Form 8-K - Current report
March 11 2024 - 8:17AM
Edgar (US Regulatory)
false
0001697532
0001697532
2024-03-11
2024-03-11
iso4217:USD
xbrli:shares
iso4217:USD
xbrli:shares
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM 8-K
CURRENT
REPORT
Pursuant
to Section 13 or 15(d)
of
the Securities Exchange Act of 1934
Date of Report (Date of
earliest event reported): March 11, 2024
APPLIED THERAPEUTICS, INC.
(Exact name of registrant as specified in its charter)
| Delaware |
|
001-38898 |
|
81-3405262 |
(State or Other Jurisdiction of
Incorporation) |
|
(Commission File Number) |
|
(I.R.S. Employer Identification
No.) |
545 Fifth Avenue, Suite 1400
New York, NY 10017 |
|
10017 |
| (Address of Principal Executive Offices) |
|
(Zip Code) |
Registrant’s telephone number,
including area code: (212) 220-9226
Check the appropriate box below if the Form 8-K
filing is intended to simultaneously satisfy the filing obligation of the registrant under any of the following provisions:
| ¨ | Written communications pursuant to Rule 425 under the Securities
Act (17 CFR 230.425) |
| | |
| ¨ | Soliciting material pursuant to Rule 14a-12 under the Exchange
Act (17 CFR 240.14a-12) |
| | |
| ¨ | Pre-commencement communications pursuant to Rule 14d-2(b) under
the Exchange Act (17 CFR 240.14d-2(b)) |
| | |
| ¨ | Pre-commencement communications pursuant to Rule 13e-4(c) under
the Exchange Act (17 CFR 240.13e-4(c)) |
Securities registered pursuant to Section 12(b) of
the Act:
| Title of each class |
|
Trading Symbol(s) |
|
Name of each exchange on which
registered |
| Common stock |
|
APLT |
|
The Nasdaq Global Market |
Indicate by check mark whether the registrant
is an emerging growth company as defined in Rule 405 of the Securities Act of 1933 (§230.405 of this chapter) or
Rule 12b-2 of the Securities Exchange Act of 1934 (§240.12b-2 of this chapter).
Emerging growth company x
If an emerging growth company, indicate by check
mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting
standards provided pursuant to Section 13(a) of the Exchange Act.
Item 7.01 Regulation FD Disclosure.
On March 11, 2024, Applied Therapeutics, Inc. (the
“Company”) released a presentation that contains company information to be used by members of management from time to time
in a series of meetings with analysts, investors and other third parties. The presentation is attached to this Current Report on Form
8-K as Exhibit 99.1 and is incorporated herein by reference.
In addition, on March 11, 2024, the Company released
a presentation that contains interim 12-month results from its ongoing Phase 3 INSPIRE trial, a registrational study evaluating the effect
of AT-007 in patients with SORD Deficiency. The presentation is attached to this Current Report on Form 8-K as Exhibit 99.2 and is incorporated
by herein by reference.
The information included in this Current Report
on Form 8-K, including Exhibits 99.1 and 99.2 incorporated by reference herein, shall not be deemed “filed” for purposes of
Section 18 of the Securities and Exchange Act of 1934, as amended (the “Exchange Act”), or otherwise subject to the liabilities
of that Section, or incorporated by reference into any filing under the Securities Act of 1933, as amended, or the Exchange Act, except
as shall be expressly set forth by specific reference in such filing.
Item 9.01 Financial Statements and Exhibits.
(d) Exhibits:
The following exhibits are attached with this current report on Form 8-K:
SIGNATURE
Pursuant to the requirements of the Securities
Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned hereunto duly authorized.
| |
APPLIED THERAPEUTICS, INC. |
| |
|
|
| Dated: March 11, 2024 |
By: |
/s/ Shoshana
Shendelman |
| |
Name: |
Shoshana Shendelman. |
| |
Title: |
President and Chief Executive Officer |
Exhibit 99.1

| Applied Therapeutics
March 2024 |

| Various statements in this presentation concerning the Company’s future expectations, plans and prospects constitute forward-looking statements. The use of
words such as “may,” “might,” “will,” “should,” “expect,” “plan,” “anticipate,” “believe,” “estimate,” “project,” “intend,” “future,” “potential,” or “continue,” the negative
of these and other similar expressions are intended to identify such forward looking statements. Such statements, based as they are on the current analysis and
expectations of management, inherently involve numerous risks and uncertainties, known and unknown, many of which are beyond the Company’s control. Such
risks include, but are not limited to: the impact of general economic conditions, general conditions in the biopharmaceutical industries, changes in the global and
regional regulatory environments in the jurisdictions in which the Company does or plans to do business, market volatility, fluctuations in costs and changes to the
competitive environment, the Company’s ability to fund its working capital requirements and expectations regarding the sufficiency of our capital resources and the
Company’s ability to achieve the anticipated benefits from the agreements entered into in connection with our partnership with Advanz Pharma. Consequently,
actual future results may differ materially from the anticipated results expressed in the forward-looking statements. In the case of forward-looking statements
regarding investigational product candidates and continuing further development efforts, specific risks which could cause actual results to differ materially from the
Company’s current analysis and expectations include: failure to demonstrate the safety, tolerability and efficacy of our product candidates; final and quality
controlled verification of data and the related analyses; the expense and uncertainty of obtaining regulatory approval, including from the U.S. Food and Drug
Administration and European Medicines Agency; the possibility of having to conduct additional clinical trials and our reliance on third parties such as our licensors
and collaboration partners regarding our suite of technologies and product candidates; the Company’s ability to take advantage of expedited regulatory pathways
for any of our product candidates; the Company’s intellectual property position and the duration of its patent rights; developments or disputes concerning the
Company’s intellectual property or other proprietary rights. Further, even if regulatory approval is obtained, biopharmaceutical products are generally subject to
stringent on-going governmental regulation, challenges in gaining market acceptance and competition.
These risks and uncertainties are described more fully under the caption ”Risk Factors” in the Company’s filings with the Securities and Exchange Commission.
Other risks and uncertainties of which the Company is not currently aware may also affect Company’s forward-looking statements. The reader should not place
undue reliance on any forward-looking statements included in this presentation. These statements speak only as of the date made and the Company is under no
obligation and disavows any obligation to update or revise such statements as a result of any event, circumstances or otherwise, unless required by applicable
legislation or regulation.
Forward Looking Statements |
| 3
Applying Science to Transform Lives
Our mission is to create transformative, life-changing treatments for patients who
desperately need them
Targeting pathways with
known roles in pathogenesis
Novel compounds with
improved potency/selectivity
SCIENCE
Clinical efficacy
confirmed via biomarkers
Pursuing expedited
regulatory pathways
Fatal or debilitating diseases
with no approved therapies
Limited / no competition
DEVELOPMENT MARKET |
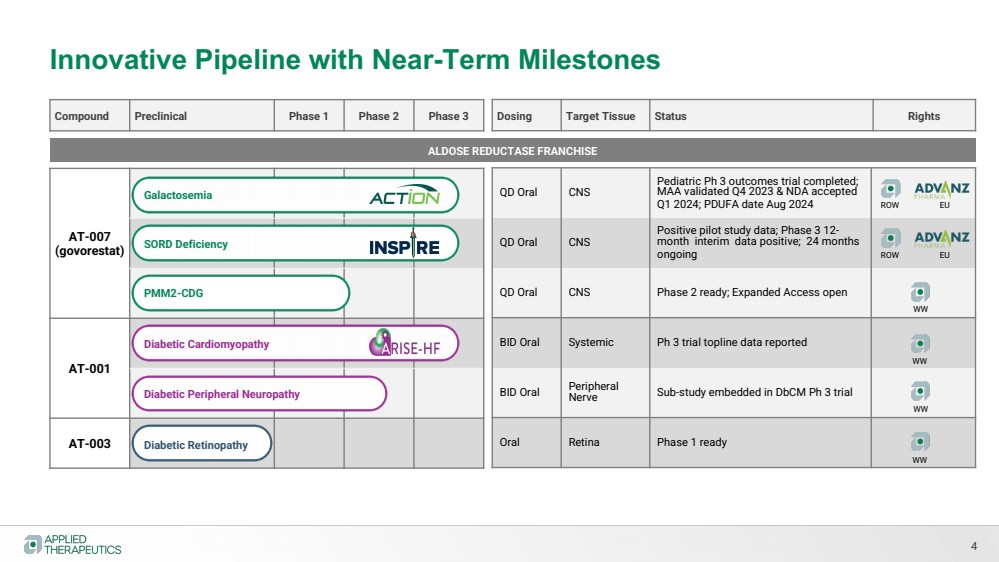
| Innovative Pipeline with Near-Term Milestones
4
Compound Preclinical Phase 1 Phase 2 Phase 3
AT-007
(govorestat)
AT-001
AT-003
QD Oral CNS
Pediatric Ph 3 outcomes trial completed;
MAA validated Q4 2023 & NDA accepted
Q1 2024; PDUFA date Aug 2024
QD Oral CNS
Positive pilot study data; Phase 3 12- month interim data positive; 24 months
ongoing
QD Oral CNS Phase 2 ready; Expanded Access open
BID Oral Systemic Ph 3 trial topline data reported
BID Oral Peripheral
Nerve Sub-study embedded in DbCM Ph 3 trial
Oral Retina Phase 1 ready
Dosing Target Tissue Status Rights
ALDOSE REDUCTASE FRANCHISE
Galactosemia
SORD Deficiency
Diabetic Cardiomyopathy
Diabetic Peripheral Neuropathy
PMM2-CDG
Diabetic Retinopathy
ROW EU
ROW EU
WW
WW
WW
WW |

| ALDOSE
REDUCTASE
First and rate limiting enzyme in the polyol pathway – an alternative metabolic
pathway activated under stress
Converts sugar to reduced sugar alcohols, which are toxic
Prior attempts to inhibit Aldose Reductase were hindered by lack of
selectivity and off-target tox issues
5
Aldose Reductase: An Enzyme Implicated in Multiple Metabolic Diseases
Leads to cell death through osmotic dysregulation, reactive oxygen species
sugar formation, and energy deficiencies
alcohol
Normal
Metabolism Energy
polyol pathway
activation
Osmotic stress
Oxidative damage
Energy depletion
Cell death
Applied Therapeutics’ compounds are 1,000X more potent than “old”
Aldose Reductase Inhibitors and highly selective; no off-target inhibition of
aldehyde reductase
sugar
sugar |

| Govorestat (AT-007)
GALACTOSEMIA
Positive adult & pediatric biomarker data
Pediatric Ph 3 clinical outcomes study completed
Regulatory submissions under review (MAA validated; NDA accepted)
• Orphan Drug Designation
• Pediatric Rare Disease Designation (PRV)
• Fast-Track Designation |

| Galactosemia is a Rare Metabolic Disease With No Approved
Therapies and Significant Unmet Need
Disease Overview
• Rare autosomal recessive metabolic disease caused by
deficiencies in the GALT or GALK enzymes
• Patients are unable to metabolize the simple sugar
galactose, which is found in foods but also synthesized
endogenously by the body
• Results in long-term CNS complications including behavior
and motor skills deficiencies, cognitive issues; tremor,
speech problems; ovarian insufficiency in females
• Progressively worsens with age
Standard of Care/ Diagnosis
• No approved therapies to treat Galactosemia
• Mandatory newborn screening in US and most EU
countries
• Galactose-restricted diet implemented immediately after
birth and adhered to for life
• Dietary restriction prevents newborn fatalities but does not
prevent long-term CNS complications due to endogenous
galactose production by the body
• Patients are primarily seen by metabolic geneticists
Mechanism of Disease
• People with Galactosemia are unable to metabolize
galactose, which accumulates in cells and tissues
• At abnormally high levels, galactose becomes a substrate
for Aldose Reductase, which converts galactose to a toxic
and aberrant metabolite, galactitol
• Galactitol is highly toxic (especially to neurons) and causes
redox derangement, cell death
• Plasma galactitol level correlates with severity of disease
Market Size/ Opportunity
• Known prevalent and addressable population (~3K US,
~7K WW)
• Small commercial footprint focused on KOLs at Centers of
Excellence
• Strong patient community engagement
• Payer feedback supports access/pricing
• Composition of matter IP through 2037 (not including
extensions)
Data on file: Symphony Claims Data, Feb. 2017-Jan. 2021. Phytila et al. JIMD Rep. 2015; 15: 79–93. Burgard et al. Report on the practices of newborn screening for rare disorders. 2011.
Swaiman et al. Pediatric Neurology. 2018. |
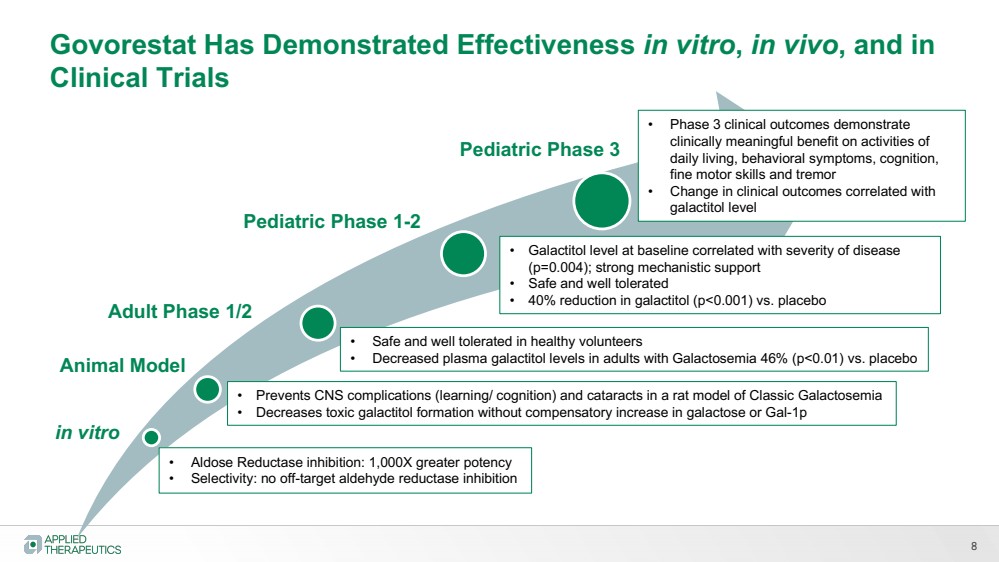
| 8
in vitro
Pediatric Phase 1-2
Pediatric Phase 3
• Aldose Reductase inhibition: 1,000X greater potency
• Selectivity: no off-target aldehyde reductase inhibition
Animal Model
• Prevents CNS complications (learning/ cognition) and cataracts in a rat model of Classic Galactosemia
• Decreases toxic galactitol formation without compensatory increase in galactose or Gal-1p
• Safe and well tolerated in healthy volunteers
• Decreased plasma galactitol levels in adults with Galactosemia 46% (p<0.01) vs. placebo
• Galactitol level at baseline correlated with severity of disease
(p=0.004); strong mechanistic support
• Safe and well tolerated
• 40% reduction in galactitol (p<0.001) vs. placebo
Govorestat Has Demonstrated Effectiveness in vitro, in vivo, and in
Clinical Trials
Adult Phase 1/2
• Phase 3 clinical outcomes demonstrate
clinically meaningful benefit on activities of
daily living, behavioral symptoms, cognition,
fine motor skills and tremor
• Change in clinical outcomes correlated with
galactitol level |

