false
0001551152
Common Stock, $0.01 Par Value
ABBV
0001551152
2023-12-06
2023-12-06
0001551152
us-gaap:CommonStockMember
exch:XNYS
2023-12-06
2023-12-06
0001551152
exch:XCHI
us-gaap:CommonStockMember
2023-12-06
2023-12-06
0001551152
abbv:SeniorNotes1.375Percentdue2024Member
exch:XNYS
2023-12-06
2023-12-06
0001551152
abbv:SeniorNotes1.250Percentdue2024Member
exch:XNYS
2023-12-06
2023-12-06
0001551152
abbv:SeniorNotes0.750Percentdue2027Member
exch:XNYS
2023-12-06
2023-12-06
0001551152
abbv:SeniorNotes2.125Percentdue2028Member
exch:XNYS
2023-12-06
2023-12-06
0001551152
abbv:SeniorNotes2.625Percentdue2028Member
exch:XNYS
2023-12-06
2023-12-06
0001551152
abbv:SeniorNotes2.125Percentdue2029Member
exch:XNYS
2023-12-06
2023-12-06
0001551152
abbv:SeniorNotes1.250Percentdue2031Member
exch:XNYS
2023-12-06
2023-12-06
iso4217:USD
xbrli:shares
iso4217:USD
xbrli:shares
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM 8-K
CURRENT REPORT
Pursuant to Section 13 or 15(d) of the
Securities Exchange Act of 1934
Date of Report (Date of earliest event reported):
December 6, 2023
ABBVIE INC.
(Exact name of registrant as specified in its charter)
| Delaware |
|
001-35565 |
|
32-0375147 |
(State or other jurisdiction
of incorporation or organization) |
|
(Commission File Number) |
|
(I.R.S Employer
Identification Number) |
1 North Waukegan Road
North Chicago, Illinois 60064-6400
(Address of principal executive offices)(Zip Code)
Registrant’s telephone number, including area code: (847) 932-7900
Former name or former address,
if changed since last report: Not Applicable
Check the appropriate box below if the Form 8-K
filing is intended to simultaneously satisfy the filing obligation to the registrant under any of the following provisions:
| ¨ |
Written communications pursuant to Rule 425 under the Securities Act (17 CFR 230.425) |
| ¨ |
Soliciting material pursuant to Rule 14a-12 under the Exchange Act (17 CFR 240.14a-12) |
| ¨ |
Pre-commencement communications pursuant to Rule 14d-2(b) under the Exchange Act (17 CFR 240.14d-2(b)) |
| ¨ |
Pre-commencement communications pursuant to Rule 13e-4(c) under the Exchange Act (17 CFR 240.13e-4(c)) |
Securities registered pursuant to Section 12(b) of the Act:
| Title
of each class |
|
Trading Symbol(s) |
|
Name of each exchange on which registered |
| Common Stock, $0.01 Par Value |
|
ABBV |
|
New York Stock Exchange
Chicago Stock Exchange |
| 1.375% Senior Notes due 2024 |
|
ABBV24 |
|
New York Stock Exchange |
| 1.250% Senior Notes due 2024 |
|
ABBV24B |
|
New York Stock Exchange |
| 0.750% Senior Notes due 2027 |
|
ABBV27 |
|
New York Stock Exchange |
| 2.125% Senior Notes due 2028 |
|
ABBV28 |
|
New York Stock Exchange |
| 2.625% Senior Notes due 2028 |
|
ABBV28B |
|
New York Stock Exchange |
| 2.125% Senior Notes due 2029 |
|
ABBV29 |
|
New York Stock Exchange |
| 1.250% Senior Notes due 2031 |
|
ABBV31 |
|
New York Stock Exchange |
Indicate by check mark whether the registrant is an emerging growth company as defined in Rule 405 of the Securities Act of 1933 (§230.405 of this chapter) or Rule 12b-2 of the Securities Exchange Act of 1934 (§240.12b-2 of this chapter).
Emerging growth company ¨
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act. ¨
| Common Stock, $0.01 Par Value |
ABBV |
| Item 7.01 |
Regulation FD Disclosure |
On December 6, 2023, AbbVie Inc. (the “Company”)
and Cerevel Therapeutics Holdings, Inc. (“Cerevel”) issued a joint press release announcing a definitive agreement
pursuant to which the Company will acquire Cerevel, on the terms and subject to the conditions set forth therein. The press release
is attached hereto as Exhibit 99.1 and incorporated by reference herein.
