Boston Scientific Gets FDA OK for Vercise Genus Deep Brain Stimulation System
January 22 2021 - 7:47AM
Dow Jones News
By Colin Kellaher
Boston Scientific Corp. on Friday said the U.S. Food and Drug
Administration approved its fourth-generation Vercise Genus Deep
Brain Stimulation System, used to reduce motor symptoms associated
with Parkinson's disease.
The Marlborough, Mass., maker of medical devices said the
portfolio, approved for conditional use in a
magnetic-resonance-imaging environment, consists of a family of
Bluetooth-enabled, rechargeable and non-rechargeable, implantable
pulse generators that power Cartesia directional leads.
Boston Scientific said the Vercise Genus system can treat the
symptoms of Parkinson's, a neurodegenerative disorder marked by
stiffness, slowness and tremors, by delivering targeted electrical
stimulation via surgically-implanted leads in the brain connected
to an implantable pulse generator.
Write to Colin Kellaher at colin.kellaher@wsj.com
(END) Dow Jones Newswires
January 22, 2021 07:32 ET (12:32 GMT)
Copyright (c) 2021 Dow Jones & Company, Inc.
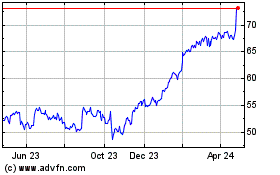
Boston Scientific (NYSE:BSX)
Historical Stock Chart
From Aug 2024 to Sep 2024
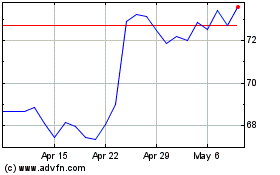
Boston Scientific (NYSE:BSX)
Historical Stock Chart
From Sep 2023 to Sep 2024