UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
Form 6-K
REPORT OF FOREIGN PRIVATE ISSUER PURSUANT TO RULE 13a-16 OR 15d-16 UNDER THE SECURITIES EXCHANGE ACT OF 1934
For the month of August 2023
Commission File Number: 001-39950
Evaxion Biotech
(Translation of registrant's name into English)
Dr. Neergaards Vej 5f
DK-2970 Hoersholm
Denmark
(Address of principal executive office)
Indicate by check mark whether the registrant files or will file annual reports under cover of Form 20-F or Form 40-F.
Form 20-F [ X ] Form 40-F [ ]
INCORPORATION BY REFERENCE
This report on Form 6-K shall be deemed to be incorporated by reference in Evaxion Biotech A/S’s registration statements on Form S-8 (File No. 333-255064), on Form F-3 (File No. 333-265132) and on Form F-1 (File No. 333-266050), including any prospectuses forming a part of such registration statements and to be a part thereof from the date on which this report is filed, to the extent not superseded by documents or reports subsequently filed or furnished.
On August 1, 2023, the Registrant issued a press release, a copy of which is attached hereto as Exhibit 99.1 and is incorporated herein by reference.
(c) Exhibit 99.1. Press release dated August 1, 2023
SIGNATURES
Pursuant to the requirements of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned, thereunto duly authorized.
| | | Evaxion Biotech |
| | | (Registrant) |
| | | |
| | | |
| Date: August 1, 2023 | | /s/ Sabine Mølleskov |
| | | Sabine Mølleskov |
| | | VP Investor Relations |
| | | |
EXHIBIT 99.1
Evaxion announces financing commitments totaling up to USD 20 million with Negma Group
- Financing, available in multiple tranches over the next three years, could extend the Company’s runway into Q4 2024
- Transaction will enable Evaxion to advance its next-generation cancer vaccine, EVX-03, towards a Phase 1 clinical trial
Copenhagen, DENMARK, Aug. 01, 2023 (GLOBE NEWSWIRE) -- Evaxion Biotech A/S (NASDAQ: EVAX) (“Evaxion” or the “Company”), a clinical-stage biotechnology company specializing in the development of AI-powered immunotherapies, today announced that it has entered into a binding agreement for a financing of up to USD 20 million with Negma Group Investment Ltd, a Paris-based leading financial institution focused on supporting growth and capturing value through a multi-strategy approach.
“We are excited to collaborate with Evaxion. At Negma Group, we invest in people who bring forth forward-thinking, data-driven solutions. We have been very impressed by both the scientific innovation and expertise of the Evaxion team, and the clinical data presented to date for the programs in oncology. We are proud to partner with Evaxion as they advance the next-generation personalized cancer vaccines to develop superior immunotherapies for patients in need,” said Sophie Villedieu, Equity Capital Market of Negma Group.
“We are pleased to be able to establish a flexible financing vehicle with Negma Group. The ability to attract such an experienced investor is a tribute to Evaxion’s AI technology and pipeline progress,” said Per Norlén, Chief Executive Officer of Evaxion.
Financing is available in tranches of up to USD 700,000 over a period of 36 months. Executing individual tranches and the timing of such tranches is at the discretion of Evaxion subject to certain terms and conditions. The convertible bonds carry a zero coupon and will be issued at a subscription price corresponding to their par value. The conversion price will be determined as 83.5% of the second lowest closing volume weighted average share price (VWAP) of at least 8 trading days immediately preceding the issuance of each conversion request by Negma.
The financing commitments are intended to cover the Company’s working capital needs, including the ongoing clinical phase 2 trial of EVX-01, and the advancement of EVX-03 towards the initiation of a Phase 1 clinical trial. If Evaxion fully utilizes the convertible note program, the company will have sufficient financing to fund its planned activities into Q4 2024.
Evaxion plans to announce the interim results of the clinical Phase 2 study with EVX-01 in patients with metastatic melanoma in Q4 2023, a program where promising Phase 1/2a data were presented at ASCO in June 2023. In addition, the Company plans to initiate a Phase 1 clinical trial with EVX-03 in patients with lung cancer and other solid tumors. EVX-03 is based on the use of Evaxion´s proprietary AI-technology and novel ObsERV™ platform, which enables the Company to identify new patient-specific tumor targets called ERVs to enhance the efficacy of its personalized cancer vaccines, with potential to broaden the utility to patients unresponsive to current immunotherapies.
About Evaxion
Evaxion Biotech A/S is a clinical-stage biotech company developing AI-powered immunotherapies. Evaxion’s proprietary and scalable AI technologies decode the human immune system to discover and develop novel immunotherapies for cancer, bacterial diseases, and viral infections. Evaxion has a broad pipeline of product candidates, including three personalized cancer immunotherapies. It is located in Hørsholm, Denmark and is listed on the Nasdaq New York stock exchange. For more information, please visit: www.evaxion-biotech.com.
Forward-looking statement
This announcement contains forward-looking statements within the meaning of Section 27A of the Securities Act of 1933, as amended, and Section 21E of the Securities Exchange Act of 1934, as amended. The words “target,” “believe,” “expect,” “hope,” “aim,” “intend,” “may,” “might,” “anticipate,” “contemplate,” “continue,” “estimate,” “plan,” “potential,” “predict,” “project,” “will,” “can have,” “likely,” “should,” “would,” “could,” and other words and terms of similar meaning identify forward-looking statements. Actual results may differ materially from those indicated by such forward-looking statements as a result of various factors, including, but not limited to, risks related to: our financial condition and need for additional capital; our development work; cost and success of our product development activities and preclinical and clinical trials; commercializing any approved pharmaceutical product developed using our AI platform technology, including the rate and degree of market acceptance of our product candidates; our dependence on third parties including for conduct of clinical testing and product manufacture; our inability to enter into partnerships; government regulation; protection of our intellectual property rights; employee matters and managing growth; our ADSs and ordinary shares, the impact of international economic, political, legal, compliance, social and business factors, including inflation, and the effects on our business from the worldwide COVID-19 pandemic and the ongoing conflict in the region surrounding Ukraine and Russia; and other uncertainties affecting our business operations and financial condition. For a further discussion of these risks, please refer to the risk factors included in our most recent Annual Report on Form 20-F and other filings with the U.S. Securities and Exchange Commission (SEC), which are available at www.sec.gov. We do not assume any obligation to update any forward-looking statements except as required by law.
For more information
Evaxion Biotech A/S
Per Norlén
Chief Executive Officer
pno@evaxion-biotech.com
Source: Evaxion Biotech
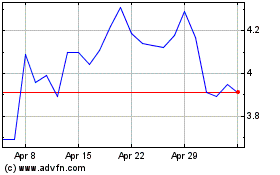
Evaxion Biotech AS (NASDAQ:EVAX)
Historical Stock Chart
From Jun 2024 to Jul 2024
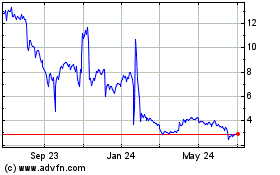
Evaxion Biotech AS (NASDAQ:EVAX)
Historical Stock Chart
From Jul 2023 to Jul 2024