On December 7, 2023, representatives of the Company will present
information about the proposed transaction to various investors of the Company. The presentation will include the slides attached hereto
as Exhibit 99.2 and incorporated by reference herein.
The information in this Item 7.01, including the exhibits referenced
herein and attached hereto, shall not be deemed “filed” for purposes of Section 18 of the Securities Exchange Act of
1934 (the “Exchange Act”), nor shall they be deemed incorporated by reference in any Company filing under the Securities Act
of 1933 or the Exchange Act, except as shall be expressly set forth by specific reference in such filing.
Forward-Looking Statements
Some statements in this Current Report on Form 8-K, including
those relating to the proposed acquisition of Cerevel by the Company, are, or may be considered, forward-looking statements for purposes
of the Private Securities Litigation Reform Act of 1995. The words “believe,” “expect,” “anticipate,”
“project” and similar expressions and uses of future or conditional verbs, generally identify forward-looking statements.
The Company cautions that these forward-looking statements are subject to risks and uncertainties that may cause actual results to differ
materially from those expressed or implied in the forward-looking statements. Such risks and uncertainties include, but are not limited
to, risks related to the satisfaction or waiver of the conditions to closing the proposed acquisition (including the failure to obtain
necessary regulatory approvals and failure to obtain the requisite vote by Cerevel stockholders) in the anticipated timeframe or at
all, including the possibility that the proposed acquisition does not close, the possibility that competing offers may be made, risks
related to the ability to realize the anticipated benefits of the proposed acquisition, including the possibility that the expected benefits
from the acquisition will not be realized or will not be realized within the expected time period, the risk that the businesses will not
be integrated successfully, disruption from the transaction making it more difficult to maintain business and operational relationships,
negative effects of this announcement or the consummation of the proposed acquisition on the market price of the Company’s common
stock and/or operating results, significant transaction costs, unknown liabilities, the risk of litigation and/or regulatory actions related
to the proposed acquisition or Cerevel’s business, risks related to the financing of the transaction, challenges to intellectual
property, competition from other products, difficulties inherent in the research and development process, adverse litigation or government
action, and changes to laws and regulations applicable to our industry. Additional information about the economic, competitive, governmental,
technological and other factors that may affect the Company’s operations is set forth in Item 1A, “Risk Factors,” of
the Company’s 2022 Annual Report on Form 10-K, which has been filed with the Securities and Exchange Commission (the “SEC”),
as updated by its subsequent Quarterly Reports on Form 10-Q and in other documents that the Company subsequently files with the SEC
that update, supplement or supersede such information. The Company undertakes no obligation, and specifically declines, to release publicly
any revisions to forward-looking statements as a result of subsequent events or developments, except as required by law.
| Item 9.01 |
Financial Statements and Exhibits |
(d) Exhibits
SIGNATURE
Pursuant to the requirements of the Securities Exchange Act of 1934,
as amended, the registrant has duly caused this report to be signed on its behalf by the undersigned hereunto duly authorized.
| |
|
|
ABBVIE INC. |
| |
|
|
| Date: December 6, 2023 |
|
By: |
/s/ Scott T. Reents |
| |
|
|
Scott T. Reents |
| |
|
|
Executive Vice President, |
| |
|
|
Chief Financial Officer |
Exhibit
99.1
PRESS RELEASE
AbbVie to Acquire Cerevel Therapeutics in Transformative Transaction
to Strengthen Neuroscience Pipeline
| · | Proposed
acquisition adds robust pipeline of assets focused on best-in-class potential for psychiatric
and neurological disorders where significant unmet needs remain |
| · | Cerevel’s
clinical-stage pipeline complements AbbVie’s current on-market portfolio and emerging
neuroscience pipeline |
| · | Emraclidine
has the potential to transform the standard of care in schizophrenia and other psychiatric
conditions |
| · | Transaction
valued at $45.00 per share in cash, for a total equity value of approximately $8.7 billion |
| · | AbbVie
to hold an investor conference call tomorrow, December 7, at 8:00 a.m. CT |
NORTH CHICAGO, Ill., and CAMBRIDGE,
Mass., Dec. 6, 2023 – AbbVie Inc. (NYSE: ABBV) and Cerevel Therapeutics (NASDAQ: CERE) today announced a definitive agreement
under which AbbVie will acquire Cerevel Therapeutics and its robust neuroscience pipeline of multiple clinical-stage and preclinical
candidates with potential across several diseases including schizophrenia, Parkinson’s disease (PD), and mood disorders. The acquisition
complements AbbVie’s neuroscience portfolio, adding a wide range of potentially best-in-class assets that may transform standards
of care across psychiatric and neurological disorders where significant unmet needs remain for patients.