| Deficiency in GALT or GALK Leads to Inability to Metabolize
Galactose; AR Converts Excess Galactose to Toxic Galactitol
Energy Production GALK GALT
NORMAL METABOLISM
Classic
Galactosemia
GALK
Deficiency
ALDOSE
REDUCTASE
galactitol
toxic accumulation
of galactitol
galactitol
no natural enzyme
to metabolize galactitol
galactitol
GALACTOSEMIA
9
• CNS Complications:
o Cognition/ Learning/ IQ/
Memory
o Behavior/ Psychiatric
o Motor Skills (Tremor,
Ataxia)
o Seizures
o Speech Deficiencies
• Other Complications:
o Ovarian Insufficiency
o Cataracts
galactose
Gal-1p
Glucose-1-P
AR = Aldose Reductase |
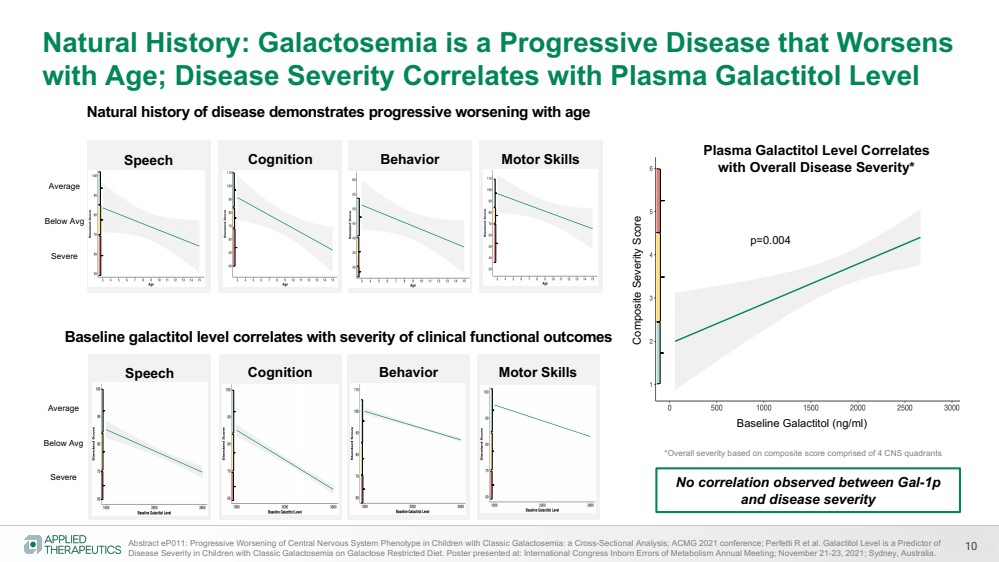
| 10
Natural History: Galactosemia is a Progressive Disease that Worsens
with Age; Disease Severity Correlates with Plasma Galactitol Level
Speech Cognition Behavior Motor Skills
Abstract eP011: Progressive Worsening of Central Nervous System Phenotype in Children with Classic Galactosemia: a Cross-Sectional Analysis; ACMG 2021 conference; Perfetti R et al. Galactitol Level is a Predictor of
Disease Severity in Children with Classic Galactosemia on Galactose Restricted Diet. Poster presented at: International Congress Inborn Errors of Metabolism Annual Meeting; November 21-23, 2021; Sydney, Australia.
Average
Below Avg
Severe
Speech Cognition Behavior Motor Skills
Average
Below Avg
Severe
Composite Severity Score
Baseline Galactitol (ng/ml)
Plasma Galactitol Level Correlates
with Overall Disease Severity*
p=0.004
*Overall severity based on composite score comprised of 4 CNS quadrants
Baseline galactitol level correlates with severity of clinical functional outcomes
Natural history of disease demonstrates progressive worsening with age
No correlation observed between Gal-1p
and disease severity |
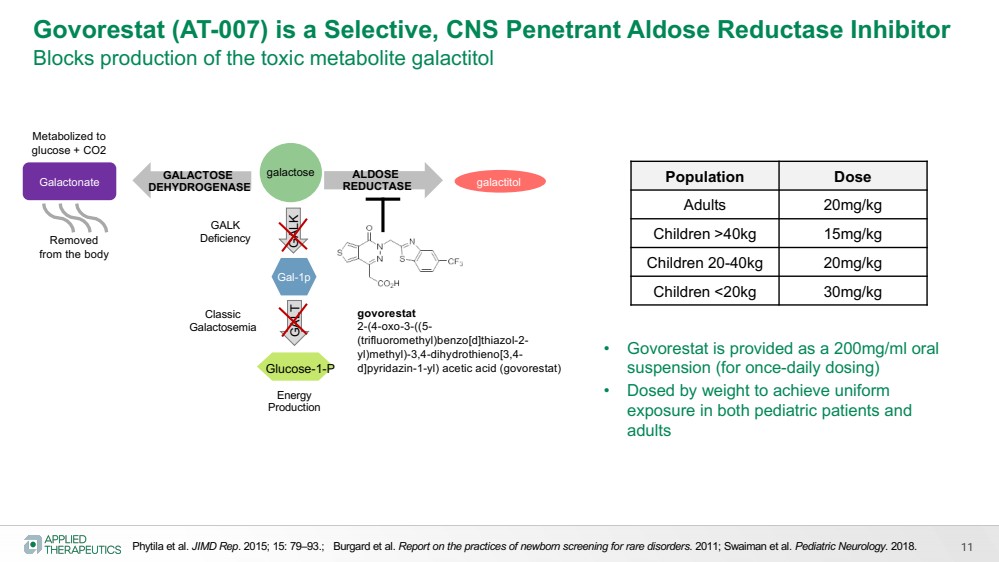
| Energy
Production GALK GALT
Classic
Galactosemia
GALK
Deficiency
ALDOSE
REDUCTASE
galactose
Gal-1p
Glucose-1-P
Govorestat (AT-007) is a Selective, CNS Penetrant Aldose Reductase Inhibitor
Blocks production of the toxic metabolite galactitol
Phytila et al. JIMD Rep. 2015; 15: 79–93.; Burgard et al. Report on the practices of newborn screening for rare disorders. 2011; Swaiman et al. Pediatric Neurology. 2018.
govorestat
2-(4-oxo-3-((5-
(trifluoromethyl)benzo[d]thiazol-2-
yl)methyl)-3,4-dihydrothieno[3,4-
d]pyridazin-1-yl) acetic acid (govorestat)
• Govorestat is provided as a 200mg/ml oral
suspension (for once-daily dosing)
• Dosed by weight to achieve uniform
exposure in both pediatric patients and
adults
Population Dose
Adults 20mg/kg
Children >40kg 15mg/kg
Children 20-40kg 20mg/kg
Children <20kg 30mg/kg
galactitol GALACTOSE
DEHYDROGENASE Galactonate
Metabolized to
glucose + CO2
Removed
from the body
11 |
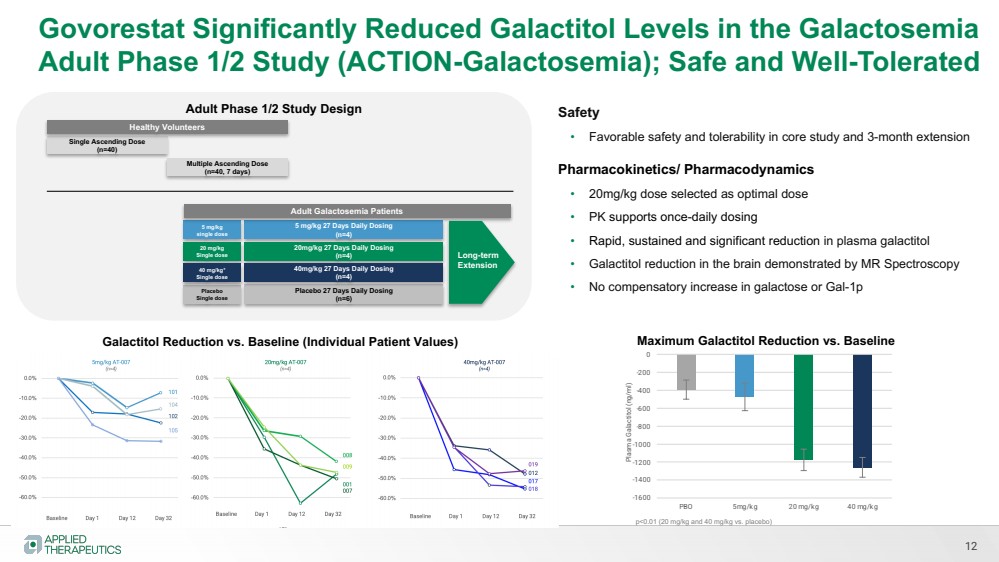
| Govorestat Significantly Reduced Galactitol Levels in the Galactosemia
Adult Phase 1/2 Study (ACTION-Galactosemia); Safe and Well-Tolerated
Single Ascending Dose
(n=40)
Multiple Ascending Dose
(n=40, 7 days)
5 mg/kg
single dose
5 mg/kg 27 Days Daily Dosing
(n=4)
Adult Galactosemia Patients
Healthy Volunteers
Placebo
Single dose
Placebo 27 Days Daily Dosing
(n=6)
20 mg/kg
Single dose
20mg/kg 27 Days Daily Dosing
(n=4)
40 mg/kg*
Single dose
40mg/kg 27 Days Daily Dosing
(n=4)
12
Adult Phase 1/2 Study Design Safety
• Favorable safety and tolerability in core study and 3-month extension
Pharmacokinetics/ Pharmacodynamics
• 20mg/kg dose selected as optimal dose
• PK supports once-daily dosing
• Rapid, sustained and significant reduction in plasma galactitol
• Galactitol reduction in the brain demonstrated by MR Spectroscopy
• No compensatory increase in galactose or Gal-1p
-1600
-1400
-1200
-1000
-800
-600
-400
-200
0
PBO 5mg/kg 20 mg/kg 40 mg/kg
Plasma Galactitol (ng/ml)
Galactitol Reduction vs. Baseline (Individual Patient Values) Maximum Galactitol Reduction vs. Baseline
Long-term
Extension
p<0.01 (20 mg/kg and 40 mg/kg vs. placebo) |
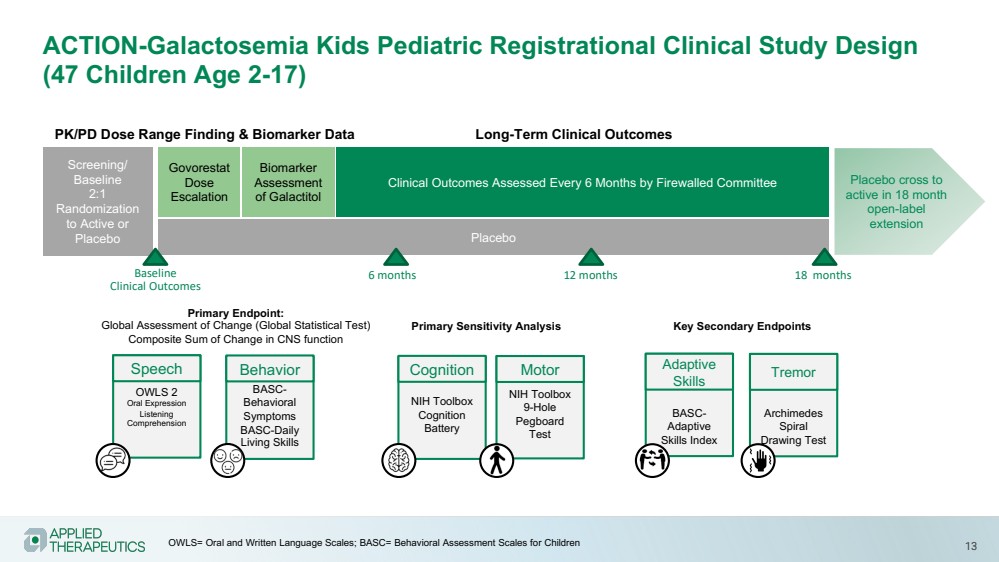
| 13
ACTION-Galactosemia Kids Pediatric Registrational Clinical Study Design
(47 Children Age 2-17)
Govorestat
Dose
Escalation
Biomarker
Assessment
of Galactitol
Placebo cross to
active in 18 month
open-label
extension
PK/PD Dose Range Finding & Biomarker Data Long-Term Clinical Outcomes
Placebo
Screening/
Baseline
2:1
Randomization
to Active or
Placebo
Clinical Outcomes Assessed Every 6 Months by Firewalled Committee
Baseline
Clinical Outcomes
OWLS 2
Oral Expression
Listening
Comprehension
Speech
NIH Toolbox
Cognition
Battery
Cognition
BASC-Behavioral
Symptoms
BASC-Daily
Living Skills
Behavior
NIH Toolbox
9-Hole
Pegboard
Test
Motor
Primary Endpoint:
Global Assessment of Change (Global Statistical Test)
Composite Sum of Change in CNS function
BASC-Adaptive
Skills Index
Adaptive
Skills
Archimedes
Spiral
Drawing Test
Tremor
Primary Sensitivity Analysis Key Secondary Endpoints
6 months 12 months 18 months
