As filed with the Securities and Exchange
Commission on December 05, 2016
Registration No. 333-
UNITED
STATES
SECURITIES
AND EXCHANGE COMMISSION
Washington, D.C. 20549
________________________
FORM S-1
REGISTRATION
STATEMENT UNDER THE SECURITIES ACT OF 1933
________________________
NUGENE INTERNATIONAL, INC.
(Exact name of registrant as specified
in its charter)
________________________
|
NEVADA
|
2844
|
46-3999052
|
(State or other jurisdiction of
incorporation)
|
(Primary Standard Industrial
Classification Code Number)
|
(IRS Employer Identification
Number)
|
17912 Cowan, Suite A
Irvine, California 92614
(949) 430-7737
(Address, including zip code, and telephone
number, including area code, of registrant’s principal executive offices)
________________________
Steven Carlson
Chief Executive Officer
NuGene International, Inc.
17912 Cowan, Suite A
Irvine, California 92614
(949) 430-7737
(Name, address, including zip code, and
telephone number, including area code, of agent for service)
________________________
Copies to:
|
Keith A. Rosenbaum
|
Darrin Ocasio, Esq.
|
|
SPECTRUM LAW GROUP, LLP
|
Jay Yamamoto, Esq.
|
|
1900 Main Street, Suite 300
|
SICHENZIA ROSS FERENCE KESNER LLP
|
|
Irvine, California 92614
|
61 Broadway
|
|
Phone: 949-851-4300
|
New York, NY 10006
|
|
Fax: 949-851-5940
|
Phone: 212-930-9700
|
|
|
Fax: 212-930-9725
|
Approximate
date of commencement of proposed sale to the public:
As soon as practicable after this Registration Statement becomes
effective.
If any of the
securities being registered on this Form are to be offered on a delayed or continuous basis pursuant to Rule 415 under the Securities
Act of 1933, check the following box.
¨
If this Form is filed to register
additional securities for an offering pursuant to Rule 462(b) under the Securities Act, please check the following box and list
the Securities Act registration statement number of the earlier effective registration statement for the same offering.
¨
If this Form is a post-effective
amendment filed pursuant to Rule 462(c) under the Securities Act, check the following box and list the Securities Act registration
statement number of the earlier effective registration statement for the same offering.
¨
If this form
is a post-effective amendment filed pursuant to Rule 462(d) under the Securities Act, check the following box and list the Securities
Act registration statement number of the earlier effective registration statement for the same offering.
¨
Indicate by
check mark whether the registrant is a large accelerated filer, an accelerated filer, a non-accelerated filer, or a smaller reporting
company. See the definitions of “large accelerated filer,” “accelerated filer” and “smaller reporting
company” in Rule 12b-2 of the Exchange Act. (Check one):
Large accelerated
filer
¨
Accelerated filer
¨
Non-Accelerated filed
¨
Smaller reporting company
þ
(Do not check
if a smaller reporting company)
________________________
CALCULATION
OF REGISTRATION FEE
|
Title of Each Class of
Securities to be Registered
|
|
Proposed
Maximum
Aggregate
Offering Price
(1)
|
|
|
Amount of
Registration Fee
(2)
|
|
|
Common Stock, $0.0001 par value per share
(2)(3)
|
|
$
|
17,250,000
|
|
|
$
|
1,999.28
|
|
|
Representative’s Warrants to Purchase Common Stock
|
|
|
—
|
|
|
|
|
(4)
|
|
Warrants to purchase common stock
|
|
$
|
17,250
|
|
|
$
|
1.99
|
(5)
|
|
Shares of common stock underlying Warrants
|
|
$
|
21,565,500
|
|
|
$
|
2,499.44
|
|
|
Common Stock Underlying Representative’s Warrants
(2)(5)
|
|
$
|
937,500
|
|
|
$
|
108.66
|
(6)
|
|
Total Registration Fee
|
|
$
|
|
|
|
$
|
4,609.40
|
|
|
|
(1)
|
Estimated solely for the purpose of calculating the Registration Fee pursuant to Rule 457(o) under the Securities Act of 1933,
as amended (the “Securities Act”).
|
|
|
(2)
|
Pursuant to Rule 416 under the Securities Act, the shares of common stock registered hereby also include an indeterminate number
of additional shares of common stock as may from time to time become issuable by reason of stock splits, stock dividends, recapitalizations
or other similar transactions.
|
|
|
(3)
|
Includes the offering price of shares of common stock the underwriters have the option to purchase to cover over-allotments,
if any.
|
|
|
(4)
|
No registration fee pursuant to Rule 457(g) under the
Securities Act.
|
|
|
(5)
|
Estimated solely for the purposes of calculating the registration fee pursuant to Rule 457(g) under the Securities Act. The
warrants are exercisable at a per share exercise price equal to 125% of the public offering price. Fee is based on maximum aggregate
offering price of $937,500, or 125% of $750,000 (5% of $15,000,000).
|
|
|
(6)
|
Estimated solely for purpose of calculating the registration
fee pursuant to Rule 457(i) under the Securities Act.
|
The Registrant
hereby amends this Registration Statement on such date or dates as may be necessary to delay its effective date until the Registrant
shall file a further amendment which specifically states that this Registration Statement shall thereafter become effective in
accordance with Section 8(a) of the Securities Act of 1933 or until the Registration Statement shall become effective on such
date as the Commission, acting pursuant to said Section 8(a), may determine.
The
information in this preliminary prospectus is not complete and may be changed. These securities may not be sold until the registration
statement filed with the Securities and Exchange Commission is effective. This preliminary prospectus is not an offer to sell these
securities and it is not soliciting offers to buy these securities in any state where the offer or sale is not permitted.
PRELIMINARY
PROSPECTUS SUBJECT TO COMPLETION DATED DECEMBER __, 2016
____________ Shares of Common Stock
Warrants to Purchase _______________
Shares of Common Stock

NuGene International, Inc.
NuGene International, Inc. is offering
shares of common stock and warrants to purchase up to an aggregate of shares
of common stock. The shares of common stock and the warrants are immediately separable and will be issued separately in this offering.
Each warrant will be exercisable for a period commencing on the date of issuance and expiring on and an exercise price equal to
$ . We expect to effect a 1-for- reverse stock split of our issued and outstanding common stock prior to the date of
this prospectus.
Our common stock is quoted on the OTCQB Marketplace
operated by OTC Markets Group, Inc under the symbol “NUGN”. We will apply to list our common stock on The NASDAQ Capital
Market under the symbol “NUGN”. No assurance can be given that our application will be approved. On December __, 2016,
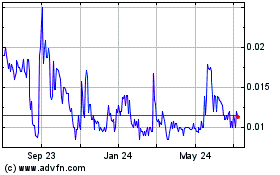
the closing sale price of our common stock, as reported by the OTCQB, was $____ per share.
Investing in our common stock involves
certain risks. Before purchasing our common stock, please review the information included in, and incorporated by reference into,
the “Risk Factors” caption beginning on page 8 of this prospectus.
Neither the Securities and Exchange
Commission nor any state securities commission has approved or disapproved of these securities or determined if this prospectus
is accurate or complete. Any representation to the contrary is a criminal offense.
|
|
|
Per Share
|
|
|
Total
|
|
|
Public offering price
|
|
$
|
|
|
|
$
|
|
|
|
Underwriting discount
(1)
|
|
$
|
|
|
|
$
|
|
|
|
Proceeds, before expenses, to us
|
|
$
|
|
|
|
$
|
|
|
|
|
(1)
|
Does not include a non-accountable expense allowance equal to 1% of the gross proceeds of this
offering payable to Joseph Gunnar & Co., the representative of the underwriters. See ‘‘Underwriting’’
for a description of compensation payable to the underwriters.
|
The underwriters may also purchase up to
additional shares of our common stock from us at the public
offering price, less the underwriting discount, within 45 days from the date of this prospectus to cover over-allotments, if any.
The underwriters expect to deliver the
shares to purchasers on or about , 2016.
Joseph Gunnar & Co.
TABLE OF CONTENTS
________________________
You should rely only on the information
contained in or incorporated by reference in this prospectus, or in any related prospectus supplement, or amendment to this prospectus
that we file with the Securities and Exchange Commission. We have not, and the underwriters have not, authorized anyone to provide
you with different information. We are not making an offer of these securities in any jurisdiction where the offer or sale is not
permitted. You should assume that the information contained in this prospectus is accurate as of the date on the front of this
prospectus only. Our business, financial conditions, results of operations and prospects may have changed since that date.
PROSPECTUS SUMMARY
The items in the following summary are described
in more detail later in this prospectus. This summary provides an overview of selected information and does not contain all of
the information you should consider before buying our common stock. Therefore, you should read the entire prospectus carefully,
especially the “Risk Factors” section beginning on page 8; “Management’s Discussion and Analysis of
Financial Condition and Results of Operations”, beginning on page 35; and, our consolidated financial statements and the
related notes beginning on page F-2. Unless otherwise stated or the context requires otherwise, references in this prospectus to
“NuGene”, the “Company”, “we”, “our”, “us”, and other similar terms
mean NuGene International, Inc. and its wholly owned subsidiaries, unless we state otherwise or the context indicates otherwise.
Business Overview
We are principally in the business of
research, development, and sales and marketing “cosmeceutical
”
skincare products. We market our NuGene
line of products to medical offices and medical spas throughout the United States (“U.S.“). We are working to
expand our direct sales of our consumer Kathy Ireland branded products. We intend to continue to have our NuGene line of
products sold domestically and internationally through professional channels, including dermatologists, plastic surgeons,
medical offices, and day and resort spas. We also expect to directly market our product through direct to consumer related
channels which may include television sales-dedicated channels. We plan to promote brand awareness through advertising, our
own sales personnel to be hired, other marketing, and public relations. We will also partially rely upon the name recognition
of Kathy Ireland secured under the License Agreement discussed below. We propose to further expand our sales efforts through
our branded website (
www.nugene.com
) and additional online commerce channels.
“Cosmeceuticals
” refer
to the combinations of cosmetics and pharmaceuticals that may offer medicinal or drug-like benefits. The term is more of a marketing
term rather than a legal term, with the cosmetics industry having adopted the term in the late 1990’s. The U.S. Food and Drug
Administration (the “FDA”) does not recognize the term. While drugs are subject to a review and approval process by
the FDA, cosmetics and cosmeceuticals are not subject to the same stringent regulatory regime and scrutiny. As such, if a product
has drug properties, it must be approved as a drug. However, cosmeceuticals are not subject to this review and approval process.
Cosmeceuticals offer consumers cosmetic-like
treatments with active ingredients, which we believe give them pharmaceutical-like efficacy. We believe that this market benefits
from the growing segment of a younger generation of consumers as well as baby boomers who want to preserve their youthful looks.
We believe the U.S. cosmeceutical market will continue to be favorably influenced by these groups who want beautiful and younger
looking skin while aging.
The use of stem cell based media as a foundation
for cosmeceutical products is designed to promote the age-defying process. Every human being has stem cells in his or her body.
These cells exist from the early stages of human development until the end of a person’s life. Throughout our lives, our
bodies continue to produce stem cells that regenerate to produce differentiated cells that make up various aspects of the body
such as skin, blood, muscles, and nerves. These are generally referred to as adult stem cells (non-embryonic). These cells are
important for the purpose of medical therapies aiming to replace lost or damaged cells or tissues or to otherwise treat disorders.
Our NuGene line of products uses human adult stem cells culture media that we believe, when applied to human skin cells, may reduce
the appearance of aging skin.
Our goal is to leverage our knowledge in
human adult stem cell technology to develop and commercialize advanced anti-aging skincare products for the retail and professional
channels. We have developed, manufactured, and market cosmetic skin care and hair products to address this significant market opportunity.
We are party to a License Agreement with
kathy ireland Worldwide
®
(“kiWW
®
”) whereby we licensed the right to utilize the
trademarks and rights to the name, likeness, and visual representations of Kathy Ireland in connection with our cosmeceutical line
of products containing adult human adipose stem cell derived or containing biologically active or biologically derived ingredients.
On March 5, 2015, we announced the launch of the NuGene kathy ireland
®
brand. The campaign includes a re-branded
internet presence and new videos featuring Ms. Ireland. We have initiated the launch of a larger multi-media campaign to promote
the product line. NuGene is obligated to utilize commercially reasonable efforts to satisfy certain minimum sales requirements
set forth in the License Agreement. We are also obligated to pay an annual fee to kiWW
®
which provides for
general advertising, good will, and promotion of the overall kathy ireland brand. NuGene paid kiWW $350,000 effective upon execution
of the License Agreement. Under the License Agreement, we are also required to pay brand participation fees for successive years
of $50,000 annually and an additional 1% of the total gross sales of licensed products of the prior year beginning in Contract
Year 4 (see “Business”).
All of our products listed below have been
formulated, fully developed, and are currently being marketed. We have formulated and are testing other products which we expect
to introduce (financial resources permitting) once we complete product testing and preparation of marketing materials. None of
our products are licensed from third parties. All of products are either protected by patents owned by us, or derived from trade
secrets and proprietary formulas.
NuGene Skincare Products
|
|
•
|
Face Wash
: As an anti-aging cleanser, our Face Wash is formulated with a unique blend of
multiple growth factors, cytokines, and rich pentapeptide complexes. NuGene’s Face Wash minimizes the visibility of pores
and softens imperfections while the natural cleansing agents gently wash away dirt, oil, and makeup leaving the skin refreshed,
smooth, clean, and hydrated.
|
|
|
•
|
Universal Cream
: Our moisturizer works to smooth fine lines and wrinkles, improve aging
skin tone and texture, increase collagen production, and reduce puffiness and dark circles—without any heaviness or oiliness.
|
|
|
•
|
Universal Serum
: Formulated with multiple human growth factors, polypeptides, vitamins,
minerals and amino acids, our signature serum encourages skin’s natural production of collagen and elastin while
boosting other vital matrix protein syntheses, enabling it to repair and prevent environmental damage, improve texture and thickness,
and restore skin’s natural luminosity.
|
|
|
•
|
Eye Serum
: Designed to prevent and correct wrinkles, fine lines, puffiness, dryness
and dark circles, this innovative product relies on our cutting-edge stem cell technology, along with multiple growth factors and
other nourishing ingredients, to help reverse skin damage, minimize the appearance of future signs of aging, and restore youthful
luminosity around the eyes, fostering skin that looks younger, smoother and fresher.
|
|
|
•
|
Light & Bright Gel
: This potent product works to break down excess melanin in the skin
without any of the harsh ingredients typically found in brightening products. In addition to quickly reducing the look of dark
spots and excess pigmentation, it works to increase overall radiance, restoring a youthful glow.
|
Hair Care Products
|
|
•
|
Anti-Hair Loss Serum
: Our stem cell based technology replenishes the hair follicles with
essential growth factors and cytokines to reduce and prevent further hair loss, as well as to stimulate follicular regeneration.
|
|
|
•
|
Regenerative Shampoo
: Rich in growth factors, polypeptide complexes and vitamins, this product
has been formulated to rejuvenate the hair and scalp. In addition to increasing hydration, vibrancy, and shine, it works to protect
hair from environmental assault, preventing and correcting breakage, thinning and split ends.
|
|
|
•
|
Regenerative Conditioner
: This hydrating, shine-enhancing conditioner utilizes additional
key ingredients, vitamins, and polypeptide complexes, to name a few, to instantly smooth even the coarsest, most damaged strands.
With regular use, it fosters stronger, shinier, younger-looking hair that is easier to style.
|
|
|
•
|
Anti-Hair Loss Serum
: Our stem cell based technology replenishes the hair follicles with
essential growth factors and cytokines to reduce and prevent further hair loss, as well as to stimulate follicular regeneration.
|
Wound Care-Wound Healing; Biopharma
Products
We have initiated research and
formulation for a new wound healing cream for the initial healing and management of cosmetic incisional scar formation in indications
such as abdominal scars in tummy tucks and voluntary C-sections. We have also initiated development of a topical cream for treatment
of burns that we expect could improve healing time and may reduce infection risk. In order to develop and bring these products
to market, we intend to work with a CGMP FDA approved contract manufacturer to combine the product with our proprietary stem cell
media technology. The preclinical FDA work we will need to have completed consists of in vitro and in vivo efficacy studies, experimental
toxicology testing, formulation study, storage condition, stability testing, and ensuring good manufacturing practices are all
followed in the manufacturing process. These topical products may require FDA approval and we can give no assurance that we will
obtain that approval.
Short Term Strategic Goals and
Objectives
During the next 12 months, our strategy
is to establish NuGene as the industry leader for adipose derived stem cell based cosmetic products. The fundamental short-term
goals and objectives of our business strategy include the following:
|
|
•
|
Complete the reformulation and stabilization of our commercial products.
|
|
|
•
|
Complete the testing of our products conducted by an independent research firm.
|
|
|
•
|
Initiate sales of our Kathy Ireland branded line of products directly to consumers.
|
|
|
•
|
Initiate an aggressive marketing campaign for our direct to consumer products in conjunction with
KBHJJ, LLC under the Services Agreement we have them. KBHJJ, LLC is owned and operated by Kevin Harrington, who is recognized as
the inventor of the infomercial and was the original Shark on the television show Shark Tank.
|
|
|
•
|
Initiate sales of a professional line of cosmeceutical products designed to be sold through doctor’s
offices, high-end med-spas, and by licensed estheticians and similarly licensed cosmetic professionals.
|
|
|
•
|
Develop a
biopharma
line of products which are more “medical” in nature and
which may require FDA approval. These products could include a topical burn cream and wound healing cream or ointment.
|
|
|
•
|
Work with a contract manufacturer to combine our stem cell media technology with other products.
|
|
|
•
|
Establish a manufacturing facility ensuring that “Good Manufacturing Practices” are followed in the
manufacturing process. Then, establish a second redundant facility for increased production capacity and back-up in the event of
a major disaster or delay.
|
|
|
•
|
Continue to leverage our intellectual property portfolio to create value. We plan to file
several new patent applications, develop, and expand our proprietary formulas and methodologies, and take other steps to strengthen,
leverage, and expand our intellectual property position.
|
|
|
•
|
Continue our collaboration with industry leading scientists, medical doctors and researchers to
support clinical trials of our stem cell media technology with other products.
|
|
|
•
|
Continue to develop and maintain strategic and development collaborations with academic laboratories,
clinical investigators, and potential commercial partners.
|
|
|
•
|
Further develop our commercialization capabilities, initially by internally developing our own
marketing and sales force. We will also seek strategic partnerships, which could include commercial partnerships, joint ventures,
or other arrangements with competitive or complementary companies.
|
Recent Developments
We have recently restructured our executive
management team to lead NuGene, including hiring Steven Carlson as our CEO. Mr. Carlson is an accomplished pharmaceutical
skin care and cosmetic products executive with broad management skills, including strategic planning and business development,
go-to-market execution, business integration, and federal health and safety compliance for consumer products. He has over 35 years
of industry experience, having worked at such notable companies as Allergan and Obagi Medical Products. Mr. Carlson has engaged
the services of a number of industry leaders and professionals to work with us. Mr. Carlson has also energized our product development
and marketing efforts through his extensive industry relationships.
Risks
Since our inception, we have incurred substantial
losses. We have generated minimal revenues to date. We will need the funding sought under this prospectus to remain a going concern,
maintain operations, and to activate our business plan, which includes, among other things, advertising, retaining channels of
distribution, retaining supplier relationships, building inventory, and recruiting experienced sales personnel.
Our business and our ability to execute
our business strategy are subject to a number of risks of which you should be aware before you decide to buy our securities. In
particular, you should carefully consider all of the risks which are discussed more fully in “Risk Factors” beginning
on page 8 of this prospectus.
Corporate Information
We were incorporated in the State of Nevada
on October 31, 2013. NuGene, Inc. (our wholly owned subsidiary) was incorporated in the State of California on December 5, 2006.
On January 20, 2015, we formed NuGene BioPharma, Inc. (“BioPharma”) in the State of California as our wholly-owned
subsidiary. On November 6, 2015, we formed The Aesthetic Group, Inc. (“TAG”) in the State of California as our wholly-owned
subsidiary. Both BioPharma and TAG have had no significant independent operations to date.
Our principal executive offices are located
at 17912 Cowan, Suite A, Irvine, California, 92614. Our telephone number is (949) 430-7737. Our website address is http://www.nugene.com.
This reference to our website is intended to be an inactive textual reference, and our website is not intended to be a part of
this prospectus.
THE OFFERING
The following summary contains basic
information about our common stock and the offering and is not intended to be complete. It does not contain all the information
that may be important to you. For a more complete understanding of our common stock, you should read the section entitled “Description
of Capital Stock” in this prospectus.
|
Common stock offered by us
|
___________ shares
|
|
|
|
|
Common stock to be outstanding
|
|
|
immediately after the offering
|
___________ shares
(1)
|
|
|
|
|
Description of Warrants
|
The warrants will have a per share exercise price equal to $ (125% of the public offering price of the common stock). The warrants are exercisable immediately and expire ______ years from the date of issuance.
|
|
|
|
|
Over-allotment option
|
We have granted the underwriters an option to purchase up to shares of our common stock at a price of $ per share solely to cover over-allotments, if any. This option is exercisable, in whole or in part, for a period of 45-days from the date of this prospectus.
|
|
|
|
|
Use of proceeds
|
We intend to use the net proceeds from this offering primarily for working capital and general corporate purposes, as well as to retire certain debt obligations which will mature upon receipt of proceeds under this offering. See “Use of Proceeds”.
|
|
|
|
|
Risk factors
|
Investing in our common stock and warrants involves risks. See “Risk Factors” beginning on page 8 of this prospectus for a discussion of risk factors that you should carefully consider before making a decision to buy shares of our common stock in this offering.
|
|
|
|
|
Lock-Ups
|
We and our directors and executive officers and certain stockholders have agreed with the underwriters not to offer for sale, issue, sell, contract to sell, pledge or otherwise dispose of any of our common stock or securities convertible into common stock for a period of 180 days after the date of this prospectus in the case of our directors and executive officers, and 90 days in the case of us and certain stockholders. See “Underwriting” beginning on page 68.
|
|
|
|
|
OTCQB symbol
|
NUGN
|
|
|
|
|
Proposed Symbol and Listing
|
We will apply to list our common stock and warrants on the NASDAQ Capital Market under the symbols “NUGN”
and “NUGNW”, respectively.
|
|
|
(1)
|
The number of shares of our common stock to be outstanding after this offering is based on
______________ shares of common stock outstanding as of December __, 2016 and assumes no exercise by (i) the underwriters of
their option to purchase additional shares of common stock to cover over-allotments, if any; and, (ii) by holders of the
warrants issued in the offering. It also specifically excludes all shares of common stock issuable upon the exercise of
outstanding convertible notes, stock options, and warrants, and the conversion of outstanding shares of convertible preferred
stock. As of September 30, 2016 we had outstanding warrants to purchase 4,679,234 shares of our common stock at a weighted
average exercise price of approximately $0.78 per share; outstanding options to purchase 4,352,619 shares of our common stock
at a weighted average exercise price of approximately $0.58 per share; issued shares of preferred stock that can be converted
into 1,917,720 shares of our common stock; and, approximately $2,601,091 outstanding principal amount and interest of
convertible debt which can be converted into shares of our common stock at a conversion price that varies depending on the
average trading price of shares of our common stock.
|
SUMMARY OF FINANCIAL DATA
The following table sets forth our summary statement of operations data for the fiscal years ended December
31, 2015 and 2014 derived from our audited consolidated financial statements and related notes included elsewhere in this prospectus.
The summary financial data for the nine months ended September 30, 2016 and 2015, and as of September 30, 2016, are derived from
our unaudited condensed consolidated financial statements appearing elsewhere in this prospectus and are not indicative of results
to be expected for the full year. Our financial statements are prepared and presented in accordance with generally accepted accounting
principles in the U.S. The results indicated below are not necessarily indicative of our future performance. You should read this
information together with the sections entitled “Capitalization,” “Management’s Discussion and Analysis
of Financial Condition and Results of Operations” and our financial statements and related notes included elsewhere in this
prospectus.
|
|
|
Nine Months Ended September 30,
|
|
|
Year Ended December 31,
|
|
|
|
|
2016
|
|
|
2015
|
|
|
2015
|
|
|
2014
|
|
|
Revenues
|
|
$
|
409,981
|
|
|
$
|
1,538,453
|
|
|
$
|
2,084,939
|
|
|
$
|
723,438
|
|
|
Cost of revenues
|
|
|
658,882
|
|
|
|
443,943
|
|
|
|
588,882
|
|
|
|
293,162
|
|
|
Gross profit(loss)
|
|
|
(248,901
|
)
|
|
|
1,094,510
|
|
|
|
1,496,057
|
|
|
|
430,276
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Operating expenses
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Advertising and promotion
|
|
|
314,469
|
|
|
|
298,738
|
|
|
|
-
|
|
|
|
-
|
|
|
Personnel
|
|
|
4,138,979
|
|
|
|
2,064,815
|
|
|
|
-
|
|
|
|
-
|
|
|
Selling, general and administrative
|
|
|
1,056,026
|
|
|
|
861,795
|
|
|
|
6,658,109
|
|
|
|
743,057
|
|
|
Research and development
|
|
|
104,072
|
|
|
|
136,248
|
|
|
|
-
|
|
|
|
-
|
|
|
Professional fees
|
|
|
1,278,866
|
|
|
|
770,889
|
|
|
|
-
|
|
|
|
-
|
|
|
Total operating expenses
|
|
|
6,892,412
|
|
|
|
4,132,485
|
|
|
|
6,658,109
|
|
|
|
743,057
|
|
|
Loss from operations
|
|
|
(7,141,313
|
)
|
|
|
(3,037,975
|
)
|
|
|
(5,162,052
|
)
|
|
|
(312,781
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Other income (expenses)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Interest income
|
|
|
5
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
Interest expense
|
|
|
(659,954
|
)
|
|
|
(76,443
|
)
|
|
|
(57,015
|
)
|
|
|
-
|
|
|
Loss on issuance of debt
|
|
|
(1,212,384
|
)
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
Change in fair value of derivative liabilities
|
|
|
257,126
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
Total other expenses
|
|
|
(1,615,207
|
)
|
|
|
(76,443
|
)
|
|
|
(57,015
|
)
|
|
|
-
|
|
|
Net loss
|
|
$
|
(8,756,520
|
)
|
|
$
|
(3,114,418
|
)
|
|
$
|
(5,219,067
|
)
|
|
$
|
(312,781
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Net loss per share - basic and diluted
|
|
$
|
(0.22
|
)
|
|
$
|
(0.08
|
)
|
|
$
|
(0.13
|
)
|
|
$
|
(0.01
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Weighted average number of common shares outstanding, basic and diluted
|
|
|
40,156,826
|
|
|
|
39,506,104
|
|
|
|
39,730,875
|
|
|
|
20,865,881
|
|
|
|
|
As of September 30, 2016
|
|
|
|
|
Actual
|
|
|
Pro Forma,
As Adjusted
|
|
|
Balance Sheet Data:
|
|
|
|
|
|
|
|
|
|
Cash
|
|
$
|
307,776
|
|
|
$
|
|
|
|
Total assets
|
|
|
585,345
|
|
|
|
|
|
|
Total liabilities
|
|
|
4,578,437
|
|
|
|
|
|
|
Total shareholders’ deficit
|
|
|
(3,993,092
|
)
|
|
|
|
|
RISK FACTORS
Investing in our common stock and warrants
involves significant risks. Before making an investment decision, you should carefully consider the risks and other information
we include or incorporate by reference in this prospectus. In particular, you should consider the risks related to this offering
described below. Our business, prospects, financial condition, and results of operations may be materially harmed as a result of
any of these risks. In this event, the market price of our common stock and warrants could decline and you could lose part or all
of your investment.
Risks Related to Our Financial Position
and Need for Additional Capital
We have incurred significant losses
since our inception. We expect to incur losses for the foreseeable future and may never achieve or maintain profitability.
Since the inception
of NuGene, Inc. (our wholly owned subsidiary) in December 2006 we have incurred significant operating losses. Our net losses were
approximately $5.2 million for the fiscal year ended December 31, 2015, and $8.8 million for the nine months ended September 30,
2016. As of September 30, 2016, we had a deficit accumulated since inception of $15.3 million. We have invested a significant portion
of our efforts and financial resources in the development of our NuGene line of products. More recently, we have begun to also
invest our efforts and financial resources in the early development of our wound care/wound healing products.
We expect to continue to incur significant
operating losses for at least the next several years. To become and remain profitable we must succeed in commercializing and marketing
our NuGene line of products. We may never succeed in these activities and may never generate revenues that are significant or large
enough to achieve profitability. Even if we do achieve profitability, we may not be able to sustain or increase profitability on
a quarterly or annual basis. Our failure to become and remain profitable could depress the market price of our common stock and
could impair our ability to raise capital, expand our business, or continue our operations. A decline in the market price of our
common stock could also cause you to lose all or a part of your investment.
Our financial statements have been
prepared assuming that our Company will continue as a going concern and we will need to obtain additional funding if we are to
continue operations.
The factors described elsewhere herein
raise substantial doubt about our ability to continue as a going concern. Our financial statements do not include any adjustments
that might result from this uncertainty. To date we have incurred significant cash losses that have materially impaired our liquidity
and working capital. We have been under severe liquidity restraints such that our prior CEO was required to personally guarantee
borrowings made by our Company from our bank. This has resulted in the substantial increase in balances outstanding and owing to
our suppliers that has put our relationships with them in jeopardy. We continue to attempt to procure the needed funding to maintain
our operations. Should we be successful, we will need to quickly reverse the historical trend of our operations through generating
significantly higher levels of revenue (at or about historical margins) and reducing our operating expenses. If we cannot generate
the revenues and gross margin at levels required to achieve profitability or obtain sufficient additional capital on acceptable
terms, we will need to substantially revise our business plan or cease operations. Should that happen, an investor could suffer
the loss of a significant portion or all of his investment in our Company.
We have a limited operating history
and investors will have no ability to gauge market acceptance for our products or the ability of management to execute on our business
plan.
We are an early-stage company with a limited
operating history and limited revenues derived from our operations. Our operations to date have been primarily focused on our formation,
the hiring of our management team, acquiring, licensing, and developing our technology and products, building and expanding our
sales force, marketing department and investor relations and commencing the commercial launch of our products.
It is difficult to predict future performance
and our ability to maintain operations is dependent upon a number of factors over which we have limited control. As a result, it
is difficult to predict our quarterly financial results and they are likely to fluctuate significantly. We are a relatively new
company with a limited operating history and our sales prospects are uncertain. We also have relatively limited experience selling
our products. Accordingly, we cannot predict with any certainty the timing or level of sales of our products in the future. If
our quarterly sales or operating results fall below the expectations of investors or securities analysts, the price of our common
stock could decline substantially. In addition to the other factors discussed under these “Risk Factors,” specific
factors that may cause fluctuations in our operating results include:
|
|
•
|
Demand and pricing for our products, including any change in wholesaler purchasing patterns for
our products;
|
|
|
•
|
Physician and patient acceptance of our products;
|
|
|
•
|
Timing of new product offerings, acquisitions, licenses or other significant events by us, our
partners or our competitors;
|
|
|
•
|
Regulatory approvals and legislative changes affecting our cosmeceutical products;
|
|
|
•
|
Any interruption in the manufacturing or distribution of our products, including events affecting
our third-party suppliers and any failure to comply with manufacturing specifications;
|
|
|
•
|
Changes in treatment practices of physicians or other providers that currently recommend our products;
|
|
|
•
|
Significant product returns and rebates;
|
|
|
•
|
Implementation of new or revised accounting or tax rules or policies; and
|
|
|
•
|
The effect of competing technological and market developments.
|
Because we have a limited operating
history, we are subject to all of the risks and uncertainties of a new business.
We initiated the rollout of the first generation
of NuGene products in 2013. We are subject to all of the risks and uncertainties normally associated with an early stage business,
including potential manufacturing issues, difficulties establishing our marketing and distribution operations, lack of name recognition,
lack of adequate capital, difficulties hiring and retaining qualified employees and difficulties in complying with all applicable
federal, state, and local regulatory and administrative requirements. As an early stage company, we expect to incur operating losses
until (if ever) we successfully release and market a line of products that will generate enough revenues and gross margin to become
profitable or thereafter maintain profitability. There is no assurance that we will be able to validate and market products that
will generate enough revenues for us to become profitable or thereafter maintain profitability. As a result, we cannot predict
when, if ever, it might achieve profitability and cannot be certain that it will be able to sustain profitability, if achieved.
Our lack of an operating history may make it difficult for you to evaluate our business prospects in connection with an investment
in our securities.
We need to raise additional capital
to continue our operations, which may not be available on commercially reasonable terms, or at all, and which materially may dilute
your investment
.
To attain profitability, we must increase
our revenues and manage our product, operating and administrative expenses, as to which each of which we can give no assurance.
Because we have been to date unable to generate sufficient revenues to pay our expenses and our existing sources of cash and cash
flows are otherwise insufficient to fund our activities, we must raise additional funds to continue our operations and to manage
our current short-term debt load. Further, our recent efforts to raise additional capital have been unsuccessful. We do not have
any arrangements in place for additional funds and no assurance can be given that required funds will become available on favorable
terms, or at all. Furthermore, if we issue equity or debt securities to raise additional funds, our existing stockholders may experience
dilution, and the new equity or debt securities may have rights, preferences and privileges senior to those of our existing stockholders.
Our current cash and cash equivalents are insufficient to fund our operations through the end of our fiscal year in 2016. If we
are unsuccessful in obtaining additional funds on commercially reasonable terms or at all, and thereafter in achieving profitability,
we may be required to curtail significantly or cease our operations, which could result in the loss of all of your investment in
our stock.
Should we be able to obtain additional
financing and thereafter be successful in growing our revenues according to our operating plans, we may not be able to manage our
growth effectively, which could adversely affect our operations and financial performance
.
The ability to manage and operate our business
as we execute our growth strategy will require further substantial capital and effective planning. Additionally, we have not been
able to maintain adequate levels of capital to fund existing operations. Significant rapid growth on top of our current operations
could greatly strain our internal resources, leading to a much lower quality of customer service, reporting problems and delays
in meeting important deadlines resulting in substantial loss of market share and other problems that could adversely affect our
financial performance. Our efforts to grow could place a significant strain on our personnel, management systems, infrastructure,
liquidity, and other resources. If we do not manage our growth effectively, our operations could be adversely affected, resulting
in slower, no or negative growth, critical shortages of cash and a failure to achieve or sustain profitability.
Significant differences between actual
and estimated demand for our products could adversely affect us.
If we overestimate demand for our products,
we may be required to write off inventories and increase our reserves for product returns or liabilities to customers in future
periods. If we underestimate demand, we may not have sufficient inventory of products to ship to our customers. Our cosmeceutical
products have expiration dates that generally range from 24 to 36 months from the date of manufacture. We need to exercise judgment
in estimating these reserves. The actual amounts could be materially different from our estimates, and differences will need to
be accounted for in the period in which they become known. If we determine that the actual amounts exceed our reserve amounts,
we will record a charge to earnings to approximate the difference. A material reduction in earnings resulting from a charge could
have a material adverse effect on our net income, results of operations and financial condition.
If we raise additional funds through
collaboration, licensing, or other similar arrangements, it may be necessary to relinquish potentially valuable rights to our current
products, potential products, or proprietary technologies, or grant licenses on terms that are not favorable to us
.
If adequate funds are not available to
us, our ability to achieve profitability or to respond to competitive pressures would be significantly limited, and we may be required
to delay, significantly curtail or eliminate the sales of one or more of our current products and/or the development of one or
more of our potential products.
Risks Related to the Development and
Commercialization of Our Product Candidates
If our product candidates are found
to cause undesirable side effects we may need to delay or abandon our development and commercialization efforts.
Any undesirable side effects that might
be caused by our product candidates could interrupt, delay or halt the commercialization and marketing of the products. In addition,
if we start selling any of our product candidates and we or others later identify undesirable side effects caused by the product.
This could prevent us from achieving or maintaining market acceptance of the affected product or could substantially increase the
costs and expenses of commercializing the product, which in turn could delay or prevent us from generating significant revenues
from its sale.
The commercial success of any product
candidates that we may develop will depend upon the degree of market acceptance by physicians, patients, and consumers.
Any products that we bring to the market
may not gain market acceptance by physicians, patients, and consumers. If these products do not achieve an adequate level of acceptance,
we may not generate significant product revenues and we may not become profitable. Physicians will not recommend our product candidates
until we can demonstrate the safety and efficacy of our product candidates as compared to other treatments. Even if the clinical
safety and efficacy of our product candidates are established, physicians may elect not to recommend these products, and consumers
may choose not to purchase our products. The degree of market acceptance of our product candidates will depend on a number of factors,
including:
|
|
•
|
the willingness and ability of patients, the healthcare community, and consumers to adopt our products;
|
|
|
•
|
the ability to manufacture our product candidates in sufficient quantities with acceptable quality
and to offer our product candidates for sale at competitive prices;
|
|
|
•
|
the perception of patients, the healthcare community, and consumers regarding the safety, efficacy
and benefits of our product candidates compared to those of competing products or therapies;
|
|
|
•
|
the convenience and ease of administration of our product candidates relative to existing treatment
methods; and
|
|
|
•
|
marketing and distribution support for our product candidates.
|
We face substantial competition in
the development of our product candidates which may result in others developing or commercializing products before or more successfully
than we do.
We are engaged in segments of the cosmeceuticals
industry that are characterized by intense competition and rapidly evolving technology. Many large companies, academic institutions,
and other public and private research organizations are pursuing the development of competing products. We face, and expect to
continue to face, intense and increasing competition as new products enter the market and advanced technologies become available.
Many of our potential competitors have significantly greater financial, technical and human resources than we have and may be better
equipped to discover, develop, manufacture, and commercialize product candidates.
Risks Related to Our Intellectual Property
If we are unable to protect our intellectual
property rights, our competitors may develop and market similar or identical products that may reduce demand for our products,
and we may be prevented from establishing collaborative relationships on favorable terms.
The following factors are important to
our success:
|
|
•
|
receiving patent protection for our product candidates;
|
|
|
•
|
maintaining our trade secrets;
|
|
|
•
|
not infringing on the proprietary rights of others; and
|
|
|
•
|
preventing others from infringing on our proprietary rights.
|
We will be able to protect our proprietary
rights from unauthorized use by third parties only to the extent that our proprietary rights are covered by valid and enforceable
patents or are effectively maintained as trade secrets. We try to protect our proprietary position by filing U.S. and foreign patent
applications related to our proprietary technology, inventions and improvements that are important to the development of our business.
Our pending patent applications, those
we may file in the future, or those we may license from third parties, may not result in patents being issued. If patents do not
issue with claims encompassing our products, our competitors may develop and market similar or identical products that compete
with ours. Even if such patents are issued, they may not provide us with proprietary protection or competitive advantages against
competitors with similar technology. Failure to obtain effective patent protection for our technology and products may reduce demand
for our products and prevent us from establishing collaborative relationships on favorable terms.
We also rely on trade secrets, know-how
and technology, which are not protected by patents, to maintain our competitive position. We try to protect this information by
entering into confidentiality agreements with parties that have access to it, such as potential corporate partners, collaborators,
employees and consultants. Any of these parties may breach the agreements and disclose our confidential information or our competitors
may learn of the information in some other way. Furthermore, others may independently develop similar technologies or duplicate
any technology that we have developed. If any trade secret, know-how or other technology not protected by a patent were to be disclosed
to or independently developed by a competitor, our business and financial condition could be materially adversely affected.
The laws of many foreign countries do not
protect intellectual property rights to the same extent as do the laws of the United States. Accordingly, the fact that we have
obtained certain patent rights in the United States does not guarantee that we will be able to obtain the same or similar rights
elsewhere. Even if we are granted patents in foreign countries, we cannot guarantee that we will be able to enforce our rights
effectively.
If we are unable to obtain and maintain
protection for our intellectual property, the value of our technology and products may be adversely affected, which would materially
affect our business
.
Patents
. Our commercial success
will continue to depend in part on the patent rights we plan to obtain related to future products we may market. Our success also
depends on our and our licensors’, collaborators’, and suppliers’ ability to maintain these patent rights against
third-party challenges to their validity, scope, or enforceability of these patent rights.
Our patent position (and those of our licensors,
collaborators, and suppliers) is subject to the same uncertainty as other pharmaceutical and consumer product companies. Our patents
and patent applications (as well as those of our licensors, collaborators and suppliers) may not protect our technologies and products
because, among other things, our pending applications may not result in issued patents; we may develop additional proprietary technologies
that are not patentable; patents issued to us may not provide a basis for future commercially viable products; and patents issued
to us may not provide us with any competitive advantage, or may be challenged, circumvented, invalidated or rendered unenforceable
by third parties. For example, the USPTO or the courts may deny, narrow, or invalidate patent claims, particularly those that concern
biotechnology and pharmaceutical inventions. Inventors or third parties of whom we are unaware, may challenge the ownership of
patents and applications we own, license or benefit from through supply agreements with our collaborators and suppliers. We, our
licensors, collaborators, and suppliers may not be successful in securing or maintaining proprietary or patent protection for our
products, and protection that is secured may be challenged and possibly lost.
Trade Secrets and Proprietary Know-how
.
We, our licensors, collaborators, and suppliers also rely upon trade secrets, proprietary know-how, and other technological innovation,
particularly when patent protection is not appropriate or available. However trade secrets are difficult to protect, and we have
limited control over the protection of trade secrets used by our licensors, collaborators, and suppliers. Although we attempt to
protect our trade secrets by requiring our employees, consultants, advisors and current and prospective business partners to enter
into confidentiality agreements prohibiting them from disclosing or taking our proprietary information and technology, these agreements
may not provide meaningful protection for our trade secrets and proprietary know-how. If our employees or consultants breach these
agreements, we may not have adequate remedies for any of these breaches. Further, third parties that are not parties to confidentiality
agreements may obtain access to our trade secrets or know-how. Others may independently develop similar or equivalent trade secrets
or know-how. If our confidential or proprietary information is divulged to third parties, including our competitors, our competitive
position in the marketplace will be harmed and our ability to successfully penetrate our target markets could be severely compromised.
Trademarks
. Our trademarks will
continue to be important to our success and competitive position. We have received U.S. trademark registration for our corporate
name, NuGene
®
, and own or have rights to use our product and component names. We also have a license for the use
of Kathy Ireland
®
who acts as our brand ambassador. We also will need to pursue trademark registration for
any new trademarks that we select. We may not be able to secure any of our trademark registrations with the PTO or comparable foreign
authorities. If we do not adequately protect our rights in our various trademarks from infringement (and we are involved in two
separate ongoing disputes with respect to trademarks), any goodwill that has been developed in those marks would be lost or impaired.
We could also be forced to cease using any of our trademarks that are found to infringe upon or otherwise violate the trademark
or service mark rights of another company, and, as a result, we could lose all the goodwill that has been developed in those marks
and could be liable for damages caused by any infringement or violation.
We may be subject to claims that
we, or our employees, have inadvertently or otherwise used or disclosed alleged trade secrets or other proprietary information
of our employees’ former employers.
We employ individuals who were previously
employed at other personal care product or nutritional supplement companies, including our competitors or potential competitors.
To the extent that our employees are involved in research areas that are similar to those in which they were involved with their
former employers, we may be subject to claims that such employees have inadvertently or otherwise used or disclosed the alleged
trade secrets or other proprietary information of the former employers. Litigation may be necessary to defend against such claims.
Claims by other parties that we infringe
or have misappropriated their proprietary technology may result in liability for damages, royalties, or other payments, or stop
our development and commercialization efforts.
Competitors and other third parties may
initiate patent litigation against us in the United States or in foreign countries based on existing patents or patents that may
be granted in the future. Many of our competitors may have obtained patents covering products and processes generally related to
our products and processes, and they may assert these patents against us. Moreover, there can be no assurance that these competitors
have not sought or will not seek additional patents that may cover aspects of our technology. As a result, there is a greater likelihood
of a patent dispute than would be expected if our competitors were pursuing unrelated technologies.
While we conduct patent searches to determine
whether the technologies used in our products infringe patents held by third parties, numerous patent applications are currently
pending and may be filed in the future for technologies generally related to our technologies, including many patent applications
that remain confidential after filing. Due to these factors and the inherent uncertainty in conducting patent searches, there can
be no guarantee that we will not violate third-party patent rights that we have not yet identified.
There may be U.S. and foreign patents issued
to third parties that relate to aspects of our product candidates. There may also be patent applications filed by these or other
parties in the United States and various foreign jurisdictions that relate to some aspects of our product candidates, which, if
issued, could subject us to infringement actions. The owners or licensees of these and other patents may file one or more infringement
actions against us. In addition, a competitor may claim misappropriation of a trade secret by an employee hired from that competitor.
Any such infringement or misappropriation action could cause us to incur substantial costs defending the lawsuit and could distract
our management from our business, even if the allegations of infringement or misappropriation are unwarranted. A need to defend
multiple actions or claims could have a disproportionately greater impact. In addition, either in response to or in anticipation
of any such infringement or misappropriation claim, we may enter into commercial agreements with the owners or licensees of these
rights. The terms of these commercial agreements may include substantial payments, including substantial royalty payments on revenues
received by us in connection with the commercialization of our products.
Payments under such agreements could increase
our operating losses and reduce our resources available for development activities. Furthermore, a party making this type of claim
could secure a judgment that requires us to pay substantial damages, which would increase our operating losses and reduce our resources
available for development activities. A judgment could also include an injunction or other court order that could prevent us from
making, using, selling, offering for sale, or importing our products or prevent our customers from using our products. If a court
determined or if we independently concluded that any of our products or manufacturing processes violated third-party proprietary
rights, our clinical trials could be delayed and there can be no assurance that we would be able to reengineer the product or processes
to avoid those rights, or to obtain a license under those rights on commercially reasonable terms, if at all.
RISKS RELATED TO REGULATORY MATTERS
While we believe that that our principal
cosmeceutical products and product candidates do not require FDA approval as new drugs, the FDA could disagree and we may be required
to conduct clinical trials to establish efficacy and safety or cease to market these product
s.
Our cosmeceutical products are marketed
on the basis that they are generally recognized as safe and effective for their intended use and thus do not require new drug approval.
The FDA has not challenged this position. The FDA may at any time disagree with our position for a variety of reasons, including
new information about the particular product or its active ingredients, how the product is promoted, if another company obtains
FDA approval for a prescription drug with the same active ingredient, or based on a change of FDA regulatory policy. This could
require us to seek new drug approval for these products to remain on the market or to withdraw a product until required clinical
trials are performed and new drug approval is obtained.
If the active ingredients of the products
are finally determined by the FDA not to be generally recognized as safe and effective for over-the-counter, or OTC use, the FDA
may seek to apply those findings to prescription products as well, leading to potential objections to the continued marketing of
the products or a demand that marketing continue only on the basis of a new drug approval. Either of these outcomes could affect
the way our products are marketed or our ability to market them at all. Further, the FDA could decide that growth factors derived
from human adipose stem cells do not come within this policy and thus must seek new drug approval to remain on the market or must
be withdrawn until approval is obtained.
Our wound healing-wound care products under
development may not be approved by the FDA or foreign regulatory authorities, and any failure or delay associated with our product
development and clinical trials or obtaining regulatory approval of these products would increase our product development costs
and time to market. We face substantial risks of failure inherent in developing stem cell growth factor derived products that may
be determined by the FDA to fall under the classification as pharmaceutical products. The pharmaceutical industry is subject to
stringent regulation by many different agencies at the federal, state, and international levels. If determined to be pharmaceutical
products, then the product candidates will be required to satisfy rigorous standards of safety and efficacy before the FDA approves
them, and before any foreign regulatory authorities approve them for commercial use in any countries outside the U.S. where we
decide to market them. Even if a regulatory filing is accepted, the FDA or foreign regulatory authorities may request additional
information from us, including data from additional clinical trials, and, ultimately, may not grant marketing approval for some
of our products or may grant approval only under conditions that are less commercially attractive than anticipated. To the extent
that these products do not perform successfully in our planned pivotal clinical trials, we may need to develop alternative candidates.
Product development is generally a long, expensive, and uncertain process. Successful development of our new wound healing-wound
care product formulations, including our incisional and or burn cream will depend on many factors, including:
|
|
•
|
Our ability to select key components, establish a stable formulation and optimize characteristics;
|
|
|
•
|
Our ability to develop a formulation that demonstrates our intended safety and efficacy profile;
and
|
|
|
•
|
Our ability to transfer from an early-stage company to commercial-scale operations and the costs
associated with commercial manufacturing.
|
If we are unable to develop suitable clinical
formulations of our wound healing-wound care product candidates or are significantly delayed in doing so, our ability to commercialize
these products will be adversely affected. Once we have manufactured a formulation that we believe is suitable for pivotal clinical
testing, we will need to complete our clinical testing, and failure can occur at any stage of testing. These clinical tests must
comply with FDA and other applicable regulations. We may suffer significant setbacks in advanced clinical trials, even after showing
promising results in earlier trials. The results of later clinical trials may not replicate the results of prior clinical trials.
Based on results at any stage of clinical trials, we may decide to discontinue development of a product candidate. We, or the FDA,
may suspend clinical trials at any time if the patients participating in the trials are exposed to unacceptable health risks or
if the FDA finds deficiencies in our applications to conduct the clinical trials or in the conduct of our trials. Moreover, not
all products in clinical testing will receive timely, or any, regulatory approval. Even if clinical trials are completed as planned,
their results may not support our assumptions or our product claims. The clinical process may fail to demonstrate that our products
are safe for humans or effective for intended uses. In addition, these failures could cause us to abandon a product entirely. If
we fail to take any current or future product candidate from the development stage to market, we will have incurred significant
expenses without the possibility of generating revenues, and our business will be adversely affected.
We will be subject to ongoing regulatory
review of products currently under development that may be marketed in the future.
Any of our pharmaceutical products under
development will be subject to extensive regulation. These regulations will impact many aspects of our operations, including the
manufacture, labeling, packaging, adverse event reporting, storage, advertising, promotion, and record keeping related to the products.
The FDA also may require post-marketing testing and surveillance to monitor the effects of approved products or place conditions
on any approvals that could restrict the commercial applications of these products. In addition, the subsequent discovery of previously
unknown problems with a product may result in restrictions on the product, including withdrawal of the product from the market.
If we fail to comply with applicable regulatory requirements, we may be subject to fines, suspension, or withdrawal of regulatory
approvals, product recalls, seizure of products, injunctions against their distribution, disgorgement of money, operating restrictions,
and criminal prosecution.
In addition to FDA restrictions on marketing
of pharmaceutical products, several other types of state and federal laws have been applied to restrict certain marketing practices
in the pharmaceutical industry in recent years. These laws include anti-kickback statutes and false claims statutes. Violations
of the federal anti-kickback statute are punishable by imprisonment, criminal fines, civil monetary penalties, and exclusion from
participation in federal healthcare programs. Although there are a number of statutory exemptions and regulatory safe harbors protecting
certain common activities from prosecution or other regulatory sanctions under the federal anti-kickback statute, the exemptions
and safe harbors are drawn narrowly, and practices that involve remuneration intended to induce prescribing, purchasing, arranging
for or recommending prescription or purchase may be subject to scrutiny if they do not qualify for a statutory exemption or safe
harbor. Federal false claims laws prohibit any person from knowingly making, or causing to be made, a false claim to the federal
government, or knowingly making, or causing to be made a false statement to have a false claim paid. The majority of states also
have statutes or regulations similar to the federal anti-kickback law and false claims laws, which apply to items and services
reimbursed under Medicaid and other state programs, or, in several states, apply regardless of the payor. Sanctions under these
federal and state laws may include civil monetary penalties, exclusion of a manufacturer’s products from reimbursement under
government programs, criminal fines, and imprisonment.
In addition, as part of the sales and marketing
process, pharmaceutical companies frequently provide samples of approved drugs to physicians. This practice is overseen by the
FDA and other governmental authorities under regulations that include, in particular, requirements concerning record keeping and
control procedures. Any failure to comply with these regulations may result in significant criminal and civil penalties as well
as damage to our credibility in the marketplace. Because of the breadth of these laws and the narrowness of the safe harbors, it
is possible that some of our business activities could be subject to challenge under one or more of these laws, which could have
a material adverse effect on our business, financial condition, and results of operations.