Under the terms of the transaction, AbbVie
will acquire all outstanding shares of Cerevel for $45.00 per share in cash. The transaction values Cerevel at a total equity value of
approximately $8.7 billion. The boards of directors of both companies have approved the transaction. This transaction is expected to
close in the middle of 2024, subject to Cerevel shareholder approval, regulatory approvals, and other customary closing conditions.
“Our existing neuroscience portfolio
and our combined pipeline with Cerevel represents a significant growth opportunity well into the next decade,” said Richard A.
Gonzalez, chairman and chief executive officer, AbbVie. “AbbVie will leverage its deep commercial capabilities, international infrastructure,
and regulatory and clinical expertise to deliver substantial shareholder value with multibillion-dollar sales potential across Cerevel’s
portfolio of assets.”
"Cerevel has always been committed
to transforming what is possible in neuroscience. With AbbVie’s long-standing expertise in developing and commercializing medicines
on a global scale, Cerevel’s novel therapies will be well positioned to reach more people living with neuroscience diseases,"
said Ron Renaud, president and chief executive officer, Cerevel Therapeutics. "The talented, passionate, and dedicated Cerevel team
has made great progress over the past five years in developing our innovative suite of potential medicines, and we are pleased that AbbVie
has recognized the tremendous potential of our pipeline. This acquisition reinforces the renaissance we are seeing in neuroscience, and
we are proud to be at the forefront."
| AbbVie
Inc. |
+1 (847)
938-9190 |
| 1
North Waukegan Road |
abbvie.com |
| North
Chicago, IL 60064 |
|
Cerevel’s late-stage asset emraclidine,
a positive allosteric modulator (PAM) of the muscarinic M4 receptor, is a potential best-in-class, next-generation antipsychotic that
may be effective in treating schizophrenia patients. Schizophrenia impacts more than five million people in the G7 (U.S., France, Germany, Italy,
Spain, United Kingdom, and Japan) and a significant opportunity for treatment innovation remains for new and better tolerated therapies.
In a Phase 1b study, emraclidine has shown promising efficacy and safety in schizophrenia and is currently completing two Phase 2 trials
that were designed to be registration enabling. In addition, emraclidine has potential in dementia-related psychosis in Alzheimer’s
disease and PD. Emraclidine is currently in a Phase 1 study in elderly healthy volunteers in support of a potential Alzheimer’s
disease psychosis program.
In addition to emraclidine, Cerevel
has multiple assets advancing in clinical development with best-in-class potential that are complementary to AbbVie’s priority
areas within neuroscience. Tavapadon, a first-in-class dopamine D1/D5 selective partial agonist for the management of PD, is currently
in Phase 3 studies and has potential for both monotherapy and adjunctive treatment. Tavapadon’s efficacy and safety-tolerability
profile could enable its utility in early PD, becoming a near-term complementary asset to AbbVie’s existing symptomatic therapies
for advanced PD. CVL-354, currently in Phase 1, is a potential best-in-class kappa opioid receptor (KOR) antagonist that has the potential
to provide significantly improved efficacy and tolerability compared to existing treatments for major depressive disorder (MDD). Darigabat,
currently in Phase 2, is an alpha 2/3/5 selective GABAA receptor PAM for treatment-resistant epilepsy and panic disorder.
Transaction Terms
AbbVie will acquire all outstanding Cerevel
common stock for $45.00 per share in cash. The proposed transaction is subject to customary closing conditions, including receipt of
regulatory approvals and approval by Cerevel shareholders. The proposed transaction is expected to be accretive to adjusted diluted earnings
per share (EPS) beginning in 2030.