OWLS= Oral and Written Language Scales; BASC= Behavioral Assessment Scales for Children |
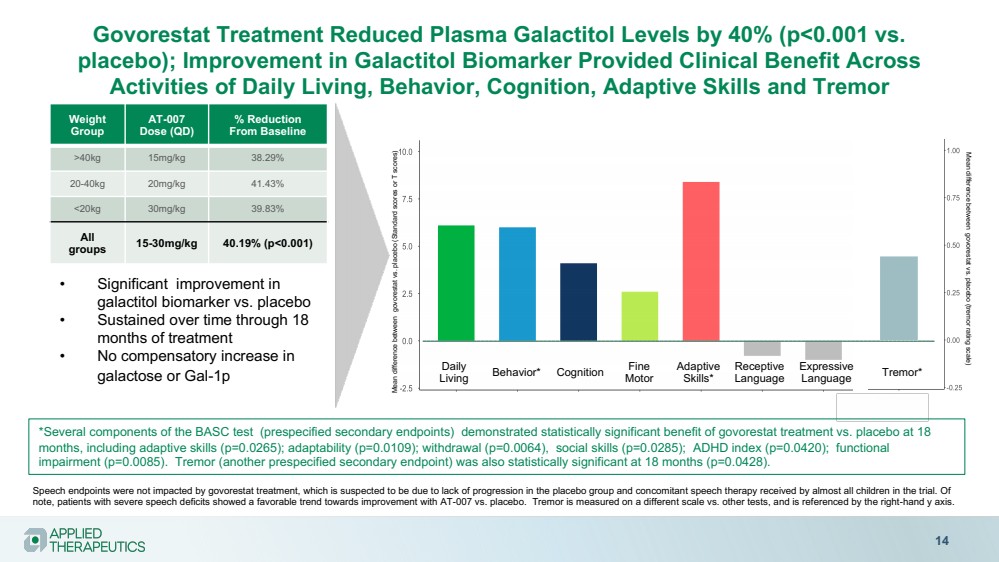
| Govorestat Treatment Reduced Plasma Galactitol Levels by 40% (p<0.001 vs.
placebo); Improvement in Galactitol Biomarker Provided Clinical Benefit Across
Activities of Daily Living, Behavior, Cognition, Adaptive Skills and Tremor
14
Weight
Group
AT-007
Dose (QD)
% Reduction
From Baseline
>40kg 15mg/kg 38.29%
20-40kg 20mg/kg 41.43%
<20kg 30mg/kg 39.83%
All
groups 15-30mg/kg 40.19% (p<0.001)
• Significant improvement in
galactitol biomarker vs. placebo
• Sustained over time through 18
months of treatment
• No compensatory increase in
galactose or Gal-1p Daily
Living Behavior* Adaptive
Skills* Cognition Receptive
Language
Expressive
Language Tremor*
Mean difference between govorestat vs. placebo (Standard scores or T scores)
Speech endpoints were not impacted by govorestat treatment, which is suspected to be due to lack of progression in the placebo group and concomitant speech therapy received by almost all children in the trial. Of
note, patients with severe speech deficits showed a favorable trend towards improvement with AT-007 vs. placebo. Tremor is measured on a different scale vs. other tests, and is referenced by the right-hand y axis.
Fine
Motor
Mean difference between govorestat vs. placebo (tremor rating scale)
*Several components of the BASC test (prespecified secondary endpoints) demonstrated statistically significant benefit of govorestat treatment vs. placebo at 18
months, including adaptive skills (p=0.0265); adaptability (p=0.0109); withdrawal (p=0.0064), social skills (p=0.0285); ADHD index (p=0.0420); functional
impairment (p=0.0085). Tremor (another prespecified secondary endpoint) was also statistically significant at 18 months (p=0.0428). |
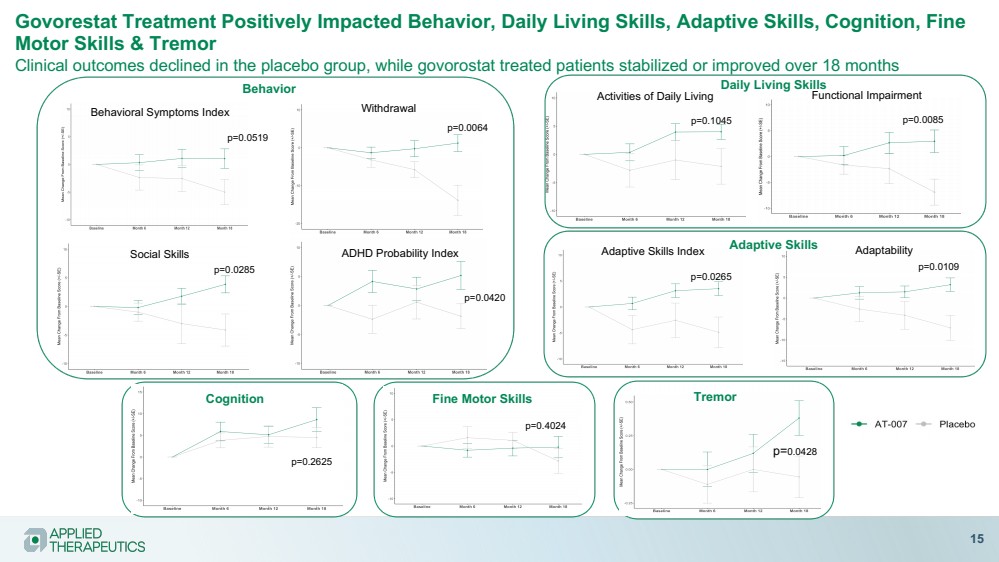
| Govorestat Treatment Positively Impacted Behavior, Daily Living Skills, Adaptive Skills, Cognition, Fine
Motor Skills & Tremor
Clinical outcomes declined in the placebo group, while govorostat treated patients stabilized or improved over 18 months
15
Behavioral Symptoms Index
Social Skills
Withdrawal
ADHD Probability Index
p=0.0519
p=0.0285
p=0.0064
p=0.0420
p=0.1045 p=0.0085
Activities of Daily Living Functional Impairment
Adaptive Skills Index
p=0.0265
Adaptability
p=0.0109
Behavior Daily Living Skills
Adaptive Skills
p=0.2625
p=0.4024
Tremor
p=0.0428
Cognition Fine Motor Skills |
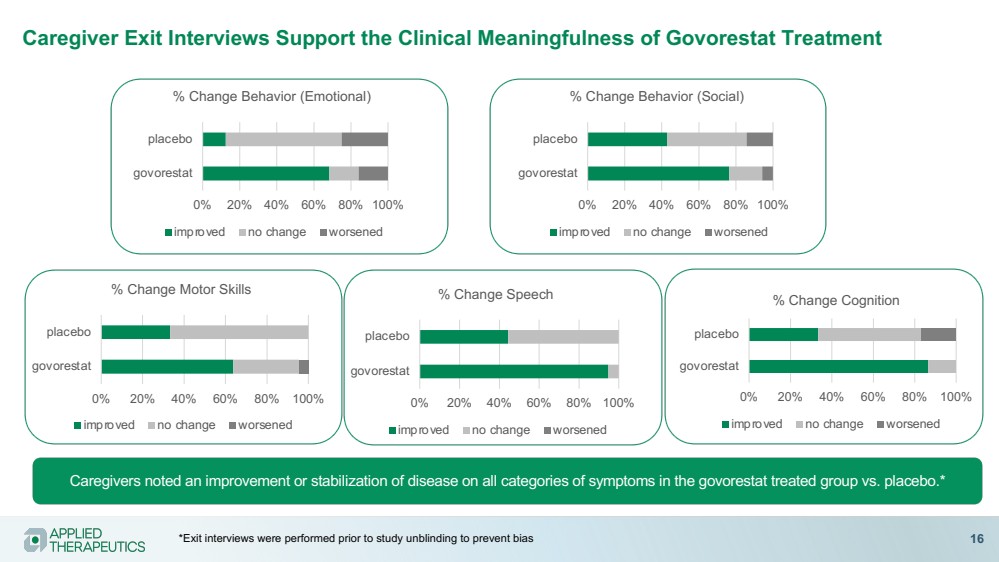
| Caregiver Exit Interviews Support the Clinical Meaningfulness of Govorestat Treatment
Caregivers noted an improvement or stabilization of disease on all categories of symptoms in the govorestat treated group vs. placebo.*
0% 20% 40% 60% 80% 100%
govorestat
placebo
% Change Behavior (Emotional)
improved no change worsened
0% 20% 40% 60% 80% 100%
govorestat
placebo
% Change Behavior (Social)
improved no change worsened
0% 20% 40% 60% 80% 100%
govorestat
placebo
% Change Cognition
improved no change worsened
0% 20% 40% 60% 80% 100%
govorestat
placebo
% Change Motor Skills
improved no change worsened
0% 20% 40% 60% 80% 100%
govorestat
placebo
% Change Speech
improved no change worsened
*Exit interviews were performed prior to study unblinding to prevent bias 16 |
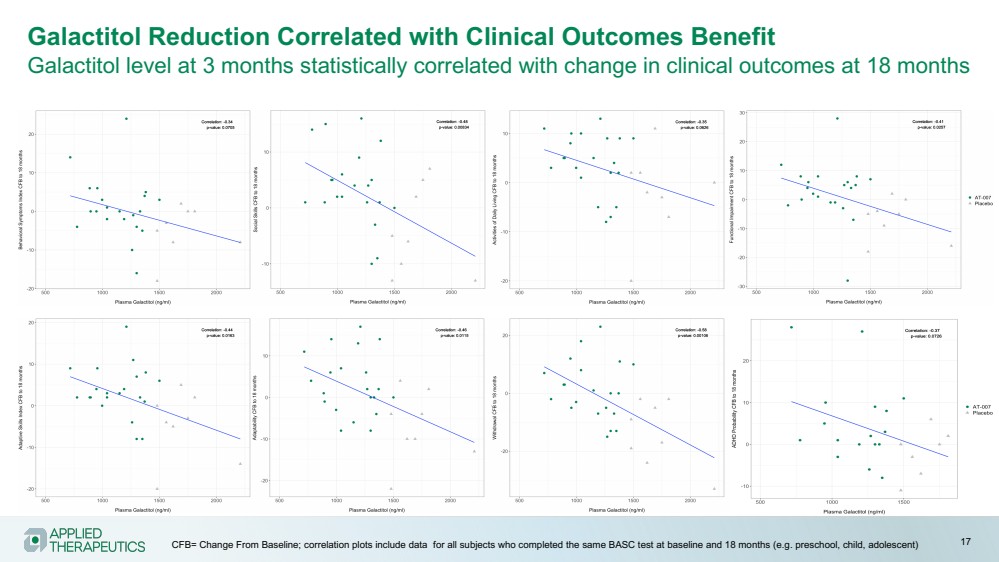
| Galactitol Reduction Correlated with Clinical Outcomes Benefit
Galactitol level at 3 months statistically correlated with change in clinical outcomes at 18 months
CFB= Change From Baseline; correlation plots include data for all subjects who completed the same BASC test at baseline and 18 months (e.g. preschool, child, adolescent) 17 |
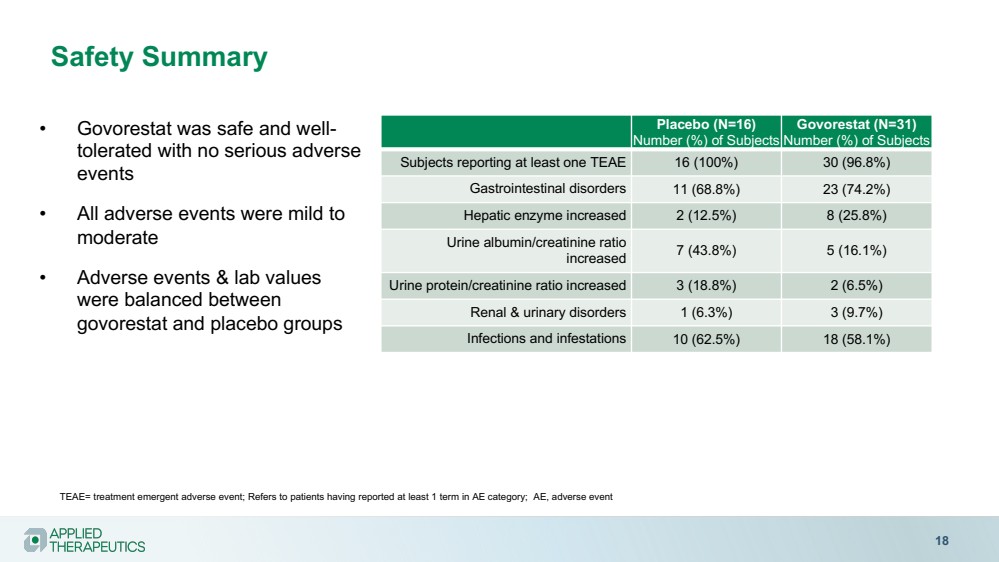
| Safety Summary
18
• Govorestat was safe and well-tolerated with no serious adverse
events
• All adverse events were mild to
moderate
• Adverse events & lab values
were balanced between
govorestat and placebo groups
Placebo (N=16)
Number (%) of Subjects
Govorestat (N=31)
Number (%) of Subjects
Subjects reporting at least one TEAE 16 (100%) 30 (96.8%)
Gastrointestinal disorders 11 (68.8%) 23 (74.2%)
Hepatic enzyme increased 2 (12.5%) 8 (25.8%)
Urine albumin/creatinine ratio
increased 7 (43.8%) 5 (16.1%)
Urine protein/creatinine ratio increased 3 (18.8%) 2 (6.5%)
Renal & urinary disorders 1 (6.3%) 3 (9.7%)
Infections and infestations 10 (62.5%) 18 (58.1%)
TEAE= treatment emergent adverse event; Refers to patients having reported at least 1 term in AE category; AE, adverse event |
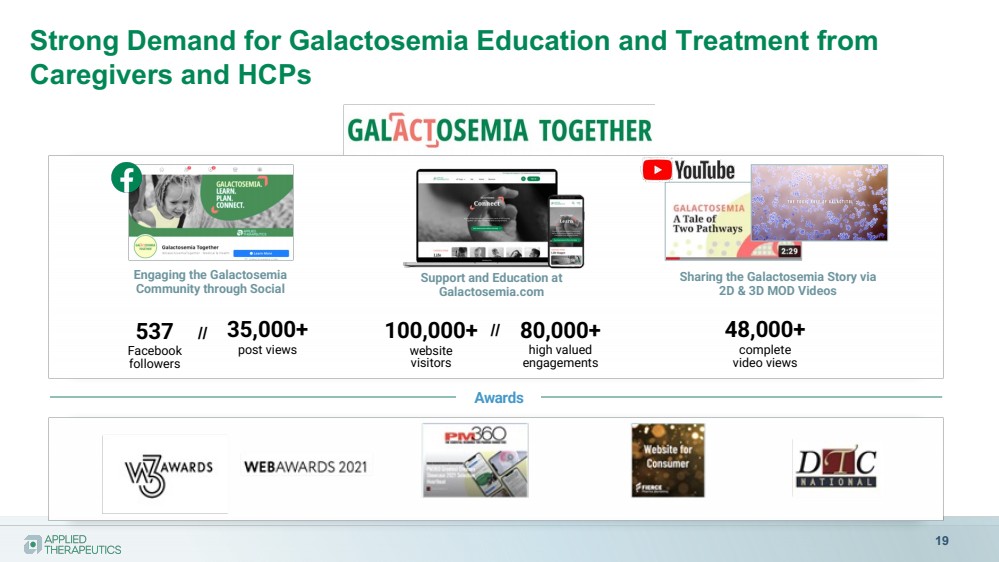
| Strong Demand for Galactosemia Education and Treatment from
Caregivers and HCPs
19
Awards
Engaging the Galactosemia
Community through Social
35,000+
post views
537
Facebook
followers
Support and Education at
Galactosemia.com
100,000+
website
visitors
Sharing the Galactosemia Story via
2D & 3D MOD Videos
48,000+
complete
video views
// 80,000+
high valued
engagements
// |