The regulatory status of our cosmeceutical
products could change, and we may be required to conduct clinical trials to establish efficacy and safety or cease to market these
products
.
The Federal Food, Drug, and Cosmetic Act
does not recognize “cosmeceuticals” as a category of products. We use the term “cosmeceuticals” as a marketing
term to describe our non-prescription, cosmetic products. The FDA does not have a premarket approval system for cosmetic products
outside of new color additives, and we believe we are permitted to market our cosmeceutical products and have them manufactured
without submitting safety or efficacy data to the FDA. However, the FDA may in the future determine to regulate what we term as
cosmeceuticals or the ingredients included in our cosmeceuticals as drugs or biologics, rather than cosmetics. If any of our products
are deemed to be drugs or biologics, rather than cosmetics, we would be required to conduct clinical trials to demonstrate the
safety and efficacy of our cosmeceutical products in order to continue to market and sell them. In such event, we may not have
sufficient resources to conduct any required clinical trials and we may not be able to establish sufficient efficacy or safety
data to resume the sale of our cosmeceutical products. Any inquiries by the FDA or any foreign regulatory authorities into the
regulatory status of our cosmeceutical products and any related interruption in the marketing and sale of our cosmeceutical products
could severely damage our brands and image in the marketplace, including our relationships with physicians and their patients.
If we choose, or are required, to
seek FDA approval to market and sell any of our products, we may be unable to demonstrate the necessary safety and efficacy to
obtain such FDA approval.
Our current business strategy is focused
on developing and commercializing product opportunities as cosmeceuticals not requiring FDA approval. In the future we, alone or
with project partners, may seek FDA regulatory approval to market and sell one or more of our products assets as an FDA-approved
drug. The FDA may also seek to classify one or more of our products as drug rather a cosmeceutical. If we choose, or are required,
to obtain FDA approval to market and sell any of our products, the approval process is costly, time consuming, uncertain, and subject
to unanticipated delays. The FDA or other regulatory agencies may not approve a product candidate on a timely basis or at all.
Before we obtain FDA approval for the sale of any product, we will be required to demonstrate through preclinical studies and clinical
trials that it is safe and effective for each intended use, which we may not be able to do. A failure to demonstrate safety and
efficacy of a product candidate to the FDA’s satisfaction would result in our failure to obtain FDA approval. Moreover, even
if the FDA were to grant regulatory approval of a product candidate, the approval may be limited to specific therapeutic areas
or limited as to its distribution, which could reduce revenue potential, and we will be subject to extensive and costly post-approval
requirements and oversight with respect to commercialization of the product candidate.
Delays in the completion of, or the
termination of, any clinical or non-clinical trials for any product candidates for which we may seek FDA approval could adversely
affect our business.
Clinical trials are very expensive, time
consuming, unpredictable and difficult to design and implement. The results of clinical trials may be unfavorable, they may continue
for several years, and they may take significantly longer to complete and involve significantly more costs than expected. Delays
in the commencement or completion of clinical testing could significantly affect product development costs and plans with respect
to any product candidate for which we seek FDA approval. The commencement and completion of clinical trials can be delayed and
experience difficulties for a number of reasons, including delays and difficulties caused by circumstances over which we may have
no control. For instance, approvals of the scope, design or trial site may not be obtained from the FDA and other required bodies
in a timely manner or at all, agreements with acceptable terms may not be reached in a timely manner or at all with clinical research
organizations to conduct the trials, a sufficient number of subjects may not be recruited and enrolled in the trials, and third-party
manufacturers of the materials for use in the trials may encounter delays and problems in the manufacturing process, including
failure to produce materials in sufficient quantities or of an acceptable quality to complete the trials. If we were to experience
delays in the commencement or completion of, or if we were to terminate, any clinical or non-clinical trials we pursue in the future,
the commercial prospects for the applicable product candidates may be limited or eliminated, which may prevent us from recouping
our investment in research and development efforts for the product candidate and would have a material adverse effect on our business,
results of operations, financial condition and prospects.
Even if we successfully develop any
product candidate into an FDA-approved drug, failure to comply with continuing federal and state regulations could result in the
loss of approvals to market the drug.
Even if we successfully develop any product
candidate into an FDA-approved drug, we will be subject to extensive continuing regulatory requirements and review, including review
of adverse drug experiences and clinical results from any post-marketing tests or continued actions required as a condition of
approval. The manufacturer and manufacturing facilities we would use to produce any drug preparations would be subject to periodic
review and inspection by the FDA. We will be reliant on third parties to maintain their manufacturing processes in compliance with
FDA and all other applicable regulatory requirements. Any changes to a product that has been approved, including the way it is
manufactured or promoted, will often require FDA approval again before the product, as modified, may be marketed and sold. In addition,
we and the manufacturers of the drug will be subject to ongoing FDA requirements for submission of safety and other post-market
information. If we or the manufacturers of the drug failed to comply with these or any other applicable regulatory requirements,
a regulatory agency may, among other things, issue warning letters, impose civil or criminal penalties, suspend or withdraw regulatory
approval, impose restrictions on our operations, close the facilities of the manufacturers, seize or detain products or require
a product recall.
Regulatory review of the manner in
which we market and promote our products could result in classification of a product as a drug, which could adversely affect our
business.
Regulatory review also covers a company’s
activities in the marketing and promotion of its products. The FDA could review the manner in which we market and promote our products,
including though not limited to claims and information provided on our packaging, in product descriptions and details, and on our
website. If the FDA determines that such marketing and promotion classifies a product as a drug, we would be forced to obtain FDA
approval for such product. We could also face significant potential penalties and restrictions for promotion of a drug for an unapproved
use. Sales and marketing programs are under scrutiny for compliance with various mandated requirements, such as illegal promotions
to health care professionals. Failure to comply with these requirements could expose us to negative publicity, fines and penalties
that could harm our business.
If our manufacturers do not comply
with U.S. and federal regulations, our supply of product could be disrupted or terminated
.
Our manufacturers must comply with U.S.
regulations and corresponding foreign standards, including the FDA’s current Good Manufacturing Practice regulations for
drug manufacturing and processing, or “cGMPs”, applicable to the manufacturing processes related to ingredients sold
to us for use in our products, and their facilities must be inspected and approved by the FDA and other regulatory agencies as
part of their business. We will have limited control over the FDA compliance of our third-party manufacturers. If any of our manufacturers
fail to meet or are found to be non-compliant with the cGMPs or any other FDA requirements or similar regulatory requirements outside
of the U.S., obtaining the required regulatory approvals, including from the FDA, to use alternative suppliers may be a lengthy
and uncertain process. A lengthy interruption in the manufacturing of one or more of our products as a result of non-compliance
could adversely affect our product inventories and supply of products available for sale which could reduce our sales, margins
and market share, as well as harm our overall business and financial results. Additionally, the Federal Drug and Cosmetic Act (“FDCA”)
may hold labelers/specification developers (brands selling a product) criminally and civilly liable for the violations, acts, and
omission of their manufacturers.
Under the FDCA, cosmetics (which we refer
to as cosmeceuticals) are defined as articles applied to the human body to cleanse, beautify, or alter the appearance. The manufacturing
of cosmetics is subject to the misbranding and adulteration sections of the FDCA applicable to cosmetics. Cosmetics are not subject
to premarket approval by the FDA but the product and ingredients must be tested to assure safety. If the product or ingredients
are not tested for safety, a specific warning is required. The FDA monitors compliance of cosmetic products through random inspections
of cosmetic manufacturers and distributors. The FDA utilizes an “intended use” doctrine to determine whether a product
is a drug or cosmetic by the labeling claims made for the product. If a cosmetic product is intended for a disease condition or
to affect the structure or any function of the human body, the FDA will regulate the product as a drug rather than as a cosmetic.
The product will then be subject to all drug requirements under the FDCA. The labeling of cosmetic products is subject to the requirements
of the FDCA, the Fair Packaging and Labeling Act and other FDA regulations.
We have only limited experience in regulatory
affairs, which may affect our ability or the time we require to obtain necessary regulatory approvals. We have only limited experience
in regulatory affairs, including the preparation and filing of applications to gain the regulatory approvals necessary for pharmaceutical
product candidates. Moreover, some of the products that are likely to result from our product development, licensing and acquisition
programs may be based on new technologies that have not been extensively tested in humans. As a result, we may experience a longer
than customary regulatory process in connection with obtaining regulatory approvals for any products that we develop, license or
acquire.
If we move forward with production
of an FDA regulated product, and we are found not to be in compliance with Good Manufacturing Practices, then our operations could
be harmed.
In the United States, FDA regulations on
Good Manufacturing Practices and Adverse Event Reporting requirements require us and our vendors to maintain good manufacturing
processes, including stringent vendor qualifications, ingredient identification, manufacturing controls and record keeping.
The ingredient identification requirement, which requires us to confirm the levels, identity, and potency of ingredients listed
on our product labels within a narrow range, is particularly burdensome and difficult for us with respect to products that contain
many different ingredients. We are also required to report serious adverse events associated with consumer use of our products.
Our operations could be harmed if regulatory authorities make determinations that we, or our vendors, are not in compliance with
these regulations or public reporting of adverse events harms our reputation for quality and safety. A finding of noncompliance
may result in administrative warnings, penalties, or actions impacting our ability to continue selling certain products.
In addition, compliance with these regulations has increased and may further increase the cost of manufacturing certain of our
products as we work with our vendors to assure they are qualified and in compliance.
RISKS RELATED TO OUR BUSINESS
Our products and product candidates
may not achieve or maintain widespread market acceptance.
We may not achieve or maintain widespread
market acceptance of our products or product candidates among physicians, patients, or healthcare providers. Our products’
success is highly dependent on physician and patient preference and market acceptance. We have a limited history of promoting our
cosmeceutical products. Our significant marketing efforts to date have been focused primarily on dermatologists and plastic surgeons.
We believe that market acceptance of our
products will depend on many factors including:
|
|
•
|
The perceived advantages of our products over competing products;
|
|
|
•
|
The effectiveness of our sales and marketing efforts;
|
|
|
•
|
The convenience and ease of administration of our products;
|
|
|
•
|
The safety and efficacy of our products and the prevalence and severity of any possible adverse
side effects;
|
|
|
•
|
The availability and success of alternative treatments;
|
|
|
•
|
Our product pricing and cost effectiveness;
|
|
|
•
|
Publicity concerning our products, product candidates or competitive products;
|
|
|
•
|
Whether or not patients routinely use our products and purchase additional product, and
|
|
|
•
|
Our ability to respond to changes in physician, aestheticians, and patient preferences for the
treatment of dermatological conditions and the improvement of the appearance of skin.
|
If our products fail to achieve or
maintain market acceptance or if new products or technologies are introduced by others that are more favorably received than our
products, are more cost effective or that otherwise render our products obsolete, we may experience a decline in the demand for
our products.
If we are unable to market and sell our
products successfully, our business, financial condition, results of operation and future growth would suffer. Our ability to compete
depends upon the success of our business development activities and our ability, and the ability of our collaborators, to innovate,
develop, and commercialize new products and product enhancements, as well as to identify new markets for our products.
Our business strategy requires us to develop
or acquire new and innovative applications of our products, identify new markets for our existing products, and develop or acquire
new technology. We are currently developing products for the treatment of burns and exploring several delivery technologies to
improve our existing products. However, our development efforts may not lead to new commercial products. To successfully expand
our product offerings, we must:
|
|
•
|
Develop or acquire new products that either add to or significantly improve our current product
lines;
|
|
|
•
|
Convince our target customers that any new cosmeceutical products or line extensions would be an
attractive revenue-generating addition to their practices;
|
|
|
•
|
Protect our products with defensible intellectual property; and
|
|
|
•
|
Satisfy and maintain all regulatory requirements for commercialization.
|
The process of developing product candidates
involves a high degree of risk and may take several years. Product candidates we may acquire or license in the future that appear
promising in the early phases of development may fail to reach the market for several reasons, including:
|
|
•
|
Pharmaceutical product candidates may fail to receive regulatory approvals required to bring the
products to market;
|
|
|
•
|
Manufacturing costs or other factors may make our pharmaceutical and cosmeceutical product candidates
uneconomical;
|
|
|
•
|
The proprietary rights of others and their competing products and technologies may prevent our
product candidates from being commercialized;
|
|
|
•
|
Success of pharmaceutical product candidates in nonclinical and early clinical studies does not
ensure that later stage clinical trials will be successful;
|
|
|
•
|
The length of time necessary to complete clinical trials and to submit an application for marketing
approval of pharmaceutical product candidates for a final decision by a regulatory authority varies significantly and may be difficult
to predict; and
|
|
|
•
|
Developing pharmaceutical and cosmeceutical product candidates is very expensive and will have
a significant impact on our operating expenses.
|
We may be unable to continue to develop
new products, enhancements to our existing products and other technologies in the near term, if at all, in part because new products
or enhancements to our existing products must meet regulatory standards and receive requisite regulatory approvals.
Our failure to introduce new products or
enhancements to our existing products could adversely affect our expected growth rate and adversely affect our overall business
and financial results.
Our marketed products and our products
under development could be rendered obsolete by technological or medical advances
.
The development of medical advances to
treat the conditions that our products are designed to address may render our marketed products and our products under development
obsolete or uneconomical. The enhancement to the appearance of skin, the regeneration of hair growth and the treatment of burns,
acne or other skin disorders are the subjects of active research and development by many potential competitors, including major
pharmaceutical companies, specialized biotechnology firms, universities and other research institutions, as well as other major
cosmeceutical companies which develop wrinkle reduction or age defying skin and hair care products. While we intend to expand our
technological capabilities to remain competitive, research and development by others may render our technology or products obsolete
or noncompetitive or result in treatments superior to any therapy we develop.
Technological advances affecting costs
of production also could adversely affect our ability to sell products. Our products could become more expensive to produce, or
not competitive, which would decrease our revenues and adversely affect our results of operations and financial condition.
Our future success depends on our
ability to retain our chief executive officer and to attract, retain and motivate qualified personnel.
We are highly dependent on Steven Carlson,
our Chief Executive Officer. The loss of the services of Mr. Carlson might impede the achievement of our research, development,
and commercialization objectives. Replacing Mr. Carlson may be difficult and time-consuming because of the limited number of individuals
in our industry with the skills and experiences required to successfully develop and commercialize our products. We generally do
not maintain key person life insurance to cover the loss of any of our employees.
Recruiting and retaining qualified scientific
personnel, clinical personnel and sales and marketing personnel will also be critical to our success. We may not be able to attract
and retain these personnel on acceptable terms, if at all, given the competition for similar personnel. In addition, we rely on
consultants and advisors, including production, marketing, medical, and clinical advisors, to assist us in formulating our research
and development and commercialization strategy. Our consultants and advisors may be employed by employers other than us and may
have commitments under consulting or advisory contracts with other entities that may limit their availability to us.
During the year ended December 31,
2015, the majority of our revenues were derived from two wholesale distributors. The loss of the distributors could have a material
negative effect on our financial condition.
During the year ended December 31, 2015,
we derived 31.1% and 15.7% of our sales from two wholesale distributors. These two distributors purchase products from us on a
purchase order basis on negotiated terms of payment. The distributors are under no obligation to continue to purchase our products.
The loss of either of the distributors, a material reduction in their purchases or the cancellation of product orders or unexpected
returns of unsold products could decrease our revenues and impede our future growth prospects. We do not have long-term purchase
commitments with our distributors. We are actively seeking to expand our products’ distribution channels in order to reduce
the impact the loss of any one distributor would have on our Company, however we can give no assurance that we will be successful
in doing so.
If we breach any of our key license
or supply agreements, we could lose exclusivity rights or the agreements could be terminated
.
We have an international licensing agreement
with kiWW for all cosmetic products for a term of eight years. These rights are important to our business, and any breach of the
related agreements could result in a termination of the respective rights, which, in turn, would prevent us from marketing the
affected products or developing the affected product candidates. Our agreements with kiWW require milestone and royalty payments,
minimum revenue requirements or minimum annual royalty payments and other obligations. If we have insufficient demand for these
products or otherwise fail to meet the minimum purchase requirements or any of the other requirements set forth in these agreements,
we could lose the exclusive nature of our right to market products under the kiWW brand. Additionally, we are overdue in payments
owed to kiWW. Should we not be able to cure this breach of our payment obligations under the agreement within a mutually agreeable
timeframe, we face the possibility that the agreement could be terminated.
If we fail to comply with any of the requirements
under our key license and supply agreements, we may lose exclusive rights under these agreements or they may be terminated in their
entirety. As of the date of this prospectus, we remain overdue in payments owing to many of the counterparties to these agreements,
which increases the risk of such loss or termination. In that event, others could obtain rights to sell products that compete directly
with our products and our revenues and market share would correspondingly decrease. The loss of any rights under any of our license
and supply agreements would adversely affect our ability to sell our products and adversely affect our revenues and results of
operations.
We face risks due to our reliance
on third parties to perform many necessary commercial services for our products, including services related to the distribution,
storage, transportation, and regulatory monitoring of our products.
We rely on third parties to perform a variety
of functions related to the sale and distribution of our cosmeceutical products. These services include distribution, logistics
management, inventory storage and transportation, invoicing and collections, the key aspects of which are out of our direct control.
If any third-party service provider fails to comply with applicable laws and regulations, fails to meet expected deadlines or otherwise
does not carry out its contractual duties to us, our ability to deliver products to meet commercial demand would be significantly
impaired. In addition, we may retain one or more third parties to perform various regulatory monitoring services for our products,
including adverse event reporting, safety database management and other product maintenance services. If the quality or accuracy
of the data maintained by these regulatory service providers is insufficient, our ability to continue to market our approved products
could be jeopardized or we could be subject to regulatory sanctions.
If our competitors develop and market
products faster than we do or if the products of our competitors are considered more desirable than our products, revenues of our
existing or new products may be adversely affected
.
The dermatology market is highly competitive
and includes a number of established, large, and mid-sized pharmaceutical and cosmeceutical companies, as well as smaller emerging
companies and specialty pharmaceutical and cosmeceutical companies, whose activities are focused on our target markets and areas
of expertise. We face and will continue to face, competition for our products and in the commercialization, development, licensing
and discovery of our product candidates. This could negatively impact our ability to achieve significant market acceptance of our
products and product candidates. Furthermore, new developments including the development of other drug technologies, delivery methods
and improved formulations, occur in the pharmaceutical industry at a rapid pace. These developments may render our currently marketed
products and product candidates or technologies obsolete or noncompetitive.
Compared to us, many of our competitors
and potential competitors have substantially greater:
|
|
•
|
Research and development resources, including personnel and technology;
|
|
|
•
|
Favorable brand name awareness
|
|
|
•
|
Clinical trial experience; and
|
|
|
•
|
Manufacturing, distribution and sales and marketing experience.
|
As a result of these factors, our competitors
may obtain regulatory approval of their products more rapidly than us. Our competitors may obtain patent protection or other intellectual
property rights that limit our ability to develop or commercialize our product candidates or technologies. Our competitors may
also develop pharmaceutical and cosmeceutical products that are more effective and less costly than ours and may also be more successful
than us in manufacturing and marketing their products.
Other competitors may invest significant
amounts in achieving production economies of scale
.
It is possible that our competitors may
be able to reduce their cost of production so that they can aggressively price their products and increase their greater market
share. Our competitors may also be able to attract and retain qualified personnel and to secure capital resources. Any of these
events could adversely affect our ability to compete and our results of operations could suffer.
Our products and product candidates
may cause undesirable side effects that could limit their use
.
Skin irritation is a reported side effect
of cosmeceutical products. Although these side effects generally are not severe, they may limit the use of our products, particularly
if physicians or patients perceive the risks to outweigh the benefits or the side effects of competitive products to be less significant.
If more severe side effects associated with any of our cosmeceutical products were to be reported or observed, we could be required
to suspend our marketing of the products, conduct additional safety tests, and potentially cease the sale of the products. In addition,
we face the potential for product liability claims from any patients who experience side effects, whether or not any action is
taken by a regulatory authority.
Undesirable side effects caused by our
product candidates could interrupt, delay or halt our development programs, including clinical trials, and could result in the
denial of any required regulatory approval by the FDA or other regulatory authorities.
We may face liability and indemnity
claims that could result in unexpected costs and damage to our reputation.
Our business exposes us to potential liability
risks that arise from the testing, manufacture and sale of our cosmeceutical products. Plaintiffs in the past have received substantial
damage awards against pharmaceutical companies based upon claims for injuries allegedly caused by the use of their products. Although
we currently maintain product liability insurance, there is no guarantee that any claims brought against us would be within our
existing or future insurance policy coverage or limits. Any judgment against us that is in excess of our policy limits would have
to be paid from our cash reserves, which would reduce our capital resources. Further, we may not have sufficient capital resources
to pay a judgment, in which case our creditors could levy against our assets. Also, it may be necessary for us to recall products
that do not meet approved specifications, which would result in adverse publicity, potentially significant costs in connection
with the recall and a loss of revenues. Any product liability claim or series of claims brought against us could harm our business
significantly, particularly if a claim results in adverse publicity or damage awards outside or in excess of our insurance policy
limits.
Failure to adequately comply with
information security policies or to safeguard against breaches of such policies could adversely affect our operations and could
damage our business, reputation, financial position, and results of operations
.
In the process of making sales using consumer
credit cards as a method of payment, we may handle and transfer such information as part of our business. These activities are
subject to laws and regulations, as well as industry standards, in the United States and other jurisdictions in which our products
and services are available. These requirements, which often differ materially and sometimes conflict among the many jurisdictions
in which we operate, are designed to protect the privacy of consumers’ personal information and to prevent that information
from being inappropriately used or disclosed. We maintain and review technical and operational safeguards designed to protect this
information and generally require others with whom we work to do so as well. However, despite those safeguards, it is possible
that hackers, employees acting contrary to our policies, third-party agents or others could improperly access relevant systems
or improperly obtain or disclose data about our consumers, or that we may be determined not to be in compliance with applicable
legal requirements and industry standards for data security, such as the Payment Card Industry guidelines. A breach or purported
breach of relevant security policies that compromises consumer data or determination of non-compliance with applicable legal requirements
or industry standards for data security could expose us to regulatory enforcement actions, card association or other monetary fines
or sanctions, or contractual liabilities, limit our ability to provide our products and services, subject us to legal action and
related costs and damage our business reputation, financial position, and results of operations.
Changes in economic conditions could
materially affect our ability to maintain or increase sales.
The cosmeceutical industry depends on consumer
discretionary spending. The United States in general or the specific markets in which we operate may suffer from depressed
economic activity, recessionary economic conditions, higher fuel or energy costs, low consumer confidence, high levels of unemployment,
reduced home values, increases in home foreclosures, investment losses, personal bankruptcies, reduced access to credit or other
economic factors that may affect consumers’ discretionary spending. Economic conditions may remain volatile and may depress
consumer confidence and discretionary spending in the future. Negative economic conditions, if and when they exist, might cause
consumers to make changes to their discretionary spending behavior, including spending currently made on our cosmeceutical line
of products. If such sales decrease, our profitability could decline as we spread fixed costs across a lower level of sales and
this could materially adversely affect our business, financial condition, or results of operations.
The cosmeceutical industry in which
we operate is highly competitive and increased competition could reduce our sales and profitability
.
We compete in different markets within
the cosmeceutical industry on the basis of the uniqueness of our product offerings, the quality of our products, customer service,
price, and distribution. Our markets are highly competitive. Our competitors vary in size and many may have greater
financial and marketing resources than we do. If we cannot maintain quality and pricing that are comparable or superior to
our competitors, we may not be able to grow our revenues and operating profits and we may lose market share. Competitive
conditions could result in our experiencing reduced revenues, gross margins, and operating results and could cause an investor
in our Company to lose a substantial amount or all of its investment in our Company.
The loss of suppliers or shortages
in ingredients could harm our business.
We acquire ingredients and products from
third-party suppliers and manufacturers. A loss of any of these suppliers and any difficulties in finding or transitioning to alternative
suppliers could harm our business. In addition, we obtain some of our products from sole suppliers that own or control the product
formulations, ingredients, or other intellectual property rights associated with such products. In the event we are unable to renew
these contracts, we may need to discontinue some products or develop substitute products, which could harm our revenue. In addition,
if we experience supply shortages or regulatory impediments with respect to the raw materials and ingredients we use in our products,
we may need to seek alternative supplies or suppliers and may experience difficulties in finding ingredients that are comparable
in quality and price. Some of our products incorporate products that may have limited supplies. If demand exceeds forecasts, we
may have difficulties in obtaining additional supplies to meet the excess demand. If we are unable to successfully respond to such
issues, our business could be harmed.
Risks Related to Our Common Stock and
Warrants
The recent public market for our
shares has been and may continue to be volatile. This volatility may affect the ability of our investors to sell their shares and
warrants as well as the price at which they sell their shares and warrants.
The market price for our shares and warrants
may be significantly affected by factors such as variations in the volume of trading activity, quarterly and yearly operating results,
general trends in the markets we serve, press releases announcing developments and changes in state or federal regulations affecting
us and our industry. Furthermore, in recent years the stock market has experienced extreme price and volume fluctuations that are
unrelated or disproportionate to the operating performance of the affected companies. Such broad market fluctuations may adversely
affect the market price of our common stock and warrants.
Because our stock price is volatile,
purchasers of our common stock and warrants could incur substantial losses.
Our stock price has been and may continue
to be volatile. The stock market in general and the market for biotechnology companies in particular have experienced extreme volatility
that has often been unrelated to the operating performance of particular companies. The market price for our common stock and warrants
may be influenced by many factors, including:
|
|
•
|
results of clinical trials of our product candidates or those of our competitors;
|
|
|
•
|
regulatory or legal developments in the United States and other countries;
|
|
|
•
|
variations in our financial results or those of companies that are perceived to be similar to us;
|
|
|
•
|
developments or disputes concerning patents or other proprietary rights;
|
|
|
•
|
the recruitment or departure of key personnel;
|
|
|
•
|
market conditions in the pharmaceutical and biotechnology sectors and issuance of new or changed
securities analysts’ reports or recommendations;
|
|
|
•
|
general economic, industry and market conditions; and
|
|
|
•
|
the other factors described in this “Risk Factors” section.
|
The application of the “penny
stock” rules to our common stock and warrants could limit the trading and liquidity of our common stock and warrants, adversely
affect the market price of our common stock and warrants, and increase your transaction costs to sell those shares and warrants
.
As long as the trading price of our common
stock is below $5 per share, and our common stock and warrants are not listed on a national securities exchange, the open-market
trading of our common stock and warrants will be subject to the “penny stock” rules, unless we otherwise qualify for
an exemption from the “penny stock” definition. The “penny stock” rules impose additional sales practice
requirements on certain broker-dealers who sell securities to persons other than established customers and accredited investors
(generally those with assets, excluding principal residence, in excess of $1,000,000 or annual income exceeding $200,000 or $300,000
together with their spouse). These regulations, if they apply, require the delivery, prior to any transaction involving a penny
stock, of a disclosure schedule explaining the penny stock market and the associated risks. Under these regulations, certain brokers
who recommend such securities to persons other than established customers or certain accredited investors must make a special written
suitability determination regarding such a purchaser and receive such purchaser’s written agreement to a transaction prior
to sale. These regulations may have the effect of limiting the trading activity of our common stock, reducing the liquidity of
an investment in our common stock and warrants, and increasing the transaction costs for sales and purchases of our common stock
as compared to other securities. The stock market in general and the market prices for penny stock companies in particular, have
experienced volatility that often has been unrelated to the operating performance of such companies. These broad market and industry
fluctuations may adversely affect the price of our stock, regardless of our operating performance. Stockholders should be
aware that, according to SEC Release No. 34-29093, the market for penny stocks has suffered in recent years from patterns of fraud
and abuse. Such patterns include 1) control of the market for the security by one or a few broker-dealers that are often related
to the promoter or issuer; 2) manipulation of prices through prearranged matching of purchases and sales and false and misleading
press releases; 3) boiler room practices involving high-pressure sales tactics and unrealistic price projections by inexperienced
sales persons; 4) excessive and undisclosed bid-ask differential and markups by selling broker-dealers; and 5) the wholesale dumping
of the same securities by promoters and broker-dealers after prices have been manipulated to a desired level, along with the resulting
inevitable collapse of those prices and with consequent investor losses. The occurrence of these patterns or practices could increase
the volatility of our share price. Although we intend to ask our shareholders to approve a reverse stock split to increase the
price per share of our common stock such that it would not be subject to the “penny stock” rules, no assurance can
be given that we will be able to effect such reverse stock split or that the per share price of our common stock will improve following
the reverse stock split such that our stock will no longer be subject to these rules.
We may not be able to attract the
attention of brokerage firms, which could have a material adverse impact on the market value of our common stock and warrants.
Security analysts of brokerage firms are
unlikely to provide coverage of our common stock warrants since there is no incentive to brokerage firms to recommend the purchase
of our common stock and warrants. The absence of such coverage limits the likelihood that an active market will develop or be maintained
for our common stock and warrants. It will also likely make it more difficult to attract new investors at times when we require
additional capital.
Future sales of our equity securities
could put downward selling pressure on our securities, and adversely affect the stock and warrant price.
There is a risk that this downward pressure
may make it impossible for an investor to sell his or her securities at any reasonable price, if at all. Future sales of substantial
amounts of our equity securities in the public market, or the perception that such sales could occur, could put downward selling
pressure on our securities, and adversely affect the market price of our common stock and warrants.
Our directors and principal stockholders
beneficially own or control a substantial portion of our outstanding common stock, which may limit the ability of our stockholders,
whether acting alone or together, to propose or direct the management on the overall direction of our Company
.
Additionally, this concentration of ownership
could discourage or prevent a potential takeover of our Company that might otherwise result in an investor receiving a premium
over the market price for his shares. A substantial portion of our outstanding shares of common stock is beneficially owned and
controlled by a group of insiders, including our officers and directors. Accordingly, our principal stockholders together with
our directors, Chief Executive Officer, and insider shareholders have the power to control the election of our directors and the
approval of actions for which the approval of our stockholders is required. If you acquire shares of our common stock, you may
have no effective voice in the management of our Company. Such concentrated control of our Company may adversely affect the
price of our common stock. Our principal stockholders may be able to control matters requiring approval by our stockholders, including
the election of directors, mergers or other business combinations. Such concentrated control may also make it difficult for our
stockholders to receive a premium for their shares of our common stock in the event we merge with a third party or enter into different
transactions that require stockholder approval. These provisions could also limit the price that investors might be willing to
pay in the future for shares of our common stock and warrants.
Our outstanding warrants may be exercised,
our convertible promissory notes may be converted, and our outstanding shares of preferred stock may be converted, in the future,
which would increase the number of shares in the public market and result in dilution to our stockholders.
We are currently authorized
to issue 100,000,000 of our common stock. As of September 30, 2016 we had 40,334,673 shares of our common stock issued and outstanding,
excluding shares of common stock earned but unissued, issuable upon exercise of our outstanding warrants, options, convertible
promissory notes, and shares of common stock into which our issued shares of preferred stock may be converted. To the extent the
shares of common stock are issued, options and warrants are exercised, convertible promissory notes are converted, or preferred
stock is converted, holders of our common stock will experience dilution. In addition, in the event of any future financing of
equity securities or securities convertible into or exchangeable for, common stock, holders of our common stock may experience
dilution. As of September 30, 2016 outstanding warrants to purchase 4,679,234 shares of our common stock at a weighted average
exercise price of approximately $0.78 per share. In addition, as of September 30, 2016 we had outstanding options to purchase 4,352,619
shares of our common stock at a weighted average exercise price of approximately $0.58 per share; and, our issued shares of preferred
stock can be converted into 1,917,720 shares of our common stock. In addition, we also had approximately $2,601,091 outstanding
principal amount and interest of convertible debt as of September 30, 2016 for which the conversion price varies depending on the
average trading price of shares of our common stock. Accordingly, if our stock price decreases, we could be required to issue a
greater number of shares of our common stock upon conversion of this debt than originally anticipated, which could lead to dilution
of your investment in our Company.
There is a limited trading market
for our common stock, and shareholders may have difficulty trading and obtaining quotations for our common stock and wrrants.
Our common stock is quoted on the OTCQB under the symbol “NUGN” and we will apply to list our
common stock on the Nasdaq Capital Market. There has been limited trading in our common stock. As a result, investors may find
it difficult to dispose of, or to obtain accurate quotations of the price of, our securities. A limited market may adversely affect
the market price of our common stock and could also impair our ability to raise capital by selling shares of capital stock and
may impair our ability to acquire other companies or assets by using common stock as consideration.
We have never paid any cash dividends
on our common stock and we do not anticipate paying any cash dividends in the foreseeable future.
We have paid no cash dividends on our common
stock to date. We currently intend to retain our future earnings, if any, to fund the development and growth of our business. In
addition, the terms of any future debt agreements may preclude us from paying dividends. As a result, we do not expect to pay any
cash dividends in the foreseeable future, and payment of cash dividends, if any, will depend on our financial condition, results
of operations, capital requirements and other factors and will be at the discretion of our board of directors. Furthermore, we
may in the future become subject to contractual restrictions on, or prohibitions against, the payment of dividends. Capital appreciation,
if any, of our common stock will be investors’ sole source of gain for the foreseeable future.
Our articles of incorporation allow
for our board to create series of preferred stock without further approval by our stockholders, which has and could further adversely
affect the rights of the holders of our common stock and warrants.
Our articles of incorporation authorize
the Board of Directors to issue up to 25,000,000 shares of “blank check” preferred stock. The preferred stock may be
issued in one or more series, the terms of which may be determined at the time of issuance by the Board of Directors without further
action by the stockholders. These terms may include preferences as to dividends and liquidation, conversion rights, redemption
rights and sinking fund provisions. We have issued 1,917,720 shares of Series A Preferred Stock to NuGene’s two founders,
both of whom also serve on our Board of Directors. The Series A Preferred Stock is initially convertible into common stock at a
ratio of one to one, has the right to elect a majority of the board of directors, and has in connection with any other vote of
shareholders three votes for every vote available to the common stock. These outstanding shares of Series A Preferred Stock diminish
and any future issuances of other series of preferred stock could further diminish the rights of holders of our common stock, and
therefore could reduce the value of such common stock and warrants. In addition, the Series A Preferred Stock could be used to
restrict our ability to merge with, or sell assets to, a third party. The Series A Preferred Stock makes it more difficult, and
could delay, discourage, prevent or make it more costly to acquire or effect a change-in-control, which in turn could prevent our
stockholders from recognizing a gain in the event that a favorable offer is extended and could materially and negatively affect
the market price of our common stock. This summary description of the Series A Preferred Stock is qualified in its entirety by
reference to the Certificate of Designations filed with Secretary of State of Nevada on December 24, 2014 and included in our Current
Report on Form 8-K filed with the SEC on January 6, 2015.
CORPORATE AND OTHER RISKS
We incur substantial costs as a result
of operating as a public company, and our management is required to devote substantial time to comply with public company regulations.
We are subject to the reporting requirements
of the Exchange Act, the Sarbanes-Oxley Act of 2002 as well as other federal and state laws. These requirements may place a strain
on our people, systems, and resources. The Exchange Act requires that we file annual, quarterly, and current reports with respect
to our business and financial condition. The Sarbanes-Oxley Act requires that we maintain effective disclosure controls and procedures
and internal controls over financial reporting. In order to maintain and improve the effectiveness of our disclosure controls and
procedures and internal controls over financial reporting, significant resources and management oversight are required. This may
divert management’s attention from other business concerns, which could have a material adverse effect on our business, financial
condition, results of operations and cash flows.
Our Chief Executive Officer and our
Chief Financial Officer have limited experience in these roles in a public company.
To serve in the roles of officer and/or
director for a public company, an individual needs to be aware of responsibilities in addition to those shouldered by the leader
of a private company. Among such additional responsibilities, a senior executive officer must be able to communicate fairly
and effectively with the stakeholders of a public company, be aware of the controls required to be maintained by a public company
and act in accordance with the legal requirements incumbent upon such senior executives and directors. While our CEO has experience
in managerial positons in large public companies and led another company through a public offering, he does not have extensive
experience in operating a public company. Our CFO has never held a positon in a public company and he is not a trained financial
expert. The absence of such extensive experience (and training in the case of our CFO) could increase our exposure to untimely
compliance with applicable regulation that could result in possible added liability and cost to the material detriment of our operations
and financial interests. Our CFO is a medical doctor by training and has assumed the title and responsibility of a chief
financial officer but has not had any prior experience in the traditional services to be rendered but has rather relied on staff
and consultants in his acting in this capacity.
We do not have an Audit Committee
and we do not have a majority of independent persons serving on our Board of Directors
.
We do not have an independent Audit Committee.
An Audit Committee qualitatively enhances a company’s internal controls over financial reporting. Among its functions, independent
Audit Committees review the financial reporting, internal controls safeguarding Company assets, interact with auditors, may oversee
material financial decisions and provide a sounding board for individuals who believe that there are irregularities in a Company’s
accounting policies and procedures. Our Board of Directors consists of our two founders and one independent director. With
our lack of an independent Audit Committee and only one independent director, we run a greater risk that a significant error or
irregularity could occur that could be materially damaging to our shareholders.
For as long as we remain an “emerging
growth company” as defined in the JOBS Act, we may take advantage of certain exemptions from various reporting requirements
that are applicable to other public companies that are not “emerging growth companies.”
These exceptions provide for, but are not
limited to, relief from the auditor attestation requirements of Section 404 of the Sarbanes-Oxley Act, less extensive disclosure
obligations regarding executive compensation in our periodic reports and proxy statements, exemptions from the requirements to
hold a nonbinding advisory vote on executive compensation and stockholder approval of any golden parachute payments not previously
approved and an extended transition period for complying with new or revised accounting standards. We may take advantage of these
reporting exemptions until we are no longer an “emerging growth company.” We may remain an “emerging growth company”
for up to five years. To the extent we use exemptions from various reporting requirements under the JOBS Act, we may be unable
to realize our anticipated cost savings from those exemptions.
Pursuant to the JOBS Act, our independent
registered public accounting firm will not be required to attest to the effectiveness of our internal control over financial reporting
pursuant to Section 404 of the Sarbanes-Oxley Act for so long as we are characterized as an “emerging growth company.”
Section 404 of the Sarbanes-Oxley Act requires annual management assessments of the effectiveness of our internal control
over financial reporting, starting with the second annual report that we file with the SEC as a public company, and generally requires
in the same report a report by our independent registered public accounting firm on the effectiveness of our internal control over
financial reporting. However, under the JOBS Act, our independent registered public accounting firm will not be required to attest
to the effectiveness of our internal control over financial reporting pursuant to Section 404 of the Sarbanes-Oxley Act until
we are no longer an “emerging growth company”.
We are also currently able to take advantage
of certain of these exemptions as a smaller reporting company. We cannot predict if investors will find our common stock less attractive
because we may rely on these exemptions. If some investors find our common stock less attractive as a result, there may be a less
active trading market for our common stock and our stock price may be more volatile.
Risks Related to this Offering
Investors in this offering will suffer
immediate and substantial dilution.
Because the price per share of our common
stock being offered is substantially higher than the net tangible book value per share of our common stock, you will suffer substantial
dilution in the net tangible book value of the common stock you purchase in this offering. If you purchase shares of common stock
in this offering, you will suffer immediate and substantial dilution of $ per share in the net tangible
book value of the common stock. See the section entitled “Dilution” below for a more detailed discussion of the dilution
you will incur if you purchase common stock in this offering.
We will have considerable discretion
over the use of the proceeds of this offering and may not realize an adequate return.
We will have considerable discretion in
the application of the net proceeds of this offering. We have not determined the amount of net proceeds that we will apply to various
corporate purposes. We may use the proceeds for purposes that do not yield a significant return, if any, for our stockholders.
Future sales or issuances of our
common stock may cause the market price of our common stock and warrants to decline.
The sale of substantial amounts of our
common stock, whether directly by us or in the secondary market by existing securityholders (including holders of our outstanding
warrants and convertible preferred stock), the perception that such sales could occur or the availability for future sale of shares
of our common stock or securities convertible into or exchangeable or exercisable for our common stock could materially and adversely
affect the market price of our common stock and warrants and our ability to raise capital through future offerings of equity or
equity related securities. Any such sales may result in significant dilution to our existing shareholders, including you. We cannot
assure you that we will be able to sell shares or other securities in any other offering at a price per share that is equal to
or greater than the price per share paid by investors in this offering, and investors purchasing shares or other securities in
the future could have rights superior to existing stockholders, which will result in additional dilution to you.
Warrants are speculative due to their
structure and the value of our common stock may never exceed the exercise price of the warrants.
The warrants do not confer any rights of
common stock ownership on their holders, such as voting rights or the right to receive dividends. Rather, the warrants merely represent
the right to acquire shares of our common stock at a fixed price for a limited period of time. Specifically, commencing on the
date of issuance, holders of the warrants may exercise their right to acquire the common stock and pay an exercise price of $ per
share (125% of public offering price of the common stock), prior to five years from the date of issuance, after which date any
unexercised warrants will expire and have no further value. Moreover, following this offering, the market value of the warrants
is uncertain and there can be no assurance that the market value of the warrants will equal or exceed their public offering price.
There can be no assurance that the market price of the common stock will ever equal or exceed the exercise price of the warrants,
and consequently, whether it will ever be profitable for holders of the warrants to exercise the warrants.
Risks Related to Our Proposed Reverse Stock
Split
We intend to effect a reverse stock split
of our outstanding common stock prior to this offering. However, the reverse stock split may not increase our stock price sufficiently
and we may not be able to list our common stock and warrants on the NASDAQ Capital Market, in which case this offering may not
be completed.
We expect that the reverse stock split of our
outstanding common stock will increase the market price of our common stock so that we will be able to meet the minimum bid price
requirement of the Listing Rules of The NASDAQ Capital Market. However, the effect of a reverse stock split upon the market price
of our common stock cannot be predicted with certainty, and the results of reverse stock splits by companies in similar circumstances
have been varied. It is possible that the market price of our common stock following the reverse stock split will not increase
sufficiently for us to be in compliance with the minimum bid price requirement. If we are unable meet the minimum bid price requirement,
we may be unable to list our shares and warrants on The NASDAQ Capital Market, in which case this offering may not be completed.
Even if the reverse stock split achieves
the requisite increase in the market price of our common stock, we cannot assure you that we will be able to continue to comply
with the minimum bid price requirement of the NASDAQ Capital Market.
Even if the reverse stock split achieves the
requisite increase in the market price of our common stock to be in compliance with the minimum bid price of the NASDAQ Capital
Market, there can be no assurance that the market price of our common stock following the reverse stock split will remain at the
level required for continuing compliance with that requirement. It is not uncommon for the market price of a company's common stock
to decline in the period following a reverse stock split. If the market price of our common stock declines following the effectuation
of a reverse stock split, the percentage decline may be greater than would occur in the absence of a reverse stock split. In any
event, other factors unrelated to the number of shares of our common stock outstanding, such as negative financial or operational
results, could adversely affect the market price of our common stock and jeopardize our ability to meet or maintain the NASDAQ
Capital Market's minimum bid price requirement. In addition to specific listing and maintenance standards, the NASDAQ Capital Market
has broad discretionary authority over the initial and continued listing of securities, which it could exercise with respect to
the listing of our common stock.
Even if the reverse stock split increases
the market price of our common stock, there can be no assurance that we will be able to comply with other continued listing standards
of the NASDAQ Capital Market.
Even if the market price of our common stock
increases sufficiently so that we comply with the minimum bid price requirement, we cannot assure you that we will be able to comply
with the other standards that we are required to meet in order to maintain a listing of our common stock on the NASDAQ Capital
Market. Our failure to meet these requirements may result in our common stock being delisted from the NASDAQ Capital Market, irrespective
of our compliance with the minimum bid price requirement.
The reverse stock split may decrease
the liquidity of the shares of our common stock.
The liquidity of the shares of our common stock
may be affected adversely by the reverse stock split given the reduced number of shares that will be outstanding following the
reverse stock split, especially if the market price of our common stock does not increase as a result of the reverse stock split.
In addition, the reverse stock split may increase the number of stockholders who own odd lots (less than 100 shares) of our common
stock, creating the potential for such stockholders to experience an increase in the cost of selling their shares and greater difficulty
effecting such sales.
Following the reverse stock split, the
resulting market price of our common stock may not attract new investors, including institutional investors, and may not satisfy
the investing requirements of those investors. Consequently, the trading liquidity of our common stock may not improve.
Although we believe that a higher market price
of our common stock may help generate greater or broader investor interest, there can be no assurance that the reverse stock split
will result in a share price that will attract new investors, including institutional investors. In addition, there can be no assurance
that the market price of our common stock will satisfy the investing requirements of those investors. As a result, the trading
liquidity of our common stock may not necessarily improve.
SPECIAL NOTE REGARDING FORWARD-LOOKING
STATEMENTS
This prospectus and the documents we incorporate
by reference in this prospectus contain forward-looking statements that involve substantial risks and uncertainties. All statements,
other than statements of historical facts, included or incorporated by reference in this prospectus and the documents we incorporate
by reference in this prospectus regarding our strategy, future operations, future financial position, future revenues, projected
costs, prospects, plans, and objectives of management are forward-looking statements. The words “anticipates,” “believes,”
“could,” “estimates,” “expects,” “intends,” “may,” “plans,”
“potential,” “predicts,” “projects,” “should,” “will,” “would”
and similar expressions are intended to identify forward-looking statements, although not all forward-looking statements contain
these identifying words.
Our forward-looking statements in this
prospectus and the documents we incorporate by reference in this prospectus are subject to a number of known and unknown risks
and uncertainties that could cause actual results, performance, or achievements to differ materially from those described or implied
in the forward-looking statements, including:
|
|
·
|
future financial and operating results;
|
|
|
·
|
our ability to fund operations and business plans, and the timing of any funding or corporate development transactions we may pursue;
|
|
|
·
|
the ability of our suppliers to provide products or services in the future of an acceptable quality on a timely and cost-effective basis;
|
|
|
·
|
our ability to continue as a going concern in the face of limited current sales, material negative stockholders’ equity and working capital;
|
|
|
·
|
the ability to manage our considerable short term debt;
|
|
|
·
|
expectations concerning market acceptance of our products;
|
|
|
·
|
current and future economic and political conditions;
|
|
|
·
|
overall industry and market trends;
|
|
|
·
|
the outcome of existing litigation described under “Legal Proceedings” below and other filings which we make with the Commission;
|
|
|
·
|
management’s goals and plans for future operations; and
|
|
|
·
|
other assumptions described in this report underlying or relating to any forward-looking statements.
|
The assumptions used for purposes of the
forward-looking statements specified in the following information represent estimates of future events and are subject to uncertainty
as to possible changes in economic, legislative, industry, and other circumstances. As a result, the identification and interpretation
of data and other information and their use in developing and selecting assumptions from and among reasonable alternatives requires
the exercise of judgment. To the extent that the assumed events do not occur, the outcome may vary substantially from anticipated
or projected results, and, accordingly, no opinion is expressed on the achievability of those forward-looking statements. We can
give no assurance that any of the assumptions relating to the forward-looking statements included in this prospectus are accurate,
and we assume no obligation to update any such forward-looking statements, except as may be required under applicable securities
laws.
We may not actually achieve the plans,
intentions, or expectations disclosed in our forward-looking statements, and you should not place undue reliance on our forward-looking
statements. Actual results or events could differ materially from the plans, intentions and expectations disclosed in the forward-looking
statements we make. We have included or incorporated by reference important factors in the cautionary statements included in this
prospectus, particularly in the “Risk Factors” section, that could cause actual results or events to differ materially
from the forward-looking statements that we make.
You should read this prospectus, the documents
incorporated by reference in this prospectus, and the documents that have been filed as exhibits to the registration statement
of which this prospectus forms a part or to any document incorporated by reference herein completely and with the understanding
that our actual future results may be materially different from what we expect. It is routine for internal projections and expectations
to change as the year, or each quarter in the year, progresses, and, therefore, it should be clearly understood that the internal
projections and beliefs upon which we base our expectations are made as of the date of this prospectus and may change prior to
the end of each quarter or the year.
USE OF PROCEEDS
We estimate that the net proceeds to us
from this offering, after deduction of the underwriting discount and payment of estimated offering expenses, will be approximately
$ (or approximately $
if the underwriters’ over-allotment option is exercised in full).
We currently intend to use the net proceeds
from this offering for operating costs, capital expenditures, retirement of those note obligations which will have an early maturity
date upon the successful closing of this offering, and for general corporate purposes, including working capital. We may also use
a portion of the net proceeds to invest in or acquire businesses or technologies that we believe are complementary to our own,
although we have no current plans, commitments, or agreements with respect to any acquisitions as of the date of this prospectus.
The expected use of the net proceeds of
the offering set forth above represents our estimates based upon our current plans and assumptions regarding industry and general
economic conditions, our future revenues and expenditures. The amounts and timing of our actual expenditures will depend upon numerous
factors, including market conditions, cash generated by our operations, business developments and related rate of growth. We may
find it necessary or advisable to use portions of the proceeds from this offering for other purposes.
From time to time, we evaluate these and
other factors and we anticipate continuing to make such evaluations to determine if the existing allocation of resources, including
the proceeds of this offering, is being optimized. Pending such uses, we intend to invest the net proceeds of this offering in
short-term, interest-bearing, investment-grade securities such as money market funds, certificates of deposit, commercial paper
and guaranteed obligations of the U.S. government.
PRICE RANGE OF COMMON STOCK
Our common stock is listed for trading
on the OTCQB under the symbol “NUGN.” On December __, 2016 the last reported sale price of our common stock was $___.
On December __, 2016 there were approximately ___ record holders of our common stock.
The following table sets forth the high
and low sale prices per share for our common stock for the periods indicated. Prior to this offering, there was no trading market
for the warrants.
|
|
|
High
|
|
|
Low
|
|
|
Fiscal Year Ended December 31, 2015
|
|
|
|
|
|
|
|
|
|
1
st
Quarter
|
|
$
|
2.50
|
|
|
$
|
1.27
|
|
|
2
nd
Quarter
|
|
$
|
4.31
|
|
|
$
|
1.98
|
|
|
3
rd
Quarter
|
|
$
|
3.87
|
|
|
$
|
1.45
|
|
|
4
th
Quarter
|
|
$
|
1.85
|
|
|
$
|
1.12
|
|
|
|
|
|
|
|
|
|
|
|
|
Fiscal Year Ending December 31, 2016
|
|
|
|
|
|
|
|
|
|
1
st
Quarter
|
|
$
|
1.14
|
|
|
$
|
0.46
|
|
|
2
nd
Quarter
|
|
$
|
0.68
|
|
|
$
|
0.42
|
|
|
3
rd
Quarter
|
|
$
|
0.85
|
|
|
$
|
0.54
|
|
|
4
th
Quarter (through December 2, 2016)
|
|
$
|
0.64
|
|
|
$
|
0.34
|
|
DIVIDEND POLICY
We have never paid or declared any cash
dividends on our common stock. Our board of directors sets our dividend policy. We currently intend to retain all available funds
and any future earnings for use in the operation and expansion of our business, but we may determine in the future to declare or
pay cash dividends on our common stock. Any future determination as to the declaration and payment of dividends will be at the
discretion of our board of directors ad will depend on then existing conditions, including our financial condition, results of
operations, contractual restrictions, capital requirements, business prospects and other factors that our board of directors considers
relevant.
DILUTION
If you invest in our common stock in this
offering, your ownership interest will be diluted to the extent of the difference between the public offering price per share and
the pro forma net tangible book value per share after this offering. We calculate net tangible book value per share by dividing
the net tangible book value, which is tangible assets less total liabilities, by the number of outstanding shares of our common
stock.
