Hologic Announces CE Mark of the Genius™ Digital Diagnostics System for Cervical Cancer Screening
November 17 2020 - 2:00AM
Business Wire
Powerful artificial intelligence combined with
digital imaging designed to transform screening
Hologic, Inc. (Nasdaq: HOLX) announced today that its new
Genius™ Digital Diagnostics System is now CE marked in Europe.
Genius Digital Diagnostics is the first digital cytology platform
to combine a new artificial intelligence (AI) algorithm with
advanced digital imaging to help cytotechnologists and pathologists
identify pre-cancerous lesions and cancer cells in women.
This press release features multimedia. View
the full release here:
https://www.businesswire.com/news/home/20201116006076/en/
Genius™ Digital Diagnostics System for
Cervical Cancer Screening (Photo: Business Wire)
For more than 30 years, Hologic has developed and brought to
market new advances in cervical cancer screening, including the
first liquid-based cytology technology, the ThinPrep® Pap Test, and
the first FDA-approved mRNA-based HPV test, the Aptima® HPV Assay.
Hologic is redefining cervical cancer screening yet again by
introducing Genius Digital Diagnostics for laboratories. The system
can rapidly analyze all cells on a ThinPrep Pap test slide,
narrowing tens of thousands of cells down to a gallery view of the
most diagnostically relevant images. This will help provide
healthcare providers with the critical information they need to
guide earlier detection and better treatment decisions for the
patients they serve.
“Application of AI requires digital images that are of
exceptional quality,” said Kevin Thornal, president, Diagnostic
Solutions Division at Hologic. “Our teams developed a breakthrough
imaging technology that converts physical glass cytology slides
into digital images with superior clarity. From this
digitalization, advanced image analysis and improved
standardization are now achievable.”
Genius Digital Diagnostics enables a more seamless and dynamic
collaboration across laboratories within a network, connecting
pathologists with remote review so each patient can benefit from
the collective knowledge of geographically dispersed experts when
needed. Digital case review promises to enhance the experience for
lab partners by improving workflow and accelerating review
time.
“As a part of our development process, we collaborated with
cytotechnologists and pathologists to ensure we were developing a
new system to meet their needs,” said Michael Quick, vice
president, R&D and Innovation at Hologic. “We’re always
innovating so we can help our lab partners detect more diseases and
save more lives every day.”
Hologic now offers the first CE-marked comprehensive cervical
cancer screening portfolio from sample collection to digital
diagnosis. Genius Digital Diagnostics consists of a digital imager
for image acquisition, an AI algorithm for analyzing images, an
image management server for storing images, and a review station
for case review. The complete system is scalable, designed to fit
the present and future needs of laboratories.
About Hologic
Hologic, Inc. is an innovative medical technology company
primarily focused on improving women’s health and well-being
through early detection and treatment. For more information on
Hologic, visit www.hologic.com.
Forward-Looking Statements
This press release may contain forward-looking information that
involves risks and uncertainties, including statements about the
use of Hologic’s Genius™ Digital Diagnostics System. There can be
no assurance these products will achieve the benefits described
herein or that such benefits will be replicated in any particular
manner with respect to an individual patient. The actual effect of
the use of the products can only be determined on a case-by-case
basis depending on the particular circumstances and patient in
question. In addition, there can be no assurance that these
products will be commercially successful or achieve any expected
level of sales. Hologic expressly disclaims any obligation or
undertaking to release publicly any updates or revisions to any
such statements presented herein to reflect any change in
expectations or any change in events, conditions or circumstances
on which any such statements are based.
Hologic, Genius Digital Diagnostics, ThinPrep, Aptima and The
Science of Sure are registered trademarks of Hologic, Inc. in the
United States and/or other countries.
SOURCE: Hologic, Inc.
View source
version on businesswire.com: https://www.businesswire.com/news/home/20201116006076/en/
Investor Contact Michael Watts Vice President, Investor
Relations and Corporate Communications (858) 410-8588
michael.watts@hologic.com
Media Contact Jane Mazur Vice President, Divisional
Communications (585) 355-5978 jane.mazur@hologic.com
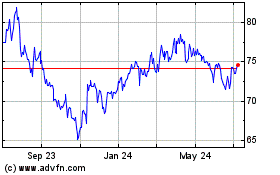
Hologic (NASDAQ:HOLX)
Historical Stock Chart
From Aug 2024 to Sep 2024
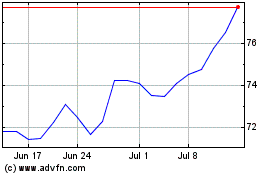
Hologic (NASDAQ:HOLX)
Historical Stock Chart
From Sep 2023 to Sep 2024