| Govorestat (AT-007)
SORD
DEFICIENCY
Preclinical proof of concept demonstrated
Positive pilot study completed
Registrational Phase 3 study positive interim 12-month data
• Orphan Drug Designation |

| SORD Deficiency is a Rare Neurological Disease with No Approved
Therapies and High Unmet Need
Disease Overview
• Sorbitol Dehydrogenase Deficiency (SORD Deficiency)
is a progressive, debilitating hereditary neuropathy that
affects peripheral nerves and motor neurons, resulting in
significant disability, loss of sensory function and
decreased mobility
• Autosomal recessive genetic disease, caused by
mutations in the SORD gene resulting in loss of SORD
enzyme function
• Average age of onset is 17 years old
Standard of Care/ Diagnosis
• No approved therapies to treat SORD Deficiency
• Genetic testing commercially available (GeneDx)
• Prior to 2020, patients were diagnosed symptomatically
as CMT2 or dHMN; new screening efforts are quickly re-categorizing CMT2/dHMN patients with SORD
• Primarily treated by neurologists/ neuromuscular
specialists at Inherited Neuropathy Consortium (INC)
Centers of Excellence
Mechanism of Disease
• Patients with SORD Deficiency are unable to metabolize
sorbitol
• Aldose Reductase converts glucose to sorbitol, which
then accumulates at up to 100X normal levels in patients
with SORD Deficiency
• Sorbitol is toxic to cells (especially neurons), resulting in
osmotic stress, redox derangement and energetic
destabilization
Market Size/ Opportunity
• ~3,300 individuals in the US with SORD Deficiency;
7,000 US+EU combined
• Small commercial footprint focused on KOLs at COEs
• Strong patient community engagement
• Payer feedback supports access/pricing
• Composition of matter IP through 2037; IP covering ARI
treatment of SORD Deficiency through 2040
CMT2 = Charcot-Marie-Tooth Type 2; dHMN = distal Hereditary Motor Neuropathy; COE = Centers of Excellence; ARI = Aldose Reductase Inhibitor |
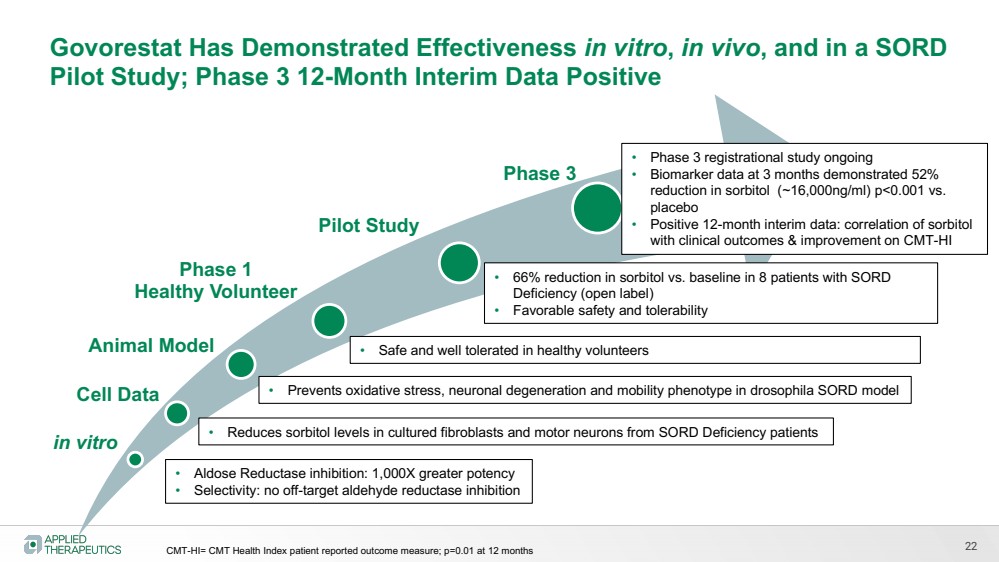
| 22
in vitro
Pilot Study
Phase 3
• Aldose Reductase inhibition: 1,000X greater potency
• Selectivity: no off-target aldehyde reductase inhibition
Animal Model
• Prevents oxidative stress, neuronal degeneration and mobility phenotype in drosophila SORD model
• Safe and well tolerated in healthy volunteers
• 66% reduction in sorbitol vs. baseline in 8 patients with SORD
Deficiency (open label)
• Favorable safety and tolerability
Govorestat Has Demonstrated Effectiveness in vitro, in vivo, and in a SORD
Pilot Study; Phase 3 12-Month Interim Data Positive
Phase 1
Healthy Volunteer
• Phase 3 registrational study ongoing
• Biomarker data at 3 months demonstrated 52%
reduction in sorbitol (~16,000ng/ml) p<0.001 vs.
placebo
• Positive 12-month interim data: correlation of sorbitol
with clinical outcomes & improvement on CMT-HI
Cell Data
• Reduces sorbitol levels in cultured fibroblasts and motor neurons from SORD Deficiency patients
CMT-HI= CMT Health Index patient reported outcome measure; p=0.01 at 12 months |
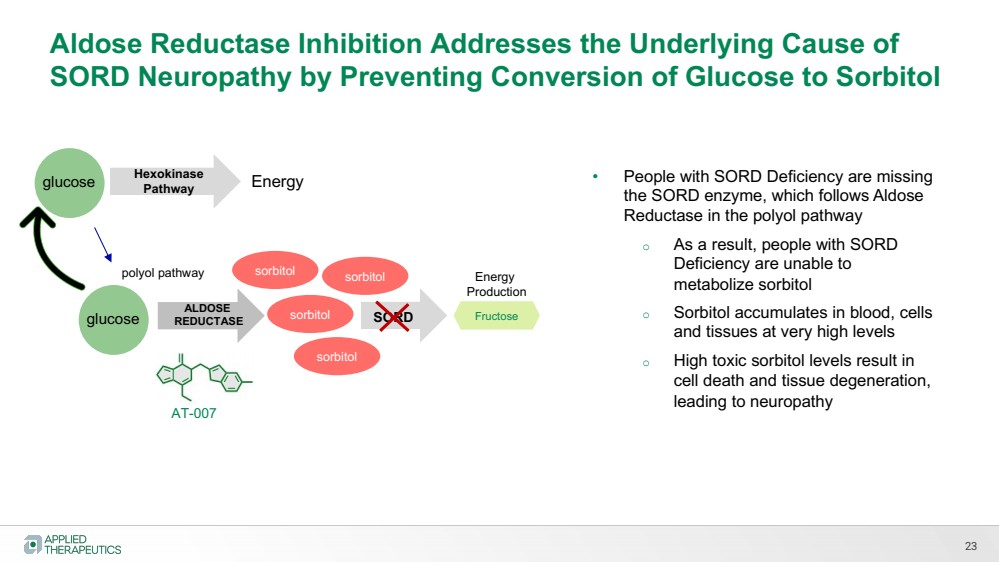
| 23
Aldose Reductase Inhibition Addresses the Underlying Cause of
SORD Neuropathy by Preventing Conversion of Glucose to Sorbitol
sorbitol
Hexokinase
Pathway
ALDOSE
REDUCTASE
Energy
polyol pathway
AT-007
• People with SORD Deficiency are missing
the SORD enzyme, which follows Aldose
Reductase in the polyol pathway
o As a result, people with SORD
Deficiency are unable to
metabolize sorbitol
o Sorbitol accumulates in blood, cells
and tissues at very high levels
o High toxic sorbitol levels result in
cell death and tissue degeneration,
leading to neuropathy
SORD Fructose
Energy
Production
sorbitol
sorbitol sorbitol
glucose
glucose |
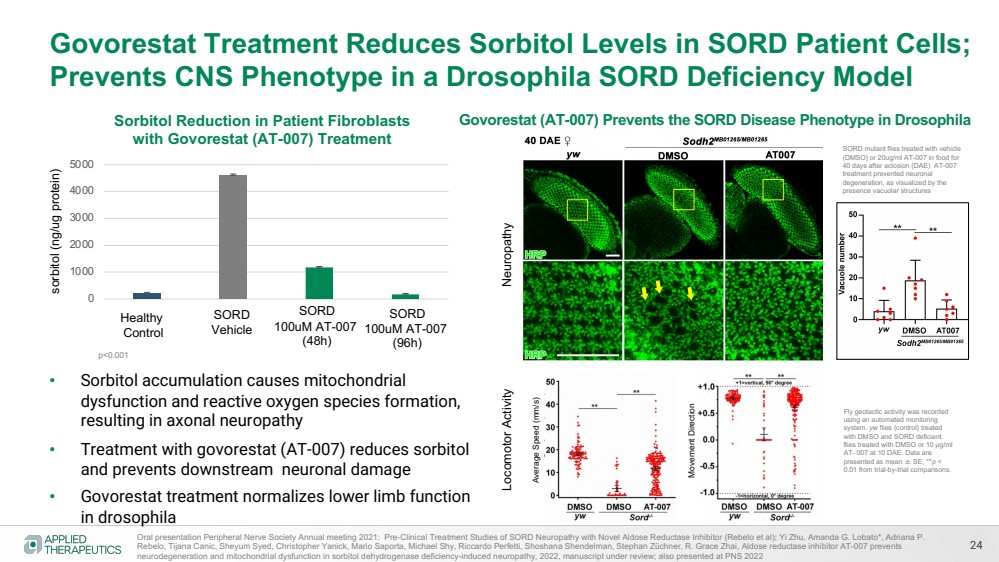
| 24
Govorestat Treatment Reduces Sorbitol Levels in SORD Patient Cells;
Prevents CNS Phenotype in a Drosophila SORD Deficiency Model
• Sorbitol accumulation causes mitochondrial
dysfunction and reactive oxygen species formation,
resulting in axonal neuropathy
• Treatment with govorestat (AT-007) reduces sorbitol
and prevents downstream neuronal damage
• Govorestat treatment normalizes lower limb function
in drosophila
sorbitol (ng/ug protein)
Oral presentation Peripheral Nerve Society Annual meeting 2021: Pre-Clinical Treatment Studies of SORD Neuropathy with Novel Aldose Reductase Inhibitor (Rebelo et al); Yi Zhu, Amanda G. Lobato*, Adriana P.
Rebelo, Tijana Canic, Sheyum Syed, Christopher Yanick, Mario Saporta, Michael Shy, Riccardo Perfetti, Shoshana Shendelman, Stephan Züchner, R. Grace Zhai, Aldose reductase inhibitor AT-007 prevents
neurodegeneration and mitochondrial dysfunction in sorbitol dehydrogenase deficiency-induced neuropathy, 2022, manuscript under review; also presented at PNS 2022
0
1000
2000
3000
4000
5000
Sorbitol Reduction in Patient Fibroblasts
with Govorestat (AT-007) Treatment
Healthy
Control
SORD
Vehicle
SORD
100uM AT-007
(48h)
SORD
100uM AT-007
(96h)
Govorestat (AT-007) Prevents the SORD Disease Phenotype in Drosophila
SORD mutant flies treated with vehicle
(DMSO) or 20ug/ml AT-007 in food for
40 days after eclosion (DAE) AT-007
treatment prevented neuronal
degeneration, as visualized by the
presence vacuolar structures
p<0.001
Average Speed (mm/s)
Movement Direction
Fly geotactic activity was recorded
using an automated monitoring
system. yw flies (control) treated
with DMSO and SORD deficient
flies treated with DMSO or 10 μg/ml
AT- 007 at 10 DAE. Data are
presented as mean ± SE, **p <
0.01 from trial-by-trial comparisons.
Neuropathy Locomotor Activity |
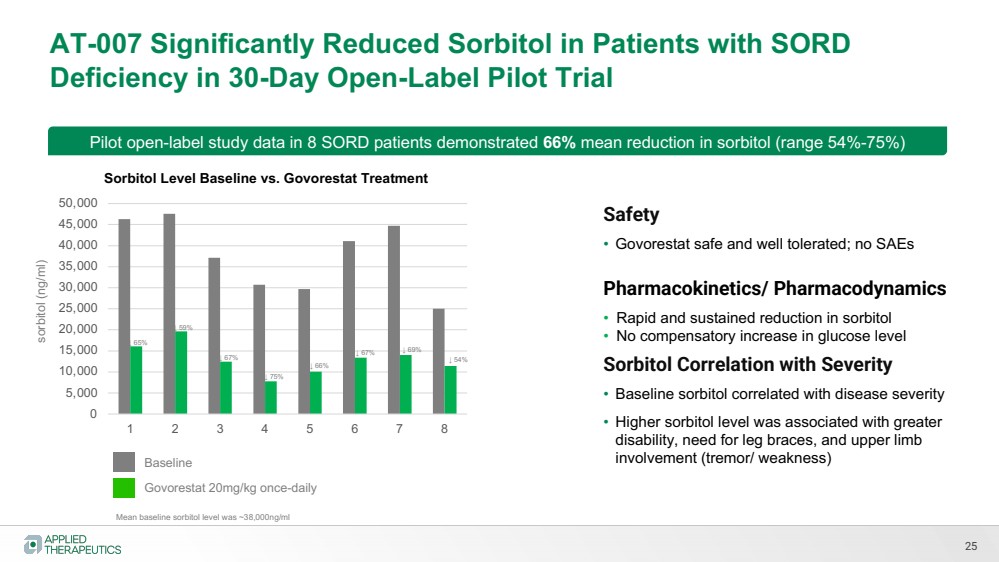
| AT-007 Significantly Reduced Sorbitol in Patients with SORD
Deficiency in 30-Day Open-Label Pilot Trial
25
Mean baseline sorbitol level was ~38,000ng/ml
Pilot open-label study data in 8 SORD patients demonstrated 66% mean reduction in sorbitol (range 54%-75%)
sorbitol (ng/ml)
0
5,000
10,000
15,000
20,000
25,000
30,000
35,000
40,000
45,000
50,000
1 2 3 4 5 6 7 8
Sorbitol Level Baseline vs. Govorestat Treatment
Baseline
Govorestat 20mg/kg once-daily
Safety
• Govorestat safe and well tolerated; no SAEs
Pharmacokinetics/ Pharmacodynamics
• Rapid and sustained reduction in sorbitol
• No compensatory increase in glucose level
Sorbitol Correlation with Severity
• Baseline sorbitol correlated with disease severity
• Higher sorbitol level was associated with greater
disability, need for leg braces, and upper limb
involvement (tremor/ weakness)
↓ 65%
↓ 59%
↓ 67%
↓ 75%
↓ 66%
↓ 67% ↓ 69%
↓ 54% |

| SORD Neuropathy Phase 3 Registrational Study (INSPIRE)
Double-Blind, Randomized, Placebo-Controlled Multi-Center Study in ~50 SORD Patients >16 years old
Primary Biomarker Efficacy:
Reduction in sorbitol vs. baseline at 3
months
Exploratory Biomarker: NFL
reduction at 3 months
CMT-FOM lower limb domain (10m walk/run)
CMT-HI (patient reported outcome)
Muscle MRI
Placebo cross to
active once
treatment effect
on outcomes is
demonstrated
Placebo
26
PHASE 3
Baseline Clinical
Outcomes &
Sorbitol;
Randomization to
Active or
Placebo (2:1)
CMT = Charcot-Marie-Tooth, FOM = Functional Outcomes Measure, HI = Health Index, MRI = Magnetic Resonance Imaging, 10MWR = 10-Meter Walk/Run
12 Month
Interim Assessment
24 Month
Clinical Outcomes
(expected Q1 2025)
outcome variable constant p value
10MWR sorbitol age p<0.05
4-stair-climb sorbitol age p<0.05
sit-to-stand sorbitol age p<0.05
3 Month
Biomarker Readout
Cross-sectional analysis of the first cohort in the INSPIRE trial confirms that sorbitol level statistically correlates with clinical outcomes
• Statistically significant correlation of sorbitol with lower limb clinical outcome measures
• Confirms sorbitol as a key driver of disease severity and disease progression over time
• Supports lower limb metrics evaluated in INSPIRE Phase 3 trial
CMT-FOM Domains and Tests |
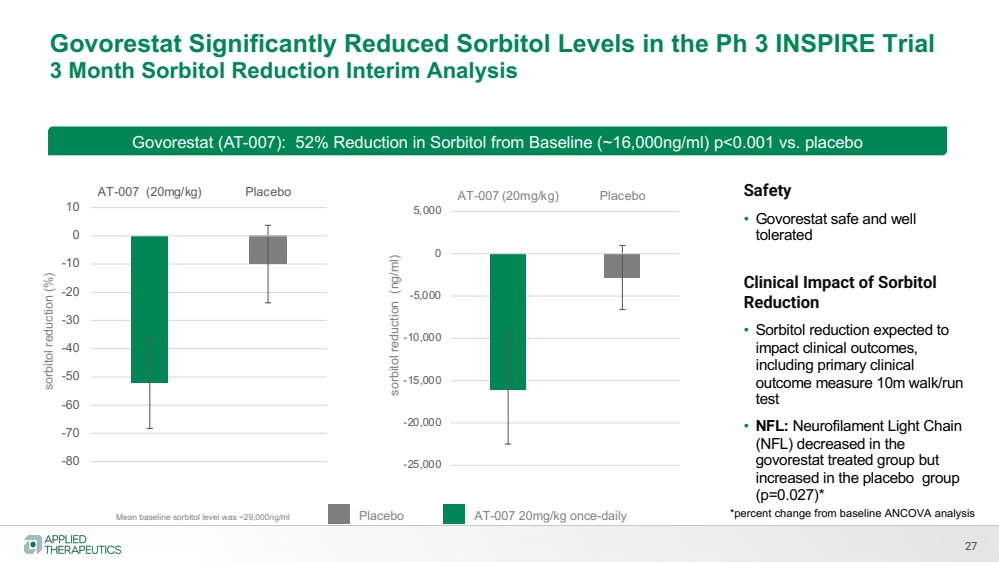
| Govorestat Significantly Reduced Sorbitol Levels in the Ph 3 INSPIRE Trial
3 Month Sorbitol Reduction Interim Analysis
27
Mean baseline sorbitol level was ~29,000ng/ml
Govorestat (AT-007): 52% Reduction in Sorbitol from Baseline (~16,000ng/ml) p<0.001 vs. placebo sorbitol reduction (ng/ml)
Placebo AT-007 20mg/kg once-daily
Safety
• Govorestat safe and well
tolerated
Clinical Impact of Sorbitol
Reduction
• Sorbitol reduction expected to
impact clinical outcomes,
including primary clinical
outcome measure 10m walk/run
test
• NFL: Neurofilament Light Chain
(NFL) decreased in the
govorestat treated group but
increased in the placebo group
(p=0.027)*
-80
-70
-60
-50
-40
-30
-20
-10
0
10
AT-007 (20mg/kg) Placebo
-25,000
-20,000
-15,000
-10,000
-5,000
0
5,000
AT-007 (20mg/kg) Placebo
sorbitol reduction (%)
*percent change from baseline ANCOVA analysis |