Our net tangible book value per share as of
September 30, 2016 was approximately ($3,993,092), or $0.10 per share. After giving effect to the sale by us of
shares of common stock offered by this prospectus at a public offering price of $ per share and after
deducting underwriting discounts and estimated offering expenses payable by us, our pro forma net tangible book value as of September
30, 2016 would have been approximately $ , or $
per share. This represents an immediate increase in pro forma net tangible book value of $ per share
to existing stockholders and an immediate dilution of $ per share to new investors purchasing our common stock
in this offering. The following table illustrates the per share dilution:
|
Assumed public offering price per share
|
|
|
|
|
|
$
|
____
|
|
|
|
|
|
|
|
|
|
|
|
|
Net tangible book value per share as of September 30, 2016
|
|
$
|
0.10
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Decrease in net tangible book value per share attributable to this offering
|
|
$
|
(0.0
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Pro forma net tangible book value per share after this offering
|
|
|
|
|
|
$
|
____
|
|
|
|
|
|
|
|
|
|
|
|
|
Dilution per share to investors participating in this offering
|
|
|
|
|
|
$
|
____
|
|
The information above assumes that the
underwriters do not exercise their over-allotment option. If the underwriters exercise their over-allotment option in full, our
net tangible book value per share after giving effect to this offering would be $ per share, and the dilution
in net tangible book value per share to investors in this offering would be $ per share.
The above discussion and table are based
on 40,334,673 shares of our common stock outstanding as of September 30, 2016 and does not include, as of that date:
|
|
•
|
4,679,234 shares underlying outstanding warrants;
|
|
|
•
|
1,917,720 shares underlying outstanding Series A Preferred Stock;
|
|
|
•
|
4,352,619 share underlying outstanding stock options; or
|
|
|
•
|
Approximately $2,601,091 outstanding principal amount and interest of convertible debt which can
be converted into shares of our common stock at a conversion price that varies depending on the average trading price of shares
of our common stock.
|
If the underwriters
exercise in full their option to purchase additional shares of common stock, the pro forma net tangible book value after this offering
would be $ per share, representing an increase in net tangible book value of $ per share to existing stockholders and immediate
dilution in net tangible book value of $ per share to purchasers in this offering at the public offering price.
CAPITALIZATION
The following table sets forth our capitalization
as of September 30, 2016 on an actual basis and on an as adjusted basis to reflect our receipt of estimated net proceeds of $ million
from the sale of shares of common stock in this offering, based on an assumed public offering price of $
per share (the last reported sales price of our common stock on December , 2016), after deducting underwriting discounts and commissions
and estimated offering expenses payable by us. The as adjusted information below is illustrative only. Our capitalization following
the closing of this offering will be adjusted based on the actual public offering price and other terms of this offering determined
at pricing.
The historical data in the table is derived
from, and should be read in conjunction with, our historical financial statements, including accompanying notes, included in the
registration statement of which this prospectus is a part. You should also read this table in conjunction with the section entitled
“Management’s Discussion and Analysis of Financial Condition and Results of Operations” and our consolidated
financial statements and the related notes thereto from our Annual Report included in this prospectus.
|
|
|
As of September 30, 2016
|
|
|
|
|
Actual
|
|
|
Pro
Forma
|
|
|
Pro
Forma
As
Adjusted
|
|
|
Notes payable
(1)
|
|
|
1,669,725
|
|
|
|
|
|
|
|
|
|
|
Stockholders’ Equity (deficiency):
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Series A Convertible preferred stock, $0.0001 par value; 25,000,000 shares authorized; 1,917,720 shares issued
and outstanding as of September June 30, 2016.
|
|
|
192
|
|
|
|
|
|
|
|
|
|
|
Common Stock – $0.0001 par value; 100,000,000 shares authorized; 40,334,673 shares issued
and outstanding as of September 30, 2016, pro forma and pro forma, as adjusted, respectively.
|
|
|
4,034
|
|
|
|
|
|
|
|
|
|
|
Additional paid-in capital
|
|
|
11,327,709
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Accumulated deficit
|
|
|
(15,325,027
|
)
|
|
|
|
|
|
|
|
|
|
Total stockholders’ deficit
|
|
|
(3,993,092
|
)
|
|
$
|
|
|
|
$
|
|
|
|
Total Capitalization
|
|
$
|
(2,323,367
|
)
|
|
|
|
|
|
|
|
|
Notes:
|
|
(1)
|
The amount represents the sum of the funded short term debt and advances from the following captions of our balance sheet:
notes payable and advances.
|
MANAGEMENT’S DISCUSSION AND ANALYSIS
OF FINANCIAL CONDITIONS AND RESULTS
OVERVIEW
We were incorporated in the State of Nevada
on October 30, 2013 under the name “Bling Marketing, Inc.” Until December 29, 2014, we were a wholesaler
of jewelry, principally earrings, rings, and pendants (“BMI Business”). We recognized a minimal amount of revenues
from operations prior to the three months ended September 30, 2014. During the three month ended September 30, 2014, we began working
with several distributors to sell our jewelry products to retail outlets and as a result, recognized sales revenue of $22,025.
On September 11, 2014, we filed a Current Report on Form 8-K indicating that we were no longer a shell company as defined by Rule12b-2
of the Exchange Act in light of our operations through the quarter ended September 30, 2014.
On December 26, 2014, we entered into an
Agreement and Plan of Merger (“Merger Agreement”) with NuGene Inc., a California corporation (“NuGene”),
and on December 29, 2014 (the “Closing Date”) we filed a certificate of merger in the State of California whereby our
subsidiary, NG Acquisition Inc. (“Acquisition Sub”) merged with NuGene. As a result, NuGene (the surviving entity)
became our wholly owned subsidiary. The transaction contemplated under the Merger Agreement is deemed to be a reverse merger. Under
reverse merger accounting, our Company (the legal acquirer) was considered to have been acquired by NuGene. The assets, liabilities,
and operations of NuGene were brought forward at their book value and no goodwill was recognized.
In connection with the Merger Agreement,
we entered into a Business Transfer and Indemnity Agreement dated December 29, 2014 (the “Indemnity Agreement”) with
our former Chief Executive Officer and Director, Dena Kurland providing for:
|
|
1.
|
The transfer of our jewelry business operations existing on the date of the Indemnity Agreement (the “BMI Business”);
|
|
|
2.
|
The assumption by Ms. Kurland of all liabilities of our Company and the indemnification by Ms. Kurland holding our Company harmless for any and all liabilities arising at or before the date of the Indemnity Agreement;
|
|
|
3.
|
The payment by NuGene to Ms. Kurland of $350,000 in cash; and
|
|
|
4.
|
The surrender by Ms. Kurland of 15,000,000 shares (before giving effect to the Stock Split discussed below) (the "Indemnity Shares") of our Company’s common stock representing 95% of the then outstanding common stock (all of which shares have been deemed cancelled by our Company).
|
Pursuant to the terms of the Merger Agreement,
our Company issued 26,052,760 shares of Company common stock and 1,917,720 Company Series A Preferred Stock to the former NuGene,
Inc. shareholders. The Series A Preferred Stock is initially convertible into common stock at a ratio of one to one. Additionally,
as long as there are a minimum of 900,000 shares of Series A Preferred Stock outstanding, the holders of the Series A Preferred
Stock have the right to elect a majority of the board of directors as long as there are a minimum of 900,000 shares of Series A
Preferred Stock outstanding. Finally, the holders of the Series A Preferred Stock, generally voting as a class with the holders
of common stock, have for each share of Series A Preferred Stock owned, three times the number of votes permitted to each share
of common stock.
On December 26, 2014, our board of directors approved a 15.04 to one stock split (“Stock Split”)
in the form of a stock dividend to holders of our common stock as of that date. To effect that board action, shareholders
received as a stock dividend 14.04 additional shares of common stock for every share of common stock held. Unless otherwise noted,
all share numbers shown in this Registration Statement give effect to the Stock Split.
On December 29, 2014, we completed the
sale of 2,000,000 shares of our common stock to 18 purchasers (“Stock Placement”) for proceeds totaling $2,000,000,
including (a) $1,625,000 of cash and (b) automatic conversion of promissory notes in the principal amount of $375,000.
At the Closing Date subsequent to the transactions
described above (and giving effect to the Stock Split), we had approximately 39,197,400 shares of common stock outstanding and
1,917,720 shares of Series A Preferred Stock outstanding. At the time of the merger, an outstanding warrant to acquire shares of
NuGene Inc. was exchanged for a warrant of Bling Marketing Inc. The warrant on its issuance, after giving effect to the Stock Split,
evidenced the right of the holder to acquire up to 500,000 shares of common stock of our Company. The warrant has a strike price
of $2.50 per share and was not exercisable for 12 months (the "Initial Exercise Date"). Any shares acquired thereunder
upon exercise thereafter cannot be sold for six months following the Initial Exercise Date.
On January 22, 2015, we were advised that
the Financial Industry Regulatory Association had approved our (i) 15.04 to one Stock Split, (ii) name change from Bling Marketing
Inc. to NuGene International, Inc., and (iii) change of trading symbol from BLMK to NUGN.
RESULTS OF OPERATIONS
The following analysis of the results of
operations for the (i) three and nine months ended September 30, 2016 and 2015; and, (ii) years ended 2014 and 2015 should be read
in conjunction with our financial statements and the notes to those financial statements that are included elsewhere in this prospectus.
Our discussion includes forward-looking statements based upon current expectations that involve risks and uncertainties, such as
our plans, objectives, expectations, and intentions. Actual results and the timing of events could differ materially from those
anticipated in these forward-looking statements because of a number of factors. An investment in our common stock involves a high
degree of risk. Readers of this prospectus should carefully consider the risks set forth in the Risk Factors section of this prospectus.
We use words such as “anticipate,” “estimate,” “plan,” “project,” “continuing,”
“ongoing,” “expect,” “believe,” “intend,” or similar expressions, variations of
those terms or the negative of those terms to identify forward-looking statements. The forward-looking statements specified in
the following information have been compiled by our management on the basis of assumptions made by management and considered by
management to be reasonable. Our future operating results, however, are impossible to predict and no representation, guaranty,
or warranty is to be inferred from those forward-looking statements.
For the three months ended September
30, 2016 as compared to the three months ended September 30, 2015
Revenues
Revenues generated during the three months
ended September 30, 2016 totaled $86,000 resulted primarily from the sale of our cosmeceutical products to wholesale distributors,
physicians and directly to consumers, including 17% of our revenues to one customer. Our customers generally purchase products
from us on a purchase order basis on standard terms but future product sales to distributors and physicians will be tied directly
to their success in reselling our products. Our customers are under no obligation to continue to purchase our products. The loss
of significant customers, a material reduction in their purchases or the cancellation of product orders or unexpected returns of
unsold products could significantly decrease our revenues and adversely affect our future growth prospects. We do not have long-term
purchase commitments from our customers.
During the three months ended September
30, 2015, we had revenue totaling $521,000, 48% and 19% of which was sold to two customers. Our revenue in 2016 decreased by $435,000
or 84% from the same period in 2015. While our revenue decrease could be attributed in part to decreases in customer demand for
our products of which we are unaware at present, we believe our severe lack of liquidity was a significant contributing factor.
Our cash constraints made it difficult to afford and to recruit qualified senior level employees, especially in the area of marketing.
Our plans for launching a television direct marketing campaign have also been delayed for the same reasons. We estimate that absent
successful procurement of the funding that we currently seek, we will be unable to build revenue in accordance with our corporate
plans and revenue could continue to be stagnant or decrease significantly.
Cost of Revenues
Cost of revenues during the three months
ended September 30, 2016 totaled approximately $555,000 and represented 647% of revenue. These costs consisted primarily of an
obsolescence charge of $437,000 as the Company is rebranding and repackaging its products, direct costs of labor and overhead of
$105,000 as production was reduced and $13,000 primarily attributed to the cost of raw materials and product packaging of our cosmeceutical
products, as well as shipping and handling. Cost of revenues during the same period in 2015 totaled approximately $150,000, or
29% of revenue. Our products sell for margins comparable with others in industries similar to ours. Our margins will reflect our
efficiency in the production of our products, the desirability of our products and our ability to grow revenue in order to scale
our operations. Our relationships with our suppliers will also be important in procuring materials at better pricing and our aforementioned
liquidity challenges could significantly affect such relationships.
Advertising and promotion
Advertising and promotion totaled approximately
$106,000 for the three months ended September 30, 2016 compared to approximately $24,000 during the same period in 2015. This represented
an increase of approximately $82,000 from 2015 to 2016. The increase was primarily attributable to $44,000 in marketing programs
and $38,000 of royalty expenses.
Personnel
Personnel expenses of approximately $3,422,000
for the three months ended September 30, 2016 increased $1,919,000 from $1,503,000 for the same period in 2015. The increase in
personnel expenses was primarily attributable to an increase in stock-based compensation expense of approximately $1,923,000 related
to the issuance stock awards and amortization of stock options partially offset by wage related expenses of $4,000.
Selling, general and administrative
Selling, general and administrative expenses
(“SGA”) totaled approximately $231,000 for the three months ended September 30, 2016 compared to approximately $356,000
for the three months ended September 30, 2015. The decrease of approximately $125,000 was primarily attributable to reduced marketing
related expenses of $128,000 and commercial and administrative related expenses of $30,000 partially offset by an increase of $30,000
in bad debt expense.
Research and development
Expenses related to research and development
totaled approximately $74,000 for the three months ended September 30, 2016 compared to approximately $98,000 for the corresponding
period in 2015. The decrease was primarily attributable to reduced clinical expenses of $65,000 partially offset by an increase
in material costs of $35,000.
Professional fees
Professional fees totaled approximately
$593,000 for the three months ended September 30, 2016 compared to approximately $417,000 for the three months ended September
30, 2015. The increase of approximately $176,000 was primarily due to an increase in marketing, operational and administrative
consulting services.
Other income (expenses)
Interest expense totaled approximately
$328,000 for the three months ended September 30, 2016 compared to $75,000 for the three months ended September 30, 2015. The increase
in interest expense was primarily attributable to the increase of our borrowings outstanding. While there was no cash interest
due during the period, interest expense resulted from the accretion to face value of borrowings advanced to the Company as well
as accruing interest on all debt outstanding.
Loss on issuance of debt of approximately
$1,212,000 arose from the recording of our derivative instrument associated with the Advances received during the three months
ended September 30, 2016.
Change in fair value of derivative liabilities
of approximately $249,000 resulted from the revaluation of our derivative instrument during the three months ended September 30,
2016.
Comparison of nine months ended September
30, 2016 and 2015
Revenues
Revenues generated during the nine months
ended September 30, 2016 totaled $410,000 resulted primarily from the sale of our cosmeceutical products to wholesale distributors,
physicians, and consumers, including 9% of our revenues to one customer. Our customers generally purchase products from us on a
purchase order basis on standard terms but future product sales to distributors and physicians will be tied directly to their success
in reselling our products. Our customers are under no obligation to continue to purchase our products. The loss of significant
customers, a material reduction in their purchases or the cancellation of product orders or unexpected returns of unsold products
could significantly decrease our revenues and adversely affect our future growth prospects. We do not have long-term purchase commitments
from our customers.
During the nine months ended September
30, 2015, we had revenue totaling $1.5 million, 45% and 21% of which was sold to one customer. Our revenue in 2016 decreased approximately
$1,128,000 or 73% from the same period in 2015. While our revenue decrease could be attributed in part to decreases in customer
demand for our products of which we are unaware at present, we believe our severe lack of liquidity was a significant contributing
factor. Our cash constraints made it difficult to afford and to recruit qualified senior level employees, especially in the area
of marketing. Our plans for launching a television direct marketing campaign have also been delayed for the same reasons. We estimate
that absent successful procurement of the funding that we currently seek, we will be unable to build revenue in accordance with
our corporate plans, and revenue could continue to be stagnant or decrease significantly.
Cost of Revenues
Cost of revenues sold during the nine months
ended September 30, 2016 totaled approximately $659,000 and represented 161% of revenues. These costs consisted primarily of an
obsolescence charge of $437,000 as the Company is rebranding and repackaging its products, direct costs of labor and overhead of
$105,000 as production was reduced during the three months ended September 30, 2016, and $117,000 primarily attributed to the cost
of raw materials, labor and overhead and product packaging of our cosmeceutical products, as well as shipping and handling. Cost
of revenue during the same period in 2015 totaled approximately $444,000, or 29% of revenue. Our products sell for margins comparable
with others in industries similar to ours. Our margins will reflect our efficiency in the production of our products, the desirability
of our products and our ability to grow revenue in order to scale our operations. Our relationships with our suppliers will also
be important in procuring materials at better pricing and our aforementioned liquidity challenges could significantly affect such
relationships.
Advertising and promotion
Advertising and promotion totaled approximately
$314,000 for the nine months ended September 30, 2016 compared to approximately $299,0000 during the same period in 2015, resulting
in a $15,000 increase from 2015 to 2016. The majority of the increase was related to marketing consultants and brand management
expenses.
Personnel
Personnel expenses of approximately $4,139,000
increased approximately $2,074,000 for the nine months ended September 30, 2016 from approximately $2,065,000 for the same prior
year period. The increase was primarily due to an increase in stock-based compensation expense of approximately $1,886,000 related
to the issuance of stock awards and amortization of stock options and $188,000 related to wage related expenses.
Selling, general and administrative
SGA totaled approximately $1,056,000 for
the nine months ended September 30, 2016 compared to approximately $862,000 for the nine months ended September 30, 2015. The increase
of approximately $194,000 was primarily related to an increase of approximately $272,000 in bad debt expense partially offset by
the reduction of commercial and administrative related expenses of approximately $78,000.
Research and development
Expenses related to research and development
totaled approximately $104,000 for the nine months ended September 30, 2016 compared to approximately $136,000 for the corresponding
period in 2015. This represented a decrease of approximately $32,000, which was primarily attributable to reduced clinical expenses
of $80,000 partially offset by an increase in material costs of $48,000.
Professional fees
Professional fees totaled approximately
$1,279,000 for the nine months ended September 30, 2016 compared to approximately $771,000 for the nine months ended September
30, 2015. The increase of $508,000 was primarily attributable to an increase of stock based compensation expense of $382,000 and
$126,000 in marketing, operational, and administrative consulting services.
Other income (expenses)
Interest expense totaled approximately
$660,000 for the nine months ended September 30, 2016 compared to $76,000 for the nine months ended September 30, 2015. The increase
in interest expense was primarily attributable to the increase of our borrowings outstanding. While there was no cash interest
due during the period, interest expense resulted from the accretion to face value of borrowings advanced to our Company as well
as accruing interest on all debt outstanding.
Loss on issuance of debt of approximately
$1,212,000 arose from the recording of our derivative instrument associated with the Advances received during the nine months ended
September 30, 2016.
Change in fair value of derivative liabilities
of approximately $257,000 resulted from the revaluation of our derivative instrument during the nine months ended September 30,
2016.
For Fiscal Year 2015 as compared
to Fiscal Year 2014
Revenues.
Revenues generated during the year ended
December 31, 2015 totaling $2,084,939 resulted primarily from the sales of our cosmeceutical products to distributors, online marketers
and consumer online sales. Sales revenues for the year ended December 31, 2014 totaled $723,438 and consisted primarily of sales
to ASP, totaling $481,000.
Cost of goods sold.
Cost of goods sold during the year ended
December 31, 2015 of $588,882 represented 28.2% of revenues. Such costs related primarily to the cost of raw materials, product
packaging, and direct labor in the manufacturing of our cosmeceutical products, as well as shipping and handling. Cost of goods
sold during the year ended December 31, 2014 totaled $293,162 or 40.5% of sales and related primarily to direct labor and the materials
used in our products.
Selling, general and administrative.
Selling, general and administrative consisted
of the following during the years ended December 31, 2015 and 2014:
|
|
|
2015
|
|
|
2014
|
|
|
|
|
Amount
|
|
|
Percentage
|
|
|
Amount
|
|
|
Percentage
|
|
|
Marketing and advertising
|
|
$
|
650,597
|
|
|
|
9.77
|
%
|
|
$
|
90,021
|
|
|
|
12.11
|
%
|
|
Bad debt
|
|
|
223,631
|
|
|
|
3.36
|
%
|
|
|
300
|
|
|
|
0.04
|
%
|
|
Business development
|
|
|
5,000
|
|
|
|
0.08
|
%
|
|
|
245,020
|
|
|
|
32.97
|
%
|
|
Corporate
|
|
|
169,831
|
|
|
|
2.55
|
%
|
|
|
2,304
|
|
|
|
0.31
|
%
|
|
Insurance
|
|
|
158,358
|
|
|
|
2.38
|
%
|
|
|
1,288
|
|
|
|
0.17
|
%
|
|
Personnel
|
|
|
691,600
|
|
|
|
10.39
|
%
|
|
|
19,044
|
|
|
|
2.56
|
%
|
|
Professional fees
|
|
|
928,392
|
|
|
|
13.94
|
%
|
|
|
142,301
|
|
|
|
19.15
|
%
|
|
Rent and utilities
|
|
|
208,517
|
|
|
|
3.13
|
%
|
|
|
33,354
|
|
|
|
4.49
|
%
|
|
Research and development
|
|
|
350,669
|
|
|
|
5.27
|
%
|
|
|
19,105
|
|
|
|
2.57
|
%
|
|
Stock based compensation and stock issued for services
|
|
|
2,989,680
|
|
|
|
44.90
|
%
|
|
|
8,833
|
|
|
|
1.19
|
%
|
|
Supplies
|
|
|
78,832
|
|
|
|
1.18
|
%
|
|
|
38,830
|
|
|
|
5.23
|
%
|
|
Taxes
|
|
|
72,020
|
|
|
|
1.08
|
%
|
|
|
21,022
|
|
|
|
2.83
|
%
|
|
Other
|
|
|
130,982
|
|
|
|
1.97
|
%
|
|
|
121,635
|
|
|
|
16.37
|
%
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
$
|
6,658,109
|
|
|
|
100.00
|
%
|
|
$
|
743,057
|
|
|
|
100.00
|
%
|
Our Company greatly increased its expenditures
on marketing and advertising as we sought to publicize the superior qualities of our products during 2015. In prior years, a significant
percentage of our revenues came from sales to related parties and did not involve cosmeceutical products. Bad debt expense arose
primarily from sales to two customers that we do not intend to sell to in the future absent satisfaction of previously due amounts.
Business development in 2014 primarily consisted of payments to a firm that attempted to employ our stem cell technology with other
companies. Corporate expenses were amounts paid to investor communications firms, news wires and investor relations consulting
firms.
Insurance expenses increased primarily
due to our Company’s carrying directors and officers liability insurance that was not previously carried as well as increased
amounts paid for general and product liability policies. Personnel expenses increased with the volume of new employees hired in
2015 to meet the demands of the product business in the areas of manufacturing, marketing, and administration as well as additional
personnel added to fulfill our Company’s responsibilities as a public company. Our founders and officers continued to work
at our Company without compensation through December 31, 2015.
Professional fees included accounting ($61,054
in 2015); cash fees paid to our Advisory Board members ($173,000 and $9,000 in 2015 and 2014, respectively); auditor fees ($25,000
and $24,000 in 2015 and 2014, respectively); consulting services ($253,516 in 2015); legal fees ($308,154 and $47,351 in 2015 and
2014, respectively); and public relations ($98,097 in 2015). Rent and utilities of $208,517 were significantly higher in 2015 over
2014’s $33,354 due to the leasing of additional space for manufacturing, administrative and sales offices required in 2015
given the different focus over that of 2014. Research and development increased significantly in 2015 and included the purchase
and charge to operations of in process technology totaling $150,000.
Stock based compensation and stock issued
for services resulted from the grant of warrants and options to purchase our common stock to consultants and employees, respectively.
The related grants and terms of the derivatives issued are described in complete detail in the accompanying notes to our financial
statements for 2015. Supplies increased proportionally with the increased number of employees and activities in 2015 as compared
to 2014. Taxes included payroll taxes ($65,653 and $14,851 in 2015 and 2014, respectively) that increased proportionally with the
number of employees and amounts paid during the year for salaries.
Interest expense.
Interest expense of $31,479 was recognized
in connection with two notes payable that were issued in September and November 2015 totaling $550,000. Additionally, we recognized
interest expense totaling $24,411 in connection with estimates of ultimate interest expense that will be owing on as yet undocumented
advances made to our Company from November 30, 2015 through the end of December 2015. Interest expense resulted from both the expected
interest that will be ultimately due to the Lenders as well as the amortization of the estimated ultimate value of the warrants
issued with the advances.
Net Loss.
During the year ended December 31, 2015,
we reported a net loss of $5,219,067 and during the year ended December 31, 2014, we reported net loss of $312,781. As discussed
above, the net loss during the year ended December 31, 2015 includes non-cash expenses totaling $2,989,680 relating to stock based
compensation and stock issued for services over the current contract period. We anticipate continuing to incur net losses as we
build new sales channels for our products prior to the realization of significant sales and gross margin from such efforts.
Related Party Transactions
During the year ended December 31, 2015,
our Company utilized employees of ASP for legal and administrative services. Management estimated the time associated with the
services provided and recognized approximately $99,390 during the year ended December 31, 2015, $70,890 of which is owing as of
December 31, 2015. We intend to pay ASP these amounts once we our current liquidity challenges are remedied (although no assurance
can be given that we will be successful in remedying these challenges).
During December 2014, ASP provided our
Company a $45,000, interest free, short-term loan to cover operating expenses. NuGene repaid the loan during January 2015.
During August 2015, the Center for Weight
Management and Plastic Surgery, Inc. (“CWM”), a company affiliated with us, provided services totaling $34,000 to our
consultants as compensation for advertising and marketing services provided to our Company. Such amounts remained an outstanding
liability at December 31, 2015.
Mr. Ali Kharazmi has purchased equipment
and paid for various expenses on behalf of NuGene. As of December 31, 2015, our Company owed Mr. Kharazmi approximately $25,641
related to expense reimbursements.
Dr. Saeed Kharazmi has incurred various
business expenses on behalf of NuGene. As of December 31, 2015, our Company owed Dr. Kharazmi approximately $2,405 in expense reimbursements.
Messrs. Ali and Saeed Kharazmi, our Chairman
of the Board and CFO, respectively, have been foregoing salaries since our Company was incorporated in 2006. Mr. Ali Kharazmi has
personally guaranteed payment of our building sublease as noted elsewhere.
Liquidity and Capital Resources.
We incurred recurring operating losses
and negative operating cash flows in 2015 and through September 30, 2016, and we expect to continue to incur operating losses and
negative operating cash flows at least through the near future. Members of our Company’s management have been required to
advance our Company funding in order to partially meet our most critical cash requirements including payroll along with those associated
with certain critical goods and services. In the process of managing these situations, our management may have made representations
implying their personal guarantee of certain of our Company’s obligations, irrespective of whether such guarantees are legally
valid and enforceable.
As a result of the aforementioned factors,
management has concluded that there is substantial doubt about our ability to continue as a going concern. Our independent registered
public accounting firm, in its report on our 2015 consolidated financial statements, raised substantial doubt about our ability
to continue as a going concern. Our consolidated financial statements as of and for the three months ended September 30, 2016 do
not contain any adjustments for this uncertainty. In response to our cash needs, we raised the following funding as described below.
Any additional amounts raised will be used for our future investing and operating cash flow needs. However, there can be no assurance
that we will be successful in raising additional amounts of financing.
Recent Financing Activities
:
During the three months ended September
30, 2016, certain investors advanced a total of $925,000 (the “Advances”) to the Company with a right to convert at
no greater than $0.40 per share. In connection with the Advances, the Company also issued warrants to purchase 462,500 shares of
our common stock at an exercise price $0.60 per share with a 5 year term. The relative fair value of the warrants compared to the
Advances was $203,467, which was recorded as a component of stockholders’ deficit with the offset recorded as a discount
on the Advances. The fair value of the warrants was determined using the Black-Scholes Model. The Company then computed the effective
conversion price of the Advances on the issuance date, noting that the Advances gave rise to a BCF of $721,533. The sum of the
relative fair value of the warrants and the BCF was recorded as a debt discount to be amortized over the term of the advances.
Subsequent to the period ended September
30, 2016: On October 21, 2106, the Company issued an investor a promissory note in the total principal amount of $267,500 with
9% annual interest rate, which will be due on October 21, 2017. The Company received $250,000 of gross proceeds and granted a warrant
to purchase 222,916 shares of common stock at $0.60 per share.
On November 2, 2016, the Company issued
certain investors promissory notes (the “Notes”) in the total principal amount of $480,000 with a 5% annual interest
rate, which will be due on April 28, 2017. The Company received $400,000 of gross proceeds from the Notes. The Company also granted
to the investors warrants to purchase 1,428,572 shares of common stock at no greater than $0.47 per share.
Off-Balance Sheet Arrangements
There are no off-balance sheet arrangements
that have or are reasonably likely to have a current or future effect on our financial condition, changes in financial condition,
revenues or expenses, results of operations, liquidity, capital expenditures or capital resources that are material to investors.
Inflation
Inflation and changing prices have had
no material effect on our net sales and revenues or on our income from continuing operations over our two most recent fiscal years.
Critical Accounting Policies and Estimates
Our management’s discussion and analysis
of our financial condition and results of operations are based on our financial statements, which have been prepared in accordance
with U.S. generally accepted accounting principles. The preparation of these financial statements requires us to make estimates
and judgments that affect the reported amounts of assets, liabilities, and expenses and the disclosure of contingent assets and
liabilities in our financial statements. On an ongoing basis, we evaluate our estimates and judgments, including those related
to the fair market value of our assets and accrued stock-based compensation expense. We base our estimates on historical
experience, known trends, and events and various other factors that are believed to be reasonable under the circumstances, the
results of which form the basis for making judgments about the carrying values of assets and liabilities that are not readily apparent
from other sources. Actual results may differ from these estimates under different assumptions or conditions. In making
estimates and judgments, management employs critical accounting policies.
For a description of our critical accounting
policies, please see Management’s Discussion and Analysis of Financial Condition and Results of Operations included in our
Annual Report on Form 10-K for the year ended December 31, 2015. There have not been any material changes to our
critical accounting policies since December 31, 2015.
BUSINESS
ORGANIZATIONAL HISTORY
We were incorporated in the State of Nevada
on October 30, 2013 under the name “Bling Marketing, Inc.” Until December 29, 2014, we were a wholesaler
of jewelry, principally earrings, rings, and pendants (“BMI Business”). We recognized a minimal amount of sales
from operations prior to the three months ended September 30, 2014.
On December 26, 2014, we entered into an
Agreement and Plan of Merger (“Merger Agreement”) with NuGene Inc., a California corporation (“NuGene”),
and on December 29, 2014 (the “Closing Date”) we filed a certificate of merger in the State of California whereby our
subsidiary, NG Acquisition Inc. (“Acquisition Sub”) merged with NuGene. As a result, NuGene (the surviving entity)
became our wholly owned subsidiary. The transaction contemplated under the Merger Agreement is deemed to be a reverse merger.
Under reverse merger accounting, our Company (the legal acquirer) is considered to have been acquired by NuGene. The assets, liabilities,
and operations of NuGene have been brought forward at their book value and no goodwill has been recognized.
NUGENE HISTORY AND BUSINESS
NuGene was incorporated in California in
December 2006 and formed and funded by our founders, Ali Kharazmi and Mohammed Kharazmi, M.D. The initial focus of the NuGene was
to develop and market customized skin care products. As part of that focus, NuGene sought to leverage the working relationships
developed by our founders with the plastic surgery community. NuGene directed significant time and resources on developing anti-aging
and scar treatment/reduction products.
In 2007 we continued to focus on age defying
products utilizing
peptide complexes
(see further description, below) and
nano-encapsulation
for
absorption into the skin (see further description, below). We introduced a limited product line under the NuGene name, and co-branded
the products with an affiliated entity, Genetic Institute of Anti Aging, Inc. (“GIAA”), which is also owned by our
founders. We utilized the services of a Korean based contract manufacturer to supply our products. This product line (the “GIAA
Line”) was based on the use of peptides and did not utilize stem cells. We had very modest sales in 2007, with our sole customer
being GIAA, a related party.
In 2008 we stopped production of the GIAA
Line and sales were limited to selling remaining inventory through medical offices and through GIAA. With the GIAA Line discontinued,
we spent the remainder of 2008 considering different formulations and methodologies for improved anti-aging products. In 2009 and
2010 we had limited activity and very limited sales. Our sales were mostly overseas and limited to the remaining inventory of the
GIAA Line. We continued to explore how we might advance our formulations and methodologies. We expended funds on research and development,
carried out mostly by scientists engaged by the Company.
In 2011 our founders decided to use adult
adipose human stem cells (undifferentiated cells found throughout the body that multiply by cell division to replenish dying cells
and regenerate tissues) as the foundation of the formulation for its products. During 2011 the Company developed a proprietary
process to extract human adult stem cells from fat cells that the Company then used in its customized NuGene line made specifically
for those client(s). Throughout 2011 we continued to provide autologous, or mature, fat-derived stem cells for use in clinical
procedures, utilizing this technology. Through this process, the Company refined its ability to culture adult human stem cells
to render human conditioned stem cell media at a proprietary concentration which is a primary ingredient in the NuGene line of
cosmeceuticals.
In 2012 we completed our initial line of
cosmeceutical products based on these adipose derived stem cells. We branded this advanced skin care line solely under the NuGene
name (the “NuGene Line”). We were able to eliminate the unpleasant odor associated with stem cells by adding a fragrance
with a very low incidence of allergic reaction. The packaging of this new product line bears no resemblance to the prior GIAA Line.
We also manufactured the NuGene Line ourselves at a small laboratory facility that we leased from an affiliated entity owned by
one of our founders. Throughout 2013, we continued to expand the product offerings of the NuGene Line. The Company focused its
stem cell work in the surgical arena and orthopedic regeneration. These services were delivered to one client, which was an affiliated
entity. Sales of the NuGene Line were limited as we were in an initial rollout and branding phase.
During 2014, we focused our efforts on
transitioning to a cosmeceutical skincare business for mass distribution. With this transition and expanded attention to our consumer
products, we sought to develop our marketing plan and distribution channels. By the end of 2014, we had wholesalers distributing
products from the NuGene Line to medical offices and medical spas throughout the United States. In addition to the NuGene Line,
we generated revenues from an affiliate, Advanced Surgical Partners (“ASP”) which is also owned by our former CEO and
Chairman of the Board, Messrs. Ali and Mohammed Kharazmi, respectively. Revenues generated from ASP resulted from NuGene providing
Plasma Rich Platelet and Stem Cell injections for orthopedic and plastic surgery procedures to ASP. We provided these products
and services to ASP as we transitioned into the commercialization of our cosmeceutical product lines. We had no such product sales
and provided no services to ASP subsequent to the Merger Agreement in December 2014.
The Company currently is principally engaged
in marketing its NuGene Line of products. We will need to seek substantial additional funding to remain a going concern, maintain
operations and to implement our business plan. Without adequate funding to support our operations for at least the next 12 months,
our ability to expend funds on advertising, retain channels of distribution, retain supplier relationships, build inventory and
recruit experienced sales personnel cannot be assured. Additionally, we were unsuccessful in several attempts to raise capital
during 2015 and early 2016 and these failures have put our Company under significant financial strain. We continue to attempt to
procure new sources of funding, whether from the issuance of debt or shares of our common stock or a combination of both. Even
if we are able to obtain such additional funding, no assurance can be given that the amounts of funding we might secure will prove
to be adequate or that we will be able to accomplish our goals within the timeframes projected. In addition, no assurance can be
given that we will be able to obtain any additional financing on commercially reasonable terms or otherwise. If we are unable to
obtain adequate funding, we may be unable to manage our existing outstanding debt, and may be required to significantly curtail
or even discontinue our operations.
OVERVIEW
“
Cosmeceuticals
” refer
to the combinations of cosmetics and pharmaceuticals that may offer medicinal or drug-like benefits. The term is more of a marketing
term rather than a legal term, with the cosmetics industry having adopted the term in the late 1990’s. The US Food and Drug
Administration (the “FDA”) does not recognize the term. While drugs are subject to a review and approval process by
the FDA, cosmetics and cosmeceuticals are not subject to the same stringent regulatory regime and scrutiny. As such, if a product
has drug properties, it must be approved as a drug. However, cosmeceuticals are not subject to this review and approval process.
Cosmeceuticals offer consumers cosmetic-like
treatments with active ingredients, which we believe give them pharmaceutical-like efficacy. Driven by aesthetic-consciousness,
according to the May 2014 RNCOS research publication entitled, “Global Cosmeceuticals Market Outlook 2018” the global
market for cosmeceutical products reached an estimated size of $35.1 billion in 2013. We believe that this market benefits from
a younger generation of consumers as well as baby boomers who want to preserve their youthful looks. In our view the U.S. cosmeceutical
market will continue to be favorably influenced by these groups who want beautiful and younger looking skin while aging.
Two of the fastest growing cosmeceuticals
segments are the anti-aging and hair restoration markets which focus on mechanisms that influence the aging of skin and prevent/restore
hair loss. Also according to the same RNCOS research, skin care garnered a material share of the cosmeceutical market in 2013 with
a 60%, $21 billion worldwide market share, followed by hair care which represented a 14%, $4.75 billion market share. The global
skincare cosmeceutical market is expected to reach $30.3 billion by 2018, accounting for a 62% share in the total cosmeceutical
markets. In particular it is anticipated that with an aging baby boomer population and increased male grooming, skin whitening,
facial care and sun-damaged products will drive the expected global demand.
The use of stem cell based media as a foundation
for cosmeceutical products is designed to promote the anti-aging process. Every human being has stem cells in his or her body.
These cells exist from the early stages of human development until the end of a person’s life. Throughout our lives, our
body continues to produce stem cells that regenerate to produce differentiated cells that make up various aspects of the body such
as skin, blood, muscles, and nerves. These are generally referred to as adult stem cells (non-embryonic). These cells are important
for the purpose of medical therapies aiming to replace lost or damaged cells or tissues or to otherwise treat disorders. Our NuGene
Line of products uses human adult stem cells culture media that we believe, when applied to human skin cells, may reduce the appearance
of aging skin. We have not yet commenced widespread marketing efforts of the NuGene Line, however we are in the process of developing
relationships with distributors to provide access to national and international channels of distribution.
STRATEGY
In our view, anti-aging products represent
(and will continue to represent) a significant portion of the facial skincare market. In key markets, we believe that the facial
skincare market is positioned for significant growth with limited downside risks from the overall condition of the economy. In
order to make claims that products can diminish the signs of aging, marketers are constantly looking for new combinations of specialty
ingredients. The category of skincare products based on biotechnology such as human stem cell is in our view just beginning to
be developed, and therefore we expect that it has significant growth potential. Our goal is to leverage our knowledge in human
adult stem cell technology to develop and commercialize advanced anti-aging skincare and hair growth products for the retail and
professional channels. We intend to develop, manufacture, and market cosmetic skin care and hair products to address this significant
market opportunity.
We plan to continue to sell our NuGene
Line of products domestically and internationally through professional channels, including dermatologists, plastic surgeons, medical
offices, and day and resort spas. We also expect to directly market our product through television sales-dedicated channels. We
plan to promote brand awareness through advertising, our own sales personnel, other marketing and public relations. We will also
partially rely upon the name recognition of Kathy Ireland secured under the License Agreement discussed below. We propose to further
expand our sales efforts through our branded website (
www.nugene.com
) and additional online commerce channels.
In addition to our product enhancement
and marketing activities, our success will also depend on our ability to develop and protect our proprietary technology. We intend
to rely on a combination of patent, trade secret and know-how, copyright and trademark laws, as well as confidentiality agreements,
licensing agreements and other agreements, to establish and protect our proprietary rights. Our success will also depend upon our
ability to avoid infringing upon the proprietary rights of others.
OUR PRODUCTS
All of our products listed below have been
formulated, fully developed and are currently being marketed. We have formulated and are testing other products which we expect
to introduce (financial resources permitting) once we complete product testing and preparation of marketing materials. None of
our products are licensed from third parties.
NuGene Skincare Products
|
|
·
|
Universal Cream
: Our moisturizer works to smooth fine lines and wrinkles, improve aging skin tone and texture, increase collagen production, and reduce puffiness and dark circles—without any heaviness or oiliness.
|
|
|
·
|
Universal Serum
: Formulated with multiple human growth factors, polypeptides, vitamins, minerals and amino acids, our signature serum encourages skin’s natural production of collagen and elastin while boosting other vital matrix protein syntheses, enabling it to repair and prevent environmental damage, improve texture and thickness, and restore skin’s natural luminosity.
|
|
|
·
|
Eye Serum
: Designed to prevent and correct wrinkles, fine lines, puffiness, dryness and dark circles, this innovative product relies on our cutting-edge stem cell technology, along with multiple growth factors and other nourishing ingredients, to help reverse skin damage, minimize the appearance of future signs of aging, and restore youthful luminosity around the eyes, fostering skin that looks younger, smoother and fresher.
|
|
|
·
|
Light & Bright Gel
: This potent product works to break down excess melanin in the skin without any of the harsh ingredients typically found in brightening products. And, in addition to quickly reducing the look of dark spots and excess pigmentation, it works to increase overall radiance, restoring a youthful glow.
|
|
|
·
|
Face Wash
: As an anti-aging cleanser, our Face Wash is formulated with a unique blend of multiple growth factors, cytokines and rich pentapeptide complexes. NuGene’s Face Wash minimizes the visibility of pores and softens imperfections while the natural cleansing agents gently wash away dirt, oil, and makeup leaving the skin refreshed, smooth, clean and hydrated.
|
Hair care Products
|
|
·
|
Anti-Hair Loss Serum: Our stem cell based technology replenishes the hair follicles with essential growth factors and cytokines to reduce and prevent further hair loss, as well as to stimulate follicular regeneration.
|
|
|
·
|
Regenerative Shampoo
: Rich in growth factors, polypeptide complexes and vitamins, this product has been formulated to rejuvenate the hair and scalp. In addition to increasing hydration, vibrancy, and shine, it works to protect hair from environmental assault, preventing and correcting breakage, thinning and split ends.
|
|
|
·
|
Regenerative Conditioner
: This hydrating, shine-enhancing conditioner utilizes additional key ingredients, vitamins, and polypeptide complexes, to name a few, to instantly smooth even the coarsest, most damaged strands. With regular use, it fosters stronger, shinier, younger-looking hair that’s easier to style.
|
|
|
·
|
Anti-Hair Loss Serum
: Our stem cell based technology replenishes the hair follicles with essential growth factors and cytokines to reduce and prevent further hair loss, as well as to stimulate follicular regeneration.
|
Wound Care-Wound Healing; Biopharma
Products
In 2014, we initiated research and formulation
for a new topical burn cream for treatment of burns that we expect may reduce infection and could improve healing time. Burns are
the fourth most common type of trauma worldwide according to RNCOS research. Despite recent advances in wound management, infection
continues to be a significant problem in treating burns. This has resulted in the routine use of prophylactic topical antimicrobial
agents. The most common topical antimicrobial agent used is silver sulfadiazine cream. However, the side effects of delayed wound
healing have remained a challenge to overcome.
Tissue healing research has demonstrated
adipose-derived stem cell culture conditioned media exhibit human dermal fibroblast proliferation and enhanced secretion of type
I, III collagen and fibronectin. These properties are well suited for dermal wound healing. In order to develop and bring this
product to market, we intend to work with a contract manufacturer to combine the product with our stem cell media technology. The
preclinical FDA work we will need to have completed consists of in vitro and in vivo efficacy studies, experimental toxicology
testing, formulation study, storage condition, stability testing, and ensuring Good Manufacturing Practices are all followed in
the manufacturing process. This topical product will require FDA approval and we can give no assurance that we will obtain that
approval.
STRATEGIC RELATIONSHIPS AND LICENSING
ARRANGEMENTS
kiWW License Agreement
In November 2014 we entered into a License
Agreement with kathy ireland Worldwide
®
("kiWW
®
") whereby we licensed the right to
utilize the trademarks and rights to the name, likeness and visual representations of Kathy Ireland in connection with our cosmeceutical
line of products containing adult human adipose stem cell derived or containing biologically active or biologically derived ingredients.
The initial term of the license is for eight years and it may be renewed for up to an additional four years.
In accordance with the License Agreement,
we are required to submit a Business Plan to the kiWW
®
within ninety days of the effective date of the License
Agreement. We have submitted our Business Plan to the Licensor within the required time frame and received the necessary approvals.
The License Agreement defines the Licensed Products as those NuGene products containing stem cell derived or containing biologically
active or biologically derived ingredients including: NuGene Face Wash, NuGene Universal Cream, NuGene Universal Serum, NuGene
Light and Bright, NuGene Eye Serum, NuGene Anti-Hair Loss Serum, NuGene Regenerative Shampoo & Conditioner, and Advanced Infusion
Serums as well as the following products which have not yet been commercially launched and/or developed: NuGene FaceMask, NuGene
Melasma Serum, NuGene Acne Serum, NuGene Revitalizing Night Cream, NuGene Toner, NuGene Body Lotion, NuGene Specialty Soap, NuGene
Neck and Décolleté Lotion, and other age defying products that are stem cell derived or which contain biologically
active or biologically derived ingredients. On March 5, 2015, we announced the launch of the NuGene kathy ireland
®
brand.
The campaign includes a re-branded internet presence and new videos featuring Ms. Ireland. We have initiated the launch of a larger
multi-media campaign to promote the product line.
NuGene is obligated to utilize commercially
reasonable efforts to meet the following minimum totals of net sales of Licensed Products for the specified periods:
|
Period
|
|
Forecasted Minimum
Net Sales
|
|
|
|
|
|
|
|
Contract Year 1
|
|
$
|
1,500,000
|
|
|
|
|
|
|
|
|
Contract Year 2
|
|
$
|
2,500,000
|
|
|
|
|
|
|
|
|
Contract Year 3
|
|
$
|
3,750.000
|
|
|
|
|
|
|
|
|
Contract Year 4
|
|
$
|
5,000,000
|
|
A "Contract Year" corresponds
to a calendar year ending December 31 and commences with the year 2015.
For each of these Contract Years we are
obligated to pay a minimum royalty as follows:
|
Period
|
|
Minimum Royalty
|
|
|
|
|
|
|
|
Contract Year 1
|
|
$
|
100,000
|
|
|
|
|
|
|
|
|
Contract Year 2
|
|
$
|
150,000
|
|
|
|
|
|
|
|
|
Contract Year 3
|
|
$
|
200,000
|
|
|
|
|
|
|
|
|
Contract Year 4
|
|
$
|
250,000
|
|
Additionally, we are obligated to pay an
annual Brand Participation fee to kiWW
®
which provides for general advertising, good will and promotion of
the overall kathy ireland brand. NuGene paid kiWW $350,000 effective upon execution of the License. This onetime fee includes a
"Brand Participation" fee for Contract Year 1. Brand Participation fees for Contract Years 2 through 8 are $50,000 annually
with an additional 1% of the total gross sales of Licensed Products of the prior year beginning in Contract Year 4.
This description of the License Agreement
is a summary and does not purport to describe all material conditions, obligations and payments that are the responsibility of
NuGene to the Licensor. The description provided of the License Agreement in this Annual Report is qualified in its entirety by
reference to the License Agreement attached as an Exhibit to our Current Report on Form 8-K filed with the Securities and Exchange
Commission (the “SEC”) on January 6, 2015, and that agreement is incorporated herein by this reference as if fully
set forth herein Report.
The Company and kiWW have recently been
in discussions to address and resolve issues and problems under the License Agreement, which includes the Company’s failure
to timely tender certain payments under the License Agreement. The Company and kiWW have reached an agreement in principle under
which both parties will release each other from all claims under the License Agreement as of the date they execute a definitive
agreement, which has not yet been executed. The definitive agreement will provide for, among other things, certain actions to be
taken by each party and a mutual commitment to proceed under the terms of the License Agreement.
Advisory Board
In an effort to improve our product line
and retain the latest and most forward looking state of the art services, the Company has assembled an Advisory Board consisting
of Board Certified Dermatologists and Plastic Surgeons consisting of medical professionals whom the Company believes are leaders
in their respective fields. We anticipate that our Advisory Board members, among other things, will provide us with strategic and
business development ideas, insight into major trends in the highly competitive cosmeceutical marketplace and input regarding their
specific expertise.
COMPETITION
NuGene experiences and will experience
intense competition from companies developing cosmeceuticals with stem cell derived active ingredients as well other ingredients.
Our pharmaceutical division competes or will compete with companies developing stem cell based pharmaceuticals. Our cosmeceutical
line of products also competes with other companies that offer a plant derived stem cell skin care line or stem-cell derived extracts.
Many of these companies have substantially greater financial, technological, research and development, marketing and personnel
resources.
GOVERNMENTAL REGULATION
U.S. Government Regulation
The health care industry is highly regulated
in the United States. The federal government, through various departments and agencies, state and local governments, and private
third-party accreditation organizations regulate and monitor the health care industry, associated products, and operations. The
following is a general overview of the laws and regulations pertaining to our business.
FDA Regulation of Stem Cell Treatment
and Products
The FDA regulates the manufacture
of human stem cell treatments and associated products under the authority of the Public Health Safety Act (“PHSA”)
and the Federal Food, Drug, and Cosmetic Act (“FDCA”). Stem cell derived products can be regulated under FDA’s
Human Cells, Tissues, and Cellular and Tissue-Based Products Regulations (“HCT/Ps”), or may also be subject to
FDA’s drug, biological product, or medical device regulations.
Cosmetic and Skin Care Regulation
Depending upon product claims and formulation,
skin care products may be regulated as cosmetics, drugs, devices, or combination cosmetics and drugs. We currently only market
cosmetic skin care products and are evaluating entry into the pharmaceutical market. The FDA has authority to regulate cosmetics
marketed in the United States under the FDCA and the Fair Packaging and Labeling Act (“FPLA”) and implementing regulations.
The Federal Trade Commission (the “FTC”) regulates the advertising of cosmetics under the FTCA.
The FDCA prohibits the marketing of adulterated
and misbranded cosmetics. Cosmetic ingredients must also comply with the FDA’s ingredient, quality, and labeling requirements
and the FTC’s requirements pertaining to truthful and non-misleading advertising. Cosmetic products and ingredients, with
the exception of color additives, are not required to have FDA premarket approval. Manufacturers of cosmetics are also not required
to register their establishments, file data on ingredients, or report cosmetic-related injuries to the FDA.
We are required to substantiate the safety
and product claims of our cosmetic products and ingredients before marketing. The FDA or FTC may disagree with our characterization
of one or more of the skin care products as a cosmetic or the product claims. This could result in a variety of enforcement actions
which could require the reformulation or relabeling of our products, the submission of information in support of the product claims
or the safety and effectiveness of our products, or more punitive action, all of which could have a material adverse effect on
our business. If the FDA determines we have failed to comply with applicable requirements under the FDCA or FPLA, it can impose
a variety of enforcement actions from public warning letters, injunctions, consent decrees, and civil penalties to seizure of our
products, total or partial shutdown of our production, and criminal prosecutions. If any of these events were to occur, it could
materially adversely affect us. If the FTC determines we have failed to substantiate our claims, it can pursue a variety of actions
including disgorgement of profits, injunction from further violative conduct, and consent decrees.
Some types of skin-care products are regulated
as both cosmetics and drugs under the FDCA. Examples of drug-cosmetic combination products are facial moisturizers that contain
sunscreen and skin protectant hand lotions. Products that are both cosmetics and drugs because of ingredients or intended use must
satisfy the regulatory requirements for both cosmetics and drugs. The drug requirements include either FDA premarket approval under
an NDA or an abbreviated new drug application (“ANDA”), or, more typically, implicit approval through conformance with
the applicable FDA final regulation (also known as an over-the-counter drug monograph) that specifies the conditions that must
be met for the drug to be generally recognized as safe and effective.
At present, we do not
anticipate that any of the products we market will be regulated as a combination cosmetic and drug or solely as a drug or device.
However, the FDA may disagree with such a determination which could result in a variety of enforcement actions and significant
additional expenditure to comply with regulations which the FDA may deem applicable to such products. We are in the process
of researching new products that may be classified as combination cosmetics and drugs which would require FDA approval.
See
“Risk Factors” beginning on page 8 of this prospectus, and in particular, on pages 14 - 18.
Domestic State and Local Government
Regulation
Some states and local governments in the
United States regulate the labeling, operation, sale, and distribution of our skin care products. To the extent additional state
or local laws apply, we intend to comply with them.
