Both RELEASE-1 and RELEASE-2 Studies Demonstrated Highly
Statistically Significant Results (P=0.006 and P=0.002
respectively) on Efficacy Endpoints
DUBLIN, Nov. 7, 2018 /CNW/ -- Endo
International plc (NASDAQ: ENDP) announced positive results today
from two identical Phase 3 RELEASE* studies of collagenase
clostridium histolyticum (CCH) for the treatment of cellulite in
the buttocks. Subjects receiving CCH showed highly statistically
significant levels of improvement in the appearance of cellulite
with treatment, as measured by the trial's primary endpoint
(RELEASE-1, p=0.006 & RELEASE-2, p=0.002), which was at least a
2-level composite improvement in cellulite severity in the target
buttock at Day 71 as compared to subjects receiving placebo. In
addition, RELEASE-1 passed 8 out of 8 key secondary endpoints and
RELEASE-2 passed 7 out of 8 key secondary endpoints. CCH was
well-tolerated in the actively-treated subjects with most adverse
events (AEs) being mild to moderate in severity and primarily
limited to the local injection area.
"Based on our review of the Phase 3 data, we remain confident in
our CCH program for cellulite—a condition that makes many women
self-conscious and prompts them to seek treatment options," said
Matthew Davis, M.D., R.Ph., Endo's
Senior Vice President and Chief Medical Officer. "If approved, CCH
has the potential to be an important new treatment for cellulite
and we are excited to take the next steps in that process."
About RELEASE-1 and RELEASE-2
RELEASE-1 and RELEASE-2
are two identical, multicenter, randomized, double-blind,
placebo-controlled studies that enrolled 845 women (423 and 422 in
each separate trial) aged 18 years or older in the United States with moderate to severe
cellulite. Each subject received up to three treatments of CCH
(0.84 mg / treatment area) or placebo with each treatment session
occurring approximately 21 days apart. Up to twelve injections were
administered during each session across each treatment area – the
left and right buttock. At both the outset and conclusion of each
treatment, cellulite severity was assessed by each patient and
clinician using two validated photonumeric cellulite severity
scales developed by Endo and third-party psychometric experts.
The primary endpoint of the Phase 3 studies was a composite
responder analysis demonstrating at least a 2-level composite
improvement independently reported by both patient and clinician on
the photonumeric scales of cellulite severity in the target
buttock. Key secondary endpoints in target buttocks included the
percentage of subjects that experience at least a 1-level or
2-level improvement in patient reported assessment percentage of
subjects with a 1-level composite improvement, the percentage of
subjects with at least a 1-level or 2-level improvement in the
global aesthetic improvement scale (GAIS), as well as percentage of
subjects with a 2-level composite improvement in non-target
buttocks and also percentage of satisfied subjects, change from
baseline in a cellulite impact scale (i.e., patients'
self-perception related to their cellulite).
"The Phase 3 results are positive for patients and consistent
with previous data that demonstrate CCH has the potential to be a
breakthrough treatment for cellulite, a condition that millions of
women experience but for which there are currently limited
effective treatment options," said Joely
Kaufman-Janette, M.D., a board certified dermatologist at
Skin Associates of South Florida,
and a CCH clinical trial investigator. "Many of my patients have
expressed that they are self-conscious about their cellulite and
looking for something to effectively treat it. The fact that CCH is
a potential new injectable option to treat cellulite will be very
meaningful for my patients and for the aesthetics industry
overall."