| INSPIRE Trial 12 Month Interim Data Overview
28
Co-primary endpoints at 12 month analysis:
• Primary clinical efficacy endpoint: Statistically significant correlation between sorbitol levels and change in clinical outcomes
at 12 months of treatment on combined measures of the CMT Functional Outcome Measures (CMT-FOM) lower limb domain
(10 meter walk-run test, 4 stair climb, and sit to stand test), 6-minute walk test and dorsiflexion (p=0.05)
• Primary pharmacodynamic/ biomarker endpoint: Sustained reduction in sorbitol level in patients treated with govorestat at
12 months, which was statistically significant compared to placebo (p<0.001).
Secondary Endpoints
• Highly statistically significant effect (p=0.01) impact of govorestat on the CMT Health Index (CMT-HI), an important patient-reported outcome measure of disease severity and well-being; aspects of the CMT-HI that demonstrated a treatment effect
included lower limb function, mobility, fatigue, pain, sensory function, and upper limb function.
• Trends (not statistically significant) on CMT-FOM measures linked to walking ability, such as 10MWR, dorsiflexion and 6 minute
walk test
o No substantial effect on stair climb or sit-to-stand test
Safety
• Govorestat was safe and well tolerated, with similar incidence of adverse events between active and placebo-treated groups
Study will continue in blinded format to 24 months |
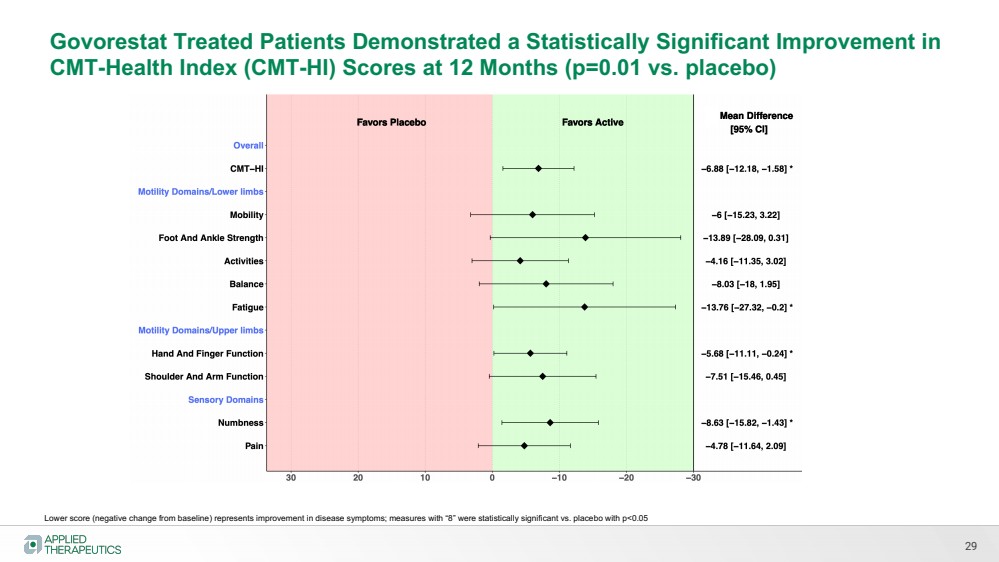
| 29
Govorestat Treated Patients Demonstrated a Statistically Significant Improvement in
CMT-Health Index (CMT-HI) Scores at 12 Months (p=0.01 vs. placebo)
Lower score (negative change from baseline) represents improvement in disease symptoms; measures with “8” were statistically significant vs. placebo with p<0.05 |
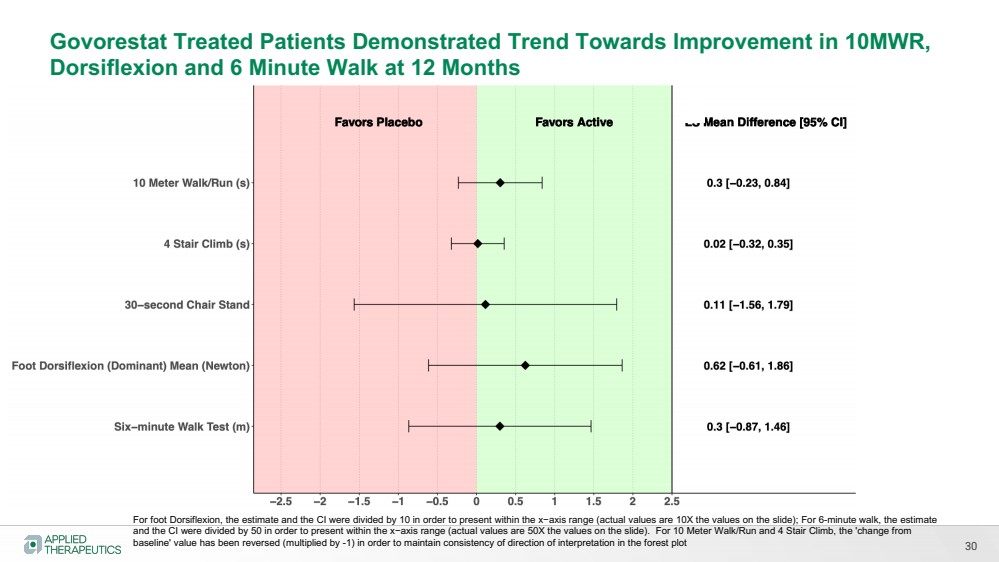
| 30
Govorestat Treated Patients Demonstrated Trend Towards Improvement in 10MWR,
Dorsiflexion and 6 Minute Walk at 12 Months
For foot Dorsiflexion, the estimate and the CI were divided by 10 in order to present within the x−axis range (actual values are 10X the values on the slide); For 6-minute walk, the estimate
and the CI were divided by 50 in order to present within the x−axis range (actual values are 50X the values on the slide). For 10 Meter Walk/Run and 4 Stair Climb, the 'change from
baseline' value has been reversed (multiplied by -1) in order to maintain consistency of direction of interpretation in the forest plot |
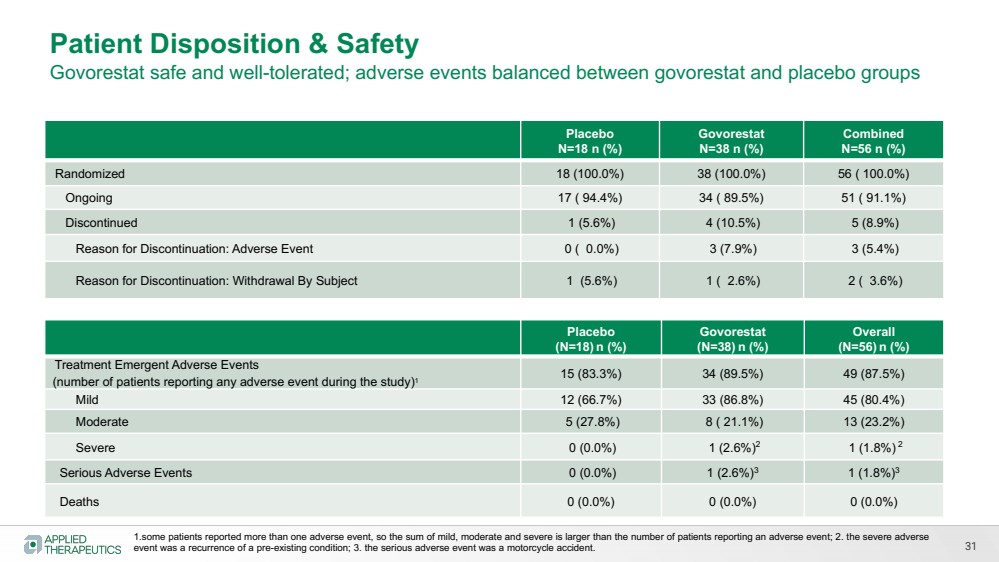
| 31
Placebo
N=18 n (%)
Govorestat
N=38 n (%)
Combined
N=56 n (%)
Randomized 18 (100.0%) 38 (100.0%) 56 ( 100.0%)
Ongoing 17 ( 94.4%) 34 ( 89.5%) 51 ( 91.1%)
Discontinued 1 (5.6%) 4 (10.5%) 5 (8.9%)
Reason for Discontinuation: Adverse Event 0 ( 0.0%) 3 (7.9%) 3 (5.4%)
Reason for Discontinuation: Withdrawal By Subject 1 (5.6%) 1 ( 2.6%) 2 ( 3.6%)
Patient Disposition & Safety
Govorestat safe and well-tolerated; adverse events balanced between govorestat and placebo groups
Placebo
(N=18) n (%)
Govorestat
(N=38) n (%)
Overall
(N=56) n (%)
Treatment Emergent Adverse Events
(number of patients reporting any adverse event during the study)1 15 (83.3%) 34 (89.5%) 49 (87.5%)
Mild 12 (66.7%) 33 (86.8%) 45 (80.4%)
Moderate 5 (27.8%) 8 ( 21.1%) 13 (23.2%)
Severe 0 (0.0%) 1 (2.6%)2 1 (1.8%) 2
Serious Adverse Events 0 (0.0%) 1 (2.6%)3 1 (1.8%)3
Deaths 0 (0.0%) 0 (0.0%) 0 (0.0%)
1.some patients reported more than one adverse event, so the sum of mild, moderate and severe is larger than the number of patients reporting an adverse event; 2. the severe adverse
event was a recurrence of a pre-existing condition; 3. the serious adverse event was a motorcycle accident. |
| 32
Inherited Neuropathy Consortium Centers of Excellence and Global
CMT Registries Exist to Support Diagnosis and Treatment
University of Pennsylvania School of Medicine
Children’s Hospital of Philadelphia
Centers participating in INSPIRE Phase 2/3 trial
www.rarediseasesnetwork.org/cms/inc/centers#CSMC.
Stanford University
Cedars-Sinai Medical Center
University of Colorado
Massachusetts General Hospital, Boston
Connecticut Children’s Medical Center
University of
Minnesota
University of Iowa, Iowa City
Wayne State University, Detroit
Johns Hopkins University,
Baltimore
University of Rochester
University of Miami
Nemours Children's Clinic
National Hospital for Neurology
University College London
Greater Ormond St.
Hospital for Children,
London
C. Fondazione IRCCS Istituto Neurologica
Carlo Besta, Milan
Charles University
Hospital, Prague
US UK & EU
UK
IT
CZ |
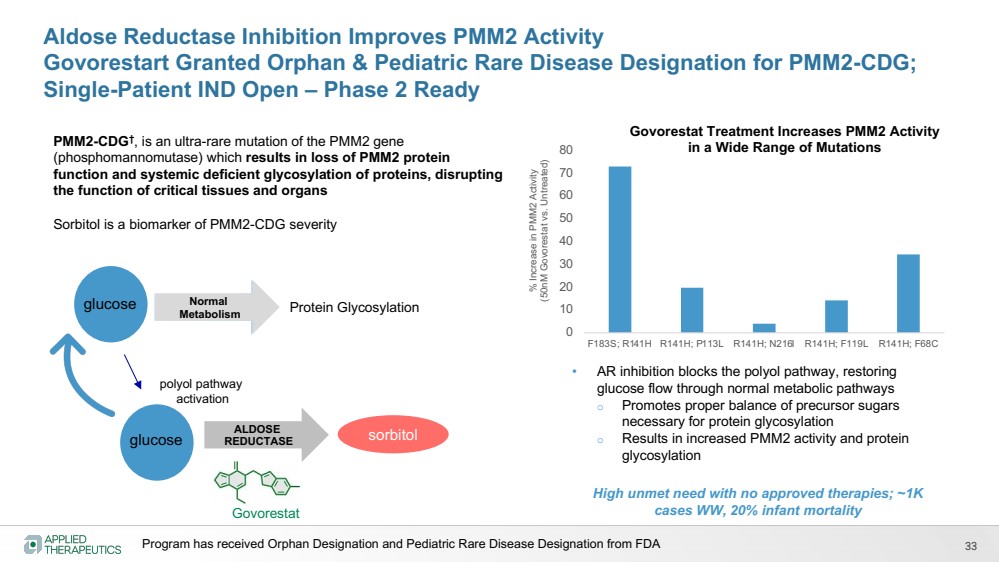
| 33
Aldose Reductase Inhibition Improves PMM2 Activity
Govorestart Granted Orphan & Pediatric Rare Disease Designation for PMM2-CDG;
Single-Patient IND Open – Phase 2 Ready
sorbitol
glucose Normal
Metabolism
ALDOSE
REDUCTASE
Protein Glycosylation
polyol pathway
activation
Govorestat
• AR inhibition blocks the polyol pathway, restoring
glucose flow through normal metabolic pathways
o Promotes proper balance of precursor sugars
necessary for protein glycosylation
o Results in increased PMM2 activity and protein
glycosylation
glucose
% Increase in PMM2 Activity
(50nM Govorestat vs. Untreated)
0
10
20
30
40
50
60
70
80
F183S; R141H R141H; P113L R141H; N216I R141H; F119L R141H; F68C
Govorestat Treatment Increases PMM2 Activity
in a Wide Range of Mutations PMM2-CDG†, is an ultra-rare mutation of the PMM2 gene
(phosphomannomutase) which results in loss of PMM2 protein
function and systemic deficient glycosylation of proteins, disrupting
the function of critical tissues and organs
Sorbitol is a biomarker of PMM2-CDG severity
High unmet need with no approved therapies; ~1K
cases WW, 20% infant mortality
Program has received Orphan Designation and Pediatric Rare Disease Designation from FDA |

| AT-001
DIABETIC
CARDIOMYOPATHY
Phase 1/2 pilot study completed
Phase 3 study completed |