Foreign Government Regulation
In general, we will need to comply with
the government regulations of each individual country in which our products are to be distributed and sold. These regulations vary
in complexity and can be as stringent, and on occasion even more stringent, than FDA regulations in the United States. The level
of complexity and stringency is not always precisely understood today for each country, creating greater uncertainty for the international
regulatory process. Furthermore, government regulations can change with little to no notice and may result in increased regulation
of our product(s), resulting in a greater regulatory burden for us. We have not yet thoroughly explored the applicable laws and
regulations that we will need to comply with in foreign jurisdictions. As a result it is possible that we may not be permitted
to sell our products in foreign markets or expand our business into one or more foreign jurisdictions.
THE SCIENCE BEHIND OUR PRODUCTS
Human Stem Cells
Cells are the basic living units that make
up humans, animals, plants and other organisms. Stem cells have two important characteristics that distinguish them from other
types of cells. First, they can renew themselves for long periods of time. Second, they are unspecialized and under certain conditions
can be induced to become cells with special functions such as metabolically active cells of the liver or transparent and protective
cells of the eye. Until recently, scientists have worked with two major kinds of stem cells,
embryonic stem cells
and
adult
stem cells
each category of which has different properties and characteristics.
Adipose Stem Cell-Derived Media
We use human fat tissue (adipose tissue)
to derive adult human stem cells. Adipose stem cell derived growth factors have been shown to contribute to exerting diverse regenerative
effects on skin cells. These growth factors help in stimulating collagen synthesis and migration of dermal fibroblasts (a major
contributor in skin repair, regeneration, and revitalization). Using a proprietary technique for the isolation of these cytokines
from adipose derived stem cells, NuGene has been able to incorporate these proteins into its cosmeceutical line of products.

Peptides
Peptides are molecular links of amino acids
that help your skin to produce the anti-wrinkle protein known as collagen. Peptides are closely related to proteins. Peptides and
proteins are present in every living cell and are responsible for many biochemical activities including acting in enzymes, hormones,
antibiotics, etc. Peptides are listed in many better skin care products, and they perform important functions in the skin. As people
age, they lose collagen which results in sagging and wrinkled skin. Since collagen is a protein composed of long chains of amino
acids linked together by peptide bonds, peptides play an important role in replacing lost collagen. As collagen breaks down, the
protein chains break up into smaller chains of peptides. The resulting peptide chains send signals to the body to produce more
collagen. Since peptides are small, they can penetrate the skin’s protective barriers. Applying peptides directly to the
skin through good skin care products tricks the skin into thinking that it has lost collagen recently and needs to produce more.
Research has identified a number of peptides which mimic the function of the naturally produced growth factors in the human body.
The advantage of using peptides over growth factors in skin care formula is their stability, but they are not complete in structural
integrity as expressed by proteins in vivo.
Production of Growth Factors
NuGene has been able to produce a sufficient
quantity of biologically active growth factors derived from adipose stem cells to meet its current product requirements. With patent-pending
technology, the process has achieved a high yield rate and superior quality of biomolecules. Our purification technique enables
NuGene to have a high concentration of growth factors and cytokines in our skincare line.
Nano-encapsulation
One of the material challenges of the skincare
science is the delivery of the active ingredients to the skin. At NuGene, we met this challenge by using proprietary nano-encapsulation
technology. This technology allowed us to effectively deliver adipose stem cell derived cytokines and growth factors through embedding
them within a nanosome or capsule. This added step enhances the penetration of these biomolecules into the skin cells. Additionally,
it prevents these growth factors/cytokines from becoming degraded by proteases present in the mixture. We believe this enhances
the effectiveness of our products.
NuGene’s Anti-Aging Mechanism
Based upon research on adipose derived
stem cell cultured media, we have discovered the following potential benefits:
|
|
·
|
Increases the proliferation of epithelial cells;
|
|
|
·
|
Increases the synthesis of hyaluronic acid;
|
|
|
·
|
Increases the synthesis of collagen and elastin;
|
|
|
·
|
Adipose stem cell-derived EGF, bFGF increases the proliferation of fibroblast which synthesizes collagen and elastin; and
|
|
|
·
|
Adipose stem cell-derived EGF, IGF, bFGF, aFGF increases the skin elasticity by up-regulation of collagen, elastin, hyaluronic acid, and other extracellular matrixes (ECMs).
|

INTELLECTUAL PROPERTY
We have invested, and continue to invest,
significantly in intellectual properties, which consist of patents, trademarks, proprietary formulas and trade secrets, and URLs.
We have a program to file applications for and obtain patents in the United States and in selected foreign countries where we believe
filing for such protection is appropriate. There are certain processes and applications which we wish to keep confidential; we
do not seek patent protection for such items. We currently have four US provisional patent applications, and four pending international
applications based on the same four US provisional patents. The principal claims made in these applications cover wound scar and
burn treatment creams, the methods of preparing stem cells and the formulations for stem cell media containing products.
The following summarizes our pending applications:
|
|
•
|
AK01 skin treatment formulations
application describes and claims dermal treatment compositions
include medium recovered from an adipose-derived stem cell culture where the medium is collected under defined conditions. The
medium includes growth factors and other materials that, when transported to living portions of the skin, improve the condition
of damaged or aged skin. The compositions include combinations of transport ingredients in controlled proportions for effective
topical application.
|
|
|
•
|
A
K03-WO burn, scar, and wound treatment creams
application describes and claims dermal treatment
compositions include an emollient base, a conditioned medium, and a nanosilver particulate. The conditioned medium may be harvested
from culture of human adipocyte-derived stem cells. The stem cells may be cultured in the presence of the nanosilver particulate.
In other embodiments, the stem cells are cultured in medium that does not contain a nanosilver particulate. Instead the conditioned
medium is compounded with the nanosilver particulate after the media is harvested from the stem cell culture.
|
|
|
•
|
AK04 burn, scar, and wound healing aids and bandages
application describes and claims bandages
combining sterile dressings with a composition including a conditioned medium. The conditioned medium may be nanoencapsulated or
may be dispersed in an emollient base and applied to the sterile dressing. The conditioned medium may be harvested from culture
of human adipocyte-derived stem cells. The nanoencapsulated conditioned medium may be washed and resuspended before applying to
the sterile dressing. The emollient base may include defined proportions of oils and emulsifying agents.
|
|
|
•
|
NG06 wound, wrinkle, and blemish treatment dressings
application claims priority (benefits
from the earlier filing of) two of the Company’s earlier US Provisional applications. The NG06 application describes and
claims inventions relating to electroactive dressings and bandages. These inventions combine electroactive sterile dressings and
the Company's proprietary human adipose-derived stem cell culture media. Human adipose-derived stem cells produce a variety of
growth-promoting and healing materials such as growth factors and cytokines. Electroactive wound dressings produce local electric
fields by providing electrical half cells in proximity to treated skin. The inventions combine the benefits of the growth factors
and the localized electric fields to a localized region of the body.
|
|
|
|
The device of the invention includes dressings having a cathode and an anode, each forming an electrical
half-cell, and a conductive treatment gel containing HADSCC media. The treatment gel may also contain human adipose-derived stem
cells. The method includes providing a treatment gel and a dressing, applying the treatment gel to the skin or to the dressing,
and applying the dressing to the skin. Other embodiments include bandages combining sterile dressings with a composition including
a conditioned medium. The conditioned medium may be nanoencapsulated or may be dispersed in an emollient base and applied to the
sterile dressing.
|
EMPLOYEES
As of September 30, 2016 we had 12 employees,
all of which were full time employees. None of our employees is represented by a labor union, and we consider our relationship
with our employees to be good. It is possible that the Company will outsource many functions so we cannot estimate whether there
will be a significant increase in the number of employees. It is likely, however, that we will need to increase the number of employees
in the general and administrative and business development areas over the next two years, as well as for our research and development
activities.
DESCRIPTION OF PROPERTY
Our corporate offices are currently located
at 17912 Cowan, Suite A, Irvine, California, 92614. We sublease our sole corporate facilities from ASP, an affiliate of our Company,
for approximately $16,637 per month (including common area maintenance), consistent with the amount that is charged to ASP by the
property owner. On February 5, 2015, ASP entered into a new five-year lease for the property with the owner beginning July 1, 2015
and subsequently amended to begin June 1, 2015. The lease was subsequently amended to increase the square footage under lease beginning
in January 2016. The lease includes annual increases in the monthly lease payments of approximately 3% each year.
LEGAL PROCEEDINGS
On July 10, 2015 Stemage Skin Care, LLC
(the “Plaintiff”) filed a complaint in the U.S. District Court for the Central District of California entitled “Stemage
Skin Care LLC, a North Carolina limited liability company vs. NuGene International, Inc. et al.” (Civil Action No.8:15-cv-01078-AG-JCG).
The complaint also names as defendants NuGene, Inc., Ali Kharazmi, Saeed Kharazmi, Kathy Ireland Worldwide, Stephen Roseberry,
Steve Rosenblum and Erik Sterling. The complaint contains allegations of damage asserted to be grounded on: (i) copyright infringement;
(ii) interference with contract; (iii) intentional interference with prospective economic advantage; (iv) negligent interference
with prospective economic advantage; and (v) conspiracy. The complaint allegedly arises out of an August 20, 2012 agreement among
the Plaintiff and kathy ireland inc. ("KI") pursuant to which KI made Kathy Ireland available to perform “Ambassador
Services" as defined within that agreement. That agreement effectively terminated in October 2014 and is the subject of a
separate arbitration with KI and Kathy Ireland before the American Arbitration Association. On or around August 4, 2016 the Company
settled the litigation. All parties to this litigation have executed a settlement agreement resolving and waiving all claims, with
a full dismissal, with prejudice, of the action. Pursuant to the settlement, the Company has paid the total sum of $50,000
to the Plaintiff.
On July 31, 2015 Star Health & Beauty,
LLC (“SH&B”) filed a complaint in the U.S. District Court for the Northern District of Georgia entitled “Star
Health & Beauty, LLC vs. NuGene, Inc. and NuGene International, Inc. Defendants” (Case No. 1:15-cv-02634-CAP). The complaint
alleges that our use of the NUGENE name and trademark infringes on SH&B’s NUGEN name. SH&B seeks cancelation of our
NUGENE trademark, as well as unspecified monetary damages. We are in the process of early discovery to assist us in evaluating
the merits of this lawsuit and intend to defend our intellectual property rights vigorously. As this matter is at an early stage,
no assurance of outcome currently can be given.
In May 2016, we were informed that the
California Labor Commissioner scheduled a hearing in connection with two individuals that claimed our Company did not fulfill its
obligations to pay a final paycheck. The two individuals are seeking back pay and penalties totaling approximately $31,000. Although
we intend to contest various aspects of each claim, we recognize our Company may be held liable and accrued our best estimate of
the eventual amount of the settlement.
On May 6, 2016 we were presented with a demand
for payment of compensation for a former executive employee (the “Executive”) pursuant to his employment contract with
our Company. The amount of the compensation claimed by the Executive totaled $49,998. Our Company’s management is evaluating
the merits of this matter. No amounts have been accrued in connection with this matter through September 30, 2016.
Other than that described above, we are
not currently a party to any other material legal proceedings. We are not aware of any pending or threatened litigation against
us that in our view would have a material adverse effect on our business, financial condition, liquidity, or operating results.
However, legal claims are inherently uncertain, and we cannot assure you that we will not be adversely affected in the future by
legal proceedings.
MANAGEMENT
Executive Officers, Directors and Key
Employees
The following are our current executive
officers and directors and their respective ages and positions as of December __, 2016:
|
Name
|
|
Age
|
|
Position
|
|
|
|
|
|
|
|
Steven Carson
|
|
59
|
|
Chief Executive Officer
|
|
|
|
|
|
|
|
Mohammad Ai Kharazmi
|
|
58
|
|
Chairman of the Board
|
|
|
|
|
|
|
|
Mohammad Saeed Kharazmi
|
|
52
|
|
Interim Chief Financial Officer; Secretary; Director
|
|
|
|
|
|
|
|
Theodore Schwarz
|
|
55
|
|
Independent Director
|
Executive Officers
Steven Carlson.
Steve was
appointed as NuGene’s Chief Executive Officer on July 25, 2016. Steve is an accomplished pharmaceutical skin care and cosmetic
products executive with broad management skills, including strategic planning and business development, go-to-market execution,
business integration, and federal health and safety compliance for consumer products. He has over 35 years of industry experience,
having worked at such notable companies as Allergan and Obagi Medical Products. Steve began his career at Allergan (NYSE: AGN).
During his 15-year tenure he rose to the position of Senior Vice President of Marketing, leading the licensing, development, and
early commercialization of one of the world’s most recognized brands - Botox®. Steve subsequently served as CEO, President,
and Director of Obagi Medical Products (OMP), a global pharmaceutical skin care line, where he successfully led the company’s
public offering in 2006. Valeant Pharmaceuticals ultimately acquired Obagi Medical Products for over $400 million.
More recently Steve served as: (i) CEO,
President, and a Director of Eyenetra Inc. (2014 to 2016), where he helped the organization develop the first smart phone based
vision testing system for providing prescriptions for glasses through telemedicine; (ii) CEO, President, and Director of Apnicure,
Inc. (2011 to 2013), where he was responsible for corporate strategy and execution for the development of a medical device alternative
to CPAP in the treatment of sleep apnea; and, (iii) CEO, President, and Director of Obagi Medical Products, Inc. (2005-2010), where
he was responsible for corporate strategies and execution for a specialty skin health pharmaceutical company focused on physician-dispensed
medical aesthetics and therapeutic dermatology. Steve is a graduate of the University of Minnesota, where he earned his B.S. degree
in Biology.
Directors
M. Ali Kharazmi.
Ali
was appointed to our Board of Directors in December, 2014. Ali Kharazmi was the co-founder of NuGene in November 2006 and was appointed
Chief Executive Officer, President and a Board Member of the Company on December 29, 2014. He has worked extensively in the medical
industry since 1986. In 1998, Ali co-founded Medical Information Networks, one of the first medical technology companies to
provide electronic prescription services and formulary control with e-detailing to physicians by pharmaceutical companies. In
2003, he co-founded Newport Beach Plastic Surgery, one of the largest staff model plastic surgery multi-site medical groups in
California. In 2005, Ali co-founded the Genetic Institute of Anti-Aging, the Center for Weight Management and the Plastic
Surgery Institute, as well as, Advanced Surgical Partners. In 2012, he founded the Regenerative Institute, a research lab that
works exclusively on stem cell related research and treatment methods and also co-founded Ortho Regeneration, a sport and orthopedic
regenerative company specializing in Platelet Rich Plasma (“PRP”) and Adult Human Adipose stem cell based regenerative
services. Ali received his Bachelors of Arts in Economics from University of California, Irvine in 1980, his Masters in Business
Administration from American Graduate School of International Management, Thunderbird in 1983 and his Juris Doctorate from Western
State University College of Law in 1997. Ali’s status as a founder of our Company, his longstanding service with us, and
his entrepreneurial investment career in early stage businesses qualify him to serve as our director.
M. Saeed Kharazmi, MD.
Saeed
was appointed to our Board of Directors in December, 2014. Saeed obtained his Medical Degree from University of California, Irvine
in 1992 and completed his Residency and Chief Residency at St. Mary’s Medical Center/UCLA in 1996. In 2014 Saeed joined University
of California, Riverside (“UCR”) as Health Services Assistant Clinical Professor and became the Medical Director of
UCR Health, a university hospitalist group. He is also the acting Medical Director for Advanced Surgical Partners, a multispecialty
surgical facility that he and his brother started in 2005. His interest in age management and research led to the development of
Genetic Institute of Anti-Aging in 2005 where he became co-founder of NuGene. Saeed led the NuGene scientific team to introduce
new active ingredients for skin rejuvenation, and wound healing. Saeed’s status as a founder of our Company, his longstanding
service with us, and his extensive business, managerial, executive and leadership experience in the healthcare industry make him
qualified to serve as our director.
Ted Schwarz.
Ted joined the
NuGene Board in June, 2016. Ted previously served as President of SkinMedica, a leading topical medical aesthetics company. During
his nine years at SkinMedica he directed the commercial, new product and R&D teams allowing for seven straight years of market
leading growth positioning SkinMedica for its successful acquisition by pharmaceutical giant Allergan for $350MM. Ted most recently
served as President of Sienna Labs where he co-developed the business plan to bring to market topical solutions based on plasmonic
resonance for treatment of both aesthetic and disease based dermatological indications. Sienna Labs recently completed $34 million
Series A financing led by ARCH Venture Partners. Ted received his BA degree in Marketing and Business from Michigan State University.
His background also includes 15 years of Pharmaceutical executive sales and marketing leadership at Galderma and La Roche-Posay,
a L’Oreal company. Ted’s extensive knowledge of our industry, experience in managing a cosmeceuticals company, and
his knowledge of and experience in the commercialization of cosmeceutical products qualify him to serve as our director.
Summary Compensation Table.
The following table sets forth the information
as to compensation paid to or earned by our Chief Executive Officer and our two other most highly compensated executive officers
during the fiscal years ended December 31, 2014 and 2015. These individuals are referred to in this registration statement as our
named executive officers. As none of our named executive officers received non-equity incentive plan compensation or nonqualified
deferred compensation earnings during the fiscal years ended December 31, 2014 and 2015 and August 31, 2016, we have omitted those
columns from the table.
|
|
|
|
|
|
|
|
|
|
|
|
Stock
|
|
|
Option
|
|
|
All Other
|
|
|
|
|
|
Name & Principal Position
|
|
Fiscal Year
|
|
|
Salary
|
|
|
Bonus
|
|
|
Awards
|
|
|
Awards
|
|
|
Compensation
|
|
|
Total
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
M. Ali Kharazmi,
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
CEO, Chairman
|
|
|
2014
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
|
|
2015
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
|
|
2016
|
|
|
|
-
|
|
|
|
-
|
|
|
|
2,000,000
|
|
|
|
|
|
|
|
2,000,000
RSU; 1,008,367 warrants
|
|
|
$
|
4,131,000
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Mo Kharazmi,
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Director/Secretary/Int. CFO
|
|
|
2014
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
|
|
2015
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
|
|
2016
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
2,000,000 RSU; 1,008,367
warrants
|
|
|
$
|
2,711,000
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Donna Queen,
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Director
|
|
|
2014
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
|
|
2015
|
|
|
$
|
17,500
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
$
|
17,500
|
|
|
|
|
|
2016
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Theodore Schwarz,
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Director
|
|
|
2014
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
|
|
2015
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
|
|
2016
|
|
|
|
-
|
|
|
|
-
|
|
|
|
(1
|
)
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Steven Carlson
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Chief Executive Officer
|
|
|
2014
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
|
|
2015
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
|
|
2016
|
|
|
$
|
41,024
|
|
|
$
|
100,000
|
|
|
|
-
|
|
|
|
2,352,619
|
|
|
|
-
|
|
|
$
|
1,127,000
|
|
(1) In June, 2016 the Company agreed to
issue Mr. Schwarz 500,000 shares of the Company’s common stock that will vest at the rate of 166,666 shares per year, over
a three-year period provided Mr. Schwarz continues to serve on the Board.
Steve Carlson Employment Agreement
On July 18, 2016 we entered into a written
employment agreement with Steve Carlson for Mr. Carlson to serve as our Chief Executive Officer (the “
Employment Agreement
”).
Under the Employment Agreement we agreed to compensate Mr. Carlson as follows: annual salary of $400,000; $100,000 draw against
salary upon closing of the first $250,000 in bridge or similar financing; annual incentive bonuses based upon performance targets
to be mutually agreed upon; and, vacation, sick leave, and other employee benefits afforded by the Company. Mr. Carlson was also
issued stock options to acquire 5% of our issued and outstanding shares, to be determined through our next significant financing
transaction or series of significant financing transactions. The exercise price of the options was fixed on July 18, 2016 and equal
to the closing price of our shares on the OTC Marketplace. The options vest ratably monthly over 3-years, and vest 100% upon a
change in control of the Company.
Ali Kharazmi Agreements
On September 22, 2016 we entered into the
following agreements with our Chairman, M. Ali Kharazmi:
(i)
Chairman
Agreement
: Under the Chairman Agreement we agreed to compensate Ali for his service as our Chairman of the Board as follows:
annual salary of $120,000; annual incentive bonuses in the discretion of the Company’s Board of Directors; and, reimbursement
of expenses. Ali was also issued 2,000,000 shares of our common stock. Ali was also issued the warrant described below, and the
restricted stock agreement described below. In the event within twelve (12) months following a change of control (as defined
in the Chairman Agreement) either: (1) Ali is removed from his position as Chairman of the Board without cause; or, (2) Ali
resigns as the Chairman, then the Company shall provide Ali with a single lump sum payment equal to the greater of (y) the amount
of compensation remaining over the remainder of the 3-year term of the agreement; or, (z) 200% of the annual compensation then
in effect, to be paid within 30-days after the change in control.
(ii)
Warrant
Agreement
: Ali was granted the right, during the first 24-months immediately following the issuance, to acquire that number
of shares of our common stock equal to the greater of (i) (1,000,000) shares; or, (ii) two and one-half percent (2.5%) of the issued
and outstanding common shares the Company, determined on an as if converted and issued basis. Upon the expiration of that 24-month
period, the number of shares available for purchase shall be fixed in accordance with the above formula, and such number shall
be fixed up to and until the expiration of the Warrant Agreement. Exercise price for the warrant is $0.71 per share, which was
the closing stock price for the Company Stock on the date of the issuance, September 22, 2016. The term of the Warrant Agreement
is 10-years, and it is non-assignable without the written consent of the Company.
(iii)
Restricted
Sock Units Agreement
: Ali was granted restricted stock units for 2,000,000 shares of the Company’s common stock (“
RSUs
”).
The RSUs vest at the rate of 33.33% each year anniversary of the issuance date (September 22, 2016). There is also 100% vesting
upon a change in control of the Company, as defined in the agreement.
Saeed Kharazmi Agreements
On September 22, 2016 we entered into the
following agreements with our Secretary, Interim CFO, and Board Member, M. Saeed Kharazmi:
(i)
Executive
Employment Agreement
: Under this Agreement we agreed to compensate Saeed for his service as our Vice-Chairman of the Board
and our Chief Medical Officer as follows: annual salary of $120,000; annual incentive bonuses in the discretion of the Company’s
Board of Directors; and, reimbursement of expenses. Saeed was also issued the warrant described below, and the restricted stock
agreement described below. In the event within twelve (12) months following a change of control (as defined in the agreement)
either: (1) Saeed is removed from his position without cause; or, (2) Saeed resigns, then the Company shall provide Saeed
with a single lump sum payment equal to the greater of (y) the amount of compensation remaining over the remainder of the 3-year
term of the agreement; or, (z) 200% of the annual compensation then in effect, to be paid within 30-days after the change in control.
(ii)
Warrant
Agreement
: Saeed was granted the right, during the first 24-months immediately following the issuance, to acquire that number
of shares of our common stock equal to the greater of (i) (1,000,000) shares; or, (ii) two and one-half percent (2.5%) of the issued
and outstanding common shares the Company, determined on an as if converted and issued basis. Upon the expiration of that 24-month
period, the number of shares available for purchase shall be fixed in accordance with the above formula, and such number shall
be fixed up to and until the expiration of the Warrant Agreement. Exercise price for the warrant is $0.71 per share, which was
the closing stock price for the Company Stock on the date of the issuance, September 22, 2016. The term of the Warrant Agreement
is 10-years, and it is non-assignable without the written consent of the Company.
(iii)
Restricted
Sock Units Agreement
: Saeed was granted restricted stock units for 2,000,000 shares of the Company’s common stock (“
RSUs
”).
The RSUs vest at the rate of 33.33% each year anniversary of the issuance date (September 22, 2016). There is also 100% vesting
upon a change in control of the Company, as defined in the agreement.
Outstanding Equity Awards at Fiscal
Year-End
None.
Director Compensation
Our officers who served as directors received
no compensation for their services as members of our Board of Directors. Donna Queen was a member of our Company’s Board
of Directors from May 26, 2015 to January 19, 2016. Her agreement with the Company called for her to be paid an annual stipend
of $30,000 and to receive 100,000 shares of our common stock that vested quarterly. She was paid $17,500 for her service as a director
through December 31, 2015.
SECURITY OWNERSHIP OF CERTAIN BENEFICIAL
OWNERS AND MANAGEMENT
The following table
sets forth certain information as of September 30, 2016, regarding the beneficial ownership of our common stock by (i) each stockholder
known by us to be the beneficial owner of more than five percent of our common stock, (ii) by each of our executive officers named
in the Summary Compensation Table and our directors and (iii) by all of our executive officers and directors as a group. Each of
the persons named in the table has sole voting and investment power with respect to common stock beneficially owned. Unless otherwise
noted in the table, the address for each of the persons identified is 17912 Cowan, Irvine, California.
|
Name and Address
of Beneficial Owner
|
|
Amount and Nature
of Beneficial
Ownership
(1)
|
|
Percent
of Common
Stock
|
|
|
M. Ali Kharazmi
(2)
Chairman
|
|
10,735,240 shares
|
|
|
26.26
|
%
(4)
|
|
|
|
|
|
|
|
|
|
M. Saeed Kharazmi
(3)
Board Member
|
|
11,235,240 shares
|
|
|
26.91
|
%
(5)
|
|
|
|
|
|
|
|
|
Steven Carlson,
CEO
|
|
|
|
|
|
|
|
All directors and named executive officers as a group (3 persons)
|
|
21,970,480 shares
|
|
|
51.37
|
%
(6)
|
|
|
(1)
|
Under Rule 13d-3, a beneficial owner of a security includes
any person who, directly or indirectly, through any contract, arrangement, understanding, relationship, or otherwise has or shares:
(i) voting power, which includes the power to vote, or to direct the voting of shares; and (ii) investment power, which includes
the power to dispose or direct the disposition of shares. Certain shares may be deemed to be beneficially owned by more than one
person (if, for example, persons share the power to vote or the power to dispose of the shares). In addition, shares are deemed
to be beneficially owned by a person if the person has the right to acquire the shares (for example, upon exercise of an option)
within 60 days of the date as of which the information is provided. In computing the percentage ownership of any person, the amount
of shares outstanding is deemed to include the amount of shares beneficially owned by such person (and only such person) by reason
of these acquisition rights. As a result, the percentage of outstanding shares of any person as shown in this table does not necessarily
reflect the person's actual ownership or voting power with respect to the number of shares of common stock actually outstanding.
|
|
|
(2)
|
Common stock beneficially owned by M. Ali Kharazmi includes
958,860 shares of common stock issuable upon conversion of Series A Preferred Stock and 1,500,000 shares of common stock held
of record by his spouse. Mr. Kharazmi disclaims ownership in these shares held by his spouse .
|
|
|
(3)
|
Common stock beneficially owned by M. Saeed Kharazmi includes
958,860 shares of common stock issuable upon conversion of Series A Preferred Stock .
|
|
|
(4)
|
Percentage of common stock beneficially owned by Ali Kharazmi
is calculated by dividing the total amount of common stock beneficially owned by 40,883,533, which includes 39,924,673 shares
of common stock outstanding and committed to be issued and 958,860 shares of common stock issuable to him upon conversion of Series
A Preferred Stock owned.
|
|
|
(5)
|
Percentage of common stock beneficially owned by M. Saeed
Kharazmi is calculated by dividing the total amount of common stock beneficially owned by 40,883,533, which includes 39,924,673
shares of common stock outstanding and committed to be issued and 958,860 shares of common stock issuable to him upon conversion
of Series A Preferred Stock owned.
|
|
|
(6)
|
Percentage of common stock beneficially owned by all named
executives is calculated by dividing the total amount of common stock beneficially owned by 41,842,393, which includes 39,924,673
shares of common stock outstanding and 1,917,720 shares of common stock issuable upon conversion of Series A Preferred Stock owned.
|
Changes in Control
. We are not aware
of any arrangements that may result in “changes in control” as that term is defined by the provisions of Item 403 of
Regulation S-K.
CERTAIN RELATIONSHIPS AND RELATED TRANSACTIONS
Director Independence
.
We have three directors, two of whom are brothers and the founders of our Company. Only one of our directors is independent within
the definition of “independence” as defined in the NASDAQ rules governing members of boards of directors.
Related Party Transactions
. Parties
are considered to be related if one party has the ability to control or exercise significant influence over the other party in
making financial and operating decisions. A related party transaction is considered to be a transfer of resources or obligations
between related parties, regardless of whether or not a price is charged.
The following individuals and entities
have been identified as related parties based on their affiliation with our former CEO and current Chairman of the Board:
|
Ali Kharazmi
|
|
Former Chief Executive Officer and President; Chairman of the Board; and, greater than 10% shareholder
|
|
Saeed Kharazmi
|
|
Board Member; Acting Chief Financial Officer and greater than 10% shareholder
|
|
Genetics Institute of Anti-Aging
|
|
Company with common ownership and management
|
|
Applied M.A.K. Enterprises, Inc. (“MAK”)
|
|
Company with common ownership and management
|
|
Advanced Surgical Partners, LLC (“ASP”)
|
|
Company with common ownership and management
|
|
Center for Weight Management & Plastic Surgery (“CWM”)
|
|
Company with common ownership and management
|
|
Center for Regenerative Science, LLC
|
|
Company with common ownership and management
|
The following amounts were owed to related
parties, affiliated with our former CEO and current Chairman of the Board, at the dates indicated:
|
|
|
September 30,
2016
|
|
|
December 31,
2015
|
|
|
December 31,
2014
|
|
|
ASP
|
|
$
|
226,188
|
|
|
$
|
70,890
|
|
|
$
|
45,000
|
|
|
MAK
|
|
|
-
|
|
|
|
-
|
|
|
|
40,084
|
|
|
CWM
|
|
|
34,000
|
|
|
|
34,000
|
|
|
|
-
|
|
|
Ali Kharazmi
|
|
|
91,175
|
|
|
|
25,641
|
|
|
|
37,000
|
|
|
Saeed Kharazmi
|
|
|
30,698
|
|
|
|
2,405
|
|
|
|
-
|
|
|
Less amounts advanced to ASP and repaid in January 2016
|
|
|
-
|
|
|
|
(95,000
|
)
|
|
|
-
|
|
|
Accounts payable - related parties
|
|
|
382,061
|
|
|
$
|
37,936
|
|
|
$
|
122,084
|
|
The amount owed to ASP at December 31,
2015 of $70,890 relates to legal and administrative services provided by ASP employees to our Company. Our Company temporarily
advanced $95,000 to ASP prior to December 31, 2015 and was repaid in full prior to January 5, 2016. Prior to December 31, 2014,
all managerial, legal, and administrative services were provided to our Company by related parties, free of charge. The amount
owed to CWM relates to medical procedures provided to NuGene consultants as compensation for advertising and marketing services
provided to NuGene. The amount owed to Ali Kharazmi and all amounts outstanding at December 31, 2015 and 2014 represent advances
that bore no interest and were due on demand or expense reimbursements incurred in the ordinary course of business. The amounts
owed to Saeed Kharazmi at December 31, 2015 related to expense reimbursements incurred in the ordinary course of business.
For the year ended December 31, 2014, our
Company recognized revenues from ASP in the amount of approximately $481,000. We had no revenues from ASP during the year ended
December 31, 2015. Beginning on December 1, 2014, our Company sublet office space from ASP. Prior to December 1, 2014, our Company
utilized corporate office space at ASP, free of charge. Messrs. Ali and Saeed Kharazmi, our former CEO and our Acting CFO, respectively,
have been foregoing salaries since NuGene, Inc. was incorporated in 2006.
DESCRIPTION OF SECURITIES
General
At the date hereof, we are authorized by
our Articles of Incorporation to issue an aggregate of 100,000,000 shares of common stock, par value $0.0001 per share, and 25,000,000
shares of “blank check” preferred stock, par value $0.0001 per share. We are incorporated in the state of Nevada.
Common Stock
We are authorized to issue up to 100,000,000
shares of common stock, $0.0001 par value. Each share of common stock entitles a stockholder to one vote on all matters upon
which stockholders are permitted to vote. Common stock does not confer on the holder any preemptive right or other similar
right to purchase or subscribe for any additional securities issued by us and is not convertible into other securities. No
shares of common stock are subject to redemption or any sinking fund provisions. All the outstanding shares of our common
stock are fully paid and non-assessable. Subject to the rights of the holders of the preferred stock, the holders of shares
of our common stock are entitled to dividends out of funds legally available when and as declared by our Board of Directors. In
the event of our liquidation, dissolution, or winding up, holders of our common stock are entitled to receive, ratably, the net
assets available to stockholders after payment of all creditors and any liquidation preference on outstanding preferred stock.
As of December __, 2016 we had 40,334,673
shares of common stock outstanding.
Warrants
The following summary of certain terms
and provisions of the warrants offered hereby is not complete and is subject to, and qualified in its entirety by the provisions
of the form of the warrant, which is filed as an exhibit to the registration statement of which this prospectus is a part of. Prospective
investors should carefully review the terms and provisions set forth in the form of warrant.
Exercisability
. The warrants
are exercisable immediately upon issuance and at any time up to the date that is five years from the date of issuance. The warrants
will be exercisable, at the option of each holder, in whole or in part by delivering to us a duly executed exercise notice accompanied
by payment in full for the number of shares of our common stock purchased upon such exercise (except in the case of a cashless
exercise as discussed below). Unless otherwise specified in the warrant, the holder will not have the right to exercise any portion
of the warrant if the holder (together with its affiliates) would beneficially own in excess of 4.99% of the number of shares of
our common stock outstanding immediately after giving effect to the exercise, as such percentage ownership is determined in accordance
with the terms of the warrants.
Cashless Exercise
. In
the event that a registration statement covering shares of common stock underlying the warrants, or an exemption from registration,
is not available for the resale of such shares of common stock underlying the warrants, the holder may, in its sole discretion,
exercise the warrant in whole or in part and, in lieu of making the cash payment otherwise contemplated to be made to us upon such
exercise in payment of the aggregate exercise price, elect instead to receive upon such exercise the net number of shares of common
stock determined according to the formula set forth in the warrant. In no event shall we be required to make any cash payments
or net cash settlement to the registered holder in lieu of issuance of common stock underlying the warrants.
Exercise Price.
The initial
exercise price per share of common stock purchasable upon exercise of the warrants is $ per share (125% of the public offering
price of the common stock). The exercise price is subject to appropriate adjustment in the event of certain stock dividends and
distributions, stock splits, stock combinations, reclassifications or similar events affecting our common stock and also upon any
distributions of assets, including cash, stock, or other property to our stockholders.
Certain Adjustments
. The
exercise price and the number of shares of common stock purchasable upon the exercise of the warrants are subject to adjustment
upon the occurrence of specific events, including stock dividends, stock splits, combinations and reclassifications of our common
stock.
Transferability
. Subject
to applicable laws, the warrants may be transferred at the option of the holders upon surrender of the warrants to us together
with the appropriate instruments of transfer.
Warrant Agent and Exchange Listing
. The
warrants will be issued in registered form under a warrant agency agreement between , as warrant agent,
and us.
Fundamental Transaction
. If,
at any time while the warrants are outstanding, (1) we consolidate or merge with or into another corporation and we are not the
surviving corporation, (2) we sell, lease, license, assign, transfer, convey or otherwise dispose of all or substantially all of
our assets, (3) any purchase offer, tender offer or exchange offer (whether by us or another individual or entity) is completed
pursuant to which holders of our shares of common stock are permitted to sell, tender or exchange their shares of common stock
for other securities, cash or property and has been accepted by the holders of 50% or more of our outstanding shares of common
stock, (4) we effect any reclassification or recapitalization of our shares of common stock or any compulsory share exchange pursuant
to which our shares of common stock are converted into or exchanged for other securities, cash or property, or (5) we consummate
a stock or share purchase agreement or other business combination with another person or entity whereby such other person or entity
acquires more than 50% of our outstanding shares of common stock, each, a “
Fundamental Transaction
”, then upon
any subsequent exercise of the warrants, the holders thereof will have the right to receive the same amount and kind of securities,
cash or property as it would have been entitled to receive upon the occurrence of such Fundamental Transaction if it had been,
immediately prior to such Fundamental Transaction, the holder of the number of warrant shares then issuable upon exercise of the
warrant, and any additional consideration payable as part of the Fundamental Transaction.
Rights as a Stockholder
. Except
as otherwise provided in the warrants or by virtue of such holder's ownership of shares of our common stock, the holder of a warrant
does not have the rights or privileges of a holder of our common stock, including any voting rights, until the holder exercises
the warrant.
Preferred Stock
We may issue up to 25,000,000 shares of
"blank check" preferred stock, $0.0001 par value, in one or more classes or series within a class as may be determined
by our Board of Directors, who may establish, from time to time, the number of shares to be included in each class or series, may
fix the designation, powers, preferences and rights of the shares of each such class or series and any qualifications, limitations
or restrictions thereof. Any preferred stock so issued by the Board of Directors may rank senior to the common stock with
respect to the payment of dividends or amounts upon liquidation, dissolution, or winding up of us, or both.
25,000,000 shares of our preferred stock
have been designated as Series A Preferred Stock, 1,917,720 of which are issued and outstanding and held by the two co-founders
of NuGene. Each share of Series A Preferred Stock is convertible at the option of the holder thereof at any time after the date
of issuance into one share of common stock subject to adjustment for certain events. No such events requiring an adjustment have
yet to occur. The Series A Preferred Stock will automatically convert into common stock on the voluntary or involuntary subsequent
sale or transfer of that preferred stock to any entity or person who is not the original holder thereof. Generally the holders
of the Series A Preferred Stock and the holders of the common stock shall vote together and not as a separate classes, with each
share of Series A Preferred Stock having three times the of votes equal to the number of shares of common stock into which the
shares of Series A preferred Stock could have been then converted. Notwithstanding the foregoing so long as there remain at least
900,000 shares of Series A Preferred Stock outstanding the holders of the Series A Preferred exclusively and as a separate class
shall be entitled to elect a majority of the board of directors of the Company. This description of the Series A Preferred Stock
is a summary that is qualified in its entirety by reference to the Certificate of Designation as filed with the Secretary of State
of Nevada on December 24, 2014, attached hereto as an exhibit and incorporated herein by this reference.
No other series or shares of preferred
stock are currently outstanding. The issuance of preferred stock, while providing desirable flexibility in connection with
possible acquisitions and other corporate purposes, could have the effect of making it more difficult for a third party to acquire,
or of discouraging a third party from acquiring, a majority of our outstanding voting stock.
The above descriptions of our capital stock
and provisions of our Articles of Incorporation and Bylaws are summaries and are qualified by reference to our second amended and
restated certificate of incorporation, as amended, and our amended and restated bylaws. We have filed copies of these documents
with the SEC as exhibits to the registration statement of which this prospectus forms a part.
Issued and Outstanding Warrants
A summary of the outstanding warrants activity
and related information as of September 30, 2016 is as follows:
|
Exercise
|
|
|
Warrants
|
|
|
Remaining
|
|
|
Warrants
|
|
|
Price
|
|
|
Outstanding
|
|
|
Life (Years)
|
|
|
Exercisable
|
|
|
$
|
0.001
|
|
|
|
1,350,000
|
|
|
|
1.19
|
|
|
|
900,000
|
|
|
$
|
0.60
|
|
|
|
462,500
|
|
|
|
4.90
|
|
|
|
462,500
|
|
|
$
|
0.71
|
|
|
|
2,016,734
|
|
|
|
9.98
|
|
|
|
2,016,734
|
|
|
$
|
1.50
|
|
|
|
50,000
|
|
|
|
1.87
|
|
|
|
50,000
|
|
|
$
|
2.00
|
|
|
|
300,000
|
|
|
|
3.87
|
|
|
|
300,000
|
|
|
$
|
2.50
|
|
|
|
500,000
|
|
|
|
4.24
|
|
|
|
500,000
|
|
|
|
|
|
|
|
4,679,234
|
|
|
|
|
|
|
|
4,229,234
|
|
In connection with this offering, we have
agreed to issue to the underwriter warrants to purchase up to a total of 5% of the number shares of common stock issued in the
offering. The warrants are exercisable at any time, and from time to time, in whole or in part, during the four-year period commencing
one year from the effective date of our initial public offering, and ending on the date that is five years from the effective date
of the offering in compliance with FINRA Rule 5110(f)(2)(G)(i). The warrants are exercisable at a per share price equal to 125%
of the price per share in this offering. The warrants provide for piggyback registration right not greater than seven years from
the effective date of the offering in compliance with FINRA Rule 5110(f)(2)(G)(v). We will bear all fees and expenses attendant
to registering the securities issuable on exercise of the warrants other than underwriting commissions incurred and payable by
the holders. The exercise price and number of shares issuable upon exercise of the warrants may be adjusted in certain circumstances
including in the event of a stock dividend or our recapitalization, reorganization, merger or consolidation. However, the warrant
exercise price or underlying shares will not be adjusted for issuances of shares of common stock at a price below the warrant exercise
price.
Anti-Takeover Effects of Various Provisions
of Nevada Law and Our Amended and Restated Articles of Incorporation.
Provisions of the Nevada Corporation Law
and our Amended and Restated Articles of Incorporation and Amended and Restated Bylaws could make it more difficult to acquire
us by means of a tender offer, a proxy contest or otherwise, or to remove incumbent officers and directors. These provisions, summarized
below, would be expected to discourage certain types of coercive takeover practices and takeover bids our board of directors may
consider inadequate and to encourage persons seeking to acquire control of us to first negotiate with us. We believe that the benefits
of increased protection of our ability to negotiate with the proponent of an unfriendly or unsolicited proposal to acquire or restructure
us will outweigh the disadvantages of discouraging takeover or acquisition proposals because, among other things, negotiation of
these proposals could result in an improvement of their terms.
Series “A” Preferred
Stock.
Our Series “A” Preferred Stock, as described in our Amended and Restated Articles of Incorporation
and the Certificate of Designation for the Series “A” Preferred Stock, grant to the holders of our Series “A”
Preferred Stock the right to approve most material transactions affecting the Company. This will negatively affect the voting power
or other rights of our common stockholders, and could delay or prevent a change of control of NuGene.
Nevada Anti-Takeover Statute.
We
are subject to Nevada’s Combination with Interested Stockholders Statute (Nevada Corporation Law Sections 78.411–78.444)
which prohibits an “interested stockholder” from entering into a “combination” with the corporation, unless
certain conditions are met. An “interested stockholder” is a person who, together with affiliates and associates, beneficially
owns (or within the prior two years, did beneficially own) 10% or more of the corporation’s capital stock entitled to vote.
No Cumulative Voting.
Our
Amended and Restated Articles of Incorporation prohibits cumulative voting in the election of directors.
Limitations on Liability and Indemnification
of Officers and Directors
Nevada Corporation Law limits or eliminates
the personal liability of directors to corporations and their stockholders for monetary damages for breaches of directors’
fiduciary duties as directors. Our Amended and Restated Articles of Incorporation and Bylaws include provisions that require the
Company to indemnify, to the fullest extent allowable under the Nevada Corporation Law, our directors or officers against monetary
damages for actions taken as a director or officer of our company, or for serving at our request as a director or officer or another
position at another corporation or enterprise, as the case may be. Our Amended and Restated Articles of Incorporation and Bylaws
also provide that we must indemnify and advance reasonable expenses to our directors and officers, subject to our receipt of an
undertaking from the indemnified party as may be required under the Nevada Corporation Law. We are also expressly authorized to
carry directors’ and officers’ insurance to protect our company, our directors, officers and certain employees for
some liabilities.
The limitation of liability and indemnification
provisions under the Nevada Corporation Law and in our Amended and Restated Articles of Incorporation and Bylaws may discourage
stockholders from bringing a lawsuit against directors for breach of their fiduciary duties. These provisions may also have the
effect of reducing the likelihood of derivative litigation against directors and officers, even though such an action, if successful,
might otherwise benefit us and our stockholders. However, these provisions do not limit or eliminate our rights, or those of any
stockholder, to seek non-monetary relief such as injunction or rescission in the event of a breach of a director’s fiduciary
duties. Moreover, the provisions do not alter the liability of directors under the federal securities laws. In addition, your investment
may be adversely affected to the extent that, in a class action or direct suit, we pay the costs of settlement and damage awards
against directors and officers pursuant to these indemnification provisions.
Authorized but Unissued Shares
Our authorized but unissued shares of common
stock and preferred stock are available for future issuance without shareholder approval. We may use additional shares for a variety
of purposes, including future public offerings to raise additional capital, to fund acquisitions and as employee compensation.
The existence of authorized but unissued shares of common stock and preferred stock could render more difficult or discourage an
attempt to obtain control of us by means of a proxy contest, tender offer, merger or otherwise.
Transfer Agent and Registrar
The registrar and transfer agent for our
common stock is Olde Monmouth Stock Transfer Co., Inc. 200 Memorial Parkway Atlantic Highlands, New Jersey 07716, Telephone: (732)
872-2727.
OTC Markets Listing
The shares of our common stock are quoted
on the OTCQB under the symbol NUGN. We will apply to list our common stock and warrants on The NASDAQ Capital Market under the
symbols “NUGN” and “NUGNW”.
UNDERWRITING
Joseph Gunnar & Co., LLC is acting
as representative of the underwriters (the “Representative”). We have entered into an underwriting agreement dated ,
2016 with the Representative. Subject to the terms and conditions of the underwriting agreement, we have agreed to sell to each
underwriter named below, and each underwriter named below has severally agreed to purchase, at the public offering price less the
underwriting discounts set forth on the cover page of this prospectus, the number of shares of common stock and warrants listed
next to its name in the following table:
|
Name of Underwriter
|
|
Number of
Shares
|
|
|
Joseph Gunnar & Co., LLC
|
|
|
|
|
|
Total
|
|
|
|
|
The underwriters are committed to purchase
all the shares of common stock and warrants offered by us other than those covered by the option to purchase additional shares
described below, if they purchase any shares. The obligations of the underwriters may be terminated upon the occurrence of certain
events specified in the underwriting agreement. Furthermore, pursuant to the underwriting agreement, the underwriters’ obligations
are subject to customary conditions, representations and warranties contained in the underwriting agreement, such as receipt by
the underwriters of officers’ certificates and legal opinions.
We have agreed to indemnify the underwriters
against specified liabilities, including liabilities under the Securities Act, and to contribute to payments the underwriters may
be required to make in respect thereof.
The underwriters are offering the shares
and warrants, subject to prior sale, when, as and if issued to and accepted by them, subject to approval of legal matters by their
counsel and other conditions specified in the underwriting agreement. The underwriters reserve the right to withdraw, cancel, or
modify offers to the public and to reject orders in whole or in part.
We have granted the underwriters an over-allotment
option. This option, which is exercisable for up to 45 days after the date of this prospectus, permits the underwriters to
purchase a maximum of additional shares (15% of the shares sold in this offering) from us to cover over-allotments, if any. If
the underwriters exercise all or part of this option, they will purchase shares covered by the option at the public offering price
that appears on the cover page of this prospectus, less the underwriting discount. If this option is exercised in full, the total
price to the public will be $ and the total net proceeds, before expenses, to us will be $ .
Discounts
The following table shows the public offering
price, underwriting discount and proceeds, before expenses, to us. The information assumes either no exercise or full exercise
by the underwriters of their over-allotment option.
|
|
|
Per Share
|
|
|
Total Without
Over-Allotment
Option
|
|
|
Total With Full
Over-Allotment
Option
|
|
|
Public offering price
|
|
$
|
|
|
|
$
|
|
|
|
$
|
|
|
|
Underwriting discount (7%)
|
|
$
|
|
|
|
$
|
|
|
|
$
|
|
|
|
Non-accountable expense allowance (1%)
|
|
$
|
|
|
|
$
|
|
|
|
$
|
|
|
|
Proceeds, before expenses, to us
|
|
$
|
|
|
|
$
|
|
|
|
$
|
|
|
The underwriters propose
to offer the shares and warrants offered by us to the public at the public offering price set forth on the cover of this prospectus.
In addition, the underwriters may offer some of the shares to other securities dealers at such price less a concession of $
per share. If all of the shares offered by us are not sold at the public offering price, the Representative may change the
offering price and other selling terms by means of a supplement to this prospectus.
We have agreed to pay
the Representative a non-accountable expense allowance of 1% of the public offering price at the closing, excluding the over-allotment
option. We have paid an expense deposit of $25,000 to the Representative, which will be applied against such non-accountable expense
allowance.
We have also agreed
to pay the following expenses of the Representative relating to the offering: (a) all fees, expenses and disbursements relating
to background checks of our officers and directors in an amount not to exceed $10,000 in the aggregate; (b) all filing fees
and communication expenses associated with the review of this offering by FINRA; (c) all fees, expenses and disbursements
relating to the registration, qualification or exemption of securities offered under the securities laws of foreign jurisdictions
designated by the Representative, including the reasonable fees and expenses of the Representative’s blue sky counsel; (d)
the fees and expenses of the underwriters’ legal counsel, not to exceed $50,000, (e) the fees costs associated with bound
volumes of the public offering materials and commemorative mementos and lucite tombstones, in such quantities as the Representative
may reasonably designate, (f) $29,500 for the underwriters’ use of Ipreo’s book-building, prospectus tracking
and compliance software for this offering; and (g) $20,000 of the Representative’s actual accountable road show expenses
for the offering.
We estimate that the
total expenses of the offering payable by us, excluding the total underwriting discount, will be approximately $ .
Discretionary Accounts
The underwriters do
not intend to confirm sales of the securities offered hereby to any accounts over which they have discretionary authority.
Lock-Up Agreements
Pursuant to “lock-up”
agreements, we, our executive officers and directors, and certain of our stockholders, have agreed, subject to limited exceptions,
without the prior written consent of the Representative not to directly or indirectly, offer to sell, sell, pledge or otherwise
transfer or dispose of any of shares of (or enter into any transaction or device that is designed to, or could be expected to,
result in the transfer or disposition by any person at any time in the future of) our common stock, enter into any swap or other
derivatives transaction that transfers to another, in whole or in part, any of the economic benefits or risks of ownership of shares
of our common stock, make any demand for or exercise any right or cause to be filed a registration statement, including any amendments
thereto, with respect to the registration of any shares of common stock or securities convertible into or exercisable or exchangeable
for common stock or any other securities of the Company or publicly disclose the intention to do any of the foregoing, subject
to customary exceptions, for a period of 180 days from the date of this prospectus, in the case of our directors and officers,
and 90 days from the date of this prospectus, in the case of us and our principal stockholders.
Representative’s Warrants
We have agreed to issue
to the Representative warrants to purchase up to a total of shares of common stock (5% of the shares of common stock
sold in this offering, excluding the over-allotment option). The warrants will be exercisable at any time, and from time to time,
in whole or in part, during the four-year period commencing one year from the effective date of the offering, which period shall
not extend further than five years from the effective date of the offering in compliance with FINRA Rule 5110(f)(2)(G). The
warrants are exercisable at a per share price equal to 125% of the public offering price per share in the offering. The warrants
have been deemed compensation by FINRA and are therefore subject to a 180 day lock-up pursuant to Rule 5110(g)(1) of
FINRA. The underwriters (or permitted assignees under Rule 5110(g)(1)) will not sell, transfer, assign, pledge, or hypothecate
these warrants or the securities underlying these warrants, nor will they engage in any hedging, short sale, derivative, put, or
call transaction that would result in the effective economic disposition of the warrants or the underlying securities for a period
of 180 days from the date of this prospectus.
The exercise price
and number of shares issuable upon exercise of the warrants may be adjusted in certain circumstances including in the event of
a stock dividend, extraordinary cash dividend or recapitalization, reorganization, merger or consolidation. However, the warrant
exercise price or underlying shares will not be adjusted for issuances of shares of common stock at a price below the warrant exercise
price.
Right of First Refusal
Until twelve months
after the closing date of the offering, the Representative will have a right of first refusal to act as lead underwriter for any
future public or private equity offering during such twelve month period.
Electronic Offer, Sale, and Distribution
of Shares and Warrants
A prospectus in electronic
format may be made available on the websites maintained by one or more of the underwriters or selling group members. The Representative
may agree to allocate a number of shares and warrants to underwriters and selling group members for sale to its online brokerage
account holders. Internet distributions will be allocated by the underwriters and selling group members that will make internet
distributions on the same basis as other allocations. Other than the prospectus in electronic format, the information on these
websites is not part of, nor incorporated by reference into, this prospectus or the registration statement of which this prospectus
forms a part, has not been approved or endorsed by us, and should not be relied upon by investors.