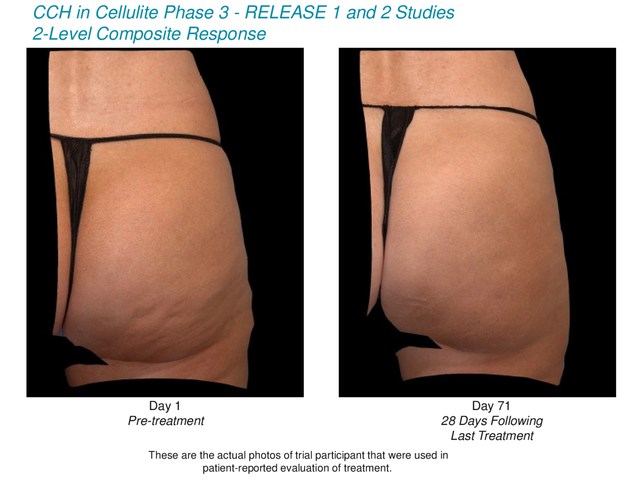
Primary Endpoint for RELEASE-1:
- 7.6 percent of subjects receiving CCH demonstrated a highly
significant (p=0.006) improvement in the composite investigators'
and patients' assessments of the appearance of cellulite, as
measured by a two-level response in both the Clinician Reported- Photonumeric Cellulite Severity
Scale (CR-PCSS) and Patient Reported- Photonumeric Cellulite
Severity Scale (PR-PCSS) scores, for the target buttock at
Day 71, compared to only 1.9 percent of placebo subjects.
Primary Endpoint for RELEASE-2:
- 5.6 percent of subjects receiving CCH demonstrated a highly
significant (0.002) improvement in the composite investigators' and
patients' assessments of the appearance of cellulite, as measured
by a two-level response in both the CR-PCSS and PR-PCSS scores, for
the target buttock at Day 71, compared to only 0.5 percent of
placebo subjects.
Key Secondary Endpoints for both RELEASE-1 and RELEASE-2:
- 37.1 percent of subjects in RELEASE-1, and 41.6 percent of
subjects in RELEASE-2 receiving CCH demonstrated a highly
significant 1-level response in the composite investigators' and
patients' assessments of the appearance of cellulite, as measured
by both the CR-PCSS and PR-PCSS scores, for the target buttock at
Day 71, compared to only 17.8 percent and 11.2 percent of placebo
subjects respectively.
- 24.3 percent of subjects in RELEASE-1, and 21.0 percent of
subjects in RELEASE-2 receiving CCH demonstrated a highly
statistically significant 2-level improvement on the patients'
assessment of the appearance of cellulite in the target buttock at
Day 71, as measured by the PR-PCSS scores compared to only 12.2
percent and 5.8 percent of placebo subjects respectively.
- 54.3 percent of subjects in RELEASE-1, and 57.9 percent of
subjects in RELEASE-2 receiving CCH demonstrated a highly
statistically significant 1-level improvement on the patients'
assessment of the appearance of cellulite in the target buttock at
Day 71, as measured by the PR-PCSS scores compared to only 36.2
percent and 29.6 percent of placebo subjects respectively.
- 48.6 percent of subjects in RELEASE-1, and 42.1 percent of
subjects in RELEASE-2 receiving CCH demonstrated a highly
statistically significant 1-level improvement on the patients'
assessment of the appearance of cellulite in the target buttock at
Day 71, as measured by the SSRS (Subject Self Rating Scale)
compared to only 22.5 percent and 15.0 percent of placebo subjects
respectively.
- 54.3 percent of subjects in RELEASE-1, and 46.8 percent of
subjects in RELEASE-2 receiving CCH reported being "Satisfied" or
"Very Satisfied" with their cellulite treatment as assessed by the
Subject Satisfaction with Cellulite Treatment Assessment at Day 71,
compared to only 25.8 percent and 13.6 percent of placebo subjects
respectively.
- 73.3 percent of subjects in RELEASE-1, and 67.8 percent of
subjects in RELEASE-2 receiving CCH were reported as "Improved" or
"Very Improved" or "Very Much Improved" in global appearance of
their cellulite area as assessed by the Subject- Global Aesthetic
Improvement Scale in the target buttock at Day 71, compared to only
43.2 percent and 24.1 percent of placebo subjects
respectively.
- Subjects receiving CCH demonstrated a statistically significant
improvement in the composite investigators' and patients'
assessments of the appearance of cellulite, as measured by a
2-level improvement in both the CR-PCSS and PR-PCSS scores, for the
non-target buttock at Day 71 for RELEASE-1 study but failed to show
statistical significance in RELEASE-2 study.