| Diabetic Cardiomyopathy (DbCM) is a Form of Heart Failure Affecting ~20%
of Diabetics; Significant Unmet Need with No Approved Treatments
Disease Overview
• Form of Heart Failure (Stage B) causing structural
cardiac damage and resulting in decreased cardiac
functional capacity
• Affects ~20% of diabetics
• Diagnosed by echocardiogram or elevated cardiac
biomarkers (NTproBNP or troponin)
Standard of Care
• No approved therapies to treat DbCM or prevent
progression to overt heart failure/ death
• Once DbCM patients have developed overt HF, they are
eligible for standard HF therapies in addition to standard
diabetes treatments
Mechanism of Disease
• Hyperactivation of the polyol pathway is a key underlying
mechanism in DbCM
• Aldose Reductase activation causes intracellular sorbitol
accumulation, osmotic stress, cell death, generation of
ROS and impaired cardiac energetics
• Previous AR inhibitors demonstrated clinical efficacy, but
were associated with off-target safety signals due to lack
of selectivity
Market Size/ Opportunity
• Blockbuster potential
• Addressable population of ~6M patients US, 5M in EU5
• Anticipated pricing in line with Entresto & SGLT2s
• Composition of matter IP through 2031 (not including
extensions)
Brownlee M. Diabetes Care. 2005;54(6):1615-1625; Miki T, et al. Heart Fail Rev. 2013;18(2):149-166. |
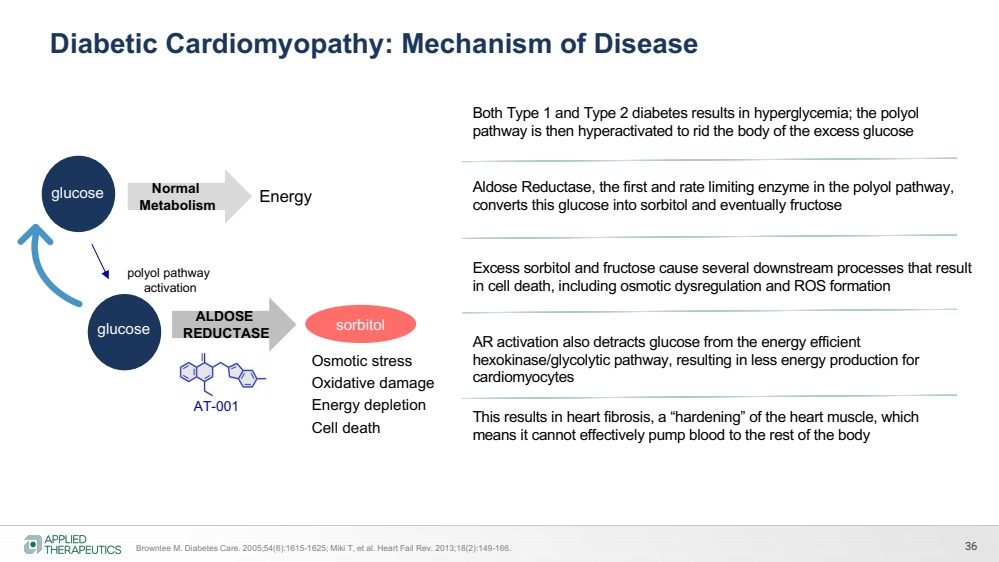
| Aldose Reductase, the first and rate limiting enzyme in the polyol pathway,
converts this glucose into sorbitol and eventually fructose
Both Type 1 and Type 2 diabetes results in hyperglycemia; the polyol
pathway is then hyperactivated to rid the body of the excess glucose
Excess sorbitol and fructose cause several downstream processes that result
in cell death, including osmotic dysregulation and ROS formation
This results in heart fibrosis, a “hardening” of the heart muscle, which
means it cannot effectively pump blood to the rest of the body
36
Diabetic Cardiomyopathy: Mechanism of Disease
Brownlee M. Diabetes Care. 2005;54(6):1615-1625; Miki T, et al. Heart Fail Rev. 2013;18(2):149-166.
AR activation also detracts glucose from the energy efficient
hexokinase/glycolytic pathway, resulting in less energy production for
cardiomyocytes
sorbitol
glucose Normal
Metabolism
ALDOSE
REDUCTASE
Energy
polyol pathway
activation
AT-001
glucose
Osmotic stress
Oxidative damage
Energy depletion
Cell death |
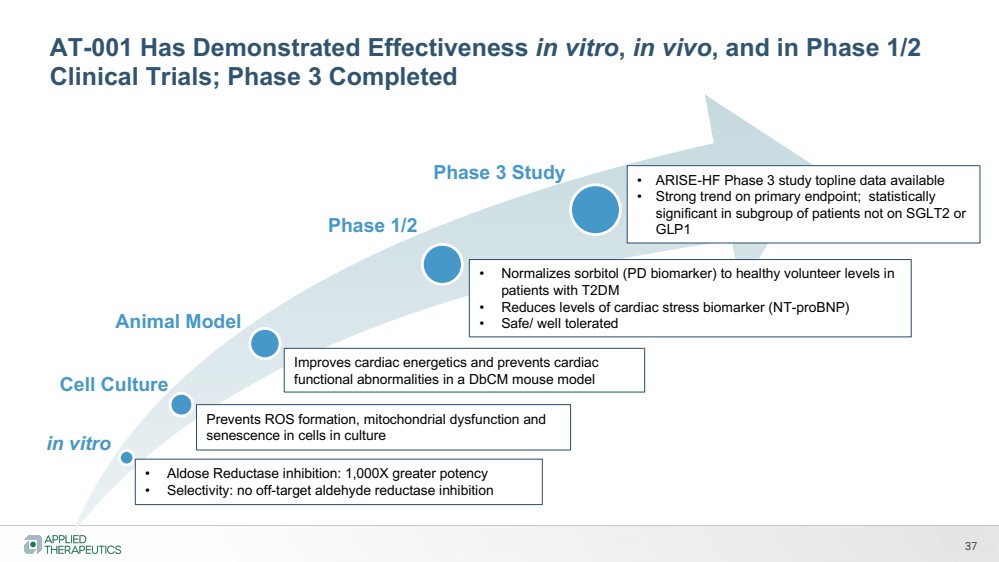
| 37
in vitro
Cell Culture
Phase 1/2
Phase 3 Study
• Aldose Reductase inhibition: 1,000X greater potency
• Selectivity: no off-target aldehyde reductase inhibition
Prevents ROS formation, mitochondrial dysfunction and
senescence in cells in culture
Animal Model
Improves cardiac energetics and prevents cardiac
functional abnormalities in a DbCM mouse model
• Normalizes sorbitol (PD biomarker) to healthy volunteer levels in
patients with T2DM
• Reduces levels of cardiac stress biomarker (NT-proBNP)
• Safe/ well tolerated
• ARISE-HF Phase 3 study topline data available
• Strong trend on primary endpoint; statistically
significant in subgroup of patients not on SGLT2 or
GLP1
AT-001 Has Demonstrated Effectiveness in vitro, in vivo, and in Phase 1/2
Clinical Trials; Phase 3 Completed |
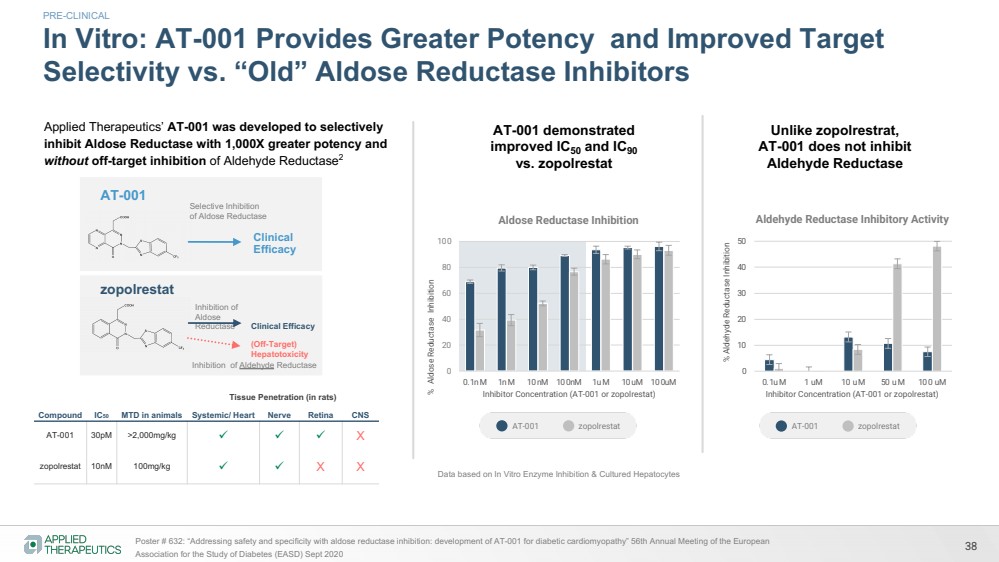
| 38 Poster # 632: “Addressing safety and specificity with aldose reductase inhibition: development of AT-001 for diabetic cardiomyopathy” 56th Annual Meeting of the European
Association for the Study of Diabetes (EASD) Sept 2020
In Vitro: AT-001 Provides Greater Potency and Improved Target
Selectivity vs. “Old” Aldose Reductase Inhibitors
0
20
40
60
80
100
0.1nM 1nM 10nM 100nM 1uM 10uM 100uM
% Aldose Reductase Inhibition
Inhibitor Concentration (AT-001 or zopolrestat)
Aldose Reductase Inhibition
0
10
20
30
40
50
0.1uM 1 uM 10 uM 50 uM 100 uM
% Aldehyde Reductase Inhibition
Inhibitor Concentration (AT-001 or zopolrestat)
Aldehyde Reductase Inhibitory Activity
PRE-CLINICAL
AT-001 demonstrated
improved IC50 and IC90
vs. zopolrestat
Unlike zopolrestrat,
AT-001 does not inhibit
Aldehyde Reductase
AT-001 zopolrestat AT-001 zopolrestat
Data based on In Vitro Enzyme Inhibition & Cultured Hepatocytes
Compound IC50 MTD in animals
Tissue Penetration (in rats)
Systemic/ Heart Nerve Retina CNS
AT-001 30pM >2,000mg/kg ü ü ü X
zopolrestat 10nM 100mg/kg ü ü X X
zopolrestat
Inhibition of
Aldose
Reductase Clinical Efficacy
Inhibition of Aldehyde Reductase
(Off-Target)
Hepatotoxicity
AT-001 Selective Inhibition
of Aldose Reductase
Clinical
Efficacy
Applied Therapeutics’ AT-001 was developed to selectively
inhibit Aldose Reductase with 1,000X greater potency and
without off-target inhibition of Aldehyde Reductase2 |
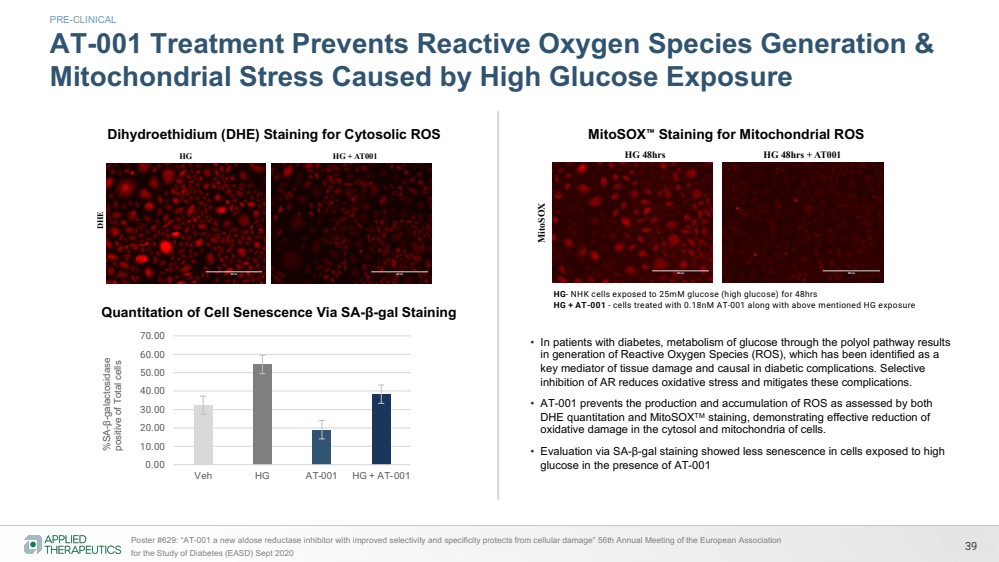
| Dihydroethidium(DHE) staining for cytosolic reactive oxygen species
HG HG + AT001
DHE HG- NHK cells exposed to 25mM glucose for 48hrs
HG + AT001 - cells treated with 0.18nM AT001 along with above mentioned HG exposure
Cytosolic reactive oxygen species attenuated by AT 001 in skin cells treated with high glucose
MitoSOX staining for Mitochondrial reactive oxygen species
MitoSOX
HG 48hrs HG 48hrs + AT001
HG- NHK cells exposed to 25mM glucose for 48hrs
HG + AT001 - cells treated with 0.18nM AT001 along with above mentioned HG exposure
Mitochondrial reactive oxygen species attenuated by AT 001 in skin cells treated with high glucose
39 Poster #629: “AT-001 a new aldose reductase inhibitor with improved selectivity and specificity protects from cellular damage” 56th Annual Meeting of the European Association
for the Study of Diabetes (EASD) Sept 2020
AT-001 Treatment Prevents Reactive Oxygen Species Generation &
Mitochondrial Stress Caused by High Glucose Exposure %SA-β-galactosidase positive of Total cells
0.00
10.00
20.00
30.00
40.00
50.00
60.00
70.00
Veh HG AT-001 HG + AT-001
Quantitation of Cell Senescence Via SA-β-gal Staining
HG- NHK cells exposed to 25mM glucose (high glucose) for 48hrs
HG + AT-001 - cells treated with 0.18nM AT-001 along with above mentioned HG exposure
• In patients with diabetes, metabolism of glucose through the polyol pathway results
in generation of Reactive Oxygen Species (ROS), which has been identified as a
key mediator of tissue damage and causal in diabetic complications. Selective
inhibition of AR reduces oxidative stress and mitigates these complications.
• AT-001 prevents the production and accumulation of ROS as assessed by both
DHE quantitation and MitoSOXTM staining, demonstrating effective reduction of
oxidative damage in the cytosol and mitochondria of cells.
• Evaluation via SA-β-gal staining showed less senescence in cells exposed to high
glucose in the presence of AT-001
Dihydroethidium (DHE) Staining for Cytosolic ROS MitoSOX™ Staining for Mitochondrial ROS
PRE-CLINICAL |
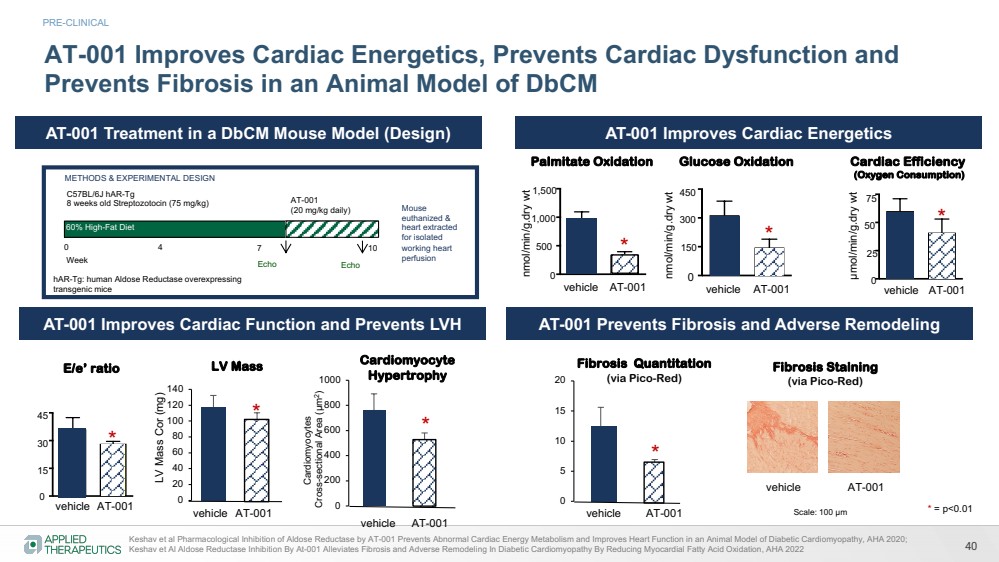
| 40
0
5
10
15
20
0
20
40
60
80
100
120
140
vehicle AT-001
METHODS & EXPERIMENTAL DESIGN
60% High-Fat Diet
Week
Mouse
euthanized &
heart extracted
for isolated
working heart
perfusion Echo Echo
AT-001
(20 mg/kg daily)
C57BL/6J hAR-Tg
8 weeks old Streptozotocin (75 mg/kg)
hAR-Tg: human Aldose Reductase overexpressing
transgenic mice
0 4 7 10
AT-001 Improves Cardiac Energetics, Prevents Cardiac Dysfunction and
Prevents Fibrosis in an Animal Model of DbCM
µmol/min/g.dry wt
Cardiac Efficiency
(Oxygen Consumption)
0
50
25
75
vehicle AT-001
Glucose Oxidation
nmol/min/g.dry wt
0
300
150
450
vehicle AT-001
nmol/min/g.dry wt
Palmitate Oxidation
0
500
1,000
1,500
vehicle AT-001
E/e’ ratio
0
15
30
45
vehicle AT-001
*
*
*
*
* = p<0.01
PRE-CLINICAL
AT-001 Improves Cardiac Energetics
AT-001 Improves Cardiac Function and Prevents LVH AT-001 Prevents Fibrosis and Adverse Remodeling
AT-001 Treatment in a DbCM Mouse Model (Design) LV Mass Cor (mg)
LV Mass
*
0
200
400
600
800
1000
*
Cardiomyocyte
Hypertrophy
Cardiomyocytes
Cross-sectional Area (µm2)
vehicle AT-001
Fibrosis Quantitation
(via Pico-Red)
*
vehicle AT-001
Keshav et al Pharmacological Inhibition of Aldose Reductase by AT-001 Prevents Abnormal Cardiac Energy Metabolism and Improves Heart Function in an Animal Model of Diabetic Cardiomyopathy, AHA 2020;
Keshav et Al Aldose Reductase Inhibition By At-001 Alleviates Fibrosis and Adverse Remodeling In Diabetic Cardiomyopathy By Reducing Myocardial Fatty Acid Oxidation, AHA 2022
Scale: 100 µm
vehicle AT-001
Fibrosis Staining
(via Pico-Red) |