No Sales of Similar Securities
We and our executive
officers and directors have agreed with the underwriters, subject to certain exceptions, not to sell or transfer any common stock
or securities convertible into, exchangeable for, or exercisable for, other than the securities which we may sell in this offering,
for 180-days after the date of this prospectus without the prior written consent of Joseph Gunnar & Co., LLC on behalf of the
underwriters. Specifically, we and our executive officers and directors have agreed not to:
• offer,
pledge, sell or contract to sell, sell any option or contract to purchase, purchase any option or contract to sell, grant any option,
right or warrant to purchase, lend, or otherwise transfer or dispose of, either directly or indirectly, any shares of common stock
or any securities convertible into or exercisable or exchangeable for common stock;
• file
or cause to become effective a registration statement under the Securities Act relating to the offer and sale of any shares of
common stock or securities convertible into or exercisable or exchangeable for shares of common stock;
• enter
into any swap or other arrangement that transfers to another, in whole or in part, any of the economic consequences of ownership
of the common stock, whether any such transaction is to be settled by delivery of common stock or such other securities, in cash
or otherwise;
• make
any demand for or exercise any right with respect to the registration of any shares of our common stock or any securities convertible
into or exercisable or exchangeable for our common stock; or
• publicly
announce an intention to effect any transaction described in this list.
The 180-day period
in all of the agreements is subject to extension if (i) during the last 17 days of the restricted period we issue an earnings
release or material news or a material event relating to us occurs or (ii) prior to the expiration of the restricted period, we
announce that we will release earnings results during the 16-day period beginning on the last day of the restricted period, in
which case the restrictions imposed in these lock-up agreements shall continue to apply until the expiration of the 18-day period
beginning on the issuance of the earnings release or the occurrence of the material news or material event. This extension will
not apply if the publication of research reports by the underwriters during the period around the expiration of the restricted
period is no longer restricted by applicable law or regulation.
We have agreed to indemnify
the underwriters and their controlling persons against certain liabilities for misstatements in the registration statement of which
this prospectus forms a part, including liabilities under the Securities Act of 1933, as amended, or to contribute to payments
the underwriters may be required to make in respect thereof. We have also agreed to reimburse the underwriters for all reasonable
third-party costs, fees and expenses not to exceed, without our consent, $ .
Stabilization
In connection with
this offering, the underwriters may engage in stabilizing transactions, over-allotment transactions, syndicate-covering transactions,
penalty bids and purchases to cover positions created by short sales.
|
|
·
|
Stabilizing
transactions permit bids to purchase shares so long as the stabilizing bids do not exceed a specified maximum,
and
are engaged in for the purpose of preventing or retarding a decline in
the market price of the shares while the offering is in progress.
|
|
|
·
|
Over-allotment transactions
involve sales by
the underwriters
of shares in excess of the number
of shares
the underwriters are
obligated to purchase. This creates
a syndicate short position which may be either a covered short position or a naked short position. In a covered short position,
the number of shares over-allotted by
the underwriters
is not
greater than the number of shares that they may purchase in the over-allotment option. In a naked short position, the number of
shares involved is greater than the number of shares in the over-allotment option. The
underwriters
may close out any short position by exercising their over-allotment option and/or purchasing shares in the open market.
|
|
|
·
|
Syndicate covering transactions
involve purchases of shares in the open market after the distribution has been completed in order to cover syndicate short positions.
In determining the source of shares to close out the short position,
the
underwriters
will consider, among other things, the price of shares available for purchase in the open market as compared
with the price at which they may purchase shares through exercise of the over-allotment option. If
the
underwriters
sell more shares than could be covered by exercise of the over-allotment option and,
therefore,
have a naked short position, the position can be closed out only by buying shares in the open market. A naked short position is
more likely to be created if the underwriters are concerned that after pricing there could be downward pressure on the price of
the shares in the open market that could adversely affect investors who purchase in the offering.
|
|
|
·
|
Penalty
bids permit the Representative to reclaim a selling concession from a syndicate member when the shares originally sold by that
syndicate member are purchased in stabilizing or syndicate covering transactions to cover syndicate short positions.
|
These stabilizing transactions,
syndicate covering transactions and penalty bids may have the effect of raising or maintaining the market price of our shares of
common stock or preventing or retarding a decline in the market price of our shares of common stock. As a result, the price of
our common stock in the open market may be higher than it would otherwise be in the absence of these transactions. Neither we nor
the underwriters make any representation or prediction as to the effect that the transactions described above may have on the price
of our common stock. These transactions may be effected on the NASDAQ Capital Market, in the over-the-counter market or otherwise
and, if commenced, may be discontinued at any time.
Passive Market Making
In connection with
this offering, underwriters and selling group members may engage in passive market making transactions in our common stock on The
NASDAQ Capital Market or on the OTCQB in accordance with Rule 103 of Regulation M under the Exchange Act, during a period
before the commencement of offers or sales of the shares and extending through the completion of the distribution. A passive market
maker must display its bid at a price not in excess of the highest independent bid of that security. However, if all independent
bids are lowered below the passive market maker’s bid, then that bid must then be lowered when specified purchase limits
are exceeded.
Other Relationships
Certain of the underwriters
and their affiliates may in the future provide various investment banking, commercial banking and other financial services for
us and our affiliates for which they may in the future receive customary fees.
Offer Restrictions Outside the United
States
Other than in the United
States, no action has been taken by us or the underwriters that would permit a public offering of the securities offered by this
prospectus in any jurisdiction where action for that purpose is required. The securities offered by this prospectus may not be
offered or sold, directly or indirectly, nor may this prospectus or any other offering material or advertisements in connection
with the offer and sale of any such securities be distributed or published in any jurisdiction, except under circumstances that
will result in compliance with the applicable rules and regulations of that jurisdiction. Persons into whose possession this prospectus
comes are advised to inform themselves about and to observe any restrictions relating to the offering and the distribution of this
prospectus. This prospectus does not constitute an offer to sell or a solicitation of an offer to buy any securities offered by
this prospectus in any jurisdiction in which such an offer or a solicitation is unlawful.
Australia
This prospectus is
not a disclosure document under Chapter 6D of the Australian Corporations Act, has not been lodged with the Australian Securities
and Investments Commission and does not purport to include the information required of a disclosure document under Chapter 6D of
the Australian Corporations Act. Accordingly, (i) the offer of the securities under this prospectus is only made to persons to
whom it is lawful to offer the securities without disclosure under Chapter 6D of the Australian Corporations Act under one or more
exemptions set out in section 708 of the Australian Corporations Act, (ii) this prospectus is made available in Australia only
to those persons as set forth in clause (i) above, and (iii) the offeree must be sent a notice stating in substance that by accepting
this offer, the offeree represents that the offeree is such a person as set forth in clause (i) above, and, unless permitted under
the Australian Corporations Act, agrees not to sell or offer for sale within Australia any of the securities sold to the offeree
within 12 months after its transfer to the offeree under this prospectus.
China
The information in
this document does not constitute a public offer of the securities, whether by way of sale or subscription, in the People’s
Republic of China (excluding, for purposes of this paragraph, Hong Kong Special Administrative Region, Macau Special Administrative
Region and Taiwan). The securities may not be offered or sold directly or indirectly in the PRC to legal or natural persons other
than directly to “qualified domestic institutional investors.”
European Economic
Area—Belgium, Germany, Luxembourg and Netherlands
The information in
this document has been prepared on the basis that all offers of securities will be made pursuant to an exemption under the Directive
2003/71/EC (“Prospectus Directive”), as implemented in Member States of the European Economic Area (each, a “Relevant
Member State”), from the requirement to produce a prospectus for offers of securities.
An offer to the public
of securities has not been made, and may not be made, in a Relevant Member State except pursuant to one of the following exemptions
under the Prospectus Directive as implemented in that Relevant Member State:
(a) to legal entities
that are authorized or regulated to operate in the financial markets or, if not so authorized or regulated, whose corporate purpose
is solely to invest in securities;
(b) to any legal entity
that has two or more of (i) an average of at least 250 employees during its last fiscal year; (ii) a total balance sheet of more
than €43,000,000 (as shown on its last annual unconsolidated or consolidated financial statements) and (iii) an annual net
turnover of more than €50,000,000 (as shown on its last annual unconsolidated or consolidated financial statements);
(c) to fewer than 100
natural or legal persons (other than qualified investors within the meaning of Article 2(1)(e) of the Prospectus Directive) subject
to obtaining the prior consent of the Company or any underwriter for any such offer; or
(d) in any other circumstances
falling within Article 3(2) of the Prospectus Directive, provided that no such offer of securities shall result in a requirement
for the publication by the Company of a prospectus pursuant to Article 3 of the Prospectus Directive.
France
This document is not
being distributed in the context of a public offering of financial securities (offre au public de titres financiers) in France
within the meaning of Article L.411-1 of the French Monetary and Financial Code (Code monétaire et financier) and Articles
211-1 et seq. of the General Regulation of the French Autorité des marchés financiers (“AMF”). The securities
have not been offered or sold and will not be offered or sold, directly or indirectly, to the public in France.
This document and any
other offering material relating to the securities have not been, and will not be, submitted to the AMF for approval in France
and, accordingly, may not be distributed or caused to distributed, directly or indirectly, to the public in France.
Such offers, sales
and distributions have been and shall only be made in France to (i) qualified investors (investisseurs qualifiés) acting
for their own account, as defined in and in accordance with Articles L.411-2-II-2° and D.411-1 to D.411-3, D. 744-1, D.754-1
and D.764-1 of the French Monetary and Financial Code and any implementing regulation and/or (ii) a restricted number of non-qualified
investors (cercle restreint d’investisseurs) acting for their own account, as defined in and in accordance with Articles
L.411-2-II-2° and D.411-4, D.744-1, D.754-1 and D.764-1 of the French Monetary and Financial Code and any implementing regulation.
Pursuant to Article
211-3 of the General Regulation of the AMF, investors in France are informed that the securities cannot be distributed (directly
or indirectly) to the public by the investors otherwise than in accordance with Articles L.411-1, L.411-2, L.412-1 and L.621-8
to L.621-8-3 of the French Monetary and Financial Code.
Ireland
The information in
this document does not constitute a prospectus under any Irish laws or regulations and this document has not been filed with or
approved by any Irish regulatory authority as the information has not been prepared in the context of a public offering of securities
in Ireland within the meaning of the Irish Prospectus (Directive 2003/71/EC) Regulations 2005 (the “Prospectus Regulations”).
The securities have not been offered or sold, and will not be offered, sold or delivered directly or indirectly in Ireland by way
of a public offering, except to (i) qualified investors as defined in Regulation 2(l) of the Prospectus Regulations and (ii) fewer
than 100 natural or legal persons who are not qualified investors.
Israel
The securities offered
by this prospectus have not been approved or disapproved by the Israeli Securities Authority (the ISA), or ISA, nor have such securities
been registered for sale in Israel. The shares may not be offered or sold, directly or indirectly, to the public in Israel, absent
the publication of a prospectus. The ISA has not issued permits, approvals or licenses in connection with the offering or publishing
the prospectus; nor has it authenticated the details included herein, confirmed their reliability or completeness, or rendered
an opinion as to the quality of the securities being offered. Any resale in Israel, directly or indirectly, to the public of the
securities offered by this prospectus is subject to restrictions on transferability and must be effected only in compliance with
the Israeli securities laws and regulations.
Italy
The offering of the
securities in the Republic of Italy has not been authorized by the Italian Securities and Exchange Commission (Commissione Nazionale
per le Societ—$$—Aga e la Borsa, “CONSOB” pursuant to the Italian securities legislation and, accordingly,
no offering material relating to the securities may be distributed in Italy and such securities may not be offered or sold in Italy
in a public offer within the meaning of Article 1.1(t) of Legislative Decree No. 58 of 24 February 1998 (“Decree No. 58”),
other than:
|
|
•
|
to Italian qualified investors, as defined in Article 100 of Decree no.58 by reference to Article
34-ter of CONSOB Regulation no. 11971 of 14 May 1999 (“Regulation no. 1197l”) as amended (“Qualified Investors”);
and
|
|
|
•
|
in other circumstances that are exempt from the rules on public offer pursuant to Article 100 of
Decree No. 58 and Article 34-ter of Regulation No. 11971 as amended.
|
Any offer, sale, or
delivery of the securities or distribution of any offer document relating to the securities in Italy (excluding placements where
a Qualified Investor solicits an offer from the issuer) under the paragraphs above must be:
|
|
•
|
made by investment firms, banks or financial intermediaries permitted to conduct such activities
in Italy in accordance with Legislative Decree No. 385 of 1 September 1993 (as amended), Decree No. 58, CONSOB Regulation No. 16190
of 29 October 2007 and any other applicable laws; and
|
|
|
•
|
in compliance with all relevant Italian securities, tax and exchange controls and any other applicable
laws.
|
Any subsequent distribution
of the securities in Italy must be made in compliance with the public offer and prospectus requirement rules provided under Decree
No. 58 and the Regulation No. 11971 as amended, unless an exception from those rules applies. Failure to comply with such rules
may result in the sale of such securities being declared null and void and in the liability of the entity transferring the securities
for any damages suffered by the investors.
Japan
The securities have
not been and will not be registered under Article 4, paragraph 1 of the Financial Instruments and Exchange Law of Japan (Law No.
25 of 1948), as amended (the “FIEL”) pursuant to an exemption from the registration requirements applicable to a private
placement of securities to Qualified Institutional Investors (as defined in and in accordance with Article 2, paragraph 3 of the
FIEL and the regulations promulgated thereunder). Accordingly, the securities may not be offered or sold, directly or indirectly,
in Japan or to, or for the benefit of, any resident of Japan other than Qualified Institutional Investors. Any Qualified Institutional
Investor who acquires securities may not resell them to any person in Japan that is not a Qualified Institutional Investor, and
acquisition by any such person of securities is conditional upon the execution of an agreement to that effect.
Portugal
This document is not
being distributed in the context of a public offer of financial securities (oferta pública de valores mobiliários)
in Portugal, within the meaning of Article 109 of the Portuguese Securities Code (Código dos Valores Mobiliários).
The securities have not been offered or sold and will not be offered or sold, directly or indirectly, to the public in Portugal.
This document and any other offering material relating to the securities have not been, and will not be, submitted to the Portuguese
Securities Market Commission (Comissão do Mercado de Valores Mobiliários) for approval in Portugal and, accordingly,
may not be distributed or caused to distributed, directly or indirectly, to the public in Portugal, other than under circumstances
that are deemed not to qualify as a public offer under the Portuguese Securities Code. Such offers, sales and distributions of
securities in Portugal are limited to persons who are “qualified investors” (as defined in the Portuguese Securities
Code). Only such investors may receive this document and they may not distribute it or the information contained in it to any other
person.
Sweden
This document has not
been, and will not be, registered with or approved by Finansinspektionen (the Swedish Financial Supervisory Authority). Accordingly,
this document may not be made available, nor may the securities be offered for sale in Sweden, other than under circumstances that
are deemed not to require a prospectus under the Swedish Financial Instruments Trading Act (1991:980) (Sw. lag (1991:980) om handel
med finansiella instrument). Any offering of securities in Sweden is limited to persons who are “qualified investors”
(as defined in the Financial Instruments Trading Act). Only such investors may receive this document and they may not distribute
it or the information contained in it to any other person.
Switzerland
The securities may
not be publicly offered in Switzerland and will not be listed on the SIX Swiss Exchange (“SIX”) or on any other stock
exchange or regulated trading facility in Switzerland. This document has been prepared without regard to the disclosure standards
for issuance prospectuses under art. 652a or art. 1156 of the Swiss Code of Obligations or the disclosure standards for listing
prospectuses under art. 27 ff. of the SIX Listing Rules or the listing rules of any other stock exchange or regulated trading facility
in Switzerland. Neither this document nor any other offering material relating to the securities may be publicly distributed or
otherwise made publicly available in Switzerland.
Neither this document
nor any other offering material relating to the securities have been or will be filed with or approved by any Swiss regulatory
authority. In particular, this document will not be filed with, and the offer of securities will not be supervised by, the Swiss
Financial Market Supervisory Authority (FINMA).
This document is personal
to the recipient only and not for general circulation in Switzerland.
United Arab Emirates
Neither this document
nor the securities have been approved, disapproved or passed on in any way by the Central Bank of the United Arab Emirates or any
other governmental authority in the United Arab Emirates, nor has the Company received authorization or licensing from the Central
Bank of the United Arab Emirates or any other governmental authority in the United Arab Emirates to market or sell the securities
within the United Arab Emirates. This document does not constitute and may not be used for the purpose of an offer or invitation.
No services relating to the securities, including the receipt of applications and/or the allotment or redemption of such shares,
may be rendered within the United Arab Emirates by the Company.
No offer or invitation
to subscribe for securities is valid or permitted in the Dubai International Financial Centre.
United Kingdom
Neither the information
in this document nor any other document relating to the offer has been delivered for approval to the Financial Services Authority
in the United Kingdom and no prospectus (within the meaning of section 85 of the Financial Services and Markets Act 2000, as amended
(“FSMA”)) has been published or is intended to be published in respect of the securities. This document is issued on
a confidential basis to “qualified investors” (within the meaning of section 86(7) of FSMA) in the United Kingdom,
and the securities may not be offered or sold in the United Kingdom by means of this document, any accompanying letter or any other
document, except in circumstances which do not require the publication of a prospectus pursuant to section 86(1) FSMA. This document
should not be distributed, published, or reproduced, in whole or in part, nor may its contents be disclosed by recipients to any
other person in the United Kingdom.
Any invitation or inducement
to engage in investment activity (within the meaning of section 21 of FSMA) received in connection with the issue or sale of the
securities has only been communicated or caused to be communicated and will only be communicated or caused to be communicated in
the United Kingdom in circumstances in which section 21(1) of FSMA does not apply to the Company.
In the United Kingdom,
this document is being distributed only to, and is directed at, persons (i) who have professional experience in matters relating
to investments falling within Article 19(5) (investment professionals) of the Financial Services and Markets Act 2000 (Financial
Promotions) Order 2005 (“FPO”), (ii) who fall within the categories of persons referred to in Article 49(2)(a) to (d)
(high net worth companies, unincorporated associations, etc.) of the FPO or (iii) to whom it may otherwise be lawfully communicated
(together “relevant persons”). The investments to which this document relates are available only to, and any invitation,
offer, or agreement to purchase will be engaged in only with, relevant persons. Any person who is not a relevant person should
not act or rely on this document or any of its contents.
LEGAL MATTERS
The validity of the securities being offered
by this prospectus is being passed upon for us by Spectrum Law Group, LLP, Irvine, California. Sichenzia Ross Ference Kesner LLP
is counsel for the underwriters in connection with this offering.
EXPERTS
The financial statements as of and for the
fiscal years ended December 31, 2014 and 2015 appearing in this prospectus and registration statement have been audited by Dov
Weinstein & Co. C.P.A. (Isr), an independent registered public accounting firm (“Dov Weinstein”), as stated in
their report appearing elsewhere herein and are included in reliance upon such report and upon the authority of such firm as experts
in accounting and auditing.
Change in Our Public Accounting Firm
On November 14, 2016, which we refer to as
the Dismissal Date, we advised Dov Weinstein that it was dismissed as our independent registered public accounting firm. Effective
November 14, 2016, we engaged Burr Pilger Mayer, Inc. as our independent registered public accounting firm to audit our consolidated
financial statements for the year ending December 31, 2016. The decision to dismiss Dov Weinstein as our independent registered
public accounting firm was approved by our Board of Directors.
The reports of Dov Weinstein on our financial
statements for the fiscal years of 2014 and 2015 contained no adverse opinion or disclaimer of opinion and were not qualified or
modified as to uncertainty, audit scope or accounting principle. In connection with its audits for the fiscal years of 2014 and
2015, there have been no disagreements with Dov Weinstein on any matter of accounting principles or practices, financial statement
disclosure, or auditing scope or procedure, which disagreements, if not resolved to the satisfaction Dov Weinstein, would have
caused them to make reference thereto in their reports on the financial statements for such years.
WHERE YOU CAN FIND MORE INFORMATION
We file reports, proxy statements and
other information with the SEC as required by the Exchange Act. You can find, copy and inspect information we file at the SEC’s
public reference room at 100 F Street, N.E., Room 1580, Washington, D.C. 20549. You can call the SEC at 1-800-SEC-0330 for further
information about the public reference room. You can review our electronically filed reports, proxy and information statements
on the SEC’s website at http://www.sec.gov or on our website at
http://www.nugene.com
. Information included on our
website is not a part of this prospectus.
This prospectus is part of a registration
statement that we filed with the SEC. The registration statement contains more information than this prospectus regarding us and
the securities, including exhibits and schedules. You can obtain a copy of the registration statement from the SEC at any address
listed above or from the SEC’s website.
NUGENE INTERNATIONAL, INC.
FINANCIAL STATEMENTS
INDEX
NUGENE INTERNATIONAL, INC. AND SUBSIDIARIES
CONDENSED
CONSOLIDATED BALANCE SHEETS
|
|
|
September 30
|
|
|
December 31
|
|
|
|
|
2016
|
|
|
2015
|
|
|
|
|
(Unaudited)
|
|
|
(1)
|
|
|
ASSETS
|
|
|
|
|
|
|
|
|
|
Current assets
|
|
|
|
|
|
|
|
|
|
Cash
|
|
$
|
307,776
|
|
|
$
|
22,907
|
|
|
Accounts receivable, net
of allowance for doubtful accounts of $532,661 and $177,140 on September
30, 2016 and December 31, 2015, respectively
|
|
|
10,181
|
|
|
|
347,048
|
|
|
Inventories
|
|
|
24,303
|
|
|
|
177,492
|
|
|
Other
current assets
|
|
|
46,751
|
|
|
|
100,555
|
|
|
Total current assets
|
|
|
389,011
|
|
|
|
648,002
|
|
|
|
|
|
|
|
|
|
|
|
|
Property and equipment, net of accumulated
depreciation of $81,551 and $40,620 at September 30, 2016 and December 31,
2015, respectively
|
|
|
171,958
|
|
|
|
208,350
|
|
|
Deposits
|
|
|
24,376
|
|
|
|
21,321
|
|
|
Total
assets
|
|
$
|
585,345
|
|
|
$
|
877,673
|
|
|
|
|
|
|
|
|
|
|
|
|
LIABILITIES AND
STOCKHOLDERS' DEFICIT
|
|
|
|
|
|
|
|
|
|
Current liabilities
|
|
|
|
|
|
|
|
|
|
Accounts payable and
accrued expenses
|
|
$
|
1,127,695
|
|
|
$
|
327,623
|
|
|
Accounts payable and
advances - related parties
|
|
|
382,061
|
|
|
|
37,936
|
|
|
Notes payable and
advances
|
|
|
1,669,725
|
|
|
|
842,504
|
|
|
Accrued interest
|
|
|
114,425
|
|
|
|
35,052
|
|
|
Derivative liabilities
|
|
|
1,061,536
|
|
|
|
-
|
|
|
Other
current liabilities
|
|
|
222,995
|
|
|
|
17,924
|
|
|
Total
current liabilities
|
|
|
4,578,437
|
|
|
|
1,261,039
|
|
|
Total
liabilities
|
|
|
4,578,437
|
|
|
|
1,261,039
|
|
|
|
|
|
|
|
|
|
|
|
|
Commitments and
contingencies (Note 10)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Stockholders' deficit
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Series A convertible preferred stock; $0.0001
par value; 25,000,000 shares authorized; 1,917,720 shares issued and
outstanding as of September 30, 2016 and December 31, 2015
|
|
|
192
|
|
|
|
192
|
|
|
Common stock; $0.0001 par value; 100,000,000
shares authorized; 40,334,673 and 39,894,673 shares issued and outstanding
as of September 30, 2016 and December 31, 2015, respectively
|
|
|
4,034
|
|
|
|
3,990
|
|
|
Additional paid-in-capital
|
|
|
11,327,709
|
|
|
|
6,180,959
|
|
|
Accumulated deficit
|
|
|
(15,325,027
|
)
|
|
|
(6,568,507
|
)
|
|
Total stockholders'
deficit
|
|
|
(3,993,092
|
)
|
|
|
(383,366
|
)
|
|
Total
liabilities and stockholders' deficit
|
|
$
|
585,345
|
|
|
$
|
877,673
|
|
See
accompanying notes to unaudited condensed consolidated financial
statements
|
(1)
|
The condensed consolidated balance
sheet as of December 31, 2015 has been derived from the audited
consolidated financial statements as of that date.
|
NUGENE INTERNATIONAL, INC. AND SUBSIDIARIES
CONDENSED
CONSOLIDATED STATEMENTS OF OPERATIONS
For
the three and nine months ended September 30, 2016 and 2015
(Unaudited)
|
|
|
Three Months Ended September 30,
|
|
|
Nine Months Ended September 30,
|
|
|
|
|
2016
|
|
|
2015
|
|
|
2016
|
|
|
2015
|
|
|
Revenues
|
|
$
|
85,888
|
|
|
$
|
520,731
|
|
|
$
|
409,981
|
|
|
$
|
1,538,453
|
|
|
Cost of revenues
|
|
|
555,450
|
|
|
|
150,411
|
|
|
|
658,882
|
|
|
|
443,943
|
|
|
Gross profit (loss)
|
|
|
(469,562
|
)
|
|
|
370,320
|
|
|
|
(248,901
|
)
|
|
|
1,094,510
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Operating expenses:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Advertising and
promotion
|
|
|
105,855
|
|
|
|
24,275
|
|
|
|
314,469
|
|
|
|
298,738
|
|
|
Personnel
|
|
|
3,421,672
|
|
|
|
1,502,559
|
|
|
|
4,138,979
|
|
|
|
2,064,815
|
|
|
Selling, general and
administrative
|
|
|
230,992
|
|
|
|
355,541
|
|
|
|
1,056,026
|
|
|
|
861,795
|
|
|
Research and
development
|
|
|
74,112
|
|
|
|
98,324
|
|
|
|
104,072
|
|
|
|
136,248
|
|
|
Professional
fees
|
|
|
592,611
|
|
|
|
416,634
|
|
|
|
1,278,866
|
|
|
|
770,889
|
|
|
Total
operating expenses
|
|
|
4,425,242
|
|
|
|
2,397,333
|
|
|
|
6,892,412
|
|
|
|
4,132,485
|
|
|
Loss
from operations
|
|
|
(4,894,804
|
)
|
|
|
(2,027,013
|
)
|
|
|
(7,141,313
|
)
|
|
|
(3,037,975
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Other income
(expenses):
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Interest income
|
|
|
4
|
|
|
|
-
|
|
|
|
5
|
|
|
|
-
|
|
|
Interest expense
|
|
|
(327,502
|
)
|
|
|
(75,000
|
)
|
|
|
(659,954
|
)
|
|
|
(76,443
|
)
|
|
Loss on issuance of
debt
|
|
|
(1,212,384
|
)
|
|
|
(75,000
|
)
|
|
|
(1,212,384
|
)
|
|
|
(76,443
|
)
|
|
Change in
fair value of derivative liabilities
|
|
|
249,226
|
|
|
|
-
|
|
|
|
257,126
|
|
|
|
-
|
|
|
Total
other income (expenses), net
|
|
|
(1,290,656
|
)
|
|
|
(75,000
|
)
|
|
|
(1,615,207
|
)
|
|
|
-
|
|
|
Net
loss
|
|
$
|
(6,185,460
|
)
|
|
$
|
(2,102,013
|
)
|
|
$
|
(8,756,520
|
)
|
|
$
|
(3,114,418
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Net loss per share - basic and diluted
|
|
$
|
(0.15
|
)
|
|
$
|
(0.05
|
)
|
|
$
|
(0.22
|
)
|
|
$
|
(0.08
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Weighted average number of common shares
outstanding, basic and diluted
|
|
|
40,334,673
|
|
|
|
39,732,934
|
|
|
|
40,156,826
|
|
|
|
39,506,104
|
|
See
accompanying notes to unaudited condensed consolidated financial
statements
NUGENE INTERNATIONAL, INC. AND SUBSIDIARIES
CONDENSED
CONSOLIDATED STATEMENTS OF CASH FLOWS
For
the nine months ended September 30, 2016 and 2015
(Unaudited)
|
|
|
Nine Months Ended September 30,
|
|
|
|
|
2016
|
|
|
2015
|
|
|
Cash flows from operating
activities
|
|
|
|
|
|
|
|
|
|
Net loss
|
|
$
|
(8,756,520
|
)
|
|
$
|
(3,114,418
|
)
|
|
Adjustments to reconcile net loss to net cash
used in operating activities:
|
|
|
|
|
|
|
|
|
|
Stock based compensation
and stock issued for services
|
|
|
4,151,367
|
|
|
|
1,731,658
|
|
|
Non-cash portion of
interest expense
|
|
|
154,373
|
|
|
|
-
|
|
|
Depreciation and
amortization
|
|
|
40,932
|
|
|
|
25,953
|
|
|
Amortization of debt
discount
|
|
|
508,926
|
|
|
|
-
|
|
|
Loss associated with
issuance of debt
|
|
|
1,212,384
|
|
|
|
-
|
|
|
Change in fair value of
derivative liabilities
|
|
|
(257,126
|
)
|
|
|
-
|
|
|
Bad debt expense
|
|
|
355,521
|
|
|
|
15,582
|
|
|
Changes in assets and
liabilities:
|
|
|
|
|
|
|
|
|
|
Accounts receivable
|
|
|
(18,654
|
)
|
|
|
(292,826
|
)
|
|
Inventories
|
|
|
211,852
|
|
|
|
(39,942
|
)
|
|
Brand participation
fee
|
|
|
-
|
|
|
|
225,000
|
|
|
Other assets
|
|
|
(4,859
|
)
|
|
|
(246,359
|
)
|
|
Accounts payable and
accrued expenses
|
|
|
800,072
|
|
|
|
283,458
|
|
|
Accounts payable and
advances - related parties
|
|
|
344,125
|
|
|
|
(16,167
|
)
|
|
Deferred revenues
|
|
|
-
|
|
|
|
(234,916
|
)
|
|
Other
current liabilities
|
|
|
205,071
|
|
|
|
-
|
|
|
Net cash used in
operating activities
|
|
|
(1,052,536
|
)
|
|
|
(1,662,977
|
)
|
|
|
|
|
|
|
|
|
|
|
|
Cash flows from investing
activities
|
|
|
|
|
|
|
|
|
|
Deposits
|
|
|
(3,055
|
)
|
|
|
(13,216
|
)
|
|
Purchase
of property and equipment
|
|
|
(4,540
|
)
|
|
|
(158,790
|
)
|
|
Net cash used in
investing activities
|
|
|
(7,595
|
)
|
|
|
(172,006
|
)
|
|
|
|
|
|
|
|
|
|
|
|
Cash flows from financing
activities
|
|
|
|
|
|
|
|
|
|
Proceeds from issuance of
notes payable
|
|
|
1,345,000
|
|
|
|
500,000
|
|
|
Proceeds
from issuance of common stock
|
|
|
-
|
|
|
|
340,000
|
|
|
Net cash provided by
investing activities
|
|
|
1,345,000
|
|
|
|
840,000
|
|
|
|
|
|
|
|
|
|
|
|
|
Net increase (decrease) in cash and cash
equivalents
|
|
|
284,869
|
|
|
|
(994,983
|
)
|
|
Cash, beginning of period
|
|
|
22,907
|
|
|
|
1,344,737
|
|
|
Cash, end of
period
|
|
$
|
307,776
|
|
|
$
|
349,754
|
|
|
|
|
|
|
|
|
|
|
|
|
Supplemental disclosure of
noncash activities:
|
|
|
|
|
|
|
|
|
|
Issuance of common shares
in connection with notes payable
|
|
$
|
26,642
|
|
|
$
|
-
|
|
|
Issuance of common
warrants in connection with notes payable
|
|
$
|
203,467
|
|
|
$
|
-
|
|
|
Derivative liabilities
|
|
$
|
106,278
|
|
|
|
|
|
|
Beneficial conversion
feature in connection with notes payable
|
|
$
|
765,316
|
|
|
$
|
-
|
|
|
Issuance of previously
earned but unissued common stock
|
|
$
|
13
|
|
|
$
|
-
|
|
|
Accrued interest converted
to notes payable
|
|
$
|
75,000
|
|
|
$
|
-
|
|
|
Transfer of prepaid
inventory to inventory
|
|
$
|
58,663
|
|
|
|
|
|
|
Issuance of common stock
for intellectual property
|
|
$
|
-
|
|
|
$
|
250,000
|
|
|
Cancellation of common
stock for intellectual property
|
|
$
|
-
|
|
|
$
|
100,000
|
|
See
accompanying notes to unaudited condensed consolidated financial
statements
NUGENE INTERNATIONAL, INC. AND SUBSIDIARIES
NOTES
TO UNAUDITED CONDENSED CONSOLIDATED FINANCIAL STATEMENTS
September
30, 2016
NOTE 1 -
NATURE OF BUSINESS AND BASIS OF PRESENTATION
NuGene
International, Inc. (also refer to as “we”, “us”, “our”, “the “Company” or
“NGI”) was incorporated in the State of Nevada on October 31, 2013. NuGene, Inc.
(our wholly owned subsidiary) was incorporated in the state of California in
December 2006. On January 20, 2015, we formed NuGene BioPharma, Inc.
(“BioPharma”) as a wholly owned California incorporated subsidiary of NGI. On
November 6, 2015, we formed The Aesthetic Group, Inc. (“TAG”) as a wholly owned
California incorporated subsidiary of NGI. Both BioPharma and TAG have had no
significant independent operations since inception.
Basis of
Presentation
The
accompanying unaudited condensed consolidated financial statements of the
Company have been prepared in accordance with accounting principles generally
accepted in the United States (“GAAP”) for interim financial information and
applicable regulations of the U.S. Securities and Exchange Commission. Certain
information and footnote disclosures normally included in financial statements
prepared in accordance with accounting principles generally accepted in the
United States have been omitted pursuant to such rules and regulations. In the
opinion of management, all adjustments (consisting only of normal recurring
adjustments) considered necessary for a fair statement of financial position and
results of operations have been included. Our operating results for the three
and nine months ended September 30, 2016 are not necessarily indicative of the
results that may be expected for the year ending December 31, 2016. The
accompanying unaudited condensed consolidated financial statements should be
read in conjunction with our audited consolidated financial statements for the
year ended December 31, 2015, which are included in our Annual Report on Form
10-K.
The
accompanying condensed consolidated financial statements reflect the accounts of
NuGene International, Inc. and its wholly owned subsidiaries. All significant
inter-company balances and transactions have been eliminated in
consolidation.
We
have incurred net losses through the date of these financial statements and have
yet to establish profitable operations. Our financial statements have been
presented on the basis that our business is a going concern, which contemplates
the realization of assets and the satisfaction of liabilities in the normal
course of business. We are subject to the risks and uncertainties associated
with a business with high short-term debt, limited commercial product revenues,
including limitations on our operating capital resources and uncertain demand
for our products. We have incurred recurring operating losses and negative
operating cash flows since inception and through September 30, 2016, and we
expect to continue to incur operating losses and negative operating cash flows
at least through the near future. Members of our Company’s management have been
required to advance our Company funding in order to partially meet our most
critical cash requirements including payroll along with those associated with
certain critical goods and services. In the process of managing these
situations, our management may have made representations implying their personal
guarantee of certain of our Company’s obligations, irrespective of whether such
guarantees are legally valid and enforceable.
As
a result of the aforementioned factors, management has concluded that there is
substantial doubt about our ability to continue as a going concern. Our
independent registered public accounting firm, in its report on our 2015
consolidated financial statements, raised substantial doubt about our ability to
continue as a going concern. Our unaudited condensed consolidated financial
statements as of and for the three and nine months ended September 30, 2016 do
not contain any adjustments for this uncertainty. In response to the Company’s
cash needs, we raised funding as described in the footnotes that follow. Any
additional amounts raised will be used for our future investing and operating
cash flow needs. However, there can be no assurance that we will be successful
in raising additional amounts of financing.
NOTE
2 - SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES
Significant
Accounting Policies
There
have been no material changes in the Company’s significant accounting policies
to those previously disclosed in the 2015 Annual Report.
Use
of Estimates
Conformity
with GAAP requires the use of estimates and judgments that affect the reported
amounts in the financial statements and accompanying notes. These estimates form
the basis for judgments we make about the carrying values of our assets and
liabilities that are not readily apparent from other sources. We base our
estimates and judgments on historical information and on various other
assumptions that we believe are reasonable under the circumstances. GAAP
requires us to make estimates and judgments in several areas, including but not
limited to, those related to revenue recognition, collectibility of accounts
receivable, contingent liabilities, fair value of financial instruments, fair
value of acquired intangible assets and goodwill, useful lives of intangible
assets and property and equipment, excess and obsolete inventory, deferred tax
asset valuation and income taxes. These estimates are based on management’s
knowledge about current events and expectations about actions we may undertake
in the future. Actual results could differ materially from those estimates.
NUGENE
INTERNATIONAL, INC. AND SUBSIDIARIES
NOTES
TO UNAUDITED CONDENSED CONSOLIDATED FINANCIAL STATEMENTS
September
30, 2016
Risks
and Uncertainties
The
Company operates in an industry that is subject to rapid change. The Company's
operations are subject to significant risk and uncertainties including
financial, operational, technological, regulatory and other risks, including the
potential of business failure.
Reclassifications
For
comparability, certain prior period amounts have been reclassified, where
appropriate, to conform to the current year’s financial statement presentation.
These reclassifications have no impact on net loss.
Inventories
The
Company’s skin and hair care inventories consist of raw materials and finished
goods and are valued at the lower of cost (first-in, first-out) or market. The
Company evaluates its inventory for excess quantities and obsolescence on a
regular basis. To determine if the cost of the Company's inventory should be
written down, current and anticipated demand, customer preferences and the age
of the merchandise are considered. For the three and nine months ended September
30, 2016 the Company recorded charges of approximately $437,000 to operations as
a result of excess and obsolete inventory related to the rebranding and
repackaging of products. For the three and nine months ended September 30, 2015,
the Company did not recognize any charges to operations associated with excess
and obsolete inventory.
Property
and Equipment
Property
and equipment consist of office furniture, computer equipment, laboratory
equipment, purchased software, website development and improvements made to our
leased offices and are stated at cost less accumulated depreciation.
Depreciation is calculated using the straight-line method over the three or five
year useful lives of the assets or the remaining term of the lease. When
property and equipment are retired or otherwise disposed of, the related cost
and accumulated depreciation are removed from the respective accounts, and any
gain or loss is included in operations. Expenditures for maintenance and repairs
are expensed as incurred.
Accounts
Payable and Accrued Expenses
Accounts
payable and accrued expenses are carried at amortized cost and represent
liabilities for goods and services provided to the Company prior to the end of
the financial year that are unpaid and arise when the Company becomes obliged to
make future payments in respect of the purchase of these goods and services.
Revenue
Recognition
In
accordance with Accounting Standards Codification (“ASC”) 605 -
Revenue
Recognition
, the Company recognizes revenue from product sales when the
product has been ordered by the customer, the selling price is fixed or
determinable, the product is shipped to the customer, title has transferred and
collectibility is reasonably assured.
Concentration
of Revenues
During
the three and nine months ended September 30, 2016, we derived 17% and 9%,
respectively, of our revenues from one customer. During the three months ended
September 30, 2015, we derived 48% and 19% of our revenues from two wholesale
distributors. During the nine months ended September 30, 2015, we derived 45%
and 21% of our revenues from two wholesale distributors. Our distributors
purchase products from us on a purchase order basis on standard terms. The
distributors are under no obligation to continue to purchase our products. The
loss of any of our major distributors, a material reduction in their purchases
or the cancellation of product orders or unexpected returns of unsold products
could significantly decrease our revenues and impede our future growth
prospects. We do not have long-term purchase commitments with our
distributors.
Cost
of Revenues
Cost
of revenues include all of the costs to manufacture the Company’s products. For
products manufactured in the Company’s own facilities, such costs include raw
materials and supplies, direct labor and factory overhead. For products
manufactured for the Company by third-party contractors, such cost represents
the amounts invoiced by the contractors.
NUGENE
INTERNATIONAL, INC. AND SUBSIDIARIES
NOTES
TO UNAUDITED CONDENSED CONSOLIDATED FINANCIAL STATEMENTS
September
30, 2016
Research
and Development
Internal
research and development costs are expensed as incurred. Third-party research
and development costs are expensed when the contracted work has been performed.
Research and development consists of consulting fees, direct labor and raw
materials associated with the development of new products to be commercialized
by our Company. Research and development expenses totaled approximately $104,000
and $136,000 for the nine months ended September 30, 2016 and 2015,
respectively. Research and development expenses totaled approximately $74,000
and $98,000 for the three months ended Septembers 30, 2016 and 2015,
respectively.
Allowance
for Doubtful Accounts
Our
Company maintains allowances for doubtful accounts for estimated losses
resulting from the inability of our customers to make required payments. Account
balances are charged off against the allowance after all means of collection
have been exhausted and the potential for recovery is considered remote. Our
allowance for doubtful accounts was approximately $533,000 and $177,000 as of
September 30, 2016 and December 31, 2015, respectively. Bad debt expense totaled
approximately $356,000 and $16,000 for the nine months ended September 30, 2016
and 2015, respectively. As of September 30, 2016, we have approximately $328,000
outstanding in gross accounts receivable from one customer. This balance
represents 60.0% of gross accounts receivable and has been fully reserved as a
doubtful account as of September 30, 2016.
Convertible
Financial Instruments
The
Company bifurcates conversion options from their host instruments and accounts
for them as free standing derivative financial instruments if certain criteria
are met. The criteria include circumstances in which (a) the economic
characteristics and risks of the embedded derivative instrument are not clearly
and closely related to the economic characteristics and risks of the host
contract, (b) the hybrid instrument that embodies both the embedded derivative
instrument and the host contract is not re-measured at fair value under
otherwise applicable generally accepted accounting principles with changes in
fair value reported in earnings as they occur and (c) a separate instrument with
the same terms as the embedded derivative instrument would be considered a
derivative instrument. An exception to this rule is when the host instrument is
deemed to be conventional, as that term is described under applicable GAAP.
When
the Company has determined that the embedded conversion options should not be
bifurcated from their host instruments, discounts are recorded for the intrinsic
value of conversion options embedded in the instruments based upon the
differences between the fair value of the underlying common stock at the
commitment date of the transaction and the effective conversion price embedded
in the instrument.
Debt
Discounts
-
Debt discounts under these arrangements are amortized to
interest expense using the interest method over the earlier of the term of the
related debt or their earliest date of redemption.
Loss on
Issuance - The conversion features of the notes were bifurcated from the host
instrument as its conversion terms were not indexed to the Company’s own stock.
In addition, the warrants associated with the debt instruments were also treated
as a free standing derivative liability. The total fair value of the embedded
conversion feature and the warrants exceeded the net proceeds received and
resulted in a loss on issuance of $1.2 million.
Common
Stock Purchase Warrants and Derivative Financial Instruments - Common stock
purchase warrants and other derivative financial instruments are classified as
equity if the contracts (1) require physical settlement or net-share settlement
or (2) give the Company a choice of net-cash settlement or settlement in its own
shares (physical settlement or net-share settlement). Contracts which (1)
require net-cash settlement (including a requirement to net cash settle the
contract if an event occurs and if that event is outside the control of the
Company), (2) give the counterparty a choice of net-cash settlement or
settlement in shares (physical settlement or net-share settlement), or (3) that
contain reset provisions that do not qualify for the scope exception are
classified as liabilities. The Company assesses classification of its common
stock purchase warrants and other derivatives at each reporting date to
determine whether a change in classification between equity and liabilities is
required.
Beneficial
conversion feature –
The issuance of the convertible debt generated a
beneficial conversion feature (“BCF”), which arises when a debt or equity
security is issued with an embedded conversion option that is beneficial to the
investor or in the money at inception because the conversion option has an
effective strike price that is less than the market price of the underlying
stock at the commitment date. The Company recognized the BCF by allocating the
intrinsic value of the conversion option, which is the number of shares of
common stock available upon conversion multiplied by the difference between the
effective conversion price per share and the fair value of common stock per
share on the commitment date, resulting in a discount on the convertible debt
(recorded as a component of additional paid-in capital).
Share
Based Payments
The
Company recognizes compensation expense for all equity-based payments in
accordance with ASC 718 -
Share-based payments
. Under ASC 718’s fair
value recognition provisions, the Company recognizes equity-based compensation
net of an estimated forfeiture rate and recognizes compensation cost only for
those shares expected to vest over the requisite service period of the
award.
NUGENE
INTERNATIONAL, INC. AND SUBSIDIARIES
NOTES
TO UNAUDITED CONDENSED CONSOLIDATED FINANCIAL STATEMENTS
September
30, 2016
Share-based
payments to employees, including grants of employee stock options, are
recognized as compensation expense in the accompanying condensed consolidated
statements of operations based on the fair values of the related payments. Such
expense is recognized over the period during which an employee is required to
provide services in exchange for the award, known as the requisite service
period (usually the vesting period).
The
Company accounts for share-based payments granted to non-employees in accordance
with ASC 505 -
Equity Based Payments to Non-Employees
. The Company
determines the fair value of the stock-based payment as either the fair value of
the consideration received or the fair value of the equity instruments issued,
whichever is more reliably measurable. If the fair value of the equity
instruments issued is used, it is measured using the stock price and other
measurement assumptions as of the earlier of either (1) the date at which a
commitment for performance by the counterparty to earn the equity instruments is
reached, or (2) the date at which the counterparty’s performance is
complete.
Net
Loss per Share of Common Stock
The
Company computes net loss per share in accordance with ASC 260 -
Earnings Per
Shar
e. ASC 260 requires presentation of both basic and diluted net loss per
share on the face of the statements of operations. Basic net loss per share is
calculated by dividing the loss attributable to common shareholders of the
Company by the weighted average number of common shares outstanding during the
period. Diluted net loss per share is determined by adjusting the loss
attributable to common shareholders and the weighted average number of common
shares outstanding for the effects of all potential dilutive common shares, such
as stock issuable pursuant to the exercise of stock warrants or the conversion
of preferred stock into common stock.
Common
stock equivalents totaling, 18,217,825 and 4,767,720 as of September 30, 2016
and 2015, respectively, were not included in the computation of diluted earnings
per share on the condensed consolidated statements of operations because the
Company reported a net loss during the three and nine months ended September 30,
2016 and 2015 and therefore the effect would be anti-dilutive.
Recent
Accounting Standards Updates
In
January 2016, the Financial Accounting Standards Board (“FASB”) issued
Accounting Standards Update (“ASU”) No. 2016-01,
Recognition and Measurement
of Financial Assets and Financial Liabilities
. ASU No. 2016-01 requires
equity investments to be measured at fair value with changes in fair value
recognized in net income; simplifies the impairment assessment of equity
investments without readily determinable fair values by requiring a qualitative
assessment to identify impairment; eliminates the requirement for public
business entities to disclose the method(s) and significant assumptions used to
estimate the fair value that is required to be disclosed for financial
instruments measured at amortized cost on the balance sheet; requires public
business entities to use the exit price notion when measuring the fair value of
financial instruments for disclosure purposes; requires an entity to present
separately in other comprehensive income the portion of the total change in the
fair value of a liability resulting from a change in the instrument-specific
credit risk when the entity has elected to measure the liability at fair value
in accordance with the fair value option for financial instruments; requires
separate presentation of financial assets and financial liabilities by
measurement category and form of financial assets on the balance sheet or the
accompanying notes to the financial statements and clarifies that an entity
should evaluate the need for a valuation allowance on a deferred tax asset
related to available-for-sale securities in combination with the entity’s other
deferred tax assets. ASU No. 2016-01 is effective for financial statements
issued for fiscal years beginning after December 15, 2017, and interim periods
within those fiscal years. The Company is currently evaluating the impact that
ASU No. 2016-01 will have on its financial statements and related
disclosures.
In
February 2016, the FASB issued ASU No. 2016-02,
Leases
(Topic
842).
ASU No. 2016-02 requires an entity to recognize right-of-use assets
and lease liabilities on its balance sheet and disclose key information about
leasing arrangements. Lessees and lessors are required to disclose qualitative
and quantitative information about leasing arrangements to enable a user of the
financial statements to assess the amount, timing and uncertainty of cash flows
arising from leases. ASU No. 2016-02 is effective for fiscal years, and interim
periods within those years, beginning after December 15, 2018. Early
adoption is permitted. The Company currently in the process of evaluating the
impact of adoption of ASU No. 2016-02 on the condensed consolidated financial
statements and related disclosures.
In
March 2016, the FASB issued ASU No. 2016-08,
Revenue from Contracts with
Customers
(Topic 606):
Principal versus Agent Considerations
.
The purpose of ASU No. 2016-08 is to clarify the implementation of guidance on
principal versus agent considerations. The amendments in ASU No. 2016-08 are
effective for interim and annual reporting periods beginning after December 15,
2017. The Company is currently assessing the impact of ASU No. 2016-08 on the
condensed consolidated financial statements and related disclosures.
NUGENE
INTERNATIONAL, INC. AND SUBSIDIARIES
NOTES
TO UNAUDITED CONDENSED CONSOLIDATED FINANCIAL STATEMENTS
September
30, 2016
In
March 2016, the FASB issued ASU No. 2016-09,
Compensation - Stock
Compensation (Topic 718): Improvements to Employee Share-Based Payment
Accounting
. The amendment is to simplify several aspects of the accounting
for share-based payment transactions including the income tax consequences,
classification of awards as either equity or liabilities, and classification on
the statement of cash flows. The amendments in ASU No. 2016-09 are effective for
interim and annual reporting periods beginning after December 15, 2016. The
Company is currently assessing the impact of ASU No. 2016-09 on the condensed
consolidated financial statements and related disclosures.
In April 2016, the FASB issued ASU No.
2016-10,
Revenue from Contracts with Customer
(“ASU No. 2016-10”)
. The new guidance is an update to ASC No. 606 and
provides clarity on identifying performance obligations and licensing
implementation. For public companies, ASU No. 2016-10 is effective for annual
periods, including interim periods within those annual periods, beginning after
December 15, 2016. The Company does not expect ASU No. 2016-10 to have a
material effect on the condensed consolidated financial statements and related
disclosures.
In August 2016, the FASB issued
ASU 2016-15,
Statement of Cash Flows - Classification of Certain
Cash Receipts and Cash Payments,
which addresses eight specific cash
flow issues with the objective of reducing the existing diversity in practice in
how certain cash receipts and cash payments are presented and classified in the
statement of cash flows. The standard is effective for fiscal years beginning
after December 15, 2017, including interim periods within those fiscal years.
Early adoption is permitted, including adoption in an interim period. The
Company is currently in the process of evaluating the impact of this new
pronouncement on its condensed consolidated statements of cash flows.
Other
accounting standards that have been issued or proposed by FASB that do not
require adoption until a future date are not expected to have a material impact
on the consolidated financial statements upon adoption. The Company does not
discuss recent pronouncements that are not anticipated to have an impact on or
are unrelated to its financial condition, results of operations, cash flows or
disclosures.
NOTE
3 - INVENTORIES
Inventories
consist of the following:
|
|
|
September
30,
2016
|
|
|
December 31,
2015
|
|
|
Raw materials
|
|
$
|
14,067
|
|
|
$
|
72,287
|
|
|
Work in process
|
|
|
-
|
|
|
|
-
|
|
|
Finished goods
|
|
|
10,236
|
|
|
|
105,205
|
|
|
Total Inventories
|
|
$
|
24,303
|
|
|
$
|
177,492
|
|
NOTE
4 - LICENSE AGREEMENT
In
November 2014, we entered into a License Agreement with kathy ireland
Worldwide
®
("kiWW
®
") under which we licensed the right to
utilize the trademarks and rights to the name, likeness and visual
representations of Kathy Ireland (“KI”) in connection with our cosmeceutical
line of products. The initial term of the license is for eight years and it may
be renewed at the option of our Company for up to an additional four years. In
accordance with the License Agreement, we will pay 5% of the net sales for all
licensed products sold and collected under the licensed marks or a minimum
guaranteed royalty of $100,000 in year one, which includes the period from
approximately November 4, 2014 through December 31, 2015 (“Contract Year 1”) of
the License Agreement. The minimum guaranteed royalty increases $50,000 each
year in years two through eight of the License Agreement. We recognized $139,000
and $68,000 in royalty fees during the nine months ended September 30, 2016 and
2015, respectively. We recognized $37,500 and $23,000 in royalty fees during the
three months ended September 20, 2016 and 2015, respectively.