AbbVie Conference Call Details
AbbVie will host an investor conference
call tomorrow, December 7, at 8:00 a.m. CT to discuss this transaction. The call will be webcast through AbbVie’s Investor
Relations website at investors.abbvie.com. An archived edition of the call will be available after 9:00 a.m. CT. Presentation materials
for the investor conference call are available here.
Cerevel Tavapadon Investor Webcast
Due to the pending transaction with AbbVie,
Cerevel will no longer be hosting its previously scheduled investor webcast to discuss tavapadon on Monday, December 11, 2023.
Advisors
AbbVie’s financial advisor is
Morgan Stanley & Co. LLC and Kirkland & Ellis LLP is serving as legal advisor. Cerevel Therapeutics’ financial
advisor is Centerview Partners LLC and Latham & Watkins LLP is serving as legal advisor.
About AbbVie in Neuroscience
At AbbVie, our commitment to preserving
personhood of people around the world living with neurological and psychiatric disorders is unwavering. With more than three decades
of experience in neuroscience, we are providing meaningful treatment options today and
| AbbVie
Inc. |
+1 (847)
938-9190 |
| 1
North Waukegan Road |
abbvie.com |
| North
Chicago, IL 60064 |
|
advancing
innovation for the future. AbbVie's Neuroscience portfolio consists of approved treatments in neurological conditions, including migraine,
movement disorders, and psychiatric disorders, along with a robust pipeline of transformative therapies. We have made a strong investment
in research and are committed to building a deeper understanding of neurological and psychiatric disorders. Every challenge makes us
more determined and drives us to discover and deliver advancements for those impacted by these conditions, their care partners, and clinicians.
For more information, visit www.abbvie.com.
About
AbbVie
AbbVie's
mission is to discover and deliver innovative medicines and solutions that solve serious health issues today and address the medical
challenges of tomorrow. We strive to have a remarkable impact on people's lives across several key therapeutic areas – immunology,
oncology, neuroscience, and eye care – and products and services in our Allergan Aesthetics portfolio. For more information about
AbbVie, please visit us at www.abbvie.com. Follow @abbvie on LinkedIn,
Facebook, Instagram, X (formerly Twitter),
and YouTube.
About
Cerevel Therapeutics
Cerevel
Therapeutics is dedicated to unraveling the mysteries of the brain to treat neuroscience diseases. The company is tackling diseases by
combining its deep expertise in neurocircuitry with a focus on targeted receptor subtype selectivity and a differentiated approach to
pharmacology. Cerevel Therapeutics has a diversified pipeline comprised of five clinical-stage investigational therapies and several
preclinical compounds with the potential to treat a range of neuroscience diseases, including schizophrenia, Alzheimer’s disease
psychosis, epilepsy, panic disorder, and Parkinson’s disease. Headquartered in Cambridge, Mass., Cerevel Therapeutics is advancing
its current research and development programs while exploring new modalities through internal research efforts, external collaborations,
or potential acquisitions. For more information, visit www.cerevel.com.
Forward-Looking
Statements
Some
statements in this news release, including those relating to the proposed acquisition of Cerevel by AbbVie, are, or may be
considered, forward-looking statements for purposes of the Private Securities Litigation Reform Act of 1995. The words
"believe," "expect," "anticipate," "project" and similar expressions and uses of future or
conditional verbs, generally identify forward-looking statements. AbbVie and Cerevel caution that these forward-looking statements
are subject to risks and uncertainties that may cause actual results to differ materially from those expressed or implied in the
forward-looking statements. Such risks and uncertainties include, but are not limited to, risks related to the satisfaction or
waiver of the conditions to closing the proposed acquisition (including the failure to obtain necessary regulatory approvals and
failure to obtain the requisite vote by Cerevel shareholders) in the anticipated timeframe or at all, including the possibility that
the proposed acquisition does not close, the possibility that competing offers may be made, risks related to the ability to realize
the anticipated benefits of the proposed acquisition, including the possibility that the expected benefits from the acquisition will
not be realized or will not be realized within the expected time period, the risk that the businesses will not be integrated
successfully, disruption from the transaction making it more difficult to maintain business and operational relationships, negative
effects of this announcement or the consummation of the proposed acquisition on the market price of AbbVie’s common stock
and/or operating results, significant transaction costs, unknown liabilities, the risk of litigation and/or regulatory actions
related to the proposed acquisition or
| AbbVie
Inc. |
+1 (847)
938-9190 |
| 1
North Waukegan Road |
abbvie.com |
| North
Chicago, IL 60064 |
|
 |  |
Cerevel’s
business, risks related to the financing of the transaction, challenges to intellectual property, competition from other products, difficulties
inherent in the research and development process, adverse litigation or government action, and changes to laws and regulations applicable
to our industry. Additional information about the economic, competitive, governmental, technological and other factors that may affect
AbbVie's and Cerevel’s operations is set forth in Item 1A, "Risk Factors," of AbbVie's 2022 Annual Report on Form 10-K,
which has been filed with the Securities and Exchange Commission (the “SEC”), as updated by its subsequent Quarterly Reports
on Form 10-Q and in Item 1A, “Risk Factors,” of Cerevel’s 2022 Annual Report on Form 10-K, which has been
filed with the SEC, as updated by its subsequent Quarterly Reports on Form 10-Q, respectively. Neither AbbVie nor Cerevel undertakes
any obligation, and each specifically declines, to release publicly any revisions to forward-looking statements as a result of subsequent
events or developments, except as required by law.