| DbCM Phase 3 Study (ARISE-HF) Design
Randomized, Placebo-Controlled Study in DbCM Patients at High Risk of Progression
Efficacy (15 Months)
Primary Endpoint: Cardiac Functional Capacity (as
measured by Peak VO2 change from baseline)
Prespecified subgroup analysis of patients not on
SGLT2 or GLP1
Secondary Endpoints:
• Clinically significant worsening (>6% D in Peak VO2)
• NT-ProBNP
• KCCQ
• PASE
1:1:1 Randomization
Placebo
• Enrolled patients with DbCM at high risk of
progression to overt heart failure
• n=675 (225/arm)
• Twice-daily oral dosing
• Add-on to standard of care diabetes
therapies
AT-001
1,000
mg
AT-001
1,500
mg
41
PHASE 3
Key Inclusion Criteria:
• Diagnosis of Type 2 Diabetes
• Age: ≥60 years, or ≥40 years with duration of diabetes >10
years
• Demonstration of DbCM/ Stage B Heart Failure
• LVEF> 45% and at least one of the following:
echocardiographic abnormalities or NTProBNP > 50 pg/ml, or
HsTNT > 6 ng/L
• RER > 1.05
• Peak VO2 <75% of age/gender predicted normal
Key Exclusion Criteria:
• Diagnosis or signs of overt/symptomatic heart failure
• Use of a loop diuretic
• History of CAD, MI, ACS, CABG, PCI, stroke
• History of severe valve disease, clinically significant
arrhythmia, or other cause of cardiomyopathy
• Severe disease impacting implementation of the protocol or
performance of a CPET
• SBP >140 mmHg or DBP >90 mmHg
• BMI >45 kg/m2
• HbA1c >8.5%
• eGFR <45 mL/min/1.73 m2 |
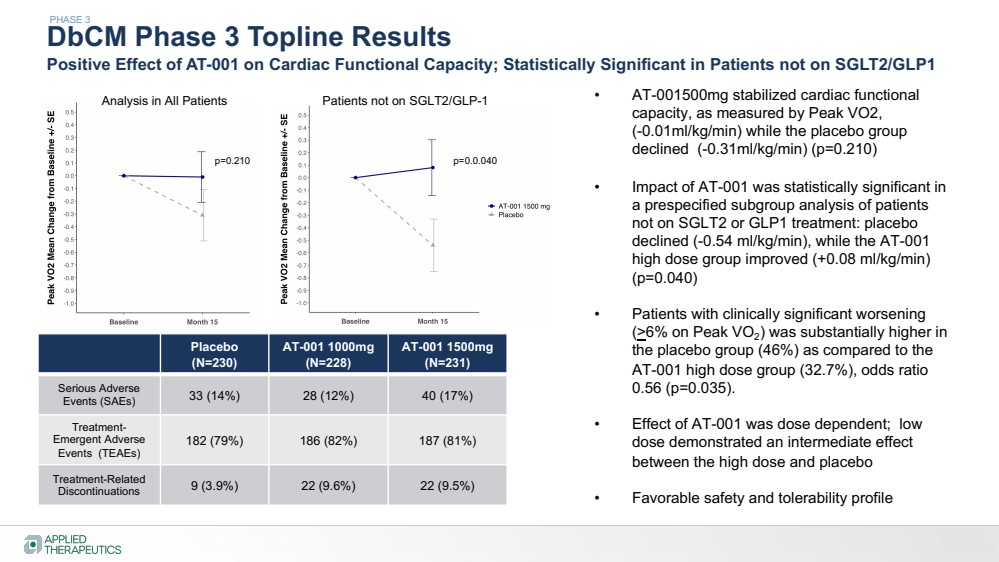
| DbCM Phase 3 Topline Results
Positive Effect of AT-001 on Cardiac Functional Capacity; Statistically Significant in Patients not on SGLT2/GLP1
PHASE 3
Placebo
(N=230)
AT-001 1000mg
(N=228)
AT-001 1500mg
(N=231)
Serious Adverse
Events (SAEs) 33 (14%) 28 (12%) 40 (17%)
Treatment-Emergent Adverse
Events (TEAEs)
182 (79%) 186 (82%) 187 (81%)
Treatment-Related
Discontinuations 9 (3.9%) 22 (9.6%) 22 (9.5%)
• AT-001500mg stabilized cardiac functional
capacity, as measured by Peak VO2,
(-0.01ml/kg/min) while the placebo group
declined (-0.31ml/kg/min) (p=0.210)
• Impact of AT-001 was statistically significant in
a prespecified subgroup analysis of patients
not on SGLT2 or GLP1 treatment: placebo
declined (-0.54 ml/kg/min), while the AT-001
high dose group improved (+0.08 ml/kg/min)
(p=0.040)
• Patients with clinically significant worsening
(>6% on Peak VO2) was substantially higher in
the placebo group (46%) as compared to the
AT-001 high dose group (32.7%), odds ratio
0.56 (p=0.035).
• Effect of AT-001 was dose dependent; low
dose demonstrated an intermediate effect
between the high dose and placebo
• Favorable safety and tolerability profile
Analysis in All Patients Patients not on SGLT2/GLP-1
p=0.210 p=0.0.040 |
| 43
Applying Science to Transform Lives
Our mission is to create transformative, life-changing treatments for patients who
desperately need them
Targeting pathways with
known roles in pathogenesis
Novel compounds with
improved potency/selectivity
SCIENCE
Clinical efficacy
confirmed via biomarkers
Pursuing expedited
regulatory pathways
Fatal or debilitating diseases
with no approved therapies
Limited / no competition
DEVELOPMENT MARKET |
Exhibit 99.2

| Applied Therapeutics
SORD 12 Month Interim Data Analysis
March 2024 |

| Various statements in this presentation concerning the Company’s future expectations, plans and prospects constitute forward-looking statements. The use of
words such as “may,” “might,” “will,” “should,” “expect,” “plan,” “anticipate,” “believe,” “estimate,” “project,” “intend,” “future,” “potential,” or “continue,” the negative
of these and other similar expressions are intended to identify such forward looking statements. Such statements, based as they are on the current analysis and
expectations of management, inherently involve numerous risks and uncertainties, known and unknown, many of which are beyond the Company’s control. Such
risks include, but are not limited to: the impact of general economic conditions, general conditions in the biopharmaceutical industries, changes in the global and
regional regulatory environments in the jurisdictions in which the Company does or plans to do business, market volatility, fluctuations in costs and changes to the
competitive environment, the Company’s ability to fund its working capital requirements and expectations regarding the sufficiency of our capital resources and the
Company’s ability to achieve the anticipated benefits from the agreements entered into in connection with our partnership with Advanz Pharma. Consequently,
actual future results may differ materially from the anticipated results expressed in the forward-looking statements. In the case of forward-looking statements
regarding investigational product candidates and continuing further development efforts, specific risks which could cause actual results to differ materially from the
Company’s current analysis and expectations include: failure to demonstrate the safety, tolerability and efficacy of our product candidates; final and quality
controlled verification of data and the related analyses; the expense and uncertainty of obtaining regulatory approval, including from the U.S. Food and Drug
Administration and European Medicines Agency; the possibility of having to conduct additional clinical trials and our reliance on third parties such as our licensors
and collaboration partners regarding our suite of technologies and product candidates; the Company’s ability to take advantage of expedited regulatory pathways
for any of our product candidates; the Company’s intellectual property position and the duration of its patent rights; developments or disputes concerning the
Company’s intellectual property or other proprietary rights. Further, even if regulatory approval is obtained, biopharmaceutical products are generally subject to
stringent on-going governmental regulation, challenges in gaining market acceptance and competition.
These risks and uncertainties are described more fully under the caption ”Risk Factors” in the Company’s filings with the Securities and Exchange Commission.
Other risks and uncertainties of which the Company is not currently aware may also affect Company’s forward-looking statements. The reader should not place
undue reliance on any forward-looking statements included in this presentation. These statements speak only as of the date made and the Company is under no
obligation and disavows any obligation to update or revise such statements as a result of any event, circumstances or otherwise, unless required by applicable
legislation or regulation.
Forward Looking Statements |