Consistent with earlier studies of CCH for the treatment of
cellulite, CCH was well-tolerated in the Phase 3 studies by all
dose groups with most adverse events (AEs) being mild to moderate
and primarily limited to the local injection area. The most common
AEs in the trial were injection site bruising, injection site pain,
injection site discoloration, injection site nodule and injection
site pruritus.
About Cellulite
Cellulite is a localized
metabolic disorder of tissue under the skin that has been reported
in 85 to 98 percent of post-pubertal females and affects women of
all races and ethnicities[i][ii]. The condition can involve the
loss of elasticity or shrinking of collagen cords, called "septae,"
that attach the skin to the muscle layers below. When fat in
cellulite prone areas swells and expands, the septae tether the
skin, which causes the surface dimpling characteristic of
cellulite[iii]. CCH is intended to target and lyse, or break, those
collagen tethers with the goal of releasing the skin dimpling and
potentially resulting in smoothing of the skin. Despite multiple
therapeutic approaches for the attempted treatment of cellulite,
there are no FDA-approved pharmacological treatments and little
scientific evidence that any current treatments are
beneficial[iv].
About Endo International plc
Endo International plc
(NASDAQ: ENDP) is a highly focused generics and specialty
branded pharmaceutical company delivering quality medicines to
patients in need through excellence in development, manufacturing
and commercialization. Endo has global headquarters in Dublin, Ireland, and U.S. headquarters in
Malvern, PA. Learn more
at www.endo.com.
Forward Looking Statements
This press release contains certain forward-looking statements
within the meaning of the Private Securities Litigation Reform Act
of 1995 and Canadian securities legislation, including, but not
limited to, the statement by Drs. Davis and Kaufman-Janette, and
other statements regarding research and development outcomes,
efficacy, adverse reactions, market and product potential and
product availability. Statements including words such as
"believes," "expects," "anticipates," "intends," "estimates,"
"plan," "will," "may," "look forward," "intend," "guidance,"
"future" or similar expressions are forward-looking statements.
Because these statements reflect Endo's current views, expectations
and beliefs concerning future events, they involve risks and
uncertainties. Although Endo believes that these forward-looking
statements and information are based upon reasonable assumptions
and expectations, readers should not place undue reliance on them,
or any other forward-looking statements or information in this news
release. Investors should note that many factors, as more fully
described in the documents filed by Endo with the Securities and
Exchange Commission and with securities regulators in Canada on the System for Electronic Document
Analysis and Retrieval, including under the caption "Risk Factors"
in Endo's Form 10-K, Form 10-Q and Form 8-K filings, and as
otherwise enumerated herein or therein, could affect Endo's future
results and could cause Endo's actual results to differ materially
from those expressed in forward-looking statements contained in
this communication. The forward-looking statements in this press
release are qualified by these risk factors. Endo assumes no
obligation to publicly update any forward-looking statements,
whether as a result of new information, future developments or
otherwise, except as may be required under applicable securities
laws.
*Randomized EvaLuation of
CEllulite Reduction by CollAgenaSE Clostridium
Histolyticum (RELEASE)
[i] Avram, Cellulite: a review of its physiology and treatment,
Journal of Cosmetic Laser Therapy 2004; 6: 181–185.
[ii] Khan MH et al. Treatment of cellulite: Part I.
Pathophysiology. J Am Acad Dermatol. 2010
Mar;62(3):361-70.
[iii] Querleux, Anatomy and physiology of subcutaneous adipose
tissue by in vivo MRI and spectroscopy: Relationship with sex and
presence of cellulite, Skin Research and Technology; 8:
118-124.
[iv] Wanner M et al. An evidence-based assessment of treatments for
cellulite. J Drugs Dermatol. 2008 Apr;7(4):341-5.
 View original content to download
multimedia:http://www.prnewswire.com/news-releases/endo-announces-positive-results-from-phase-3-studies-of-collagenase-clostridium-histolyticum-cch-in-patients-with-cellulite-300745339.html
View original content to download
multimedia:http://www.prnewswire.com/news-releases/endo-announces-positive-results-from-phase-3-studies-of-collagenase-clostridium-histolyticum-cch-in-patients-with-cellulite-300745339.html
SOURCE Endo International plc