Additional
Information and Where to Find It
This
news release may be deemed solicitation material in respect of the proposed acquisition of Cerevel. A special shareholder meeting will
be announced soon to obtain Cerevel shareholder approval in connection with the proposed acquisition. Cerevel expects to file with the
SEC a proxy statement and other relevant documents in connection with the proposed acquisition. Cerevel shareholders are urged to read
the definitive proxy statement and other relevant materials carefully and, in their entirety, when they become available because they
will contain important information about Cerevel and the proposed acquisition. Investors may obtain a free copy of these materials (when
they are available) and other documents filed by Cerevel with the SEC at the SEC’s website at www.sec.gov, and at Cerevel’s
website at www.cerevel.com.
No
Offer or Solicitation
This
news release is for information purposes only and is not intended to and does not constitute, or form part of, an offer, invitation or
the solicitation of an offer or invitation to purchase, otherwise acquire, subscribe for, sell or otherwise dispose of any securities,
or the solicitation of any vote or approval in any jurisdiction, pursuant to the proposed transaction or otherwise, nor shall there be
any sale, issuance or transfer of securities in any jurisdiction in contravention of applicable law.
Participants
in the Solicitation
Cerevel
and its directors, executive officers and certain employees and other persons may be deemed to be participants in soliciting proxies
from its shareholders in connection with the proposed acquisition. Information regarding Cerevel’s directors and executive officers
is set forth in Cerevel’s proxy statement on Schedule 14A for its 2023 Annual Meeting of Stockholders, which was filed with the
SEC on April 28, 2023, and in Cerevel’s Current Reports on Form 8-K filed with the SEC on May 3, 2023 and May 10,
2023. Additional information regarding the persons who may, under the rules of the SEC, be considered to be participants in the
solicitation of Cerevel’s shareholders in connection with the proposed acquisition and any direct or indirect interests they may
have in the proposed acquisition will be set forth in Cerevel’s definitive proxy statement for its special shareholder meeting
when it is filed with the SEC.