| INSPIRE Trial 12 Month Interim Topline Data
3
Co-primary endpoints at 12 month analysis:
• Primary clinical efficacy endpoint: Statistically significant correlation between sorbitol levels and change in clinical outcomes
at 12 months of treatment on combined measures of the CMT Functional Outcome Measures (CMT-FOM) lower limb domain
(10 meter walk-run test, 4 stair climb, and sit to stand test), 6-minute walk test and dorsiflexion (p=0.05)
• Primary pharmacodynamic/ biomarker endpoint: Sustained reduction in sorbitol level in patients treated with govorestat at
12 months, which was statistically significant compared to placebo (p<0.001).
Secondary Endpoints
• Highly statistically significant effect (p=0.01) impact of govorestat on the CMT Health Index (CMT-HI), an important patient-reported outcome measure of disease severity and well-being; aspects of the CMT-HI that demonstrated a treatment effect
included lower limb function, mobility, fatigue, pain, sensory function, and upper limb function.
• Trends (not statistically significant) on CMT-FOM measures linked to walking ability, such as 10MWR, dorsiflexion and 6 minute
walk test
o No substantial effect on stair climb or sit-to-stand test
Safety
• Govorestat was safe and well tolerated, with similar incidence of adverse events between active and placebo-treated groups
Study will continue in blinded format to 24 months |
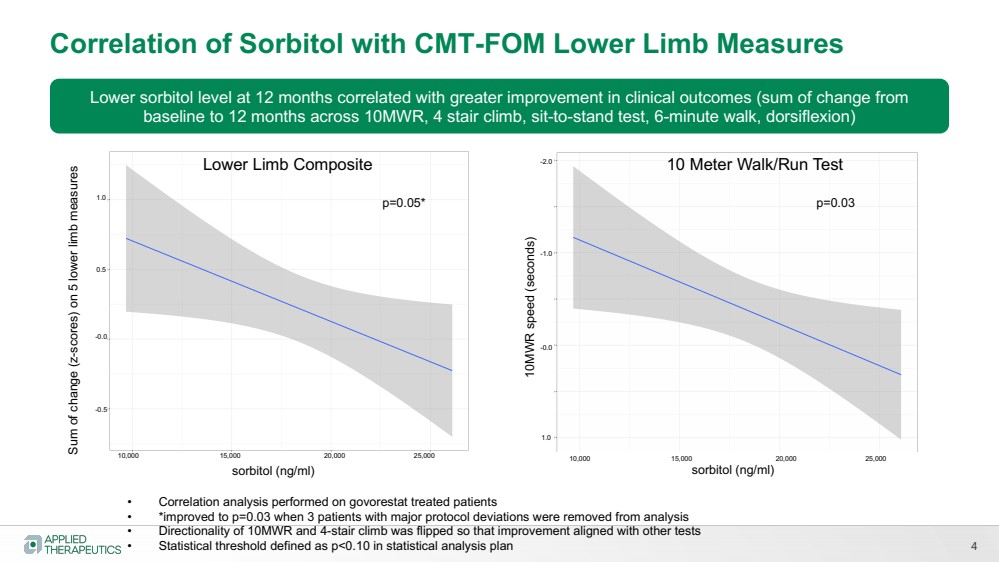
| 4
Correlation of Sorbitol with CMT-FOM Lower Limb Measures
sorbitol (ng/ml)
Sum of change (z-scores) on 5 lower limb measures
Lower sorbitol level at 12 months correlated with greater improvement in clinical outcomes (sum of change from
baseline to 12 months across 10MWR, 4 stair climb, sit-to-stand test, 6-minute walk, dorsiflexion)
• Correlation analysis performed on govorestat treated patients
• *improved to p=0.03 when 3 patients with major protocol deviations were removed from analysis
• Directionality of 10MWR and 4-stair climb was flipped so that improvement aligned with other tests
• Statistical threshold defined as p<0.10 in statistical analysis plan
p=0.05*
10,000 15,000 20,000 25,000
-0.5
-0.0
0.5
1.0
Lower Limb Composite
sorbitol (ng/ml)
10MWR speed (seconds)
10 Meter Walk/Run Test
p=0.03
10,000 15,000 20,000 25,000
1.0
-0.0
-1.0
-2.0 |
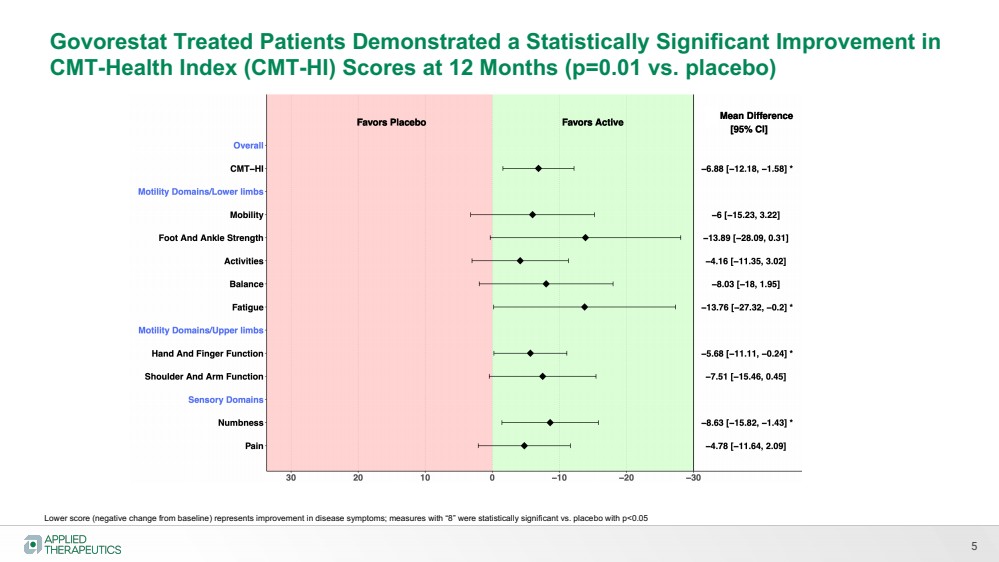
| 5
Govorestat Treated Patients Demonstrated a Statistically Significant Improvement in
CMT-Health Index (CMT-HI) Scores at 12 Months (p=0.01 vs. placebo)
Lower score (negative change from baseline) represents improvement in disease symptoms; measures with “8” were statistically significant vs. placebo with p<0.05 |
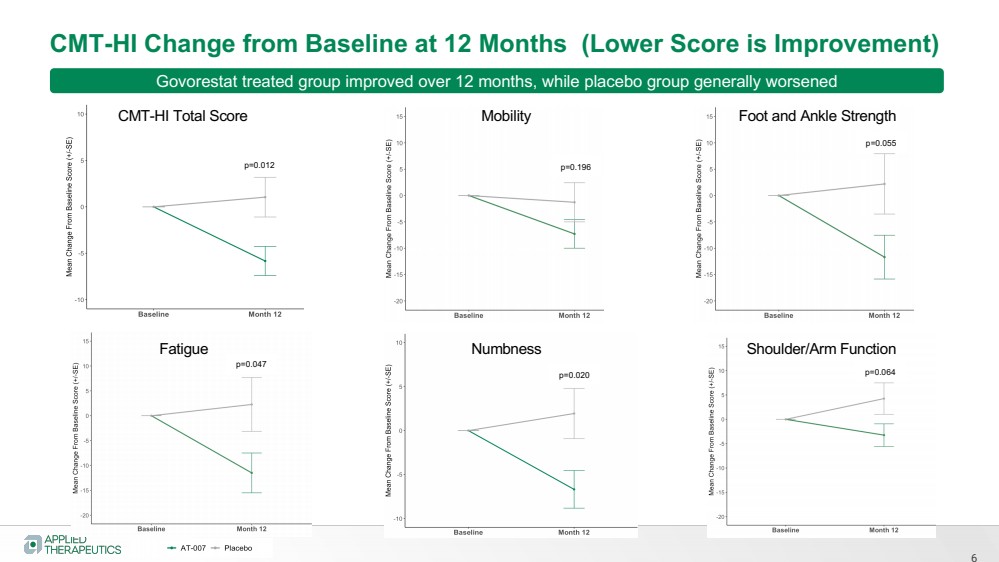
| 6
CMT-HI Change from Baseline at 12 Months (Lower Score is Improvement)
CMT-HI Total Score Mobility Foot and Ankle Strength
Fatigue Numbness Shoulder/Arm Function
p=0.012 p=0.196
p=0.055
p=0.064 p=0.020
p=0.047
Govorestat treated group improved over 12 months, while placebo group generally worsened |
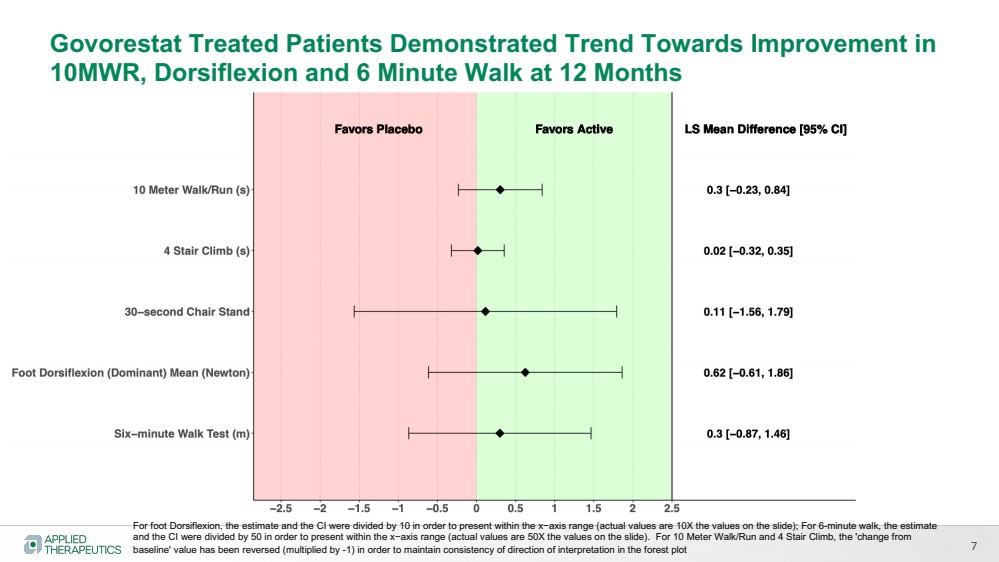
| 7
Govorestat Treated Patients Demonstrated Trend Towards Improvement in
10MWR, Dorsiflexion and 6 Minute Walk at 12 Months
For foot Dorsiflexion, the estimate and the CI were divided by 10 in order to present within the x−axis range (actual values are 10X the values on the slide); For 6-minute walk, the estimate
and the CI were divided by 50 in order to present within the x−axis range (actual values are 50X the values on the slide). For 10 Meter Walk/Run and 4 Stair Climb, the 'change from
baseline' value has been reversed (multiplied by -1) in order to maintain consistency of direction of interpretation in the forest plot |
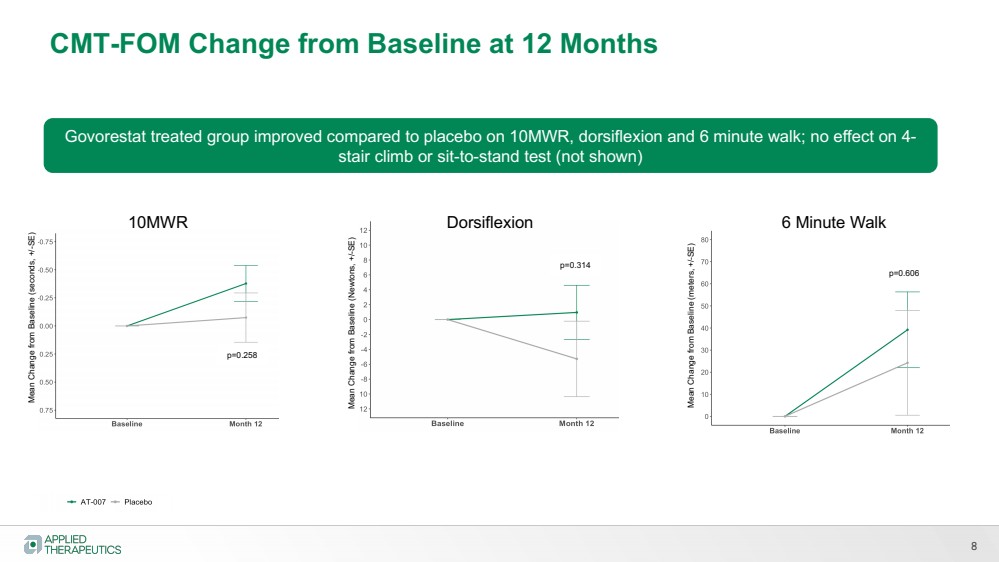
| 8
CMT-FOM Change from Baseline at 12 Months
10MWR
Mean Change from Baseline (seconds, +/-SE)
Mean Change from Baseline (Newtons, +/-SE)
Dorsiflexion 6 Minute Walk Mean Change from Baseline (meters, +/-SE)
p=0.258
p=0.314 p=0.606
Govorestat treated group improved compared to placebo on 10MWR, dorsiflexion and 6 minute walk; no effect on 4-
stair climb or sit-to-stand test (not shown) |
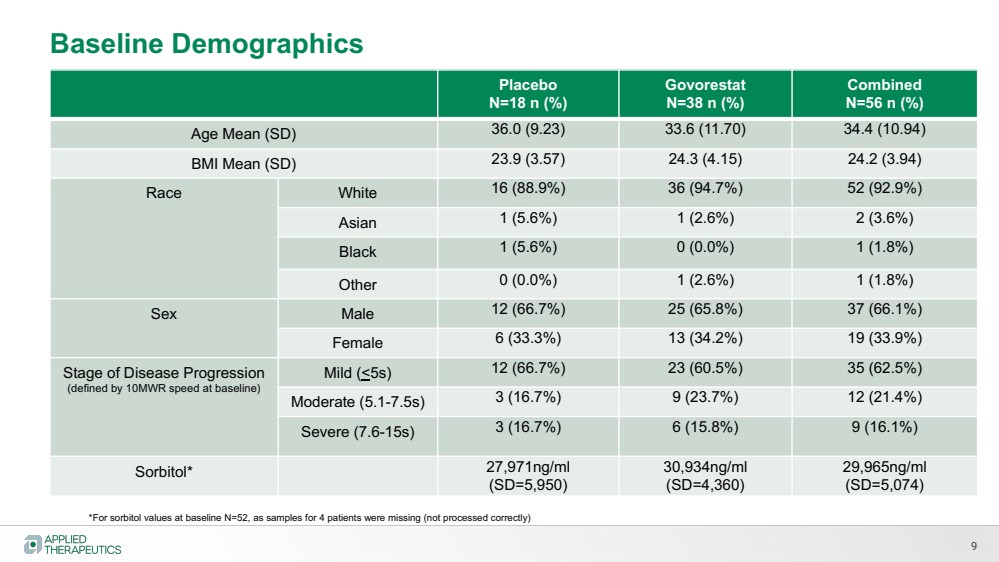
| 9
Placebo
N=18 n (%)
Govorestat
N=38 n (%)
Combined
N=56 n (%)
Age Mean (SD) 36.0 (9.23) 33.6 (11.70) 34.4 (10.94)
BMI Mean (SD) 23.9 (3.57) 24.3 (4.15) 24.2 (3.94)
Race White 16 (88.9%) 36 (94.7%) 52 (92.9%)
Asian 1 (5.6%) 1 (2.6%) 2 (3.6%)
Black 1 (5.6%) 0 (0.0%) 1 (1.8%)
Other 0 (0.0%) 1 (2.6%) 1 (1.8%)
Sex Male 12 (66.7%) 25 (65.8%) 37 (66.1%)
Female 6 (33.3%) 13 (34.2%) 19 (33.9%)
Stage of Disease Progression
(defined by 10MWR speed at baseline)
Mild (<5s) 12 (66.7%) 23 (60.5%) 35 (62.5%)
Moderate (5.1-7.5s) 3 (16.7%) 9 (23.7%) 12 (21.4%)
Severe (7.6-15s) 3 (16.7%) 6 (15.8%) 9 (16.1%)
Sorbitol* 27,971ng/ml
(SD=5,950)
30,934ng/ml
(SD=4,360)
29,965ng/ml
(SD=5,074)
Baseline Demographics
*For sorbitol values at baseline N=52, as samples for 4 patients were missing (not processed correctly) |
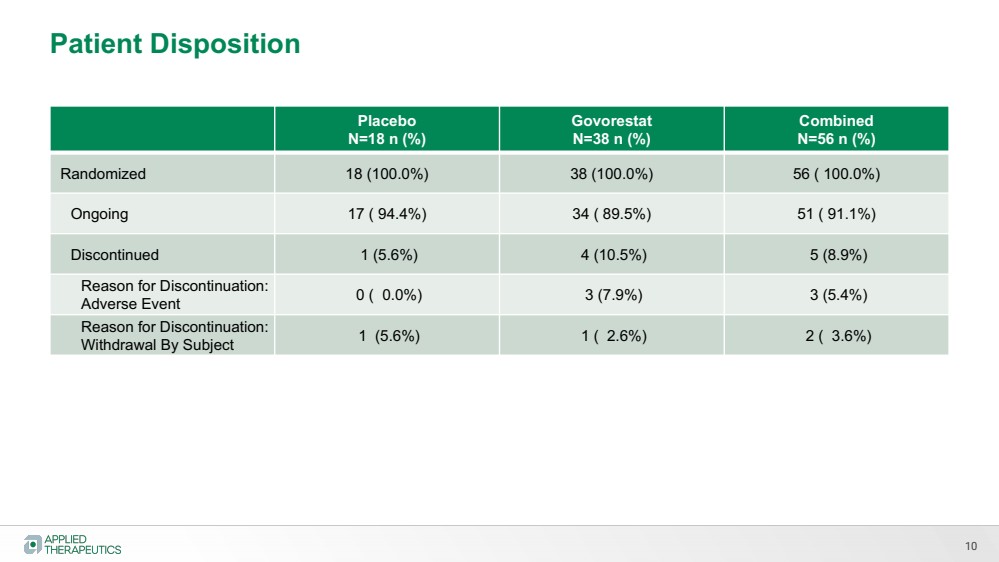
| 10
Placebo
N=18 n (%)
Govorestat
N=38 n (%)
Combined
N=56 n (%)
Randomized 18 (100.0%) 38 (100.0%) 56 ( 100.0%)
Ongoing 17 ( 94.4%) 34 ( 89.5%) 51 ( 91.1%)
Discontinued 1 (5.6%) 4 (10.5%) 5 (8.9%)
Reason for Discontinuation:
Adverse Event 0 ( 0.0%) 3 (7.9%) 3 (5.4%)
Reason for Discontinuation:
Withdrawal By Subject 1 (5.6%) 1 ( 2.6%) 2 ( 3.6%)
Patient Disposition |
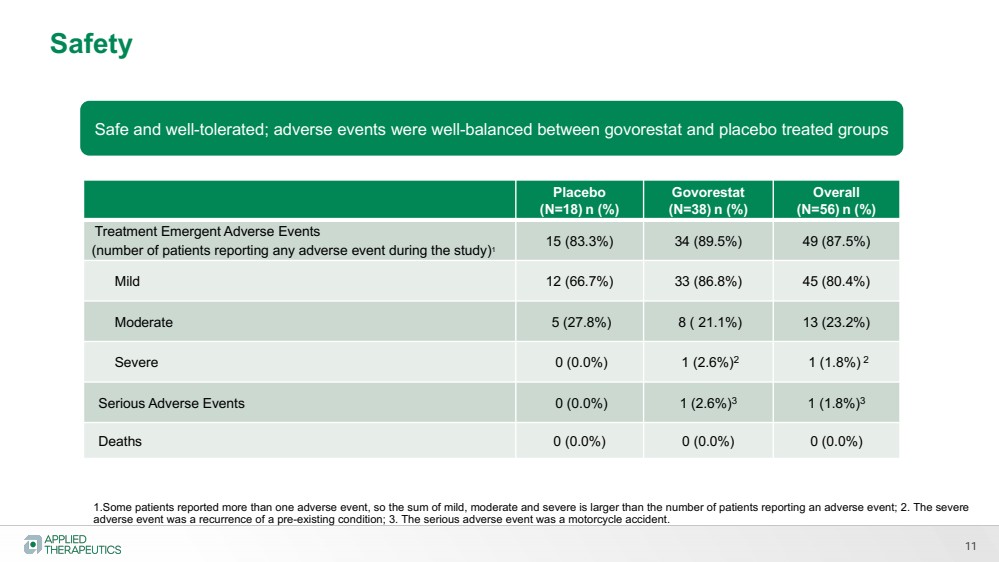
| 11
Placebo
(N=18) n (%)
Govorestat
(N=38) n (%)
Overall
(N=56) n (%)
Treatment Emergent Adverse Events
(number of patients reporting any adverse event during the study)1 15 (83.3%) 34 (89.5%) 49 (87.5%)
Mild 12 (66.7%) 33 (86.8%) 45 (80.4%)
Moderate 5 (27.8%) 8 ( 21.1%) 13 (23.2%)
Severe 0 (0.0%) 1 (2.6%)2 1 (1.8%) 2
Serious Adverse Events 0 (0.0%) 1 (2.6%)3 1 (1.8%)3
Deaths 0 (0.0%) 0 (0.0%) 0 (0.0%)
Safety
1.Some patients reported more than one adverse event, so the sum of mild, moderate and severe is larger than the number of patients reporting an adverse event; 2. The severe
adverse event was a recurrence of a pre-existing condition; 3. The serious adverse event was a motorcycle accident.
Safe and well-tolerated; adverse events were well-balanced between govorestat and placebo treated groups |
| X |
- DefinitionBoolean flag that is true when the XBRL content amends previously-filed or accepted submission.
| Name: |
dei_AmendmentFlag |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionFor the EDGAR submission types of Form 8-K: the date of the report, the date of the earliest event reported; for the EDGAR submission types of Form N-1A: the filing date; for all other submission types: the end of the reporting or transition period. The format of the date is YYYY-MM-DD.
| Name: |
dei_DocumentPeriodEndDate |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:dateItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe type of document being provided (such as 10-K, 10-Q, 485BPOS, etc). The document type is limited to the same value as the supporting SEC submission type, or the word 'Other'.
| Name: |
dei_DocumentType |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:submissionTypeItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionAddress Line 1 such as Attn, Building Name, Street Name
| Name: |
dei_EntityAddressAddressLine1 |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionAddress Line 2 such as Street or Suite number
| Name: |
dei_EntityAddressAddressLine2 |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- Definition
+ References
+ Details
| Name: |
dei_EntityAddressCityOrTown |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionCode for the postal or zip code
| Name: |
dei_EntityAddressPostalZipCode |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionName of the state or province.
| Name: |
dei_EntityAddressStateOrProvince |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:stateOrProvinceItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionA unique 10-digit SEC-issued value to identify entities that have filed disclosures with the SEC. It is commonly abbreviated as CIK. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b-2
| Name: |
dei_EntityCentralIndexKey |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:centralIndexKeyItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionIndicate if registrant meets the emerging growth company criteria. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b-2
| Name: |
dei_EntityEmergingGrowthCompany |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionCommission file number. The field allows up to 17 characters. The prefix may contain 1-3 digits, the sequence number may contain 1-8 digits, the optional suffix may contain 1-4 characters, and the fields are separated with a hyphen.
| Name: |
dei_EntityFileNumber |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:fileNumberItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionTwo-character EDGAR code representing the state or country of incorporation.
| Name: |
dei_EntityIncorporationStateCountryCode |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:edgarStateCountryItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe exact name of the entity filing the report as specified in its charter, which is required by forms filed with the SEC. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b-2
| Name: |
dei_EntityRegistrantName |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe Tax Identification Number (TIN), also known as an Employer Identification Number (EIN), is a unique 9-digit value assigned by the IRS. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b-2
| Name: |
dei_EntityTaxIdentificationNumber |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:employerIdItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionLocal phone number for entity.
| Name: |
dei_LocalPhoneNumber |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionBoolean flag that is true when the Form 8-K filing is intended to satisfy the filing obligation of the registrant as pre-commencement communications pursuant to Rule 13e-4(c) under the Exchange Act. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 13e
-Subsection 4c
| Name: |
dei_PreCommencementIssuerTenderOffer |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionBoolean flag that is true when the Form 8-K filing is intended to satisfy the filing obligation of the registrant as pre-commencement communications pursuant to Rule 14d-2(b) under the Exchange Act. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 14d
-Subsection 2b
| Name: |
dei_PreCommencementTenderOffer |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionTitle of a 12(b) registered security. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b
| Name: |
dei_Security12bTitle |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:securityTitleItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionName of the Exchange on which a security is registered. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection d1-1
| Name: |
dei_SecurityExchangeName |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:edgarExchangeCodeItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionBoolean flag that is true when the Form 8-K filing is intended to satisfy the filing obligation of the registrant as soliciting material pursuant to Rule 14a-12 under the Exchange Act. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Section 14a
-Number 240
-Subsection 12
| Name: |
dei_SolicitingMaterial |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionTrading symbol of an instrument as listed on an exchange.
| Name: |
dei_TradingSymbol |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:tradingSymbolItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionBoolean flag that is true when the Form 8-K filing is intended to satisfy the filing obligation of the registrant as written communications pursuant to Rule 425 under the Securities Act. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Securities Act
-Number 230
-Section 425
| Name: |
dei_WrittenCommunications |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
Applied Therapeutics (NASDAQ:APLT)
Historical Stock Chart
From Apr 2024 to May 2024
Applied Therapeutics (NASDAQ:APLT)
Historical Stock Chart
From May 2023 to May 2024