Additionally,
we are obligated to pay an annual Brand Participation Fee to kiWW
®
which provides for general advertising, good will and promotion of the KI brand.
NuGene prepaid kiWW $350,000 effective upon execution of the License as a Brand
Participation fee for Contract Year 1. The Brand Participation Fee for Contract
Years 2 through 8 is $50,000 annually (Year 2 having been completely recognized
in the three months ended March 31, 2016) with an additional 1% of the total
gross sales of Licensed Products of the prior year beginning in Contract Year 4.
Our Company is currently in arrears with respect to the payment of the Year 2
fee that was due in November 2015. Such payment arrears could be construed as an
event of default under the License Agreement and could be a cause for its
annulment. As of September 30, 2016 and December 31, 2015, $50,000 and $222,000
due under this agreement, respectively, is included in current liabilities in
the condensed consolidated balance sheets.
NUGENE
INTERNATIONAL, INC. AND SUBSIDIARIES
NOTES
TO UNAUDITED CONDENSED CONSOLIDATED FINANCIAL STATEMENTS
September
30, 2016
NOTE
5 - PROPERTY AND EQUIPMENT
Property
and equipment, net consist of the following:
|
|
|
September 30,
2016
|
|
|
December 31,
2015
|
|
|
Software / website development
|
|
$
|
8,219
|
|
|
$
|
8,219
|
|
|
Equipment
|
|
|
141,125
|
|
|
|
136,585
|
|
|
Leasehold
improvements
|
|
|
104,166
|
|
|
|
104,166
|
|
|
Property and equipment, gross
|
|
|
253,510
|
|
|
|
248,970
|
|
|
Accumulated
depreciation
|
|
|
(81,552
|
)
|
|
|
(40,620
|
)
|
|
Property and
equipment, net
|
|
$
|
171,958
|
|
|
$
|
208,350
|
|
Depreciation
expense for the three months ended September 30, 2016 and 2015 was approximately
$14,000 and $12,000, respectively.
Depreciation
expense for the nine months ended September 30, 2016 and 2015 was approximately
$41,000 and $26,000, respectively.
NOTE
6 - PROMISSORY NOTES PAYABLE AND ADVANCES
Borrowings
under notes payable and advances as of September 30, 2016 and December 31, 2015
are summarized as follows:
|
|
|
Company Proceeds
|
|
|
Carrying Value as of
September 30, 2016
|
|
|
Carrying Value as of
December 31, 2015
|
|
|
Accrued Interest
as of September 30,
2016
|
|
|
Accrued Interest
as of December 31,
2015
|
|
|
Principal Value
at Maturity
|
|
|
15%
Note
|
|
$
|
500,000
|
|
|
$
|
-
|
|
|
$
|
500,000
|
|
|
$
|
-
|
|
|
$
|
31,479
|
|
|
$
|
-
|
|
|
10% Note
|
|
|
50,000
|
|
|
|
50,000
|
|
|
|
50,000
|
|
|
|
4,466
|
|
|
|
1,383
|
|
|
|
50,000
|
|
|
10% Note
(2015)
|
|
|
87,500
|
|
|
|
108,055
|
|
|
|
62,949
|
|
|
|
8,860
|
|
|
|
548
|
|
|
|
110,000
|
|
|
10% Note (2015 Advances)
|
|
|
322,500
|
|
|
|
361,110
|
|
|
|
229,555
|
|
|
|
28,750
|
|
|
|
1,642
|
|
|
|
361,110
|
|
|
10% Note (2016 Advances)
|
|
|
1,065,000
|
|
|
|
332,795
|
|
|
|
-
|
|
|
|
16,256
|
|
|
|
-
|
|
|
|
1,080,556
|
|
|
8% Convertible Note
|
|
|
245,000
|
|
|
|
207,765
|
|
|
|
-
|
|
|
|
11,090
|
|
|
|
-
|
|
|
|
275,000
|
|
|
15% Convertible Note
|
|
|
35,000
|
|
|
|
610,000
|
|
|
|
-
|
|
|
|
45,002
|
|
|
|
-
|
|
|
|
610,000
|
|
|
|
|
$
|
2,305,000
|
|
|
$
|
1,669,725
|
|
|
$
|
842,504
|
|
|
$
|
114,425
|
|
|
$
|
35,052
|
|
|
$
|
2,486,666
|
|
On
April 4, 2016, we issued a note payable to Canyon Assets Holdings, Inc.
(“Lender”) dated March 28, 2016 in the principal amount of $575,000. The note
was issued to the Lender in consideration for the Lender’s having satisfied on
the Company’s behalf an outstanding note payable (the “CAH Note”) of the Company
(the “JTS Note”). The JTS Note issued September 25, 2015 was repaid in full
($500,000 face value and accrued interest of $75,000) by the Lender with the
payment of $575,000 in cash. The terms of the CAH Note call for an interest rate
of 15%, due one year from the date of the CAH Note. However, all accrued and
unpaid interest and all other amounts payable under the CAH Note are due to the
Lender within ten (10) business days after the closing by the Company of an
equity or convertible debt financing in one or more series of transactions, with
aggregate gross proceeds of at least $1 million.
On
April 4, 2016, we issued a $275,000 face value note payable and 50,000 shares of
our common stock to Gemini Master Fund, LTD (“Gemini”) pursuant to a Security
Purchase Agreement dated March 30, 2016 (the date the funds were received by our
Company). Under the terms of the related note payable (the “Gemini Note”), the
Company received $245,000, net of costs and original issue discount. Other
significant terms of the Gemini Note include:
|
|
·
|
A maturity date of December 31, 2016 in
the absence of events triggering mandatory early repayment (as summarized
below);
|
|
|
·
|
Interest accrues at the rate of 8% on the
$275,000 face value (18% in the event of an event of default as defined in
the Gemini Note);
|
|
|
·
|
The Gemini Note is convertible in part
(subject to a $10,000 minimum) at the option of Gemini into shares of the
Company’s common stock at the rate of $0.70 per share (subject to
adjustment summarized below);
|
|
|
·
|
The Company has the option to prepay the
Gemini Note;
|
|
|
·
|
All prepayments of the Gemini Note,
whether effected at the option of the Company or subject to mandatory
early repayment (as summarized herein), require the Company to repay
Gemini 112% of the outstanding principal and all outstanding accrued
interest through the date of prepayment;
|
|
|
·
|
All principal and interest outstanding
under the Gemini Note are required to be immediately repaid should the
Company complete a financing or series of financings totaling $1.5 million
or more;
|
|
|
·
|
The Conversion price of the Gemini Note is
adjusted for the following: 1) loss of Company DTC eligibility -
conversion price adjusts to $0.25 per share; 2) stock dividends and splits
- as described in the Gemini Note; 3) a rights offering below the market
price (as defined) - as described in the Gemini Note; 4) fundamental
transactions (as defined) - as described in the Gemini Note; 5) subsequent
equity sales below $0.70 per share - as more particularly detailed and
described in the Gemini Note; and
|
|
|
·
|
The Gemini Note is convertible into
392,857 shares of common stock.
|
NUGENE
INTERNATIONAL, INC. AND SUBSIDIARIES
NOTES
TO UNAUDITED CONDENSED CONSOLIDATED FINANCIAL STATEMENTS
September
30, 2016
The
embedded conversion feature of the Gemini Note was bifurcated and valued at
$106,278. Management used a Monte Carlo valuation model to estimate the
fair value of the embedded conversion option at issuance of the convertible note
issued, with the following weighted average key inputs:
|
|
|
At issuance
|
|
|
Stock price
|
|
$
|
0.68
|
|
|
Term (years)
|
|
|
0.76
|
|
|
Volatility
|
|
|
98.9
|
%
|
|
Risk-free rate of interest
|
|
|
0.5
|
%
|
|
Dividend yield
|
|
|
0.0
|
%
|
The
embedded conversion feature is separately measured at fair value, with changes
in fair value recognized in current operations. The embedded
conversion has been reduced by $9,098 during the nine months ended September 30,
2016.
In
connection with the issuance of the Gemini Note, we recorded additional debt
discounts related to the following:
|
|
·
|
Issuance of 50,000 shares of our common
stock resulting in the relative common stock fair value of approximately
$27,000;
|
|
|
·
|
Recording of a beneficial conversion
feature of approximately $44,000; and
|
|
|
·
|
Original issuance discount of
$25,000.
|
The
debt discounts are amortized to interest expense using the effective
interest method over the term of the notes. During the three and nine months
ended September 30, 2016, the Company recognized interest expense of $67,235 and
$134,470, respectively, from the amortization of the debt discount, resulting in
a net Gemini Note balance of $207,765.
During
January through June 2016, three individuals (the “Lenders”) advanced a total of
$140,000 to the Company. The borrowings were not accompanied by documentation of
the nature of the borrowings. However, there were general discussions as to the
repayment terms and the interest expected of 10% per annum to be paid to the
Lenders in connection with the borrowings. Accordingly, we have accounted for
the borrowings based on estimates of their still undocumented final terms.
On
July 19, 2016, we issued a 60 day 15% promissory note payable to a purchaser for
cash proceeds totaling $35,000. In the event that we secure any future financing
with aggregate gross proceeds of at least $500,000 while the promissory note is
outstanding, the promissory note and all accrued interest therefrom will be
immediately due and payable within ten business days of the closing of such
financing.
During
the three months ended September 30, 2016, certain investors advanced a total of
$925,000 (the “Advances”) to the Company with a right to convert at no greater
than $0.40 per share. In connection with the Advances,
the Company also issued warrants to purchase
462,500 shares of our common stock at an exercise price $0.60 per share with a 5
year term. The relative fair value of the warrants compared to the Advances was
$203,467, which was recorded as a component of stockholders’ deficit with the
offset recorded as a discount on the Advances. The fair value of the warrants
was determined using the Black-Scholes Model. The Company then computed the
effective conversion price of the Advances on the issuance date, noting that the
Advances gave rise to a
BCF of $721,533. The sum of the relative fair
value of the warrants and the BCF was recorded as a debt discount to be
amortized over the term of the advances.
The
debt discounts are amortized to interest expense using the effective
interest method over the term of the Advances. During the three and nine months
ended September 30, 2016, the Company recognized interest expense of $137,022,
from the amortization of the debt discount, resulting in the Advances balance of
$177,240.
The Advances contain an embedded conversion
feature that the Company has determined is a derivative requiring bifurcation in
accordance with the provisions of ASC 815. The embedded conversion feature of
the Notes was valued at inception using the Monte Carlo simulation (see Note 7)
at $1,212,384 and was recorded as a loss on the condensed consolidated
statements of operations. As of September 30, 2016, the derivative was
remeasured to $964,356 and the difference was recorded as a change in fair value
of derivative liabilities on the statement of operations.
Management
used a Monte Carlo valuation model to estimate the fair value of the
embedded conversion option at issuance of the Advances, with the following
weighted average key inputs:
|
|
|
At issuance
|
|
|
Stock price
|
|
$
|
0.74
|
|
|
Term (years)
|
|
|
0.75
|
|
|
Volatility
|
|
|
116
|
%
|
|
Risk-free rate of interest
|
|
|
0.5
|
%
|
|
Dividend yield
|
|
|
0.0
|
%
|
NOTE
7 - FAIR VALUE MEASUREMENTS
The
Company’s financial liabilities that are measured at fair value on a recurring
basis as of September 30, 2016 by level within the fair value hierarchy, are as
follows:
|
|
|
Quoted
prices in active
markets
|
|
|
Significant
other
observable
inputs
|
|
|
Significant
unobservable
inputs
|
|
|
|
|
(Level 1)
|
|
|
(Level 2)
|
|
|
(Level 3)
|
|
|
Embedded conversion option
|
|
$
|
-
|
|
|
$
|
-
|
|
|
$
|
1,061,536
|
|
There
were no transfers between Level 1, 2 or 3 during the nine months ended September
30, 2016. As of September 30, 2016, the embedded conversion option has an
aggregate fair value of $1,061,536.
NUGENE
INTERNATIONAL, INC. AND SUBSIDIARIES
NOTES
TO UNAUDITED CONDENSED CONSOLIDATED FINANCIAL STATEMENTS
September
30, 2016
The
following table presents additional information about Level 3 liabilities
measured at fair value. Both observable and unobservable inputs may be used to
determine the fair value of positions that the Company has classified within the
Level 3 category. As a result, the unrealized gains and losses for liabilities
within the Level 3 category may include changes in fair value that were
attributable to both observable (e.g., changes in market interest rates) and
unobservable (e.g., changes in unobservable long-dated volatilities) inputs.
Changes
in Level 3 liabilities measured at fair value for the nine months ended
September, 2016 were as follows:
|
|
|
Balance
as of
December
31,
|
|
|
|
|
|
Change
in Fair
Market
|
|
|
Balance
as of
September
30,
|
|
|
|
|
2015
|
|
|
Issuance
|
|
|
Value
|
|
|
2016
|
|
|
Embedded conversion option
|
|
$
|
-
|
|
|
$
|
1,318,662
|
|
|
$
|
(257,126
|
)
|
|
$
|
1,061,536
|
|
The
Company’s derivative liabilities are measured at fair value using the Monte
Carlo simulation valuation methodology. A summary of the weighted average (in
aggregate) significant unobservable inputs (Level 3 inputs) used in measuring
the Company’s derivative liabilities that are categorized within Level 3 of the
fair value hierarchy as of September 30, 2016 is as follows:
|
|
|
As of September 30, 2016
|
|
|
Stock price
|
|
$
|
0.64
|
|
|
Contractual life (years)
|
|
|
0.6
|
|
|
Volatility
|
|
|
118
|
%
|
|
Risk-free rate of interest
|
|
|
0.5
|
%
|
|
Dividend yield
|
|
|
0.0
|
%
|
NOTE
8 - STOCKHOLDERS’ DEFICIT
Common
Stock
The
Company has Advisory Agreements with members of its Advisory Board. The terms of
the individual Advisory Agreements vary and provide for up to 50,000 initial
sign-on shares vesting for a maximum of an 18-month period, and up to 50,000
shares of common stock per annum issued on the anniversary of the effective date
of the agreement. Expenses for the issuance of common stock for services related
to these share issuances is recognized over the service period in which the
shares are earned or over the respective vesting period, as applicable, and is
calculated based on the average closing price per share of our common stock,
during the respective quarter, as quoted on the OTC Marketplace. The Advisory
Board earned 297,282 and 188,950 shares of common stock through September 30,
2016 and December 31, 2015, respectively. No shares of common stock earned by
the Advisory Board have been issued as of September 30, 2016. Selling, general
and administrative expenses (“SGA”) recognized in connection with the Advisory
Agreements totaled approximately $76,000 and $467,000 for the nine months ended
September 30, 2016 and 2015, respectively, and approximately $13,000 and
$170,000 for the three months ended September 30, 2016 and 2015,
respectively.
In
June 2016, Theodore Schwarz executed an Offer Letter whereby Mr. Schwarz agreed
to become a member of the Company’s Board of Directors (the “Board”). Under the
Offer Letter the Company has agreed to pay Mr. Schwarz $2,500 for each Board
meeting attended. Additionally, Mr. Schwarz may be issued 500,000 shares of the
Company’s common stock in the future that may vest at the rate of 166,666 shares
per year, over a three-year period provided Mr. Schwarz continues to serve on
the Board.
Restricted
Stock Awards
In
November 2015, the Company entered into a consulting agreement with a third
party for consulting services over six months. The Company agreed to issue the
consultant 90,000 shares of the Company’s restricted stock, which were issued
over six months. The Company issued 30,000 shares of restricted stock in
February 2016, and the remaining 60,000 shares were issued in June 2016,
resulting in $378,000 of stock based compensation expense for the nine months
ended September 2016.
NUGENE
INTERNATIONAL, INC. AND SUBSIDIARIES
NOTES
TO UNAUDITED CONDENSED CONSOLIDATED FINANCIAL STATEMENTS
September
30, 2016
On
March 17, 2016, we entered into a consulting agreement for services over one
year. In connection with the agreement, we granted the consultant 200,000 shares
of our fully vested common stock as consideration for the consultant’s services.
The Company recorded prepaid consulting of approximately $128,000 in the first
quarter of 2016 and recognized stock based compensation totaling approximately
$91,000 and $128,000 for the three and nine months ended September 30, 2016,
respectively.
In
April, 2016, the Company entered into a consulting agreement with a third party
for consulting services over a year. The 200,000 shares of restricted stock
award vested on April 25, 2016. The grant date fair value of restricted stock
award was $118,000 and was immediately recognized as stock based
compensation.
On
September 22, 2016, the Company entered into new compensation agreements and
arrangements with two of its directors. As part of the agreements, our Chairman
was issued 2,000,000 shares of our fully vested common stock. The grant date
fair value of stock award was $1,420,000 and was immediately recognized as stock
based compensation.
Restricted
Stock Units
In
connection with the agreements the Company entered into with two of its
directors, each director was granted restricted stock units for 2,000,000 shares
of the Company’s common stock (“
RSUs
”). The RSUs vest at the rate of
33.33% each year anniversary of the issuance date (September 22, 2016). There is
also 100% vesting upon a change in control of the Company.
The
grant date fair value of the RSUs were $2,840,000. The Company recognized stock
based compensation of $20,749 for the three and nine months ended September 30,
2016.
Common
Stock Options
On
August 14, 2015, we granted two employees with an option to each purchase up to
1,000,000 shares of our common stock. On May 2, 2016, we reduced the exercise
price from $1.50 per share to $0.54 per share for these options. All other terms
of the previous option agreements remained unchanged. We remeasured the
options on the modification date using the Black-Scholes Model. The incremental
fair value of vested options was approximately $71,000 and was recognized
immediately. The sum of the incremental compensation cost and the remaining
unrecognized compensation cost for the unvested option shares
was approximately $1.7 million on the modification date and will be
amortized ratably over the remaining vesting
period. We recognized approximately $320,000 and $997,000 of
Personnel expense as stock based compensation in connection with the vesting of
the options during the three and nine months ended September 30, 2016,
respectively.
On
July 18, 2016 we granted our Chief Executive Office a stock option to acquire
2,352,619 shares of our common stock, which represented approximately 5% of our
fully-diluted issued and outstanding shares on the date of grant. The exercise
price of the option was $0.61 per share, which was the closing price for the
Company stock at the date of issuance, and will vest ratably monthly over 3
years, with 100% vesting upon a change in control of the Company. The aggregate
grant date fair value of these options was calculated by using the Black-Scholes
Model and was approximately $1,127,000. In accordance with the employment
agreement, the Chief Executive Officer will be issued stock options to acquire
5% of our fully-diluted issued and outstanding shares through our next
significant financing transaction or series of significant financing
transactions.
A
summary of the stock options activity for the nine months ended September 30,
2016 is as follows:
|
|
|
Number of Shares
|
|
|
Weighted Average
Exercise Price
|
|
|
Weighted Average
Contractual
Life
|
|
|
Outstanding as of December 31, 2015
|
|
|
2,000,000
|
|
|
$
|
0.54
|
|
|
|
3.87
|
|
|
Granted
|
|
|
2,352,619
|
|
|
|
0.61
|
|
|
|
9.80
|
|
|
Outstanding as of September 30, 2016
|
|
|
4,352,619
|
|
|
|
0.58
|
|
|
|
7.07
|
|
|
Options vested
|
|
|
1,214,034
|
|
|
$
|
0.55
|
|
|
|
4.51
|
|
Stock-based
compensation associated with stock options was approximately $383,000 and
$159,000 for the three months ended September 30, 2016 and 2015, respectively
and was approximately $1,060,000 and $159,000 for the nine months ended
September 30, 2016 and 2015, respectively. Unamortized stock-based
compensation expense amounted to approximately $1,624,000 as of September 30,
2016, and will be amortized over 3 years.
NUGENE
INTERNATIONAL, INC. AND SUBSIDIARIES
NOTES
TO UNAUDITED CONDENSED CONSOLIDATED FINANCIAL STATEMENTS
September
30, 2016
Common
Stock Warrants
In
connection with the agreements the Company entered into with two of its
directors, we issued a fully vested warrant with a ten year term to each
director to purchase shares of common stock equal to the greater of 1,000,000
shares or 2.5% of the issued and outstanding common shares of the Company during
the first 24 months immediately following the issuance. On the date of issuance,
each director was issued a warrant to purchase 1,008,367 shares of common stock
at an exercise price of $0.71 per share, which was the closing price for the
Company stock at the date of issuance. The aggregate grant date fair value of
these warrants was calculated by using the Black-Scholes Model and was
approximately $1,291,000. The entire far value was recorded as stock based
compensation expense as of September 30, 2016.
In
connection with the Advances (Note 6),
the
Company issued warrants to purchase 462,500 shares of our common stock at an
exercise price $0.60 per share with a 5 year term. The relative fair value of
the warrants compared to the Advances was $203,467, which was recorded as a
component of stockholders’ deficit with the offset recorded as a discount on the
Advances. The fair value of the warrants was determined using the Black-Scholes
Model.
A
summary of the outstanding warrants activity and related information as of
September 30, 2016 is as follows:
|
Exercise
|
|
|
Warrants
|
|
|
Remaining
|
|
|
Warrants
|
|
|
Price
|
|
|
Outstanding
|
|
|
Life (Years)
|
|
|
Exercisable
|
|
|
$
|
0.001
|
|
|
|
1,350,000
|
|
|
|
1.19
|
|
|
|
900,000
|
|
|
$
|
0.60
|
|
|
|
462,500
|
|
|
|
4.90
|
|
|
|
462,500
|
|
|
$
|
0.71
|
|
|
|
2,016,734
|
|
|
|
9.98
|
|
|
|
2,016,734
|
|
|
$
|
1.50
|
|
|
|
50,000
|
|
|
|
1.87
|
|
|
|
50,000
|
|
|
$
|
2.00
|
|
|
|
300,000
|
|
|
|
3.87
|
|
|
|
300,000
|
|
|
$
|
2.50
|
|
|
|
500,000
|
|
|
|
4.24
|
|
|
|
500,000
|
|
|
|
|
|
|
|
4,679,234
|
|
|
|
|
|
|
|
4,229,234
|
|
NOTE
9 - RELATED PARTY TRANSACTIONS
The
following individuals and entities have been identified as related parties based
on their affiliation with our Chairman of the Board:
|
Ali Kharazmi
|
|
Chairman of the Board and greater than 10%
shareholder
|
|
|
|
|
|
Mohammad Saeed Kharazmi
|
|
Board Member and greater than 10% shareholder
|
|
|
|
|
|
Genetics Institute of Anti-Aging
|
|
Company with common ownership and management
|
|
Applied M.A.K. Enterprises, Inc. (“MAK”)
|
|
Company with common ownership and management
|
|
Advanced Surgical Partners, LLC (“AdvSP”)
|
|
Company with common ownership and management
|
|
Center for Weight Management & Plastic Surgery (“CWM”)
|
|
Company with common ownership and management
|
|
Center for Regenerative Science, LLC
|
|
Company with common ownership and management
|
The
following amounts were owed to related parties, affiliated with the CEO and
Chairman of the Board, at the dates indicated:
|
|
|
September
30,
2016
|
|
|
AdvSP
|
|
$
|
226,188
|
|
|
CWM
|
|
|
34,000
|
|
|
Ali Kharazmi
|
|
|
91,175
|
|
|
Applied M.A.K.
|
|
|
-
|
|
|
Mohammad Saeed
Kharazmi
|
|
|
30,698
|
|
|
Accounts payable -
related parties
|
|
$
|
382,061
|
|
The
amount owed to AdvSP relates to legal and administrative services provided by
AdvSP employees to the Company. The Company temporarily advanced $95,000 to
AdvSP prior to December 31, 2015 and was repaid in full prior to January 5,
2016. The amount owed to CWM relates to medical procedures provided to NuGene
consultants as compensation for advertising and marketing services provided to
NuGene. The amount owed to Ali and Saeed Kharazmi and all amounts outstanding
represent advances that bore no interest and were due on demand or expense
reimbursements incurred in the ordinary course of business.
Subsequent
to September 30, 2016, the amounts owed to the related parties totaling $382,061
were sold to two unrelated investors (See Note 11).
Subsequent
to September 30, 2016, the amounts owed to the related parties totaling $382,061
were sold to two unrelated investors. The Company issued a note to each investor
in the amount of $191,030. The notes have an interest rate of 5% per annum and
mature on the earlier of: (i) May 7, 2017; or (ii) the date the Company raises a
total of $2 million, subsequent to the issuance of these notes. The notes have a
conversion feature under which the note holder can convert at any time at a
conversion price of $0.40 per share.
NUGENE
INTERNATIONAL, INC. AND SUBSIDIARIES
NOTES
TO UNAUDITED CONDENSED CONSOLIDATED FINANCIAL STATEMENTS
September
30, 2016
NOTE 10 -
COMMITMENTS AND CONTINGENCIES
Commitments
We
occupy our sole corporate facilities at 17912 Cowan, Suite A, Irvine,
California, 92614 from AdvSP, an affiliate of our Company, for approximately
$16,637 per month (including common area maintenance), consistent with the
amount that is charged to AdvSP by the property owner. On February 5, 2015,
AdvSP entered into a new five-year lease for the property with the owner
beginning July 1, 2015 and subsequently amended to begin June 1, 2015. The lease
was subsequently amended to increase the square footage under lease beginning in
January 2016. The lease includes annual increases in the monthly lease payments
of approximately 3% each year.
At
September 30, 2016, aggregate future minimum payments under the lease, including
common area maintenance costs, are as follows:
|
2016
|
|
$
|
49,027
|
|
|
2017
|
|
|
198,935
|
|
|
2018
|
|
|
204,585
|
|
|
2019
|
|
|
210,234
|
|
|
2020
|
|
|
106,530
|
|
|
Total
|
|
$
|
769,311
|
|
During
the three months ended September 30, 2016 and 2015, we incurred rent expense
totaling approximately $51,000 and $37,000, respectively. During the nine months
ended September 30, 2016 and 2015, we incurred rent expense totaling
approximately $151,000 and $112,000, respectively.
Delinquent
Payroll Tax Payments/Returns
The
Company is delinquent in filing certain payroll tax returns resulting in the
non-payment of the related withholdings and employer taxes. The delinquency and
non-payments are for the quarterly period ended June 30, 2016. The total amount
of money owed (excluding potential late filing and late payment penalties) as of
September 30, 2016 is approximately $50,000 (which is included in “accounts
payable and accrued expenses” in the accompanying condensed consolidated balance
sheet as of September 30, 2016).
Legal
Proceedings
On July 10, 2015 Stemage Skin Care,
LLC (the “Plaintiff”) filed a complaint in the U.S. District Court for the
Central District of California entitled “Stemage Skin Care LLC, a North Carolina
limited liability company vs. NuGene International, Inc. et al.” (Civil Action
No.8:15-cv-01078-AG-JCG). The complaint also names as defendants NuGene, Inc.,
Ali Kharazmi, Saeed Kharazmi, Kathy Ireland Worldwide, Stephen Roseberry, Steve
Rosenblum and Erik Sterling. The complaint contains allegations of damage
asserted to be grounded on: (i) copyright infringement; (ii) interference with
contract; (iii) intentional interference with prospective economic advantage;
(iv) negligent interference with prospective economic advantage; and (v)
conspiracy. The complaint allegedly arises out of an August 20, 2012 agreement
among the Plaintiff and kathy ireland inc. ("KI") pursuant to which KI made
Kathy Ireland available to perform “Ambassador Services" as defined within that
agreement. That agreement effectively terminated in October 2014 and is the
subject of a separate arbitration with KI and Kathy Ireland before the American
Arbitration Association. We filed a response denying all claims and based on our
review of the matter, we believe that the complaint is without merit. Early
stages of discovery have been completed and we have agreed to stay further
discovery and motions during settlement discussions between Plaintiff and KI.
Notwithstanding, no assurance of outcome currently can be given.
The matter had been settled for a total payment of
$50,000 to Plaintiff. Subsequent to September 30, 2016 the Company agreed to pay
the $50,000 to settle the matter. A definitive Settlement Agreement is being
executed by and between all involved parties. The Settlement Agreement should be
fully executed before the end of November, 2016, with the Company paying the
$50,000 within that same time-frame (See Note 11).
NUGENE
INTERNATIONAL, INC. AND SUBSIDIARIES
NOTES
TO UNAUDITED CONDENSED CONSOLIDATED FINANCIAL STATEMENTS
September
30, 2016
On July 31, 2015 Star Health & Beauty, LLC
(“SH&B”) filed a complaint in the U.S. District Court for the Northern
District of Georgia entitled “Star Health & Beauty, LLC vs. NuGene, Inc. and
NuGene International, Inc. Defendants” (Case No. 1:15-cv-02634-CAP). The
complaint alleges that our use of the NUGENE name and trademark infringes on
SH&B’s NUGEN name. SH&B seeks cancelation of our NUGENE trademark, as
well as unspecified monetary damages.
The
matter is proceeding very slowly and discovery is ongoing. Plaintiff claims use
to the NUGENE name only for a hair growth/restoration product. We intend to use
a different name for our hair growth/restoration product. As such, we believe
that our intellectual property rights to use of the name NUGENE for all products
other than our hair growth/restoration product will be vindicated. However, no
assurance of outcome currently can be given.
In October 2015, NSE Products, Inc., (“NSE”) a Delaware
corporation based in Provo, Utah, initiated actions in the US Patent and
Trademark Office contesting several of the Company's trademark registrations and
applications. These actions, including Oppositions to trademark applications and
Petitions to Revoke registered marks, rely on assertions made by NSE regarding
the purported likelihood of confusion and dilution of NSE's trademarks that
include the words NU SKIN.
In
July 2016,
this litigation was settled. The Company agreed to abandon all rights to use the
marks NUGENE REGEN, NUCELL, NUCELL HAIR, NUCLEANSE, NUEYE, NUSTEM, and NUMEDIA.
The Company will also discontinue any and use of the marks NUCELL, NUCELL HAIR,
NUCLEANSE, NUEYE, and NULIGHT. The Company is entitled to use and register
without objection by NSE (i) the unitary word mark NUGENE or the design mark
used by NuGene, without limitation; and, (ii) the unitary word marks NUMEDIA and
NUSTEM for stem cell media in International Class 1. NuGene will not attempt to
use or register any other trademark that is either similar to NU SKIN or that
contains NU for personal care, nutrition, and technology products, including but
not limited to goods that fall with International Classes 3 and 5. NuGene, or
any entity or company with which NuGene is or becomes affiliated, will not
attempt to use or register as a trademark NU SKIN.
As
part of the settlement, NSE agreed to the above terms. NSE, or any entity or
company with which NSE is or becomes affiliated, will not directly or indirectly
object to or challenge NuGene's use or registration of the unitary word mark
NuGene or the design mark currently used by Nugene. NSE further agreed that NSE,
or any entity or company with which NSE is or becomes affiliated, will not
attempt to use or register as a trademark NUGENE, NUSTEM, or NUMEDIA.
In
May 2016, we were informed that the California Labor Commissioner scheduled a
hearing in connection with two individuals that claimed the Company did not
fulfill its obligations to pay a final paycheck. The two individuals are seeking
back pay and penalties totaling approximately $31,000. The Company intends to
contest various aspects of each claim. However, the Company recognizes that it
may be held liable and has accrued its best estimate of the eventual amount of
the settlement. It should be noted that the California Labor Board is slow to
process these claims. The Company will also attempt to informally settle each
claim prior to any hearing or further action. Limited settlement discussions
have been initiated as of this time.
On
May 6, 2016, we were presented with a demand for payment of compensation for a
former executive employee (the “Executive”) pursuant to his employment contract
with the Company. The amount of the compensation claimed by the Executive
totaled $49,998, which includes a claim for retaliatory termination. The
Executive also filed with the Labor Board seeking payment of approximately
$12,000 with regard to issues with his last check and a bounced check.. The
Executive was an “at-will” employee, and the Company’s management is evaluating
the merits of this matter. No amounts have been accrued in connection with this
matter through September 30, 2016. We have been in active settlement discussions
with the Executive. We are hopeful we can settle for a cash payment less that
what the Executive is seeking and an issuance of common stock of the
Company.
From
time to time, we may be involved in routine legal proceedings, as well as
demands, claims and threatened litigation that arise in the normal course of our
business. The ultimate amount of liability, if any, for any claims of any type
(either alone or in the aggregate) may materially and adversely affect our
financial condition, results of operations and liquidity. In addition, the
ultimate outcome of any litigation is uncertain. Any outcome (including any for
the actions described above), whether favorable or unfavorable, may materially
and adversely affect us due to legal costs and expenses, diversion of management
attention and other factors. We expense legal costs in the period incurred. We
cannot assure you that additional contingencies of a legal nature or
contingencies having legal aspects will not be asserted against us in the
future, and these matters could relate to prior, current or future transactions
or events.
Other
than that described above, we are not currently a party to any other material
legal proceedings. We are not aware of any pending or threatened litigation
against us that in our view would have a material adverse effect on our
business, financial condition, liquidity, or operating results. However, legal
claims are inherently uncertain, and we cannot assure you that we will not be
adversely affected in the future by legal proceedings.
NOTE
11 - SUBSEQUENT EVENTS
On
October 21, 2106, the Company issued an investor a promissory note in the total
principal amount of $267,500 with 9% annual interest rate, which will be due on
October 21, 2017. The Company received $250,000 of gross proceeds and granted a
warrant to purchase 222,916 shares of common stock at $0.60 per share.
On November 2, 2016, the
Company issued certain investors promissory notes (the “Notes”) in the total
principal amount of $480,000 with a 5% annual interest rate, which will be due
on April 28, 2017. The Company received $400,000 of gross proceeds from the
Notes.
The
Company also granted to the investors warrants to purchase 1,428,572 shares of
common stock at no greater than $0.47 per share.
Subsequent
to September 30, 2016, the Company agreed to pay $50,000 to settle the Stemage
Skin Care matter. A definitive Settlement Agreement is being executed by and
between all involved parties. The Settlement Agreement should be fully executed
before the end of November, 2016, with the Company paying the $50,000 within
that same time-frame.
REPORT OF THE INDEPENDENT REGISTERED
PUBLIC ACCOUNTING FIRM
To the Board of Directors and Stockholders
of
NuGene International, Inc.
We have audited the accompanying consolidated
balance sheets of NuGene International, Inc. and subsidiaries (“the Company”) as of December 31, 2015 and 2014 and
the related consolidated statements of operations, changes in stockholders’ deficit and cash flows for the years then ended.
These financial statements are the responsibility of the Company’s management. Our responsibility is to express an opinion
on these financial statements based on our audits.
We conducted our audit in accordance with
standards of the Public Company Accounting Oversight Board (United States). Those standards require that we plan and perform the
audit to obtain reasonable assurance about whether the financial statements are free of material misstatement. The Company is not
required to have, nor were we engaged to perform, an audit of its internal control over financial reporting. Our audit included
consideration of internal control over financial reporting as a basis for designing audit procedures that are appropriate in the
circumstances, but not for the purpose of expressing an opinion on the effectiveness of the Company’s internal control over
financial reporting. Accordingly, we express no such opinion. An audit includes examining, on a test basis, evidence supporting
the amounts and disclosures in the financial statements. An audit also includes assessing the accounting principles used and significant
estimates made by management, as well as evaluating the overall financial statement presentation. We believe that our audit provides
a reasonable basis for our opinion.
In our opinion, the financial statements
referred to above present fairly, in all material respects, the consolidated financial position of NuGene International, Inc. and
subsidiaries as of December 31, 2015 and 2014, and the results of its operations and cash flows for the periods described above
in conformity with accounting principles generally accepted in the United States of America.
The accompanying financial statements have
been prepared assuming that the Company will continue as a going concern. As discussed in the notes to the financial statements,
the Company has not established sufficient revenues to cover its operating costs and has suffered from continued losses from operations.
As of December 31, 2015, the Company has a working capital deficit and does not have the sufficient cash resources available to
meet its planned business objectives. These and other factors raise substantial doubt about the Company’s ability to continue
as a going concern. Management’s plan regarding these matters is also described in the notes to the financial statements.
The financial statements do not include any adjustments that might result from the outcome of this uncertainty.
/s/ Dov Weinstein & Co. C.P.A. (Isr)
www.wcpa.co.il
Jerusalem, Israel
March 30, 2016
NuGene International, Inc.
and Subsidiaries
Consolidated Balance
Sheets
December 31, 2015 and 2014
|
|
|
2015
|
|
|
2014
|
|
|
|
|
|
|
|
|
|
|
Assets
|
|
|
|
|
|
|
|
|
|
Current assets:
|
|
|
|
|
|
|
|
|
|
Cash
|
|
$
|
22,907
|
|
|
$
|
1,344,737
|
|
|
Accounts receivable, net of allowance for doubtful accounts of $177,140 and $3,000 in 2015 and 2014, respectively
|
|
|
347,048
|
|
|
|
15,567
|
|
|
Inventories
|
|
|
177,492
|
|
|
|
110,953
|
|
|
Brand participation fee, net
|
|
|
-
|
|
|
|
300,000
|
|
|
Other current assets
|
|
|
100,555
|
|
|
|
15,520
|
|
|
|
|
|
|
|
|
|
|
|
|
Total current assets
|
|
|
648,002
|
|
|
|
1,786,777
|
|
|
|
|
|
|
|
|
|
|
|
|
Property and equipment, net of accumulated depreciation of $40,620 and $1,255 at 2015 and 2014, respectively
|
|
|
208,350
|
|
|
|
60,674
|
|
|
Deposits
|
|
|
21,321
|
|
|
|
12,800
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
$
|
877,673
|
|
|
$
|
1,860,251
|
|
|
|
|
|
|
|
|
|
|
|
|
Liabilities and Stockholders' Equity (Deficiency)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Current liabilities:
|
|
|
|
|
|
|
|
|
|
Accounts payable and accrued expenses
|
|
$
|
327,623
|
|
|
$
|
135,563
|
|
|
Accounts payable - related parties
|
|
|
37,936
|
|
|
|
122,084
|
|
|
Deferred revenues
|
|
|
-
|
|
|
|
234,916
|
|
|
Notes payable
|
|
|
842,504
|
|
|
|
-
|
|
|
Accrued interest
|
|
|
35,052
|
|
|
|
-
|
|
|
Other current liabilities
|
|
|
17,924
|
|
|
|
-
|
|
|
|
|
|
|
|
|
|
|
|
|
Total current liabilities
|
|
|
1,261,039
|
|
|
|
492,563
|
|
|
|
|
|
|
|
|
|
|
|
|
Commitments and contingencies
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Stockholders
’
equity (deficiency):
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Series A convertible preferred stock; $0.0001 par value; 25,000,000 shares authorized; 1,917,720 shares issued and outstanding on December 31, 2015 (1,917,720 at December 31, 2014)
|
|
|
192
|
|
|
|
192
|
|
|
Common stock; $0.0001 par value; 100,000,000 shares authorized; 39,894,673 and 39,197,400 shares issued and outstanding on December 31, 2015 and 2014, respectively
|
|
|
3,990
|
|
|
|
3,920
|
|
|
Common stock earned but unissued; 302,283 shares as of December 31, 2015
|
|
|
30
|
|
|
|
-
|
|
|
Additional paid-in capital
|
|
|
6,180,929
|
|
|
|
2,713,016
|
|
|
Accumulated deficit
|
|
|
(6,568,507
|
)
|
|
|
(1,349,440
|
)
|
|
|
|
|
|
|
|
|
|
|
|
Total stockholders' equity (deficiency)
|
|
|
(383,366
|
)
|
|
|
1,367,688
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
$
|
877,673
|
|
|
$
|
1,860,251
|
|
See accompanying notes.
NuGene International, Inc.
and Subsidiaries
Consolidated Statements of Operations
For the years ended December
31, 2015 and 2014
|
|
|
2015
|
|
2014
|
|
|
|
|
|
|
|
|
|
|
|
Revenues
|
|
$
|
2,084,939
|
|
|
$
|
723,438
|
|
|
|
|
|
|
|
|
|
|
|
|
Cost of goods sold
|
|
|
588,882
|
|
|
|
293,162
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
1,496,057
|
|
|
|
430,276
|
|
|
|
|
|
|
|
|
|
|
|
|
Operating expenses:
|
|
|
|
|
|
|
|
|
|
Selling, general and administrative
|
|
|
6,658,109
|
|
|
|
743,057
|
|
|
|
|
|
|
|
|
|
|
|
|
Loss from operations
|
|
|
(5,162,052
|
)
|
|
|
(312,781
|
)
|
|
|
|
|
|
|
|
|
|
|
|
Interest expense, net
|
|
|
(57,015
|
)
|
|
|
—
|
|
|
|
|
|
|
|
|
|
|
|
|
Net loss
|
|
$
|
(5,219,067
|
)
|
|
$
|
(312,781
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Weighted average number of common shares
|
|
|
|
|
|
|
|
|
|
outstanding - basic and diluted
|
|
|
39,730,875
|
|
|
|
20,865,881
|
|
|
|
|
|
|
|
|
|
|
|
|
Net loss per share - basic and diluted
|
|
$
|
(0.13
|
)
|
|
$
|
(0.01
|
)
|
|
|
|
|
|
|
|
|
|
|
See accompanying notes.
NuGene International, Inc. and Subsidiaries
Consolidated Statements of Changes in Stockholders' Equity (Deficiency)
For the years ended December 31, 2015
and 2014
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Total
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Common Stock Earned but
|
|
|
Additional
|
|
|
|
|
|
|
|
|
Stockholders'
|
|
|
|
|
Preferred
Stock
|
|
|
Common
Stock
|
|
|
Unissued
|
|
|
Paid-In
|
|
|
Treasury
|
|
|
Accumulated
|
|
|
Equity
|
|
|
|
|
Shares
|
|
|
Par Value
|
|
|
Shares
|
|
|
Par Value
|
|
|
Shares
|
|
|
Par
Value
|
|
|
Capital
|
|
|
Stock
|
|
|
Deficit
|
|
|
(Deficiency)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Balance at December 31, 2013
|
|
|
-
|
|
|
$
|
-
|
|
|
|
20,552,760
|
|
|
$
|
2,055
|
|
|
|
-
|
|
|
$
|
-
|
|
|
$
|
1,054,907
|
$
|
|
|
-
|
|
|
$
|
(1,036,659
|
)
|
|
$
|
20,303
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Issuance of common stock
|
|
|
-
|
|
|
|
-
|
|
|
|
4,000,000
|
|
|
|
400
|
|
|
|
-
|
|
|
|
-
|
|
|
|
933
|
|
|
|
-
|
|
|
|
-
|
|
|
|
1,333
|
|
|
Stock based compensation
|
|
|
-
|
|
|
|
-
|
|
|
|
1,500,000
|
|
|
|
150
|
|
|
|
-
|
|
|
|
-
|
|
|
|
8,683
|
|
|
|
-
|
|
|
|
-
|
|
|
|
8,833
|
|
|
Issuance of common stock
from debt conversion
|
|
|
-
|
|
|
|
-
|
|
|
|
375,000
|
|
|
|
38
|
|
|
|
-
|
|
|
|
-
|
|
|
|
374,962
|
|
|
|
-
|
|
|
|
-
|
|
|
|
375,000
|
|
|
Issuance of common stock
in private placement
|
|
|
-
|
|
|
|
-
|
|
|
|
1,625,000
|
|
|
|
163
|
|
|
|
-
|
|
|
|
-
|
|
|
|
1,624,837
|
|
|
|
-
|
|
|
|
-
|
|
|
|
1,625,000
|
|
|
Recapitalization
|
|
|
1,917,720
|
|
|
|
192
|
|
|
|
11,144,640
|
|
|
|
1,114
|
|
|
|
-
|
|
|
|
-
|
|
|
|
(1,306
|
)
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
Treasury stock acquired
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
(350,000
|
)
|
|
|
-
|
|
|
|
(350,000
|
)
|
|
Cancellation of treasury
stock
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
(350,000
|
)
|
|
|
350,000
|
|
|
|
-
|
|
|
|
-
|
|
|
Net
loss
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
(312,781
|
)
|
|
|
(312,781
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Balance at December 31, 2014
|
|
|
1,917,720
|
|
|
|
192
|
|
|
|
39,197,400
|
|
|
|
3,920
|
|
|
|
-
|
|
|
|
-
|
|
|
|
2,713,016
|
|
|
|
-
|
|
|
|
(1,349,440
|
)
|
|
|
1,367,688
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Issuance of common stock
for services
|
|
|
-
|
|
|
|
-
|
|
|
|
460,000
|
|
|
|
46
|
|
|
|
302,283
|
|
|
|
30
|
|
|
|
1,505,737
|
|
|
|
-
|
|
|
|
-
|
|
|
|
1,505,813
|
|
|
Stock based compensation
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
1,483,867
|
|
|
|
-
|
|
|
|
-
|
|
|
|
1,483,867
|
|
|
Issuance of warrants in
connection with notes payable
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
138,333
|
|
|
|
-
|
|
|
|
-
|
|
|
|
138,333
|
|
|
Issuance of common stock
in exchange for cash
|
|
|
-
|
|
|
|
-
|
|
|
|
237,273
|
|
|
|
24
|
|
|
|
-
|
|
|
|
-
|
|
|
|
339,976
|
|
|
|
-
|
|
|
|
-
|
|
|
|
340,000
|
|
|
Net
loss
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
(5,219,067
|
)
|
|
|
(5,219,067
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Balance at December
31, 2015
|
|
|
1,917,720
|
|
|
$
|
192
|
|
|
|
39,894,673
|
|
|
$
|
3,990
|
|
|
|
302,283
|
|
|
$
|
30
|
|
|
$
|
6,180,929
|
$
|
|
|
-
|
|
|
$
|
(6,568,507
|
)
|
|
$
|
(383,366
|
)
|
See accompanying notes.
NuGene International, Inc. and Subsidiaries
Consolidated Statements of Cash Flows
For the years ended December 31, 2015
and 2014
|
|
|
2015
|
|
|
2014
|
|
|
|
|
|
|
|
|
|
|
Cash flows from operating activities:
|
|
|
|
|
|
|
|
|
|
Net loss
|
|
$
|
(5,219,067
|
)
|
|
$
|
(312,781
|
)
|
|
Adjustments to reconcile net loss to cash flows from operating activities:
|
|
|
|
|
|
|
|
|
|
Stock based compensation and stock issued for services
|
|
|
2,989,680
|
|
|
|
8,833
|
|
|
Noncash portion of interest expense
|
|
|
55,889
|
|
|
|
-
|
|
|
Depreciation and amortization
|
|
|
39,365
|
|
|
|
51,255
|
|
|
Bad debt expense
|
|
|
223,631
|
|
|
|
3,000
|
|
|
Changes in operating assets and liabilities:
|
|
|
|
|
|
|
|
|
|
Accounts receivable
|
|
|
(555,112
|
)
|
|
|
(11,315
|
)
|
|
Inventories
|
|
|
(66,539
|
)
|
|
|
(110,953
|
)
|
|
Brand participation fee
|
|
|
300,000
|
|
|
|
(350,000
|
)
|
|
Other assets
|
|
|
(93,556
|
)
|
|
|
(28,320
|
)
|
|
Accounts payable and accrued expenses
|
|
|
192,060
|
|
|
|
134,312
|
|
|
Accounts payable - related parties
|
|
|
(84,148
|
)
|
|
|
122,085
|
|
|
Deferred revenues
|
|
|
(234,916
|
)
|
|
|
234,916
|
|
|
Other liabilities
|
|
|
17,924
|
|
|
|
-
|
|
|
|
|
|
|
|
|
|
|
|
|
Cash flows from operating activities
|
|
|
(2,434,789
|
)
|
|
|
(258,968
|
)
|
|
|
|
|
|
|
|
|
|
|
|
Cash flows from investing activities:
|
|
|
|
|
|
|
|
|
|
Purchases of property and equipment
|
|
|
(187,041
|
)
|
|
|
(61,929
|
)
|
|
|
|
|
|
|
|
|
|
|
|
Cash flows from financing activities:
|
|
|
|
|
|
|
|
|
|
Acquisition of treasury stock
|
|
|
-
|
|
|
|
(350,000
|
)
|
|
Proceeds from notes payable
|
|
|
960,000
|
|
|
|
-
|
|
|
Proceeds from issuance of common stock
|
|
|
340,000
|
|
|
|
1,626,333
|
|
|
Proceeds from issuance of convertible debt
|
|
|
-
|
|
|
|
375,000
|
|
|
|
|
|
|
|
|
|
|
|
|
Cash flows from financing activities
|
|
|
1,300,000
|
|
|
|
1,651,333
|
|
|
|
|
|
|
|
|
|
|
|
|
Change in cash
|
|
|
(1,321,830
|
)
|
|
|
1,330,436
|
|
|
|
|
|
|
|
|
|
|
|
|
Cash, beginning of year
|
|
|
1,344,737
|
|
|
|
14,301
|
|
|
|
|
|
|
|
|
|
|
|
|
Cash, end of year
|
|
$
|
22,907
|
|
|
$
|
1,344,737
|
|
|
|
|
|
|
|
|
|
|
|
|
Supplemental disclosure of cash flow information:
|
|
|
|
|
|
|
|
|
|
Cash paid for:
|
|
|
|
|
|
|
|
|
|
Interest
|
|
$
|
1,126
|
|
|
$
|
-
|
|
|
Income taxes
|
|
$
|
-
|
|
|
$
|
-
|
|
See accompanying notes.
NUGENE INTERNATIONAL, INC
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
December 31, 2015
NOTE 1 – NATURE OF BUSINESS AND BASIS OF PRESENTATION
NuGene International, Inc. (the “Company”
or “NGI”) was incorporated in the State of Nevada on October 31, 2013. On January 22, 2015, we changed our name to
NuGene International, Inc. and effected a 15.04 to one stock split in the form of a stock dividend (“Stock Split”).
All amounts shown for common stock and additional paid in capital included in these financial statements have been adjusted to
reflect the Stock Split. NuGene, Inc. (our wholly owned subsidiary) was incorporated in the state of California in December 2006.
On January 20, 2015, we formed NuGene BioPharma, Inc. (“BioPharma”) as a wholly owned California incorporated subsidiary
of NGI. On November 6, 2015, we formed The Aesthetic Group, Inc. (“TAG”) as a wholly owned California incorporated
subsidiary of NGI. Both BioPharma and TAG had no significant independent operations during 2015.
Basis of Presentation
The accompanying consolidated financial
statements of our Company have been prepared in accordance with accounting principles generally accepted in the United States.
The consolidated financial statements reflect the accounts of NuGene International, Inc. and its wholly owned subsidiaries BioPharma
and TAG. All significant inter-company balances and transactions have been eliminated in consolidation.
We have incurred net losses through the date of these financial statements and have yet to establish profitable
operations. Our financial statements have been presented on the basis that our business is a going concern, which contemplates
the realization of assets and the satisfaction of liabilities in the normal course of business. We are subject to the risks and
uncertainties associated with a business with high short-term debt, limited commercial product revenues, including limitations
on our operating capital resources and uncertain demand for our products. We have incurred recurring operating losses and negative
operating cash flows in 2015, and we expect to continue to incur operating losses and negative operating cash flows in the near
future. As a result, management has concluded that there is substantial doubt about our ability to continue as a going concern.
Our independent registered public accounting firm, in its report on our 2015 consolidated financial statements, has raised substantial
doubt about our ability to continue as a going concern.
Our consolidated financial statements as
of and for the year ended December 31, 2015 do not contain any adjustments for this uncertainty. In response to our Company’s
cash needs, we raised funding as described in our footnotes that follow. Any additional amounts raised will be used for our future
investing and operating cash flow needs. However, there can be no assurance that we will be successful in raising additional amounts
of financing.
NOTE 2 – SUMMARY OF SIGNIFICANT
ACCOUNTING POLICIES
Use of Estimates
Conformity with Generally Accepted Accounting
Principles (“GAAP”) requires the use of estimates and judgments that affect the reported amounts in the financial
statements and accompanying notes. These estimates form the basis for judgments we make about the carrying values of our assets
and liabilities that are not readily apparent from other sources. We base our estimates and judgments on historical information
and on various other assumptions that we believe are reasonable under the circumstances. GAAP requires us to make estimates and
judgments in several areas, including but not limited to, those related to revenue recognition, collectability of accounts receivable,
contingent liabilities, fair value of financial instruments, fair value of acquired intangible assets and goodwill, useful lives
of intangible assets and property and equipment, excess and obsolete inventory, deferred tax asset valuation and income taxes.