| AbbVie
Inc. |
+1 (847)
938-9190 |
| 1
North Waukegan Road |
abbvie.com |
| North
Chicago, IL 60064 |
|
| AbbVie
Contacts: |
Cerevel
Contacts: |
| |
|
| Media: |
Media: |
| Gabrielle
Tarbert |
Anna
Robinson |
| gabrielle.tarbert@abbvie.com |
anna.robinson@cerevel.com |
| |
|
| Investors: |
Investors: |
| Liz
Shea |
Matthew
Calistri |
| liz.shea@abbvie.com |
matthew.calistri@cerevel.com |
| AbbVie
Inc. |
+1 (847)
938-9190 |
| 1
North Waukegan Road |
abbvie.com |
| North
Chicago, IL 60064 |
|
Exhibit 99.2

| 1
Acquisition of Cerevel
December 6, 2023 |

| December 6, 2023 2
Forward-Looking Statements and Non-GAAP Financial Information
Some statements in this presentation, including those relating to the proposed acquisition of Cerevel Therapeutics Holding, Inc. by AbbVie Inc. are, or may be considered, forward-looking statements for purposes of the Private Securities Litigation Reform Act of 1995. The words "believe," "expect," "anticipate," "project" and similar expressions and uses of
future or conditional verbs, generally identify forward-looking statements. AbbVie cautions that these forward-looking statements are subject to risks and uncertainties that may
cause actual results to differ materially from those expressed or implied in the forward-looking statements. Such risks and uncertainties include, but are not limited to, risks related
to the satisfaction or waiver of the conditions to closing the proposed acquisition (including the failure to obtain necessary regulatory approvals and failure to obtain the requisite
vote by Cerevel stockholders) in the anticipated timeframe or at all, including the possibility that the proposed acquisition does not close, the possibility that competing offers may
be made, risks related to the ability to realize the anticipated benefits of the proposed acquisition, including the possibility that the expected benefits from the acquisition will not be
realized or will not be realized within the expected time period, the risk that the businesses will not be integrated successfully, disruption from the transaction making it more difficult
to maintain business and operational relationships, negative effects of this announcement or the consummation of the proposed acquisition on the market price of AbbVie’s
common stock and/or operating results, significant transaction costs, unknown liabilities, the risk of litigation and/or regulatory actions related to the proposed acquisition or
Cerevel’s business, risks related to the financing of the transaction, challenges to intellectual property, competition from other products, difficulties inherent in the research and
development process, adverse litigation or government action, and changes to laws and regulations applicable to our industry. Additional information about the economic,
competitive, governmental, technological and other factors that may affect AbbVie's operations is set forth in Item 1A, "Risk Factors," of AbbVie's 2022 Annual Report on Form 10-
K, which has been filed with the Securities and Exchange Commission, as updated by its subsequent Quarterly Reports on Form 10-Q. AbbVie undertakes no obligation, and
specifically declines, to release publicly any revisions to forward-looking statements as a result of subsequent events or developments, except as required by law.
This presentation contains certain non-GAAP financial measures. Non-GAAP financial measures are adjusted for certain non-cash items and for factors that are unusual or
