engage in transactions that stabilize, maintain or otherwise affect the price of the securities. This may include over-allotments or short sales of the securities, which involve the sale by persons participating in the offering of more securities than were sold to them. In these circumstances, these persons would cover such over-allotments or short positions by making purchases in the open market or by exercising their over-allotment option, if any. In addition, these persons may stabilize or maintain the price of the securities by bidding for or purchasing securities in the open market or by imposing penalty bids, whereby selling concessions allowed to dealers participating in the offering may be reclaimed if securities sold by them are repurchased in connection with stabilization transactions. The effect of these transactions may be to stabilize or maintain the market price of the securities at a level above that which might otherwise prevail in the open market. These transactions may be discontinued at any time.
If indicated in the applicable prospectus supplement, underwriters or other persons acting as agents may be authorized to solicit offers by institutions or other suitable purchasers to purchase the securities at the public offering price set forth in the prospectus supplement, pursuant to delayed delivery contracts providing for payment and delivery on the date or dates stated in the prospectus supplement. These purchasers may include, among others, commercial and savings banks, insurance companies, pension funds, investment companies and educational and charitable institutions. Delayed delivery contracts will be subject to the condition that the purchase of the securities covered by the delayed delivery contracts will not at the time of delivery be prohibited under the laws of any jurisdiction in the United States to which the purchaser is subject. The underwriters and agents will not have any responsibility with respect to the validity or performance of these contracts.
We may engage in at-the-market offerings into an existing trading market in accordance with rule 415(a)(4) under the Securities Act. In addition, we may enter into derivative transactions with third parties, or sell securities not covered by this prospectus to third parties in privately negotiated transactions. If the applicable prospectus supplement so indicates, in connection with those derivatives, the third parties may sell securities covered by this prospectus and the applicable prospectus supplement, including in short sale transactions. If so, the third party may use securities pledged by us, or borrowed from us or others to settle those sales or to close out any related open borrowings of common stock, and may use securities received from us in settlement of those derivatives to close out any related open borrowings of our common stock. In addition, we may loan or pledge securities to a financial institution or other third party that in turn may sell the securities using this prospectus and an applicable prospectus supplement. Such financial institution or other third party may transfer its economic short position to investors in our securities or in connection with a concurrent offering of other securities.
The underwriters, dealers and agents may engage in transactions with us, or perform services for us, in the ordinary course of business for which they receive compensation.
LEGAL MATTERS
Unless otherwise indicated in the applicable prospectus supplement, certain legal matters in connection with the offering and the validity of the securities offered by this prospectus, and any supplement thereto, will be passed upon by Sichenzia Ross Ference LLP.
EXPERTS
The audited consolidated financial statements of the Company and its subsidiaries, as of and for the years ended August 31, 2021 and 2020 included in this prospectus have been so included in reliance upon the report of Davidson & Company LLP, independent registered public accountants, upon the authority of said firm as experts in accounting and auditing.

UP TO $5,925,000 SHARES OF COMMON STOCK
Maxim Group LLC
August 12, 2022
Lexaria Bioscience (NASDAQ:LEXX)
Historical Stock Chart
From Aug 2024 to Sep 2024
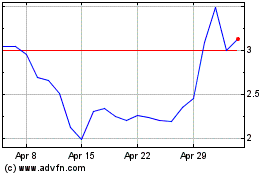
Lexaria Bioscience (NASDAQ:LEXX)
Historical Stock Chart
From Sep 2023 to Sep 2024