These estimates are based on management’s knowledge about current events and expectations about actions we may undertake
in the future. Actual results could differ materially from those estimates.
Risks and Uncertainties
Our Company operates in an industry that
is subject to rapid change. Our Company's operations are subject to significant risk and uncertainties including financial, operational,
technological, regulatory and other risks, including the potential of business failure.
Reclassifications
For comparability, certain prior period
amounts have been reclassified, where appropriate, to conform to the current year’s financial statement presentation. These
reclassifications have no impact on net loss.
Cash Equivalents
Cash equivalents include investments with
initial maturities of three months or less. Our Company maintains its cash balances at credit-worthy financial institutions that
are insured by the Federal Deposit Insurance Corporation up to $250,000.
Inventories
Our Company’s skin and hair care
inventories consist of raw materials and finished goods and are valued at the lower of cost (first-in, first-out) or market. Our
Company evaluates its inventory for excess quantities and obsolescence on a regular basis. To determine if the cost of our Company's
inventory should be written down, current and anticipated demand, customer preferences and the age of the merchandise are considered.
For the years ended December 31, 2015 and 2014, our Company did not recognize any significant charges to operations associated
with excess and obsolete inventory.
Property and Equipment
Property and equipment consist of office
furniture, computer equipment, laboratory equipment, purchased software, website development and improvements made to our leased
offices. Property and equipment is stated at cost less accumulated depreciation. Depreciation is calculated using the straight-line
method over the three-year useful life of the assets or the remaining term of the lease. When property and equipment are retired
or otherwise disposed of, the related cost and accumulated depreciation are removed from the respective accounts, and any gain
or loss is included in operations. Expenditures for maintenance and repairs are expensed as incurred.
Website Development
Costs and expenses incurred during the
planning and operating stages of our Company’s website development are expensed as incurred. Our Company accounts for the
development of its website by expensing all costs associated with the planning of the website as incurred and capitalizing the
costs to develop the website. Once the website is available for use, the related costs will be amortized over their estimated
useful life on a straight-line basis (generally estimated to be 3 years) and tested for impairment annually.
Intangible Assets
Identifiable intangible assets with indefinite
lives are not amortized but instead are tested for impairment annually or more frequently if circumstances indicate a possible
impairment may exist. Intangible assets with estimable useful lives are amortized over their respective estimated useful lives,
generally on a straight-line basis, and are reviewed for impairment whenever events or changes in circumstances indicate that
the carrying value may not be recoverable.
Deferred Revenue
As of December 31, 2015, we had no deferred
revenue. As of December 31, 2014, we had a remaining balance of $234,916 in deferred revenues included in current liabilities
related to the sale of products undelivered at that date that was ultimately recognized in 2015.
Revenue Recognition
Our Company recognizes revenue from product
sales when the product has been ordered by the customer, the selling price is fixed or determinable, the product is shipped to
the customer, title has transferred and collectability is reasonably assured. During 2014, we generated revenues from an affiliate,
Advanced Surgical Partners (“AdvSP”). Revenues generated from AdvSP resulted from NuGene providing plasma rich platelet
and adipose derived stem cells for orthopedic and plastic surgery procedures to AdvSP.
Concentration of Revenues
During the year ended December 31, 2015, we derived 31.1% and 15.7% of our revenues from two wholesale
distributors (ending balances in accounts receivable totaling $173,790 and $327,893, respectively). During the year ended December
31, 2014, we derived 66% or $481,000 of our revenues from AdvSP. Excluding sales to ASP, 51% of our revenues during the year ended
December 31, 2014 were derived from a single wholesale distributor. Our distributors purchase products from us on a purchase order
basis on standard terms. The distributors are under no obligation to continue to purchase our products. The loss of any of our
major distributors, a material reduction in their purchases or the cancellation of product orders or unexpected returns of unsold
products could significantly decrease our revenues and impede our future growth prospects. We do not have long-term purchase commitments
with our distributors.
Cost of Goods Sold
Cost of goods sold include all of the
costs to manufacture our Company’s products. For products manufactured in our Company’s own facilities, such costs
include raw materials and supplies, direct labor and factory overhead. For products manufactured for our Company by third-party
contractors, such cost represents the amounts invoiced by the contractors.
Royalty Expense
We recognize royalty expenses in accordance
with the terms of our license agreement with a celebrity product endorser.
Royalties are expensed in the statements of
operation in the period that the related revenues are recognized, in cost of goods sold.
Research and Development
Internal research and development costs
are expensed as incurred. Third-party research and development costs are expensed when the contracted work has been performed.
Research and development consists of consulting fees, direct labor and raw materials associated with the development of new products
to be commercialized by our Company. Research and development expenses totaled $350,669 and $65,830 in 2015 and 2014, respectively.
Allowance for Doubtful Accounts
Our Company maintains allowances for doubtful
accounts for estimated losses resulting from the inability of our customers to make required payments. Account balances are charged
off against the allowance after all means of collection have been exhausted and the potential for recovery is considered remote.
Our allowance for doubtful accounts was $177,140 and $3,000 at December 31, 2015 and 2014, respectively. Bad debt expense totaled
$223,631 and $300, for the years ended December 31, 2015 and 2014, respectively.
Share Based Payments
We recognize equity–based compensation
net of an estimated forfeiture rate and recognize compensation cost only for those shares expected to vest over the requisite
service period of the award. Share-based payments to employees, including grants of employee stock options, are recognized as
compensation expense in the accompanying statements of operations based on the fair values of the related payments. Such expense
is recognized over the period during which an employee is required to provide services in exchange for the award, known as the
requisite service period (usually the vesting period). We account for share–based payments granted to non–employees
by calculating the fair value of the stock–based payment as either the fair value of the consideration received or the fair
value of the equity instruments issued, whichever is more reliably measurable. If the fair value of the equity instruments issued
is used, it is measured using the stock price and other measurement assumptions as of the earlier of either (1) the date at which
a commitment for performance by the counterparty to earn the equity instruments is reached, or (2) the date at which the counterparty’s
performance is complete.
Earnings per Share of Common Stock
We present both basic and diluted earnings
per share (“EPS”) on the face of the statement of operations. Basic EPS is calculated by dividing the profit or loss
attributable to our common shareholders by the weighted average number of common shares outstanding during the period. Diluted
EPS is determined by adjusting the profit or loss attributable to common shareholders and the weighted average number of common
shares outstanding for the effects of all potential dilutive common shares (common stock equivalents or “CSE”).
Income Taxes
We account for income taxes under the
asset and liability method. Deferred tax assets and liabilities are recognized for the future consequences of differences between
the financial statement carrying amounts of existing assets and liabilities and their respective tax bases (temporary differences).
Deferred tax assets and liabilities are measured using tax rates expected to apply to taxable income in the years in which those
temporary differences are recovered or settled. Valuation allowances for deferred tax assets are established when it is more likely
than not that some portion or all of the deferred tax assets will not be realized. We determined that as of December 31, 2015,
no additional accrual for income taxes was necessary. The federal and state income tax returns of our Company are subject to examination
by the IRS and state taxing authorities, generally for three years after filing.
Fair Value of Financial Instruments
We measure assets and liabilities at fair
value based on an expected exit price as defined by the authoritative guidance on fair value measurements. Fair value represents
the estimated amount that would be received upon the sale of an asset or paid to transfer a liability in an orderly transaction
between market participants. As such, fair value may be based on assumptions that market participants would use in pricing an
asset or liability. The authoritative guidance on fair value measurements establishes a consistent framework for measuring fair
value on either a recurring or nonrecurring basis whereby inputs used in valuation techniques are assigned a hierarchical level.
The following are the hierarchical levels of inputs to measure fair value:
|
|
- Level 1:
|
Quoted prices in active markets for identical instruments;
|
|
|
- Level 2:
|
Other significant observable inputs (including quoted prices
in active markets for similar instruments);
|
|
|
- Level 3:
|
Significant unobservable inputs (including assumptions
in determining the fair value of certain investments).
|
The carrying values for cash and cash
equivalents, accounts receivable, other current assets, accounts payable and accrued liabilities, and deferred revenue approximate
their fair value due to their short maturities.
Recent Accounting Standards Updates
In February 2016, the FASB issued ASU
No.
2016-02
-
Leases (Topic 842): Simplifying the Measurement of Inventory
(“Topic 842”). Topic 842 requires lessees to recognize a right-of-use asset and lease liability for virtually
all of their leases. The liability will be equal to the present value of lease payments. The asset will be based on the liability,
subject to adjustment, such as for initial direct costs. For income statement purposes, the FASB retained a dual model, requiring
leases to be classified as either operating or finance. Operating leases will result in straight-line expense while finance leases
will result in a front-loaded expense pattern (similar to current capital leases). Classification will be based on criteria that
are largely similar to those applied in current lease accounting, but without explicit bright lines. Topic 842 is effective for
our Company on January 1, 2019 and includes interim periods within that year. Topic 842 will be applied using a modified retrospective
transition, providing for certain practical expedients. Early application is permitted and our Company is currently evaluating
the potential impact that adoption may have on its financial statements.
In July 2015, the FASB issued ASU No.
2015-11
-
Inventory (Topic 330): Simplifying the Measurement of Inventory
(“Topic 330”). Topic 330 requires measuring
inventory at the lower of cost and net realizable value based on estimated selling prices in the ordinary course of business,
less reasonably predictable costs of completion, disposal and transportation (changed from the previous guidance of lower of cost
or market). This update also clarified various other inventory measurement and disclosure requirements. The update does not apply
to inventory measured using the LIFO or retail inventory methods. The guidance is effective for annual reporting periods beginning
after December 15, 2016, including interim periods within that reporting period, and should be applied prospectively. Early application
is permitted and our Company is currently evaluating the potential impact that adoption may have on its financial statements.
In May 2014, the FASB issued ASU No. 2014-09
-
Revenue from Contracts with Customers
(“ASU 2014-09). ASU 2014-09 outlines a single comprehensive model for entities
to use in accounting for revenue arising from contracts with customers and supersedes most current revenue recognition guidance,
including industry-specific guidance. ASU 2014-09 is based on the principle that an entity should recognize revenue to depict
the transfer of goods or services to customers in an amount that reflects the consideration to which the entity expects to be
entitled in exchange for those goods or services. The ASU also requires additional disclosure about the nature, amount, timing
and uncertainty of revenue and cash flows arising from customer contracts, including significant judgments and changes in judgments
and assets recognized from costs incurred to fulfill a contract. Entities have the option of using either a full retrospective
or a modified retrospective approach for the adoption of the new standard. This standard is effective for fiscal years beginning
after December 15, 2017, including interim periods within that reporting period. Our Company is currently evaluating the
new guidance to determine the impact it will have on its consolidated financial statements.
Other accounting standards that have been
issued or proposed by FASB that do not require adoption until a future date are not expected to have a material impact on the
consolidated financial statements upon adoption. Our Company does not discuss recent pronouncements that are not anticipated to
have an impact on or are unrelated to its financial condition, results of operations, cash flows or disclosures.
NOTE 3 – INVENTORIES
Inventories consist of the following:
|
|
|
December 31,
2015
|
|
|
December 31, 2014
|
|
|
Raw materials
|
|
$
|
72,287
|
|
|
$
|
52,045
|
|
|
Finished goods
|
|
|
105,205
|
|
|
|
58,908
|
|
|
Total Inventories
|
|
$
|
177,492
|
|
|
$
|
110,953
|
|
NOTE 4 – INTANGIBLE ASSETS
Licensee Agreement
In November 2014, we entered into a License
Agreement with kathy ireland Worldwide
®
("kiWW
®
") under which we licensed the right to
utilize the trademarks and rights to the name, likeness and visual representations of Kathy Ireland (“KI”) in connection
with our cosmeceutical line of products. The initial term of the license is for eight years and it may be renewed at the option
of our Company for up to an additional four years. In accordance with the License Agreement, we will pay 5% of the net sales for
all licensed products sold and collected under the licensed marks or a minimum guaranteed royalty of $100,000 in year one, which
includes the period from approximately November 4, 2014 through December 31, 2015 (“Contract Year 1”) of the License
Agreement. The minimum guaranteed royalty increases $50,000 each year in years two through eight of the License Agreement. We
recognized $100,000 in royalty fees during the year ended December 31, 2015.
Additionally, we are obligated to pay
an annual Brand Participation Fee to kiWW
®
which provides for general advertising, good will and promotion of the
KI brand. NuGene prepaid kiWW $350,000 effective upon execution of the License as a Brand Participation fee for Contract Year
1. Accordingly, we amortized this initial Brand Participation Fee over the 14-month period of Contract Year 1. The net carrying
value of the Brand Participation Fee at December 31, 2014 was $300,000. The Brand Participation Fee for Contract Years 2 through
8 is $50,000 annually with an additional 1% of the total gross sales of Licensed Products of the prior year beginning in Contract
Year 4.
SkinGuardian Intellectual Property
On March 17
,
2015, we entered into an
Intellectual Property Asset Purchase Agreement (“IP Agreement”) with an individual (“IP Seller”) to acquire
all of the rights, title and interest in certain intellectual property connected with a product named
SkinGuardian
(“SGIP”).
Upon the acquisition of SGIP, our Company issued 150,000 shares of restricted common stock (net, as amended) to the IP Seller valued
at $150,000. The consideration paid for SGIP was expensed during the year ended December 31, 2015.
NOTE 5 – PROPERTY AND EQUIPMENT
Property and equipment, net consist of
the following:
|
|
|
December 31, 2015
|
|
|
December 31, 2014
|
|
|
Software / website development
|
|
$
|
8,219
|
|
|
$
|
2,139
|
|
|
Equipment
|
|
|
136,585
|
|
|
|
35,698
|
|
|
Leasehold improvements
|
|
|
104,166
|
|
|
|
24,092
|
|
|
|
|
|
|
|
|
|
|
|
|
Property and equipment, gross
|
|
|
248,970
|
|
|
|
61,929
|
|
|
Accumulated depreciation
|
|
|
(40,620
|
)
|
|
|
(1,255
|
)
|
|
Property and equipment, net
|
|
$
|
208,350
|
|
|
$
|
60,674
|
|
Depreciation expense for the years ended
December 31, 2015 and 2014 was $39,365 and $1,255, respectively.
NOTE 6 – PROMISSORY NOTES PAYABLE
AND ADVANCES
On September 25, 2015, we entered into
a Securities Purchase Agreement and issued a 15% Promissory Note with the principal face value of $500,000 (the “15% Note”)
to an accredited investor. Under the terms of the 15% Note, all principal and related accrued interest outstanding are due and
payable to the noteholder upon the earlier of: (i) September 25, 2016; or (ii) within ten business days after the consummation
of an equity or convertible debt financing with aggregate gross proceeds of at least $1,000,000.
Borrowings made pursuant to the 15% Note
bear interest at the annual rate of 15% (or $75,000), irrespective of whether paid at or prior to September 25, 2016. If
any amount payable pursuant to the note payable is not paid when due (without regard to any applicable grace periods as set forth
in the Note), whether at stated maturity, by acceleration or otherwise, such overdue amount shall bear interest at the rate of
15% from the date of such non-payment until such amount is paid in full. We have recognized $31,479 in interest expense through
December 31, 2015 based on an estimated repayment date of May 15, 2016.
During November 2015, we issued a one
year 10% promissory notes payable (the “10% Note”) to a purchaser for cash proceeds totaling $50,000. In the event
that we secure any future financing with aggregate gross proceeds of at least $1 million while the 10% Note is outstanding, the
10% Note and all accrued interest therefrom will be immediately due and payable within ten business days of the closing of such
financing. The holder may also convert any unpaid principal under the 10% Note into any funding instrument entered into by our
Company for a period of 180 days after the date of the 10% Note. The full interest of 10% of the borrowings under the 10% Note
($5,000) is due irrespective of whether paid at maturity or when required to be prepaid. We have recognized $1,383 in interest
expense through December 31, 2015 based on an estimated repayment date of May 15, 2016.
On November 30, 2015 and on four subsequent
dates during December 2015, four individuals (the “Lenders”) advanced a total of $410,000 to the Company. The borrowings
(“Advances”) were not accompanied by documentation of the nature of the Advances. However, there were general discussions
as to the repayment terms, the interest expected to be paid to the Lenders and additional equity of the Company to be issued to
the Lenders in connection with the Advances. Accordingly, we have accounted for the Advances based on estimates of their final
terms.
In connection with the expected value
of the warrants, we have recorded an equity contribution of $138,333 in December 2015 ($15,259 of which was amortized and charged
to interest expense through December 31, 2015). We calculated an original issue discount of $51,111 on the borrowings ($5,578
of which was charged to interest expense through December 31, 2015) and we have recognized $2,190 of accrued interest through
December 31, 2015 for the expected interest to be paid on the non-discounted face value of the borrowings. The borrowings are
carried at $430,838 as of December 31, 2015.
Borrowings under notes payable and advances
as of December 31, 2015 are summarized as follows:
|
|
|
Company
Proceeds
|
|
|
Carrying Value
at December
31, 2015
|
|
|
Accrued
Interest at
December 31,
2015
|
|
|
Principal Value
at Maturity
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
15% Note
|
|
$
|
500,000
|
|
|
$
|
500,000
|
|
|
$
|
31,479
|
|
|
$
|
500,000
|
|
|
10% Note
|
|
|
50,000
|
|
|
|
50,000
|
|
|
|
1,383
|
|
|
|
50,000
|
|
|
Advances
|
|
|
410,000
|
|
|
|
292,504
|
|
|
|
2,190
|
|
|
|
461,111
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
$
|
960,000
|
|
|
$
|
842,504
|
|
|
$
|
35,052
|
|
|
$
|
1,011,111
|
|
NOTE 7 – STOCKHOLDERS’
EQUITY
Preferred Stock
Our Company’s articles of incorporation
authorize 25,000,000 shares of Preferred Stock with a par value of $0.0001. We designated 2,000,000 shares of Preferred Stock
as Series A Convertible Preferred Stock. The Series A Preferred Stock is convertible into common stock at a ratio of one to one.
Additionally, as long as there are a minimum of 900,000 shares of Series A Preferred Stock outstanding, the holders of the Series
A Preferred Stock have the right to elect a majority of our Company’s Board of Directors. Finally, in all manners brought
to a vote of the shareholders, the holders of the Series A Preferred Stock have three votes to every one vote of common stock.
As of December 31, 2015 and December 31, 2014, there were 1,917,720 shares of Series A Preferred Stock outstanding which were
issued to the founders of NuGene, Ali and Saeed Kharazmi. Ali and Saeed Kharazmi each own 50% of the outstanding Series A Preferred
Stock and are our Company’s Chief Executive Officer and Chairman of the Board, respectively.
Common Stock Split
We have 100,000,000 shares of common stock
authorized with a par value of $0.0001. On December 26, 2014, our Board of Directors approved a 15.04 to one stock split (“Stock
Split”) in the form of a stock dividend to holders of our common stock as of that date. To effect that Board action,
we delivered to each recipient of the stock dividend 14.04 additional shares of common stock for every share of common stock held.
Unless otherwise noted, all share numbers shown in these condensed consolidated financial statements give effect to the Stock
Split as approved by our Board of Directors.
Merger Agreement
On December 29, 2014, we completed a merger
(the “Merger”) whereby NuGene, Inc. became a wholly owned subsidiary of our Company. In connection with the
merger, we entered into a Business Transfer and Indemnity Agreement with our former Chief Executive Officer and Director providing
for:
|
|
1.
|
The transfer of our jewelry business
operations existing on the date of the Indemnity Agreement;
|
|
|
2.
|
The assumption by our former CEO
of all liabilities of our Company and her indemnification holding our Company harmless
for all liabilities arising at or before December 29, 2014;
|
|
|
3.
|
The payment of $350,000 in cash
to our former CEO; and
|
|
|
4.
|
The surrender by our former CEO
of 15,000,000 shares (before giving effect to the Stock Split discussed below) (the "Indemnity
Shares") of our Company’s common stock representing 95% of the then outstanding
common stock (all of which shares have been cancelled by our Company and are now included
in our Company’s pool of authorized but unissued shares).
|
At the time of the Merger, all of the
issued and outstanding shares of NuGene, Inc. were exchanged (after giving effect to the Stock Split), for 26,052,760 shares of
our common stock and 1,917,720 our Series A Preferred Stock. On December 29, 2014, we completed the sale of 2,000,000 shares of
our common stock to 18 purchasers (“Stock Placement”) for proceeds totaling $2,000,000, including (a) $1,625,000 of
cash and (b) automatic conversion of $375,000 of outstanding promissory notes. Prior to the Merger, NuGene, Inc. received proceeds
totaling $1,333 in exchange for 4,000,000 shares of its common stock from five persons and issued 1,500,000 shares of common stock,
valued at $500, to a consultant for services rendered during the last quarter of 2014.
On March 31, 2015, we entered into a stock
purchase agreement with a shareholder of our Company (the “Buyer”) resulting in the issuance of 50,000 shares of common
stock to the Buyer for proceeds of $50,000. Under the agreement, we extended to the Buyer an option to purchase an additional
60,000 shares of common stock at $1 per share within 45 days of the Closing. On May 8, 2015, the Buyer exercised his option to
purchase the remaining shares under the stock purchase agreement and accordingly, we issued the Buyer an additional 60,000 shares
of our common stock for cash proceeds of $60,000. On April 20, 2015, our Company entered into a stock purchase agreement with
an unaffiliated accredited investor for the sale of 27,273 shares of common stock at $1.10 per share, resulting in total cash
proceeds of $30,000. On June 2, 2015, our Company entered into a stock purchase agreement with an unaffiliated accredited investor
for the sale of 100,000 shares of common stock at $2.00 per share, resulting in total cash proceeds of $200,000.
Common Stock
As of December 31, 2015, our Company had
entered into Advisory Agreements with members of its Advisory Board. The terms of the individual Advisory Agreements
vary and provide for up to 50,000 initial sign-on shares vesting for a maximum of an 18-month period, and up to 50,000 shares
of common stock per annum issued on the anniversary of the effective date of the agreement. Expenses for the issuance of common
stock for services related to these share issuances was recognized over the service period in which the shares are earned or over
the respective vesting period, as applicable, and was calculated based on the average closing price per share of our common stock,
during the respective quarter, as quoted on the OTC Marketplace. Selling, general and administrative expenses (“SGA”)
recognized in connection with the Advisory Agreements totaled $572,216 for the year ended December 31, 2015.
During 2015, offered positions on our
Company’s Board of Directors to two individuals, although one of the offers was later rescinded. In addition to annual cash
stipends of $30,000, the candidates were granted a total of 200,000 shares of our Company’s common stock vesting 25% on
the third, sixth, ninth and twelfth month following the inception of the proposed Board service. SGA totaling $164,416 was recognized
in connection with these shares over the service periods in which the shares were earned and was calculated based on the average
closing price per share of our common stock, during the respective quarter, as quoted on the OTC Marketplace.
In February 2015, we entered into a six-month
consulting agreement with an investor relations firm. In addition to a $27,000 retainer, the consulting agreement (as amended)
called for the vesting of 110,000 shares of our common stock over the six-month term, issuable upon the conclusion of the agreement.
On August 17, 2015, we issued the 110,000 shares of common stock owed to the investor relations firm. We recognized $167,092 of
SGA in connection with the consulting agreement during the year ended December 31, 2015.
In March 2015, we purchased technology
from an individual in exchange for 150,000 (as amended) shares of our common stock. The common stock was valued at the closing
price per share of our common stock, on the date of issuance resulting in the recognition of $150,000 in SGA for the year ended
December 31, 2015. During 2015, we issued 200,000 fully vested shares of our common stock to three consultants as compensation
for services rendered. The common stock was valued at the closing price per share of our common stock, on the date of issuance,
as quoted on the OTC Marketplace resulting in the recognition of $452,100 in stock based compensation for the year ended December
31, 2015.
Common Stock Options
On August 14,
2015, we granted an option to purchase up to 1,000,000 shares of our common stock to each of two employees. The options have a
strike price of $1.50 per share, have a term of five years from the date of grant, vest evenly over 24 months and have a cashless
exercise feature.
The fair value of these options was estimated using the Black-Scholes option-pricing model based on the
following assumptions: expected dividend yield 0%, expected volatility 130%, risk-free interest rate 1.61% and expected life of
5 years.
We recognized $483,719 of SGA as stock based compensation in connection with the vesting
of the options during year ended December 31, 2015.
Common Stock Warrants
Our Company has issued warrants to purchase
shares of our common stock to accredited investors and consultants as compensation for services rendered, as well as, in conjunction
with the purchase of our common stock. A summary of the Company’s warrants activity and related information as of December
31, 2015 is provided below.
On August 14,
2015, we granted a five-year fully vested warrant to purchase up to 150,000 shares of our common stock to each of two consultants
as compensation for financial services rendered. The warrants have a strike price of $2.00 per share and have a cashless exercise
feature. The fair value of the warrants was estimated to be $377,327 using the Black-Scholes option-pricing model based on the
following assumptions: expected dividend yield 0%, expected volatility 130%, risk-free interest rate 1.61%, and expected life of
5 years and is included in SGA as stock based compensation in the accompanying statement of operations.
On August 14,
2015, we granted a three-year fully vested warrant to purchase up to 50,000 shares of our common stock to a former consultant of
our Company in satisfaction of amounts previously owed to him. The warrant has a strike price of $1.50 per share and has a cashless
exercise feature. The fair value of the warrants was estimated to be $55,821 using the Black-Scholes option-pricing model based
on the following assumptions: expected dividend yield 0%, expected volatility 130%, risk-free interest rate 1.08%, and expected
life of 3 years and is included in SGA as stock based compensation in the accompanying statement of operations.
On December 11, 2015, we entered into a service
agreement (the “SA”) with KBHJJ, LLC (“KBHJJ”). KBHJJ will provide media awareness services to the Company
over the five-year life of the SA. In consideration for these services, our Company agreed to compensate KBHJJ as follows:
|
|
·
|
The
grant of a two-year warrant to purchase up to 1,350,000 shares at an exercise price of
$0.001 per share of the Company’s Common Stock vested as follows: 450,000 shares
upon execution of the agreement; 450,000 shares upon the six-month anniversary of the
SA; and 450,000 shares upon the one-year anniversary of the SA, contingent upon the completion
of certain conditions.
|
|
|
·
|
Payment
of $50,000 upon execution of the SA.
|
|
|
·
|
Payment
of $50,000 due January 4, 2016.
|
|
|
·
|
Up
to 500,000 shares of the Company’s Common Stock that can be earned over the first
18 months of the SA, contingent on hitting benchmarks defined in the SA.
|
We recognized $567,000 of SGA as stock
based compensation in connection with the SA in the accompanying statement of operations in the year ended December 31, 2015.
We have a warrant outstanding held by
an investor to purchase up to 500,000 shares of our common stock at a price of $2.50 per share through December 26, 2020, exercisable
beginning on December 26, 2015. Any shares acquired thereunder upon exercise may not be sold until June 26, 2016.
|
Stock Warrants Outstanding as of December 31, 2015
|
|
|
Exercise
|
|
|
Warrants
|
|
|
Remaining
|
|
|
Warrants
|
|
|
Price
|
|
|
Granted
|
|
|
Life (Years)
|
|
|
Exercisable
|
|
|
$
|
0.001
|
|
|
|
1,350,000
|
|
|
|
1.97
|
|
|
|
450,000
|
|
|
$
|
1.50
|
|
|
|
50,000
|
|
|
|
2.87
|
|
|
|
50,000
|
|
|
$
|
2.00
|
|
|
|
300,000
|
|
|
|
4.88
|
|
|
|
300,000
|
|
|
$
|
2.50
|
|
|
|
500,000
|
|
|
|
5.25
|
|
|
|
-
|
|
|
|
|
|
|
|
2,200,000
|
|
|
|
|
|
|
|
800,000
|
|
NOTE 8 – RELATED PARTY TRANSACTIONS
The following individuals and entities
have been identified as related parties based on their affiliation with our CEO and Chairman of the Board:
|
Ali Kharazmi
|
|
Chief Executive Officer,
President, Board Member and greater than 10% shareholder
|
|
Saeed Kharazmi
|
|
Chairman of the Board, Acting Chief
Financial Officer and greater than 10% shareholder
|
|
Genetics Institute of Anti-Aging
|
|
Company with common ownership and management
|
|
Applied M.A.K. Enterprises, Inc. (“MAK”)
|
|
Company with common ownership and management
|
|
Advanced Surgical Partners, LLC (“AdvSP”)
|
|
Company with common ownership and management
|
|
Center for Weight Management & Plastic
Surgery (“CWM”)
|
|
Company with common ownership and management
|
|
Center for Regenerative Science, LLC
|
|
Company with common ownership and management
|
The following amounts were owed to related
parties, affiliated with the CEO and Chairman of the Board, at the dates indicated:
|
|
|
December 31,
2015
|
|
|
December 31,
2014
|
|
|
AdvSP
|
|
$
|
70,890
|
|
|
$
|
45,000
|
|
|
MAK
|
|
|
-
|
|
|
|
40,084
|
|
|
CWM
|
|
|
34,000
|
|
|
|
-
|
|
|
Ali Kharazmi
|
|
|
25,641
|
|
|
|
37,000
|
|
|
Saeed Kharazmi
|
|
|
2,405
|
|
|
|
-
|
|
|
Less amounts advanced to AdvSP and repaid in January 2016
|
|
|
(95,000
|
)
|
|
|
-
|
|
|
Accounts payable - related parties
|
|
$
|
37,936
|
|
|
$
|
122,084
|
|
The amount owed to AdvSP at December 31, 2015
of $70,890 relates to legal and administrative services provided by AdvSP employees to our Company. Our Company temporarily advanced
$95,000 to AdvSP prior to December 31, 2015 and was repaid in full prior to January 5, 2016. Prior to December 31, 2014, all managerial,
legal and administrative services were provided to our Company by related parties, free of charge. The amount owed to CWM relates
to medical procedures provided to NuGene consultants as compensation for advertising and marketing services provided to NuGene.
The amount owed to Ali Kharazmi and all amounts outstanding at December 31, 2015 and December 31, 2014 represent advances that
bore no interest and were due on demand or expense reimbursements incurred in the ordinary course of business. The amounts owed
to Saeed Kharazmi at December 31, 2015 related to expense reimbursements incurred in the ordinary course of business.
For the year ended December 31, 2014,
our Company recognized revenues from AdvSP in the amount of approximately $481,000. We had no revenues from AdvSP during the year
ended December 31, 2015. Beginning on December 1, 2014, our Company sublet office space from AdvSP. Prior to December 1, 2014,
our Company utilized corporate office space at AdvSP, free of charge. Messrs. Ali and Saeed Kharazmi, our CEO and our Acting CFO,
respectively, have been foregoing salaries since NuGene, Inc. was incorporated in 2006.
NOTE 9 – COMMITMENTS AND CONTINGENCIES
Commitments
We sub-lease our sole
corporate facilities at 17912 Cowan, Suite A, Irvine, California, 92614 from AdvSP, an affiliate of our Company, for approximately
$16,637 per month (including common area maintenance), consistent with the amount that is charged to AdvSP by the property owner.
On February 5, 2015, AdvSP entered into a new five-year lease for the property with the owner beginning July 1, 2015 and subsequently
amended to begin June 1, 2015. The lease was subsequently amended to increase the square footage under lease beginning in January
2016. The lease includes annual increases in the monthly lease payments of approximately 3% each year.
At December 31, 2015,
aggregate future minimum payments under the lease, including common area maintenance costs, are as follows:
|
2016
|
|
$
|
193,285
|
|
|
2017
|
|
|
198,935
|
|
|
2018
|
|
|
204,585
|
|
|
2019
|
|
|
210,234
|
|
|
2020
|
|
|
106,530
|
|
|
Total
|
|
$
|
913,569
|
|
During the years ended December 31, 2015
and 2014, we incurred rent expense totaling $167,454 and $31,314, respectively.
Legal Proceedings
On July
10, 2015 Stemage Skin Care, LLC (the “Plaintiff”) filed a complaint in the U.S. District Court for the Central District
of California entitled “Stemage Skin Care LLC, a North Carolina limited liability company vs. NuGene International, Inc.
et al.”
(Civil Action No.8:15-cv-01078-AG-JCG).
The complaint also names as defendants
NuGene, Inc., Ali Kharazmi, Saeed Kharazmi, Kathy Ireland Worldwide, Stephen Roseberry, Steve Rosenblum and Erik Sterling. The
complaint contains allegations of damage asserted to be grounded on: (i) copyright infringement; (ii) interference with contract;
(iii) intentional interference with prospective economic advantage; (iv) negligent interference with prospective economic advantage;
and (v) conspiracy. The complaint allegedly arises out of an August 20, 2012 agreement among the Plaintiff and kathy ireland inc.
("KI") pursuant to which KI made Kathy Ireland available to perform “Ambassador Services" as defined within
that agreement. That agreement effectively terminated in October 2014 and is the subject of a separate arbitration with KI and
Kathy Ireland before the American Arbitration Association.
We filed a
response denying all claims and based on our review of the matter, we believe that the complaint is without merit. These proceedings
are at an early stage of discovery and of motions and no assurance of outcome currently can be given.
On
July 31, 2015 Star Health & Beauty, LLC (“SH&B”) filed a complaint in the U.S.
District Court for the
Northern District of Georgia entitled “Star Health & Beauty, LLC vs. NuGene, Inc. and NuGene International, Inc. Defendants”
(Case No. 1:15-cv-02634-CAP). The complaint alleges that our use of the NUGENE name and trademark infringes on SH&B’s
NUGEN name. SH&B seeks cancelation of our NUGENE trademark, as well as unspecified monetary damages. We are in the process
of early discovery to assist us in evaluating the merits of this lawsuit and intend to defend our intellectual property rights
vigorously.
As this matter is at an early stage, no assurance of outcome
currently can be given.
In October 2015, NSE Products, Inc., (“NSE”)
a Delaware corporation based in Provo, Utah, initiated actions in the US Patent and Trademark Office contesting several of the
Company's trademark registrations and applications. These actions, including Oppositions to trademark applications and Petitions
to Revoke registered marks, rely on assertions made by NSE regarding the purported likelihood of confusion and dilution of NSE's
trademarks that include the words NU SKIN. The Company is defending its registered marks and pending applications against these
actions.
From time to time, we may be involved
in routine legal proceedings, as well as demands, claims and threatened litigation that arise in the normal course of our business.
The ultimate amount of liability, if any, for any claims of any type (either alone or in the aggregate) may materially and adversely
affect our financial condition, results of operations and liquidity. In addition, the ultimate outcome of any litigation is uncertain.
Any outcome (including any for the actions described above), whether favorable or unfavorable, may materially and adversely affect
us due to legal costs and expenses, diversion of management attention and other factors. We expense legal costs in the period
incurred. We cannot assure you that additional contingencies of a legal nature or contingencies having legal aspects will not
be asserted against us in the future, and these matters could relate to prior, current or future transactions or events.
Other than that described above, we are
not currently a party to any other material legal proceedings. We are not aware of any pending or threatened litigation against
us that in our view would have a material adverse effect on our business, financial condition, liquidity, or operating results.
However, legal claims are inherently uncertain, and we cannot assure you that we will not be adversely affected in the future
by legal proceedings.
NOTE 10 – SUBSEQUENT EVENTS
Subsequent to December 31,
2015 and through the date of these financial statements, our Company suffered continued losses from operations with accompanying
lack of liquidity. In order to fund operations on a temporary basis while our Company attempted to procure a more permanent form
of financing, our Chief Executive Officer arranged for our bank to cover amounts overdrawn on our bank accounts up to $60,000 (the
“Temporary Financing”). The Temporary Financing is personally guaranteed by our Chief Executive Officer until such
time as all amounts advanced are repaid to the bank. Our Company’s officers have also personally advanced our Company over
$57,700 in funding through the date of this Annual Report.
REPORT OF THE INDEPENDENT REGISTERED
PUBLIC ACCOUNTING FIRM
To the Board of Directors and Stockholders
of
NuGene International, Inc
We have audited the accompanying consolidated
balance sheets of NuGene International, Inc and subsidiaries (“the Company”) as of December 31, 2014 and 2013 and the
related consolidated statements of operations, changes in stockholders’ equity and cash flows for the years then ended. These
consolidated financial statements are the responsibility of the Company’s management. Our responsibility is to express an
opinion on these financial statements based on our audits.
We conducted our audit in accordance with standards
of the Public Company Accounting Oversight Board (United States). Those standards require that we plan and perform the audit to
obtain reasonable assurance about whether the financial statements are free of material misstatement. The Company is not required
to have, nor were we engaged to perform, an audit of its internal control over financial reporting. Our audit included consideration
of internal control over financial reporting as a basis for designing audit procedures that are appropriate in the circumstances,
but not for the purpose of expressing an opinion on the effectiveness of the Company’s internal control over financial reporting.
Accordingly, we express no such opinion. An audit includes examining, on a test basis, evidence supporting the amounts and disclosures
in the financial statements. An audit also includes assessing the accounting principles used and significant estimates made by
management, as well as evaluating the overall financial statement presentation. We believe that our audit provides a reasonable
basis for our opinion.
In our opinion, the consolidated financial
statements referred to above present fairly, in all material respects, the consolidated financial position of NuGene International,
Inc and subsidiaries as of December 31, 2014 and 2013, and the results of its operations and cash flows for the periods described
above in conformity with accounting principles generally accepted in the United States of America.
/s/ Dov Weinstein & Co. C.P.A. (Isr)
www.wcpa.co.il
Jerusalem, Israel
March 30, 2015
NUGENE INTERNATIONAL, INC.
CONSOLIDATED BALANCE SHEETS
|
|
|
December 31,
|
|
|
|
|
2014
|
|
|
2013
|
|
|
|
|
|
|
|
|
|
|
ASSETS
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Current assets:
|
|
|
|
|
|
|
|
|
|
Cash and cash equivalents
|
|
$
|
1,344,737
|
|
|
$
|
14,301
|
|
|
Accounts receivable, net
|
|
|
15,567
|
|
|
|
7,252
|
|
|
Inventories
|
|
|
110,953
|
|
|
|
-
|
|
|
Brand participation fee, net
|
|
|
300,000
|
|
|
|
-
|
|
|
Other assets
|
|
|
15,520
|
|
|
|
-
|
|
|
Total current assets
|
|
|
1,786,777
|
|
|
|
21,553
|
|
|
Property and equipment, net
|
|
|
60,674
|
|
|
|
-
|
|
|
Security deposits
|
|
|
12,800
|
|
|
|
-
|
|
|
|
|
|
|
|
|
|
|
|
|
Total assets
|
|
$
|
1,860,251
|
|
|
$
|
21,553
|
|
|
|
|
|
|
|
|
|
|
|
|
LIABILITIES AND STOCKHOLDERS’ EQUITY
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Current liabilities:
|
|
|
|
|
|
|
|
|
|
Accounts payable and accrued liabilities
|
|
$
|
135,563
|
|
|
$
|
1,250
|
|
|
Accounts payable - related parties
|
|
|
122,084
|
|
|
|
-
|
|
|
Deferred revenues
|
|
|
234,916
|
|
|
|
-
|
|
|
Total current liabilities
|
|
|
492,563
|
|
|
|
1,250
|
|
|
Total liabilities
|
|
|
492,563
|
|
|
|
1,250
|
|
|
|
|
|
|
|
|
|
|
|
|
Commitments
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Stockholders’ equity:
|
|
|
|
|
|
|
|
|
|
Preferred stock, $0.0001 par value; 25,000,000 shares authorized; 1,917,720 and 0 shares issued and outstanding at December 31, 2014 and 2013, respectively
|
|
|
192
|
|
|
|
-
|
|
|
|
|
|
|
|
|
|
|
|
|
Common stock, $0.0001 par value; 100,000,000 shares authorized; 39,197,400 and 20,552,760 shares issued and outstanding at December 31, 2014 and 2013, respectively
|
|
|
3,920
|
|
|
|
2,055
|
|
|
Additional paid-in capital
|
|
|
2,713,016
|
|
|
|
1,054,907
|
|
|
Accumulated deficit
|
|
|
(1,349,440
|
)
|
|
|
(1,036,659
|
)
|
|
|
|
|
|
|
|
|
|
|
|
Total stockholders’ equity
|
|
|
1,367,688
|
|
|
|
20,303
|
|
|
|
|
|
|
|
|
|
|
|
|
Total liabilities and stockholders’ equity
|
|
$
|
1,860,251
|
|
|
$
|
21,553
|
|
The accompanying notes are an integral part of these financial
statements.
NUGENE INTERNATIONAL, INC.
CONSOLIDATED STATEMENTS OF OPERATIONS
|
|
|
Years Ended December 31,
|
|
|
|
|
2014
|
|
|
2013
|
|
|
|
|
|
|
|
|
|
|
Sales
|
|
$
|
723,438
|
|
|
$
|
155,197
|
|
|
|
|
|
|
|
|
|
|
|
|
Cost of goods sold
|
|
|
293,162
|
|
|
|
137,243
|
|
|
|
|
|
|
|
|
|
|
|
|
Gross profit
|
|
|
430,276
|
|
|
|
17,954
|
|
|
|
|
|
|
|
|
|
|
|
|
Operating expenses:
|
|
|
|
|
|
|
|
|
|
Wages and professional fees
|
|
|
167,927
|
|
|
|
-
|
|
|
Research and development
|
|
|
65,830
|
|
|
|
2,370
|
|
|
General and administrative
|
|
|
509,300
|
|
|
|
8,571
|
|
|
Total operating expenses
|
|
|
743,057
|
|
|
|
10,941
|
|
|
|
|
|
|
|
|
|
|
|
|
Net (loss) / income
|
|
$
|
(312,781
|
)
|
|
$
|
7,013
|
|
|
|
|
|
|
|
|
|
|
|
|
Weighted average number of common shares outstanding - basic and diluted
|
|
|
20,865,881
|
|
|
|
20,552,760
|
|
|
|
|
|
|
|
|
|
|
|
|
Net loss per share - basic and diluted
|
|
$
|
(0.01
|
)
|
|
$
|
0.00
|
|
The accompanying notes are an integral part of these financial
statements.
NUGENE INTERNATIONAL, INC.
STATEMENT OF STOCKHOLDERS' EQUITY
For the years ended December 31, 2014 and
2013
|
|
|
Preferred
Stock
|
|
|
Common
Stock
|
|
|
Additional
|
|
|
Treasury
|
|
|
Accumulated
|
|
|
Total
Stockholders’
|
|
|
|
|
Shares
|
|
|
Amount
|
|
|
Shares
|
|
|
Amount
|
|
|
Paid-In Capital
|
|
|
Stock
|
|
|
Deficit
|
|
|
Equity
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Balance, January 1, 2013
|
|
|
-
|
|
|
$
|
-
|
|
|
|
20,552,760
|
|
|
$
|
2,055
|
|
|
$
|
1,054,907
|
|
|
$
|
-
|
|
|
$
|
(1,043,672
|
)
|
|
$
|
13,290
|
|
|
Net income
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
7,013
|
|
|
|
7,013
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Balance, December 31, 2013
|
|
|
-
|
|
|
|
-
|
|
|
|
20,552,760
|
|
|
|
2,055
|
|
|
|
1,054,907
|
|
|
|
-
|
|
|
|
(1,036,659
|
)
|
|
|
20,303
|
|
|
Issuance of common stock
|
|
|
-
|
|
|
|
-
|
|
|
|
4,000,000
|
|
|
|
400
|
|
|
|
933
|
|
|
|
|
|
|
|
-
|
|
|
|
1,333
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Stock based compensation
|
|
|
-
|
|
|
|
-
|
|
|
|
1,500,000
|
|
|
|
150
|
|
|
|
8,683
|
|
|
|
-
|
|
|
|
-
|
|
|
|
8,833
|
|
|
Issuance of common stock resulting from debt conversion
|
|
|
-
|
|
|
|
-
|
|
|
|
375,000
|
|
|
|
38
|
|
|
|
374,962
|
|
|
|
-
|
|
|
|
-
|
|
|
|
375,000
|
|
|
Issuance of common stock in private placement
|
|
|
-
|
|
|
|
-
|
|
|
|
1,625,000
|
|
|
|
163
|
|
|
|
1,624,837
|
|
|
|
-
|
|
|
|
-
|
|
|
|
1,625,000
|
|
|
Recapitalization
|
|
|
1,917,720
|
|
|
|
192
|
|
|
|
11,144,640
|
|
|
|
1,114
|
|
|
|
(1,306
|
)
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
Treasury stock acquired
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
(350,000
|
)
|
|
|
-
|
|
|
|
(350,000
|
)
|
|
Cancellation of treasury stock
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
(350,000
|
)
|
|
|
350,000
|
|
|
|
-
|
|
|
|
-
|
|
|
Net loss
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
-
|
|
|
|
(312,781
|
)
|
|
|
(312,781
|
)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Balance, December 31, 2014
|
|
|
1,917,720
|
|
|
$
|
192
|
|
|
|
39,197,400
|
|
|
$
|
3,920
|
|
|
$
|
2,713,016
|
|
|
$
|
-
|
|
|
$
|
(1,349,440
|
)
|
|
$
|
1,367,688
|
|
The accompanying notes are an integral part of these financial
statements.
NUGENE INTERNATIONAL, INC.
STATEMENTS OF CASH FLOWS
|
|
|
Years ended December 31,
|
|
|
|
|
2014
|
|
|
2013
|
|
|
Cash flows from operating activities
|
|
|
|
|
|
|
|
|
|
Net (loss) / income
|
|
$
|
(312,781
|
)
|
|
$
|
7,013
|
|
|
|
|
|
|
|
|
|
|
|
|
Adjustments to reconcile net loss to net cash (used in) provided by operating activities
|
|
|
|
|
|
|
|
|
|
Acquisition of treasury stock
|
|
|
(350,000
|
)
|
|
|
-
|
|
|
Bad debt expense
|
|
|
3,000
|
|
|
|
-
|
|
|
Depreciation and amortization
|
|
|
51,255
|
|
|
|
|
|
|
Stock based compensation
|
|
|
8,833
|
|
|
|
-
|
|
|
Changes in operating assets and liabilities
|
|
|
|
|
|
|
|
|
|
Accounts receivable
|
|
|
(11,315
|
)
|
|
|
(7,252
|
)
|
|
Inventories
|
|
|
(110,953
|
)
|
|
|
-
|
|
|
Prepaid expenses
|
|
|
(3,500
|
)
|
|
|
-
|
|
|
Other current assets
|
|
|
(12,020
|
)
|
|
|
-
|
|
|
Accounts payable and accrued liabilities
|
|
|
134,312
|
|
|
|
1,250
|
|
|
Accounts payable - related parties
|
|
|
122,085
|
|
|
|
-
|
|
|
Deferred revenue
|
|
|
234,916
|
|
|
|
-
|
|
|
|
|
|
|
|
|
|
|
|
|
Net cash (used in) provided by operating activities
|
|
|
(246,168
|
)
|
|
|
1,011
|
|
|
|
|
|
|
|
|
|
|
|
|
Cash flows from investing activities
|
|
|
|
|
|
|
|
|
|
Deposits
|
|
|
(12,800
|
)
|
|
|
-
|
|
|
Brand participation fee
|
|
|
(350,000
|
)
|
|
|
-
|
|
|
Purchase of property and equipment
|
|
|
(61,929
|
)
|
|
|
-
|
|
|
|
|
|
|
|
|
|
|
|
|
Net cash used in investing activities
|
|
|
(424,729
|
)
|
|
|
-
|
|
|
|
|
|
|
|
|
|
|
|
|
Cash flows from financing activities
|
|
|
|
|
|
|
|
|
|
Proceeds from issuance of common stock
|
|
|
1,626,333
|
|
|
|
-
|
|
|
Proceeds from issuance of convertible debt
|
|
|
375,000
|
|
|
|
-
|
|
|
|
|
|
|
|
|
|
|
|
|
Net cash provided by investing activities
|
|
|
2,001,333
|
|
|
|
-
|
|
|
Net change in cash
|
|
|
1,330,436
|
|
|
|
1,011
|
|
|
Cash, beginning of year
|
|
|
14,301
|
|
|
|
13,290
|
|
|
Cash, end of year
|
|
$
|
1,344,737
|
|
|
$
|
14,301
|
|
|
|
|
|
|
|
|
|
|
|
|
Supplemental disclosure of cash flow information:
|
|
|
|
|
|
|
|
|
|
Income taxes paid
|
|
$
|
-
|
|
|
$
|
-
|
|
|
Interest paid
|
|
$
|
-
|
|
|
$
|
-
|
|
The accompanying notes are an integral part of these financial
statements.
NUGENE INTERNATIONAL, INC
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
For the period ended December 31, 2014
NOTE 1 – NATURE OF BUSINESS AND BASIS OF PRESENTATION
NuGene International, Inc. (the “Company”
or “NuGene Int’l”) was incorporated in the State of Nevada on October 31, 2013 under the name “Bling Marketing,
Inc.” Until December 29, 2014, we were a wholesaler of jewelry, principally earrings, rings and pendants (“BMI
Business”). On December 26, 2014 we entered into an Agreement and Plan of Merger (“Merger Agreement”)
with NuGene Inc., a California corporation (“NuGene”), and on December 29, 2014 (the “Closing Date”) we
filed a certificate of merger in the State of California whereby our subsidiary, NG Acquisition Inc. merged with NuGene. As a result,
NuGene, which is the surviving entity, became our wholly owned subsidiary. The transaction contemplated under the Merger
Agreement is deemed to be a reverse merger, whereby the Company (the legal acquirer) is considered the accounting acquiree and
NuGene (the legal acquiree) is considered the accounting acquirer. On January 22, 2015 we changed our name to NuGene International,
Inc., effected a 15.04 to one stock split (“Stock Split”) and changed our trading symbol from BLMK to NUGN. All amounts
shown for common stock and additional paid in capital included in these financial statements are presented post Stock Split.
On January 20, 2015, we formed NuGene BioPharma,
Inc (“BioPharma”), a wholly-owned subsidiary, in the state of California. BioPharma’s primary focus will be on
stem cell derived treatments that may require FDA approval. The Company owns 100% of the issued and outstanding capital stock of
NuGene and BioPharma.
NuGene, our wholly
owned subsidiary, was incorporated in the state of
California in December 2006. We have since developed our initial line
of cosmeceutical products based on adipose derived stem cells (undifferentiated cells found throughout the body that multiply by
cell division to replenish dying cells and regenerate tissues) as the foundation of the formulation for our products. The Company
has developed the ability to culture adult human stem cells to render human conditioned stem cell media at a proprietary concentration
which is the primary ingredient in the NuGene line of cosmeceuticals. We currently have branded this advanced skin care line solely
under the NuGene name (the “NuGene Line”).
During 2014, we focused our efforts on transitioning
to a cosmeceutical skincare business for mass distribution. With this transition and expanded attention to our consumer products
we sought to develop our marketing plan and distribution channels.
Basis of Presentation
The accompanying consolidated financial statements
have been prepared in accordance with accounting principles generally accepted in the United States. The accompanying consolidated
financial statements reflect the accounts of NuGene International, Inc. and its wholly owned subsidiaries. All significant inter-company
balances and transactions have been eliminated.
NOTE 2 – SUMMARY OF SIGNIFICANT ACCOUNTING
POLICIES
The principal accounting policies are set
out below, these policies have been consistently applied to the period presented, unless otherwise stated:
Use of Estimates
Conformity with Generally Accepted Accounting
Principles (“GAAP”) requires the use of estimates and judgments that affect the reported amounts in the financial statements
and accompanying notes. These estimates form the basis for judgments we make about the carrying values of our assets and liabilities,
which are not readily apparent from other sources. We base our estimates and judgments on historical information and on various
other assumptions that we believe are reasonable under the circumstances. GAAP requires us to make estimates and judgments in several
areas, including, but not limited to, those related to revenue recognition, collectability of accounts receivable, contingent liabilities,
fair value of financial instruments, fair value of acquired intangible assets and goodwill, useful lives of intangible assets and
property and equipment, excess and obsolete inventory, deferred tax asset valuation and income taxes. These estimates are based
on management’s knowledge about current events and expectations about actions we may undertake in the future. Actual results
could differ materially from those estimates.
Risks and Uncertainties
The Company operates in an industry that is
subject to rapid change. The Company's operations are subject to significant risk and uncertainties including financial, operational,
technological, regulatory and other risks, including the potential of business failure.
Cash and cash equivalents
Cash and cash equivalents include investments
with initial maturities of three months or less. The Company maintains its cash balances at credit-worthy financial institutions
that are insured by the Federal Deposit Insurance Corporation up to $250,000.
Inventories
The Company’s skincare inventories consist
of raw materials and finished goods and are valued at the lower of cost (first-in, first-out) or net realizable value. The Company
evaluates its inventory for excess and obsolescence on a regular basis. To determine if the cost of the Company's inventory should
be written down, current and anticipated demand, customer preferences and age of the merchandise are considered. For the years
ending December 31, 2014 and 2013 the Company did not recognize any charges to expense associated with excess and obsolete inventory
cost adjustments.
Property, Plant and Equipment
Property and equipment consist of office furniture,
computer equipment, laboratory equipment and improvements made to our leased office and are stated at cost less accumulated depreciation.
Depreciation is calculated using the straight-line method over the three year useful life of the assets or the remaining term of
the lease. When property and equipment are retired or otherwise disposed of, the related cost and accumulated depreciation are
removed from the respective accounts, and any gain or loss is included in operations. Expenditures for maintenance and repairs
are expensed as incurred.
Intangible Assets
Identifiable intangible assets with indefinite
lives are not amortized, but instead are tested for impairment annually, or more frequently if circumstances indicate a possible
impairment may exist. Intangible assets with estimable useful lives are amortized over their respective estimated useful lives,
generally on a straight-line basis, and are reviewed for impairment whenever events or changes in circumstances indicate that the
carrying value may not be recoverable.
Website Development
Under Accounting Standards Codification (“ASC”)
350-50 –
Intangibles – Goodwill and Other – Website Development Costs
, costs and expenses incurred during
the planning and operating stages of the Company’s website development are expensed as incurred through research and development.
The Company accounts for the development of its website by expensing all costs associated with the planning of the website as incurred
and capitalizing the costs to develop the website. Once the website is available for use, the asset will be amortized over its
useful life on a straight line basis, estimated to be 3 years, and tested for impairment annually.
Accounts payable and accrued expenses
Accounts payable and accrued expenses are carried
at amortized cost and represent liabilities for goods and services provided to the Company prior to the end of the financial year
that are unpaid and arise when the Company becomes obliged to make future payments in respect of the purchase of these goods and
services.
Deferred Revenue
As of December 31, 2014, we recorded $234,916
in deferred revenues under current liabilities related to the sale of products undelivered as of year-end. Deferred revenues will
be recognized as sales in 2015.
Revenue Recognition
In addition to revenues recognized through
the sale of our NuGene Line of skin care products, we generated revenues from an affiliate, Advanced Surgical Partners (“ASP”)
which is also owned by our CEO and Chairman of the Board, Messrs. M. Ali and M. Saeed Kharazmi, respectively. Revenues generated
from ASP resulted from NuGene providing Plasma Rich Platelet and Stem Cell injections for orthopedic and plastic surgery procedures
to ASP. We provided these services to ASP as we transitioned into the cosmeceutical side of NuGene and expect to minimize these
services to ASP in early 2015.
In accordance with ASC 605 -
Revenue Recognition
,
the Company recognizes revenue from product sales when the product has been ordered by the customer, the selling price is fixed
or determinable, the product is shipped to the customer, title has transferred and collectability is reasonably assured.
Concentration of Revenues
For the period ending December 31, 2014 and 2013, the Company recognized
revenues from ASP in the amount of approximately $481,000 and $135,000 or 66% and 87% of total revenues, respectively. We provided
these services to ASP as we transitioned into the cosmeceutical side of NuGene and expect to minimize these services to ASP in
early 2015.
Excluding sales to ASP, 51% of our revenues
during the year ended December 31, 2014 were derived from a single wholesale distributor. We do not have long-term purchase commitments
from this wholesaler. This distributor purchases products from us on a purchase order basis on its standard terms. The distributor
is under no obligation to continue to purchase our products. The loss of the wholesaler, a material reduction in its purchases
or the cancellation of product orders or unexpected returns of unsold products could decrease our revenues and impede our future
growth prospects.
Cost of Sales
Cost of sales includes all of the costs to
manufacture the Company’s products. For products manufactured in the Company’s own facilities, such costs include raw
materials and supplies, direct labor and factory overhead. For products manufactured for the Company by third-party contractors,
such cost represents the amounts invoiced by the contractors.
Royalty Expense
The Company recognizes royalty expenses according
to its license agreement. The licensee will pay the licensor up to 5% of net sales or a minimum guaranteed royalty of $100,000
beginning in year one and increasing in annual increments of $50,000 for the first eight years of the contract.
Royalties
are expensed in the statements of operation in the period that the related revenues are recognized, in cost of goods sold.
Research and Development
Internal research and development costs are
expensed as incurred. Third-party research and development costs are expensed when the contracted work has been performed. Research
and development consists of website development expenses.
Allowance for Doubtful Accounts
The Company maintains allowances for doubtful
accounts for estimated losses resulting from the inability of its customers to make required payments. Account balances are charged
off against the allowance after all means of collection have been exhausted and the potential for recovery is considered remote.
Our allowance for doubtful accounts was $3,000 and $0 at December 31, 2014 and 2013, respectively.
Share Based Payments
The Company recognizes compensation expense
for all equity–based payments in accordance with ASC 718 -
Share-based payments
. Under fair value recognition provisions,
the Company recognizes equity–based compensation net of an estimated forfeiture rate and recognizes compensation cost only
for those shares expected to vest over the requisite service period of the award.
Share-based payments to employees, including
grants of employee stock options, are recognized as compensation expense in the financial statements based on their fair values.
That expense is recognized over the period during which an employee is required to provide services in exchange for the award,
known as the requisite service period (usually the vesting period).
The Company accounts for share–based
payments granted to non–employees in accordance with ASC 505 -
Equity Based Payments to Non–Employees
. The Company
determines the fair value of the stock–based payment as either the fair value of the consideration received or the fair value
of the equity instruments issued, whichever is more reliably measurable. If the fair value of the equity instruments issued is
used, it is measured using the stock price and other measurement assumptions as of the earlier of either (1) the date at which
a commitment for performance by the counterparty to earn the equity instruments is reached, or (2) the date at which the counterparty’s
performance is complete.
Earnings Per Share of Common Stock
The Company computes earnings per share in
accordance with ASC 260 -
Earnings Per Shar
e. ASC 260 requires presentation of both basic and diluted earnings per share
(“EPS”) on the face of the income statement. Basic EPS is calculated by dividing the profit or loss attributable to
common shareholders of the Company by the weighted average number of common shares outstanding during the period. Diluted EPS is
determined by adjusting the profit or loss attributable to common shareholders and the weighted average number of common shares
outstanding for the effects of all potential dilutive common shares, such as stock issuable pursuant to the exercise of stock warrants
or the conversion of preferred stock into common stock.
Common stock equivalents totaling, 2,417,720
on December 31, 2014 were not included in the computation of diluted earnings per share in 2014 on the consolidated statement of
operations due to the fact that the Company reported a net loss in 2014 and to do so would be anti-dilutive.
Income Taxes
Income taxes are accounted for in accordance
with ASC Topic 740 -
Income Taxes
. Under the asset and liability method, deferred tax assets and liabilities are recognized
for the future consequences of differences between the financial statement carrying amounts of existing assets and liabilities
and their respective tax bases (temporary differences). Deferred tax assets and liabilities are measured using tax rates expected
to apply to taxable income in the years in which those temporary differences are recovered or settled. Valuation allowances for
deferred tax assets are established when it is more likely than not that some portion or all of the deferred tax assets will not
be realized.
Management evaluates its tax positions on an
annual basis and has determined that as of December 31, 2014 no additional accrual for income taxes is necessary. The federal and
state income tax returns of the Company are subject to examination by the IRS and state taxing authorities, generally for three
years after they were filed.
Fair
Value of Financial Instruments
The Company measures assets and liabilities
at fair value based on an expected exit price as defined by the authoritative guidance on fair value measurements, which represents
the amount that would be received on the sale of an asset or paid to transfer a liability, as the case may be, in an orderly transaction
between market participants. As such, fair value may be based on assumptions that market participants would use in pricing an asset
or liability. The authoritative guidance on fair value measurements establishes a consistent framework for measuring fair value
on either a recurring or nonrecurring basis whereby inputs, used in valuation techniques, are assigned a hierarchical level.
The following are the hierarchical levels of
inputs to measure fair value:
|
|
- Level 1:
|
Quoted prices in active markets for identical instruments;
|
|
|
- Level 2:
|
Other significant observable inputs (including quoted prices
in active markets for similar instruments);
|
|
|
- Level 3:
|
Significant unobservable inputs (including assumptions
in determining the fair value of certain investments).
|
The carrying values for cash and cash equivalents,
accounts receivable, other current assets, accounts payable and accrued liabilities, and deferred revenue approximate their fair
value due to their short maturities.
Recent Accounting Standards Updates
In June 2014, the Financial Accounting Standards
Board (“FASB”) issued Accounting Standards Update (“ASU”) No. 2014-10—
Development Stage Entities
(Topic 915): Elimination of Certain Financial Reporting Requirements, Including an Amendment to Variable Interest Entities Guidance
in Topic 810, Consolidation
. ASU 2014-10 eliminates several of the reporting requirements for development stage entities, including
the requirement to present inception to date information in the statements of income, cash flows, and shareholder equity, and to
label the financial statements as those of a development stage entity. ASU 2014-10 also clarifies that the guidance in ASC Topic
275
Risks and Uncertainties
, is applicable to entities that have not commenced principal operations, and eliminates an exception
to the sufficiency-of-equity risk criterion for development stage entities, and will require all reporting entities that have an
interest in development stage enterprises to apply consistent consolidation guidance for variable interest entities. ASU 2014-10
is effective for all annual reporting periods beginning after December 15, 2014, with early adoption permitted. The Company has
elected to early adopt these amendments and accordingly has not labeled the financial statements as those of a development stage
entity and has not presented inception-to-date information on the respective financial statements.
Other accounting standards that have been issued
or proposed by FASB that do not require adoption until a future date are not expected to have a material impact on the consolidated
financial statements upon adoption. The Company does not discuss recent pronouncements that are not anticipated to have an impact
on or are unrelated to its financial condition, results of operations, cash flows or disclosures.
NOTE 3 – INVENTORIES
Inventories consist of the following:
|
|
|
December 31,
|
|
|
|
|
2014
|
|
|
2013
|
|
|
Raw materials
|
|
$
|
52,045
|
|
|
$
|
-
|
|
|
Finished goods
|
|
|
58,908
|
|
|
|
-
|
|
|
Total Inventories
|
|
$
|
110,953
|
|
|
$
|
-
|
|
NOTE 4 – INTANGIBLE ASSETS
Licensee Agreement
In November 2014 we entered into a License
Agreement with kathy ireland Worldwide
®
("kiWW
®
") whereby we licensed the right to utilize
the trademarks and rights to the name, likeness and visual representations of Kathy Ireland in connection with our cosmeceutical
line of products containing adult human adipose stem cell derived or containing biologically active or biologically derived ingredients.
The initial term of the license is for eight years and it may be renewed for up to an additional four years.
In accordance with the License Agreement, we
will pay 5% of the net sales for all licensed products sold and collected under the licensed marks or a minimum guaranteed royalty
of $100,000 in year one of the License Agreement. The minimum guaranteed royalty increases $50,000 each year in years two through
eight of the License Agreement.
Additionally, we are obligated to pay an annual
Brand Participation Fee to kiWW
®
which provides for general advertising, good will and promotion of the overall
kathy ireland brand. NuGene paid kiWW $350,000 effective upon execution of the License as a Brand Participation fee for Contract
Year 1. The Brand Participation Fee for Contract Years 2 through 8 is $50,000 annually with an additional 1% of the total gross
sales of Licensed Products of the prior year beginning in Contract Year 4. The net carrying value of the Brand Participation Fee
at December 31, 2014 was $300,000, net of amortization expense of $50,000.
NOTE 5 – PROPERTY AND EQUIPMENT
Property and equipment, net consist of the
following:
|
|
|
December 31,
|
|
|
|
|
2014
|
|
|
2013
|
|
|
Software
|
|
$
|
2,139
|
|
|
$
|
-
|
|
|
Equipment
|
|
|
35,698
|
|
|
|
-
|
|
|
Leasehold improvements
|
|
|
24,092
|
|
|
|
-
|
|
|
|
|
|
|
|
|
|
|
|
|
Property and equipment, gross
|
|
|
61,929
|
|
|
|
-
|
|
|
Accumulated depreciation
|
|
|
(1,255
|
)
|
|
|
-
|
|
|
Property and equipment, net
|
|
$
|
60,674
|
|
|
$
|
-
|
|
Depreciation expense for the year ended December
31, 2014 was $1,255.
NOTE 6 – STOCKHOLDERS’ EQUITY
Merger Agreement
On December 26, 2014 we entered into a Merger
Agreement with NuGene and on December 29, 2014 (the “Closing Date”) we filed a certificate of merger in the State of
California whereby our subsidiary, NG Acquisition Inc. merged with NuGene. As a result, NuGene, which is the surviving entity,
became our wholly owned subsidiary.
In connection with the Merger Agreement, we
entered into a Business Transfer and Indemnity Agreement dated December 29, 2014 (the “Indemnity Agreement”) with our
former Chief Executive Officer and Director, Dena Kurland providing for:
|
|
1.
|
The transfer of our jewelry business operations existing on the date of the Indemnity Agreement;
|
|
|
2.
|
The assumption by Ms. Kurland of all liabilities of our Company and the indemnification by Ms.
Kurland holding our Company harmless for any and all liabilities arising at or before the date of the Indemnity Agreement;
|
|
|
3.
|
The payment by NuGene to Ms. Kurland of $350,000 in cash; and
|
|
|
4.
|
The surrender by Ms. Kurland of 15,000,000 shares (before giving effect to the Stock Split discussed
below) (the "Indemnity Shares") of our Company’s common stock representing 95% of the then outstanding common stock
(all of which shares have been cancelled by the Company and are now included in the Company’s pool of authorized but unissued
shares.).
|
In conjunction with the Merger Agreement, all
of the issued and outstanding shares of NuGene at December 29, 2014 were exchanged for 26,052,760 shares of NuGene Int’l
common stock and 1,917,720 NuGene Int’l Series A Preferred Stock.
Common Stock
On December 23, 2014, we amended our articles
of incorporation to provide for the authorization of 100,000,000 shares of Company common stock with a par value of $0.0001.
On December 26, 2014 our board of directors
approved a 15.04 to one stock split (“Stock Split”) in the form of a stock dividend to holders of our common stock
as of that date. To effect that board actions, each recipient of the stock dividend would receive 14.04 additional shares
of common stock for every share of common stock held. Unless otherwise noted, all share numbers shown in these consolidated financial
statements reflect the effects of the Stock Split as approved by our board of directors.
On December 29, 2014 we completed the sale
of 2,000,000 shares of our common stock to 18 purchasers (“Stock Placement”) for proceeds totaling $2,000,000, including
(a) $1,625,000 of cash and (b) automatic conversion of $375,000 of NuGene promissory notes. Stock Placement proceeds totaling $350,000
were used as payment to Ms. Kurland in accordance with the Indemnity Agreement and $75,000 were used as repayment of a short term
loan associated with the license with kathy ireland Worldwide, Inc. The remaining proceeds of approximately $1.2 million will be
used to continue funding our operations and grow our product line.
Prior to the Merger Agreement, NuGene received
proceeds totaling $1,333 for the purchase of 4,000,000 shares of common stock from 5 investors and issued 1,500,000 shares of common
stock, valued at $500, to a consultant for services rendered. As noted above, these shares were exchanged for shares of the Company
in conjunction with the Merger Agreement.
Preferred Stock
On December 23, 2014, we amended our articles
of incorporation to provide for the authorization of 25,000,000 shares of Preferred Stock with a par value of $0.0001. We designated
2,000,000 shares of Preferred Stock as “Series A Convertible Preferred Stock.” The Series A Preferred Stock is (i)
initially convertible into common stock at a ratio of one to one, (ii) as long as there are a minimum of 900,000 shares of Series
A Preferred Stock outstanding, the holders of the Series A Preferred Stock have the right to elect a majority of the board of directors
and (iii) the holders of the Series A Preferred Stock, generally voting as a class with the holders of common stock, have three
times the number of votes available for every vote of available common stock. As of December 31, 2014, we had 1,917,720 shares
of Series A Preferred Stock outstanding which were issued to the founders of NuGene, Messrs. M. Ali and M. Saeed Kharazmi, in conjunction
with the Merger Agreement. M. Ali and M. Saeed Kharazmi each own 50% of the outstanding Series A Preferred Stock and are the Company’s
Chief Executive Officer and Chairman of the Board, respectively.
Warrants
At the time of the merger, an outstanding warrant
to acquire shares of NuGene Inc. was exchanged for a warrant of Bling Marketing Inc. The warrant on its issuance, after giving
effect to the Stock Split, evidenced the right of the holder to acquire up to 500,000 shares of common stock of the Company, had
a strike price of $2.50 per share, was not exercisable for 12 months (the "Initial Exercise Date"), and any shares acquired
thereunder upon exercise thereafter would be locked up and could not be sold for six months following the Initial Exercise Date.
The warrant was issued to an entity that is not directly or indirectly an affiliate of either NuGene or of the Company.
NOTE 7 – CONVERTIBLE PROMISSORY NOTES
During the fourth quarter of 2014, we entered
into three convertible promissory notes (the “Notes”) totaling $375,000. Proceeds from the Notes were used primarily
to pay for the Brand Participation Fee discussed in Note 4. The Notes were converted into 375,000 shares of Company common stock
in conjunction with the Stock Placement discussed in Note 6.
NOTE 8 – INCOME TAXES
Prior to the Merger Agreement, NuGene had elected
to be treated as a "sub-chapter S corporation". Under the Internal Revenue Code NuGene paid no federal taxes and generally
paid a state of California income tax of about 1.5% of taxable income. NuGene terminated their sub-chapter S election in December
2014 and accordingly is now subject to taxation as a C corporation.
The provision (benefit) for income taxes for
the years ended December 31, 2014 and 2013 differs from the amount which would be expected as a result of applying the statutory
tax rates to the losses before income taxes due primarily to changes in the valuation allowance to fully reserve net deferred tax
assets.
Realization of deferred tax assets is dependent
upon sufficient future taxable income during the period that deductible temporary differences and carry-forwards are expected to
be available to reduce taxable income.
|
|
|
December 31,
|
|
|
Deferred tax assets:
|
|
2014
|
|
|
2013
|
|
|
Net operating loss before non-deductible items
|
|
$
|
(312,781
|
)
|
|
$
|
-
|
|
|
Tax rate
|
|
|
34
|
%
|
|
|
34
|
%
|
|
Total deferred tax assets
|
|
|
106,345
|
|
|
|
-
|
|
|
Less: Valuation allowance
|
|
|
(106,345
|
)
|
|
|
-
|
|
|
|
|
|
|
|
|
|
|
|
|
Net deferred tax assets
|
|
$
|
-
|
|
|
$
|
-
|
|
The Company has provided a valuation allowance
against the full amount of the deferred tax asset due to management’s uncertainty about its realization. As of December 31,
2014, the Company had approximately $459,550 in tax loss carryforwards that can be utilized future periods to reduce taxable income,
and expire by the year 2033.
NOTE 9 – RELATED PARTY TRANSACTIONS
Parties are considered to be related if one
party has the ability to control or exercise significant influence over the other party in making financial and operating decisions.
A related party transaction is considered to
be a transfer of resources or obligations between related parties, regardless of whether or not a price is charged.
The following entities have been identified
as related parties:
|
Ali Kharazmi
|
|
Chief Executive Officer, President, Board Member and greater than 10% shareholder
|
|
Mohammed Kharazmi
|
|
Chairman of the Board, Acting Chief Financial Officer and greater than 10% shareholder
|
|
H&K Global Enterprises, Inc
|
|
Company with common ownership and management
|
|
Genetics Institute of Anti Aging
|
|
Company with common ownership and management
|
|
Applied M.A.K. Enterprises, Inc (“MAK”)
|
|
Company with common ownership and management
|
|
Advanced Surgical Partners, LLC (“ASP”)
|
|
Company with common ownership and management
|
|
Center for Weight Management
|
|
Company with common ownership and management
|
|
Center for Regenerative Science, Inc
|
|
Company with common ownership and management
|
The following material related party transactions
have taken place during the year:
|
|
|
December 31,
|
|
|
|
|
2014
|
|
|
2013
|
|
|
ASP
|
|
$
|
45,000
|
|
|
$
|
-
|
|
|
MAK
|
|
|
40,084
|
|
|
|
-
|
|
|
Ali Kharazmi
|
|
|
37,000
|
|
|
|
-
|
|
|
Accounts payable - Related Parties
|
|
$
|
122,084
|
|
|
$
|
-
|
|
From time to time, the Company is loaned funds
from its various related parties for working capital purposes. These advances bear no interest and are due on demand. The total
amounts loaned by ASP and MAK and $25,000 of the amount loaned by Ali Kharazmi have been paid back as of March 28, 2015.
|
|
|
Year ended December 31,
|
|
|
|
|
2014
|
|
|
2013
|
|
|
ASP
|
|
$
|
481,000
|
|
|
$
|
135,000
|
|
|
Related Party Sales included in Total Sales
|
|
$
|
481,000
|
|
|
$
|
135,000
|
|
As discussed previously, revenues generated
from ASP resulted from NuGene providing Plasma Rich Platelet and Stem Cell injections for orthopedic and plastic surgery procedures
to ASP. We provided these services to ASP as we transitioned into the cosmeceutical side of NuGene and expect to minimize these
services to ASP in early 2015. The transactions are on terms no more favorable than between two parties at an arm’s length.
The Company subleases office space from ASP
as discussed further in Note 10.
The Company does not have employment contracts
with its two key employees, the controlling shareholders, who are officers and directors of the Company.
NOTE 10 – COMMITMENTS AND CONTINGENCIES
We entered into a sub-lease
of our corporate offices in Orange County, California, on December 1, 2014. The property consists of approximately 11,000 square
feet of office, manufacturing and assembly space. After completing various leasehold improvements, we moved in to the property
in early March 2015. We currently sublease the property from ASP, an affiliate of the Company, for $12,000 per month, consistent
with the amount that is charged to ASP by the landlord. Effective February 5, 2015, ASP entered into a new five year lease for
the property beginning July 1, 2015. The lease calls for a base rent of $9,968 per month plus payment of approximately $2,000 per
month for common area maintenance. The lease includes annual increases in the monthly lease payments of approximately 3% each year.
Effective December 1, 2014, the Company entered
into three consulting agreements. The terms of each agreement is 36 months. Each consultant is entitled to base compensation of
$3,000 per month and 33,333 shares of restricted shares of common stock per annum, issued on the anniversary of the effective date
of the agreement.
From time to time, we may be involved in routine
legal proceedings, as well as demands, claims and threatened litigation that arise in the normal course of our business. The ultimate
amount of liability, if any, for any claims of any type (either alone or in the aggregate) may materially and adversely affect
our financial condition, results of operations and liquidity. In addition, the ultimate outcome of any litigation is uncertain.
Any outcome, whether favorable or unfavorable, may materially and adversely affect us due to legal costs and expenses, diversion
of management attention and other factors. We expense legal costs in the period incurred. We cannot assure you that additional
contingencies of a legal nature or contingencies having legal aspects will not be asserted against us in the future, and these
matters could relate to prior, current or future transactions or events.
We are not currently a party to any material
legal proceedings. We are not aware of any pending or threatened litigation against us that in our view would have a material adverse
effect on our business, financial condition, liquidity, or operating results. However, legal claims are inherently uncertain, and
we cannot assure you that we will not be adversely affected in the future by legal proceedings.
NOTE 11 – SUBSEQUENT EVENTS
On January 20, 2015, the Company established
a wholly owned operating subsidiary called NuGene BioPharma, Inc, which will primarily focus on stem cell derived treatments that
may require FDA approval.
On February 5, 2015 the Company amended its
office sublease effective July 1, 2015, to a five year term ending June 30, 2020, with base rental per month of $9,968.
On March 17
,
2015, BioPharma entered
into an Intellectual Property Asset Purchase Agreement (“IP Agreement”) with Dr. H.R. Christopher O'Brien, PharmD,
an individual, thereby acquiring all of the rights, title and interest in and to SkinGuardian, an FDA-approved (monographed) skin
protectant, antiseptic, and moisturizing lotion, including but not limited to (i) Patent (Pub. No.: US 2009/0105195), (ii) the
Trademark (Registration No.: 85082446); (iii) all formulae and derivatives; (iv) FSA work product, clinical trial data, etc.; (v)
the goodwill; and (vi) any and all other rights, title, and interest in and to the SkinGuardian™ product (collectively, the
“Intellectual Property”) and Intellectual Property Rights, free and clear of any liabilities, claims, liens or encumbrances.
As consideration for the purchase of the Intellectual
Property, including the tangible and intangible assets primarily used or associated with the Intellectual Property, the Company
has agreed to issue 250,000 shares of common stock to Mr. O’Brien. The common stock is exempt from registration pursuant
to Section 4(a)(2) of the Securities Act of 1933, as amended, and includes a lock-up agreement whereby those shares may not be
sold for a period of 18 months following the closing of the IP Agreement.
In accordance with ASC 855-10, Company management reviewed all material
events through the date of this report and determined that there are no additional material subsequent events to report.
Shares
Common
Stock
Warrants
to Purchase _____________ Shares of Common Stock

PROSPECTUS
Joseph Gunnar & Co.
PART II
INFORMATION NOT REQUIRED IN PROSPECTUS
|
|
Item 13.
|
Other Expenses of Issuance and Distribution.
|
The following table sets forth the fees
and expenses to be incurred in connection with the registration of the securities being registered hereby, all of which will be
borne by the registrant. Except for the SEC registration fee, all amounts are estimates.
|
Description
|
|
Amount
|
|
|
SEC registration fee
|
|
$
|
|
|
|
FINRA filing fee
|
|
|
*
|
|
|
NASDQ listing fee
|
|
|
*
|
|
|
Accounting fees and expenses
|
|
|
*
|
|
|
Legal fees and expenses
|
|
|
*
|
|
|
Transfer agent and registrar fees and expenses
|
|
|
*
|
|
|
Printing and engraving expenses
|
|
|
*
|
|
|
Miscellaneous expenses
|
|
|
*
|
|
|
Total expenses
|
|
$
|
*
|
|
*
To be filed by amendment.
|
|
Item 14.
|
Indemnification of Directors and Officers.
|
Subsection 1 of Section 78.7502 of
the Nevada Revised Statutes empowers a corporation to indemnify any person who was or is a party or is threatened to be made a
party to any threatened, pending or completed action, suit or proceeding, whether civil, criminal, administrative or investigative
(other than an action by or in the right of the corporation) by reason of the fact that he or she is or was a director, officer,
employee or agent of the corporation, or is or was serving at the request of the corporation as a director, officer, employee or
agent of another corporation or other enterprise, against expenses (including attorneys’ fees), judgments, fines and amounts
paid in settlement actually and reasonably incurred by him or her in connection with such action, suit or proceeding if he or she
(i) is not liable pursuant to Section 78.138 of the Nevada Revised Statutes or (ii) acted in good faith and in a
manner which he or she reasonably believed to be in or not opposed to the best interests of the corporation, and, with respect
to any criminal action or proceeding, had no reasonable cause to believe his or her conduct was unlawful. Section 78.138 of
the Nevada Revised Statutes provides that, with certain exceptions, a director or officer is not individually liable to the corporation
or its stockholders or creditors for any damages as a result of any act or failure to act in his or her capacity as a director
or officer unless it is proven that (i) his or her act or failure to act constituted a breach of his or her fiduciary duties
as a director or officer, and (ii) his or her breach of those duties involved intentional misconduct, fraud or a knowing violation
of law.
Subsection 2 of Section 78.7502 empowers
a corporation to indemnify any person who was or is a party or is threatened to be made a party to any threatened, pending or completed
action or suit by or in the right of the corporation to procure a judgment in its favor by reason of the fact that such person
acted in any of the capacities set forth above against expenses, including amounts paid in settlement and attorneys’ fees
actually and reasonably incurred by him or her in connection with the defense or settlement of such action or suit if he or she
(i) is not liable pursuant to Section 78.138 of the Nevada Revised Statutes, or (ii) acted in good faith and in
a manner which he or she reasonably believes to be in or not opposed to the best interests of the corporation, except that no indemnification
may be made in respect of any claim, issue or matter as to which such person shall have been adjudged by a court of competent jurisdiction
to be liable to the corporation or for amounts paid in settlement to the corporation, unless and only to the extent that the court
in which such action or suit was brought or other court of competent jurisdiction determines that, in view of all the circumstances
of the case, such person is fairly and reasonably entitled to indemnity for such expenses as the court deems proper.
Section 78.7502 further provides that
to the extent that a director, officer, employee or agent of a corporation has been successful in the defense of any action, suit
or proceeding referred to in subsections (1) and (2), or in the defense of any claim, issue or matter therein, he or she shall
be indemnified against expenses (including attorneys’ fees) actually and reasonably incurred by him or her in connection
with the defense. Subsection 3 of Section 78.751 of the Nevada Revised Statutes provides that the indemnification provided
for by Section 78.7502 does not exclude any other rights to which the indemnified party may be entitled (except that indemnification
will generally not be available to a director or officer if a final adjudication establishes that his or her acts or omissions
involved intentional misconduct, fraud or a knowing violation of the law and was material to the cause of action) and that the
indemnification shall continue for directors, officers, employees or agents who have ceased to hold such positions, and inures
to the benefit of their heirs, executors and administrators.
Section 78.752 of the Nevada Revised
Statutes empowers the corporation to purchase and maintain insurance or make other financial arrangements on behalf of a person
who is or was a director, officer, employee or agent of the corporation, or is or was serving at the request of the corporation
as a director, officer, employee or agent of another corporation or other enterprise, for any liability asserted against him or
her and expenses incurred by him or her in any such capacity or arising out of his or her status as such whether or not the corporation
has the power to indemnify him or her against such liabilities or expenses. We maintain a customary directors’ and officers’
liability insurance policy.
Our Amended and Restated Articles of Incorporation
provide for indemnification of our directors and officers, substantially identical in scope to that permitted under applicable
Nevada law. Our Amended and Restated Articles of Incorporation provide, compliant with Subsection 2 of Section 78.751, that
the expenses of our directors and officers incurred in defending any action, suit or proceeding, whether civil, criminal, administrative
or investigative, must be paid by us as they are incurred and in advance of the final disposition of the action, suit or proceeding,
upon delivery, if required by applicable Nevada law, of an undertaking by or on behalf of the director or officer to repay all
amounts so advanced if it is ultimately determined that the director or officer is not entitled to be indemnified by us.
|
|
Item 15.
|
Recent Sales of Unregistered Securities.
|
In December, 2014 we issued securities
in connection with the merger transaction described above in the Overview Section of Management’s Discussion and Analysis
of Financial Conditions and Results.
On December 29, 2014, we completed the
sale of 2,000,000 shares of our common stock to 18 purchasers for proceeds totaling $2,000,000, including (a) $1,625,000 of cash
and (b) automatic conversion of promissory notes in the principal amount of $375,000.
On August 14, 2015, we granted an option
to purchase up to 1,000,000 shares of our common stock to each of two employees.
On September 25, 2015, we entered into
a Securities Purchase Agreement and issued a 15% Promissory Note with the principal face value of $500,000 (the “15% Note”)
to an accredited investor. Under the terms of the 15% Note, all principal and related accrued interest outstanding are due and
payable to the noteholder upon the earlier of: (i) September 25, 2016; or (ii) within ten business days after the consummation
of an equity or convertible debt financing with aggregate gross proceeds of at least $1,000,000.
In November 2015, the Company entered into
a consulting agreement with a third party for consulting services over six months. The Company agreed to pay the consultant 90,000
in shares of the Company’s restricted stock, which were issued over six months. The Company issued 30,000 shares of restricted
stock in February 2016, and the remaining 60,000 shares were issued in June 2016.
During November 2015, we issued a one year
10% promissory notes payable (the “10% Note”) to a purchaser for cash proceeds totaling $50,000. In the event that
we secure any future financing with aggregate gross proceeds of at least $1 million while the 10% Note is outstanding, the 10%
Note and all accrued interest therefrom will be immediately due and payable within ten business days of the closing of such financing.
The holder may also convert any unpaid principal under the 10% Note into any funding instrument entered into by our Company for
a period of 180 days after the date of the 10% Note. The full interest of 10% of the borrowings under the 10% Note ($5,000) is
due irrespective of whether paid at maturity or when required to be prepaid.
On November 30, 2015 and on four subsequent
dates during December 2015, four accredited investors (the “Lenders”) advanced a total of $410,000 to the Company.
The borrowings (“Advances”) were not accompanied by documentation of the terms of the Advances. However, there were
various discussions as to the repayment terms, the interest expected to be paid to the Lenders and additional equity of our Company
to be issued to the Lenders in connection with the Advances.
On March 17, 2016, we entered into a consulting
agreement for services over one year. In connection with the agreement, we granted the consultant 200,000 shares of our fully vested
common stock as consideration for the consultant’s services.
On March 30, 2016, an accredited investor
advanced our Company $244,975. The advance was undocumented. The advance was subsequently documented on April 4, 2016 in the form
of our $275,000 promissory note. We also issued 50,000 shares of our common stock to the investor. The note bears interest at rate
of 8% per annum and is convertible into shares of our common stock at the rate of $0.70 per share.
On April 4, 2016, we issued our promissory
note in the amount of $575,000 to an accredited investor in exchange for an aggregate purchase price of $575,000. The terms of
the Note call for an interest rate of 15%, due one year from the date of issuance, though all accrued and unpaid interest and all
other amounts payable under the Note are due to the investor within ten (10) business days after the closing by the Company of
an equity or convertible debt financing in one or more series of transactions, with aggregate gross proceeds of at least $1 million.
In April, 2016, the Company entered into
a consulting agreement with a third party for consulting services over a year. The 200,000 shares of restricted stock award vested
on April 25, 2016.
In May 2016, we entered into agreements
with two employees that hold options to purchase up to 1,000,000 shares of our common stock each. The agreements modified the strike
price of their outstanding options from the previous strike prices of $1.50 to $0.55 per share. All other terms of the previous
option agreements remained unchanged.
During January through June 2016, three
accredited investors (the “Lenders”) advanced a total of $140,000 to the Company. The borrowings were not accompanied
by documentation of the nature of the borrowings. However, there were general discussions as to the repayment terms and the interest
expected of 10% per annum to be paid to the Lenders in connection with the borrowings.
On July 19, 2016, we issued a sixty day
15% promissory note payable to an accredited investor for cash proceeds totaling $35,000. In the event that we secure any future
financing with aggregate gross proceeds of at least $500,000 while the promissory note is outstanding, the promissory note and
all accrued interest therefrom will be immediately due and payable within ten business days of the closing of such financing.
During July and August 2016, four accredited
investors (the “Lenders”) advanced a total of $435,000 to the Company. The borrowings (“Advances”) were
not accompanied by documentation of the nature of the Advances. However, there were general discussions as to the repayment terms
and the interest expected to be paid to the Lenders in connection with the Advances.
During the three months ended September
30, 2016, certain accredited investors advanced a total of $925,000 (the “Advances”) to the Company with a right to
convert at no greater than $0.40 per share. We received aggregate proceeds in the amount of $925,000. In connection with the Advances, the
Company also issued warrants to purchase 462,500 shares of our common stock at an exercise price $0.60 per share with a 5 year
term. The Advances were not accompanied by documentation. However, there were general discussions as to the repayment terms and
the interest expected of 10% per annum to be paid to the Lenders in connection with the borrowings.
On September 22, 2016, the Company entered
into new compensation agreements and arrangements with two of its directors. Our Chairman was issued (i) 2,000,000 shares of our
fully vested common stock; (ii) restricted stock units for 2,000,000 shares of the Company’s common stock; and, (iii) a stock
option to acquire 2,352,619 shares of our common stock, with an exercise price of $0.61 per share. Our interim chief financial
officer/board member was issued (i) restricted stock units for 2,000,000 shares of the Company’s common stock; and, (iii)
a stock option to acquire 2,352,619 shares of our common stock, with an exercise price of $0.61 per share.
On October 21, 2106, the Company issued
an accredited investor a promissory note in the total principal amount of $267,500 with 9% annual interest rate, which will be
due on October 21, 2017. The Company received $250,000 of gross proceeds and granted a warrant to purchase 222,916 shares of common
stock at $0.60 per share.
On November 2, 2016, the Company issued
certain accredited investors promissory notes (the “Notes”) in the total principal amount of $480,000 with a 5% annual
interest rate, which will be due on April 28, 2017. The Company received $400,000 of gross proceeds from the Notes. The Company
also granted to the investors warrants to purchase 1,428,572 shares of common stock at no greater than $0.47 per share.
No underwriters were used in the foregoing
transactions. The securities were issued in reliance on the exemptions from registration provided by Section 4(a)(2) of the Securities
Act and/or Rule 701 promulgated under Section 3(b) of the Securities Act as a transaction pursuant to a compensatory benefit plan
or contract relating to compensation. Each purchaser received written disclosures that the securities had not been registered under
the Securities Act and that any resale must be made pursuant to a registration statement or an available exemption from registration.
All of the foregoing securities were deemed restricted securities for the purposes of the Securities Act.
|
|
Item 16.
|
Exhibits and Financial Statement Schedules.
|
(a)
Exhibits
.
See the Exhibit Index immediately following the signature page hereto, which is incorporated into this Item 16 by reference.
(b)
Financial
Statements Schedules
. See Page F-1.
|
|
(a)
|
The undersigned registrant hereby undertakes:
|
(1) To file, during any period in which offers or sales are being made, a post-effective amendment to this registration statement:
(i) To include any prospectus required by section 10(a)(3) of the Securities Act of 1933;
(ii) To reflect in the prospectus any facts or events arising after the effective date of the registration statement (or the most recent posteffective amendment thereof) which, individually or in the aggregate, represent a fundamental change in the information set forth in the registration statement. Notwithstanding the foregoing, any increase or decrease in volume of securities offered (if the total dollar value of securities offered would not exceed that which was registered) and any deviation from the low or high end of the estimated maximum offering range may be reflected in the form of prospectus filed with the Commission pursuant to Rule 424(b) of this chapter) if, in the aggregate, the changes in volume and price represent no more than a 20% change in the maximum aggregate offering price set forth in the “Calculation of Registration Fee” table in the effective registration statement; and
(iii) To include any material information with respect to the plan of distribution not previously disclosed in the registration statement or any material change to such information in the registration statement.
(2) That, for the purposes of determining any liability under the Securities Act of 1933, each such post-effective amendment shall be deemed to be a new registration statement relating to the securities offered therein, and the offering of the securities at that time shall be deemed to be the initial
bona fide
offering thereof.
(3) To remove from registration by means of a post-effective amendment any of the securities being registered which remain unsold at the termination of the offering.
(4) That, for the purpose of determining liability under the Securities Act of 1933 to any purchaser, each prospectus filed pursuant to Rule 424(b) as part of a registration statement relating to an offering, other than registration statements relying on Rule 430B or other than prospectuses filed in reliance on Rule 430A, shall be deemed to be part of and included in the registration statement as of the date it is first used after effectiveness. Provided, however, that no statement made in a registration statement or prospectus that is part of the registration statement or made in a document incorporated or deemed incorporated by reference into the registration statement or prospectus that is part of the registration statement will, as to a purchaser with a time of contract of sale prior to such first use, supersede or modify any statement that was made in the registration statement or prospectus that was part of the registration statement or made in any such document immediately prior to such date of first use.
(5) For the purpose of determining liability of the registrant under the Securities Act of 1933 to any purchaser in the initial distribution of the securities in a primary offering of securities of the undersigned registrant pursuant to this registration statement, regardless of the underwriting method used to sell the securities to the purchaser, if the securities are offered or sold to such purchaser by means of any of the following communications, the undersigned registrant will be a seller to the purchaser and will be considered to offer or sell such securities to such purchaser:
(i) Any preliminary prospectus or prospectus of the undersigned registrant relating to the offering required to be filed pursuant to Rule 424;
(ii) Any free writing prospectus relating to the offering prepared by or on behalf of the undersigned registrant or used or referred to by the undersigned registrant;
(iii) The portion of any other free writing prospectus relating to the offering containing material information about the undersigned registrant or its securities provided by or on behalf of the undersigned registrant; and
(iv) Any other communication that is an offer in the offering made by the undersigned registrant to the purchaser.
SIGNATURES
Pursuant to the requirements
of the Securities Act of 1933, the registrant has duly caused this Registration Statement to be signed on its behalf by the undersigned,
thereunto duly authorized in the City of Irvine, State of California, on December 05, 2016.
|
|
NUGENE INTERNATIONAL, INC.
|
|
|
|
|
|
|
By:
|
/s/ Steven Carlson
|
|
|
Name:
|
Steven Carlson
|
|
|
Title:
|
Chief Executive Officer
|
POWER OF ATTORNEY
We, the undersigned
officers and directors of NuGene International, Inc., a Nevada corporation, hereby severally constitute and appoint Steven Carlson
our true and lawful attorney-in-fact and agents, with full power of substitution and resubstitution for him and in his name, place
and stead, and in any and all capacities, to sign for us and in our names in the capacities indicated below any and all amendments
(including post-effective amendments) to this registration statement (or any other registration statement for the same offering
that is to be effective upon filing pursuant to Rule 462(b) under the Securities Act of 1933, as amended), and to file the same,
with all exhibits thereto and other documents in connection therewith, with the Securities and Exchange Commission, granting unto
said attorney-in-fact and agent, full power and authority to do and perform each and every act and thing requisite or necessary
to be done in and about the premises, as full to all intents and purposes as he might or could do in person, hereby ratifying and
confirming all that said attorney-in-fact and agent, or his substitute or substitutes, may lawfully do or cause to be done by virtue
hereof.
Pursuant to the requirements
of the Securities Act of 1933, this Registration Statement has been signed by the following persons in the capacities and on the
date indicated.
|
Signature
|
|
Title
|
|
Date
|
|
|
|
|
|
|
|
/s/ Steven Carlson
|
|
Chief Executive Officer
|
|
December 05, 2016
|
|
Steven Carlson
|
|
(Principal Executive Officer)
|
|
|
|
|
|
|
|
|
|
/s/ Mohammad Ali Kharazmi
|
|
Chairman of the Board of Directors
|
|
December 05, 2016
|
|
Mohammad Ali Kharazmi
|
|
|
|
|
|
|
|
|
|
|
|
/s/ Mohammad Saeed Kharazmi
|
|
Director; Chief Financial Officer
|
|
December 05, 2016
|
|
Mohammad Saeed Kharazmi
|
|
(Principal Financial and Accounting Officer)
|
|
|
|
|
|
|
|
|
|
/s/ Theodore Schwarz
|
|
Independent Director
|
|
December 05, 2016
|
|
Theodore Schwarz
|
|
|
|
|
EXHIBIT INDEX
|
|
|
|
|
Incorporated
by Reference
|
|
Exhibit No.
|
|
Description
|
|
Form
|
|
Exhibit
|
|
Filing
Date
|
|
2.1
|
|
Agreement and Plan of Merger dated December 26, 2014,
among the Company, NG Acquisition, Inc. and NuGene Inc.
|
|
8-K
|
|
2.1
|
|
01/06/2015
|
|
|
|
|
|
|
|
|
|
|
|
3.1
|
|
Certificate of Amendment of Articles of Incorporation
of the Company (increase of authorized, new class of preferred)
|
|
8-K
|
|
3.1
|
|
01/06/2015
|
|
|
|
|
|
|
|
|
|
|
|
3.2
|
|
Certificate of Amendment of Articles of Incorporation of the
Company, name change
|
|
8-K
|
|
3.2
|
|
01/06/2015
|
|
|
|
|
|
|
|
|
|
|
|
4.1
|
|
Certificate of Designation of Series A
|
|
|
|
|
|
|
|
|
|
Preferred Stock
|
|
8-K
|
|
4.1
|
|
01/06/2015
|
|
|
|
|
|
|
|
|
|
|
|
4.2
|
|
Form of Non-Convertible Promissory Note
|
|
8-K
|
|
4.1
|
|
11/08/2016
|
|
|
|
|
|
|
|
|
|
|
|
5.1*
|
|
Opinion of Spectrum Law Group, LLP
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
10.1
|
|
Form of Lock Up Agreement-
|
|
|
|
|
|
|
|
|
|
NuGene Shareholders
|
|
8-K
|
|
10.1
|
|
01/06/2015
|
|
|
|
|
|
|
|
|
|
|
|
10.2
|
|
Form of Lock Up/Leak Out Agreement-
|
|
|
|
|
|
|
|
|
|
Stock Placement Investors
|
|
8-K
|
|
10.2
|
|
01/06/2015
|
|
|
|
|
|
|
|
|
|
|
|
10.3
|
|
License Agreement between the Company and
|
|
|
|
|
|
|
|
|
|
kathy ireland Worldwide, Inc.
|
|
8-K
|
|
10.3
|
|
01/06/2015
|
|
|
|
|
|
|
|
|
|
|
|
10.4
|
|
Sublease Agreement between the Company and Advanced Surgical
Partners, Inc.
|
|
8-K
|
|
10.4
|
|
01/06/2015
|
|
|
|
|
|
|
|
|
|
|
|
10.5
|
|
Form of Convertible Promissory Note.
|
|
8-K
|
|
10.5
|
|
01/06/2015
|
|
|
|
|
|
|
|
|
|
|
|
10.6
|
|
Business Transfer and Indemnity Agreement, dated December
29, 2014, between the Company and Dena Kurland
|
|
8-K
|
|
10.6
|
|
01/06/2015
|
|
|
|
|
|
|
|
|
|
|
|
10.7
|
|
Form of Stock Purchase Agreement
|
|
10-Q
|
|
10.7
|
|
05/15/2015
|
|
|
|
|
|
|
|
|
|
|
|
10.8+
|
|
Offer Letter to join Board of Directors between The Company
and Donna Queen
|
|
8-K
|
|
10.8
|
|
05/29/2015
|
|
|
|
|
|
|
|
|
|
|
|
10.9+
|
|
Form of Option Agreement granted to employees on August
14, 2015
|
|
8-K
|
|
10.9
|
|
08/21/2015
|
|
|
|
|
|
|
|
|
|
|
|
10.10+
|
|
Form of Warrant Agreement granted to consultants on August 14,
2015
|
|
8-K
|
|
10.10
|
|
08/21/2015
|
|
|
|
|
|
|
|
|
|
|
|
10.11+
|
|
Form of Warrant Agreement granted to consultant on August
14, 2015
|
|
8-K
|
|
10.11
|
|
08/21/2015
|
|
|
|
|
|
|
|
|
|
|
|
10.12
|
|
Form of Note Payable entered into on September
25, 2015
|
|
8-K
|
|
10.12
|
|
09/30/2015
|
|
|
|
|
|
|
|
|
|
|
|
10.13
|
|
Form of Security Purchase Agreement entered into on September
25, 2015
|
|
8-K
|
|
10.13
|
|
09/30/2015
|
|
|
|
|
|
|
|
|
|
|
|
10.14
|
|
Form of Securities Purchase Agreement entered into on
December 11, 2015
|
|
8-K
|
|
10.14
|
|
12/17/2015
|
|
|
|
|
|
|
|
|
|
|
|
10.15
|
|
Form of Note Payable issued on December 11, 2015
|
|
8-K
|
|
10.15
|
|
12/17/2015
|
|
|
|
|
|
|
|
|
|
|
|
10.16
|
|
Form of Warrant issued on December 11, 2015
|
|
8-K
|
|
10.16
|
|
12/17/2015
|
|
|
|
|
|
|
|
|
|
|
|
10.17
|
|
Form of Notes issued in November, 2015
|
|
8-K
|
|
10.17
|
|
12/17/2015
|
|
|
|
|
|
|
|
|
|
|
|
10.18
|
|
Service Agreement with KBHJJ
|
|
8-K
|
|
10.18
|
|
12/17/2015
|
|
|
|
|
|
|
|
|
|
|
|
10.19
|
|
Warrant Agreement with KBHJJ
|
|
8-K
|
|
10.19
|
|
12/17/2015
|
|
|
|
|
|
|
|
|
|
|
|
10.20
|
|
Form of Security Purchase Agreement
|
|
8-K
|
|
10.20
|
|
04/07/2016
|
|
|
|
|
|
|
|
|
|
|
|
10.21
|
|
Form of Note Payable to Gemini Master Fund, LTD
|
|
8-K
|
|
10.21
|
|
04/07/2016
|
|
|
|
|
|
|
|
|
|
|
|
10.22
|
|
Form of Note Payable to Canyon Assets Holdings, Inc.
|
|
8-K
|
|
10.22
|
|
04/07/2016
|
|
|
|
|
|
|
|
|
|
|
|
10.23+
|
|
Offer Letter to join Board of Directors between The Company
and Theodore Schwarz
|
|
8-K
|
|
10.8
|
|
06/21/2016
|
|
|
|
|
|
|
|
|
|
|
|
10.24+
|
|
Employment Agreement with Steven R. Carlson, dated 18 July 2016
|
|
8-K
|
|
10.1
|
|
07/29/2016
|
|
|
|
|
|
|
|
|
|
|
|
10.25+
|
|
Chairman Agreement with M. Ali Kharazmi, dated September 22,
2016
|
|
8-K
|
|
10.1
|
|
09/28/2016
|
|
|
|
|
|
|
|
|
|
|
|
10.26+
|
|
Warrant Agreement with M. Ali Kharazmi, dated September 22,
2016
|
|
8-K
|
|
10.2
|
|
09/28/2016
|
|
|
|
|
|
|
|
|
|
|
|
10.27+
|
|
Restricted Units Agreement with M. Ali Kharazmi, dated September
22, 2016
|
|
8-K
|
|
10.3
|
|
09/28/2016
|
|
|
|
|
|
|
|
|
|
|
|
10.28+
|
|
Executive Employment Agreement with M. Saeed Kharazmi, dated
September 22, 2016
|
|
8-K
|
|
10.4
|
|
09/28/2016
|
|
|
|
|
|
|
|
|
|
|
|
10.29+
|
|
Warrant Agreement with M. Saeed Kharazmi, dated September 22,
2016
|
|
8-K
|
|
10.5
|
|
09/28/2016
|
|
|
|
|
|
|
|
|
|
|
|
10.30+
|
|
Restricted Units Agreement with M. Saeed Kharazmi, dated September
22, 2016
|
|
8-K
|
|
10.6
|
|
09/28/2016
|
|
|
|
|
|
|
|
|
|
|
|
10.31
|
|
Form of Note Purchase Agreement
|
|
8-K
|
|
10.1
|
|
11/08/2016
|
|
|
|
|
|
|
|
|
|
|
|
10.32
|
|
Form of Common Stock Warrant
|
|
8-K
|
|
10.2
|
|
11/08/2016
|
|
|
|
|
|
|
|
|
|
|
|
21.1
|
|
List of subsidiaries
|
|
10-K
|
|
21.1
|
|
03/30/2015
|
|
|
|
|
|
|
|
|
|
|
|
23.1*
|
|
Consent of Weinstein & Co., independent registered public
accounting firm
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
23.2
|
|
Consent of Spectrum Law Group, LLP (included in Exhibit 5.1)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
24.1*
|
|
Power of Attorney (included on applicable signature pages)
|
|
|
|
|
|
|
* Filed herewith.
+ Indicates management contract or compensatory
plan or arrangement.
NuGene (PK) (USOTC:NUGN)
Historical Stock Chart
From Apr 2024 to May 2024
NuGene (PK) (USOTC:NUGN)
Historical Stock Chart
From May 2023 to May 2024