unpredictable, and exclude those costs, expenses and other specified items presented in AbbVie’s reconciliation tables. AbbVie’s management believes non-GAAP financial
measures provide useful information to investors regarding AbbVie’s results of operations and assist management, analysts and investors in evaluating the performance of the
business. Non-GAAP financial measures should be considered in addition to, and not as a substitute for, measures of financial performance prepared in accordance with GAAP.
AbbVie does not provide a reconciliation of forward-looking non-GAAP financial measures to the most directly comparable GAAP reported financial measures on a forward-looking
basis because it is unable to predict with reasonable certainty the ultimate outcome of unusual gains and losses, certain acquisition-related expenses, gains and losses from equity
securities, actuarial gains and losses from pension and postretirement plan remeasurements, potential future asset impairments and pending litigation without unreasonable effort.
These items are uncertain, depend on various factors and could have a material impact on GAAP reported results for the guidance period.
This presentation is intended for the investor community only; materials are not intended to promote the products referenced herein or otherwise influence healthcare prescribing
decisions. |

| December 6, 2023 3
Strong Strategic Fit for AbbVie
Leverages AbbVie’s commercial capabilities, international
infrastructure, and regulatory and clinical expertise to
maximize Cerevel’s high-value assets
A unique opportunity to acquire a pipeline of potentially
best-in-class assets focused on treating neurological and
psychiatric diseases
Potential for substantial shareholder value creation
with multibillion-dollar sales potential across the
portfolio of assets |

| December 6, 2023 4
AbbVie Neuro-Psychiatry
Developing Innovative Therapies for Mood, Thought and Anxiety Disorders
Psychiatry Represents a Large
and Underserved Opportunity
AbbVie Aspires to be a Leader in Mood, Thought, and
Anxiety Disorders with High Unmet Need
Most prevalent psychiatric conditions in the G7
(U.S., EU5, Japan)
Psychosis
Mood
Disorders
Anxiety
Disorders
• Generalized Anxiety
• Social Anxiety
• PTSD
• OCD
• Schizophrenia
• Psychosis/Agitation
Associated with
Alzheimer’s and
Parkinson’s Diseases
• Depression
• Bipolar Disorder
48.9M Major Depressive Disorder
9.4M Bipolar Disorder
21.1M Post-Traumatic Stress Disorder
17.3M Generalized Anxiety Disorder
5.6M Schizophrenia
8.0M Opioid Use Disorder
2.9M Autism Spectrum Disorder
Source: Clarivate DRG and AbbVie estimates
PTSD: Post-traumatic stress disorder; OCD: Obsessive-compulsive disorder; EU5: France, Germany, Italy, Spain, United Kingdom |
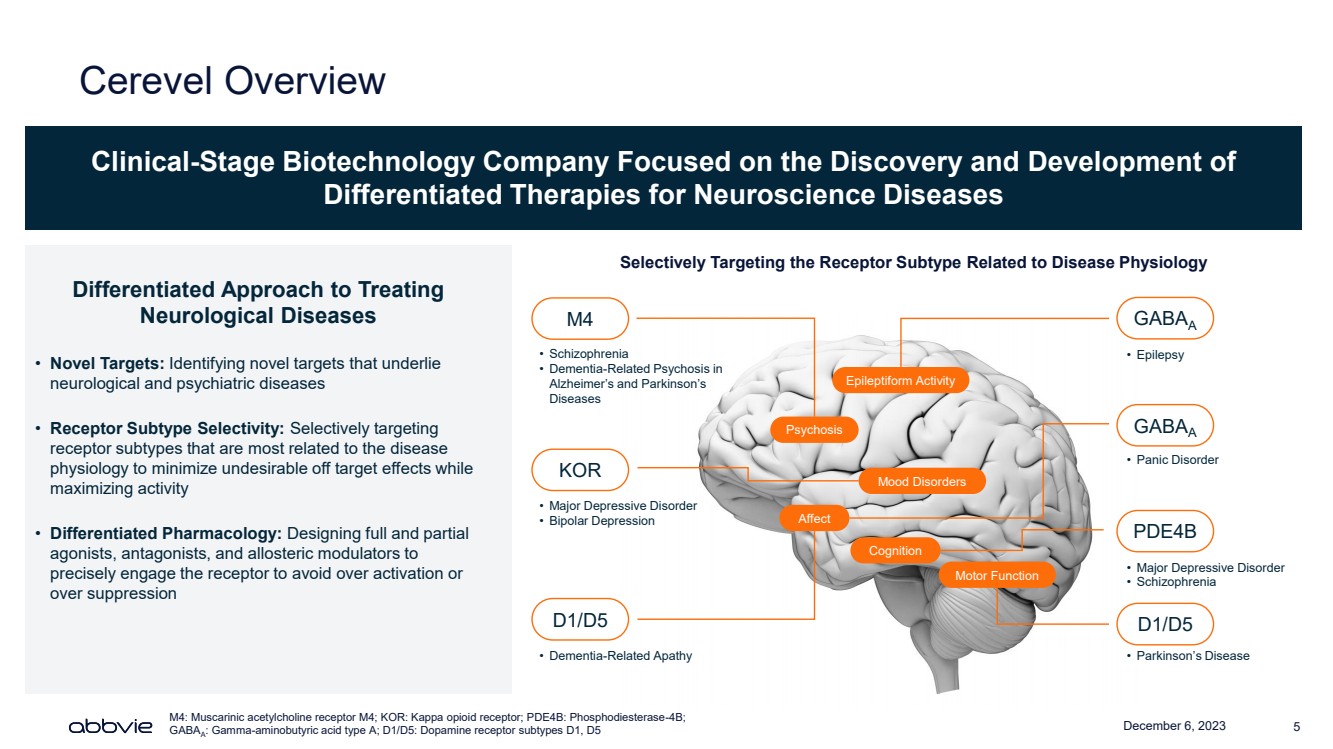
| December 6, 2023 5
Cerevel Overview
Clinical-Stage Biotechnology Company Focused on the Discovery and Development of
Differentiated Therapies for Neuroscience Diseases
Differentiated Approach to Treating
Neurological Diseases
• Novel Targets: Identifying novel targets that underlie
neurological and psychiatric diseases
• Receptor Subtype Selectivity: Selectively targeting
receptor subtypes that are most related to the disease
physiology to minimize undesirable off target effects while
maximizing activity
• Differentiated Pharmacology: Designing full and partial
agonists, antagonists, and allosteric modulators to
precisely engage the receptor to avoid over activation or
over suppression
M4
GABAA
PDE4B
D1/D5
GABAA
KOR
D1/D5
• Parkinson’s Disease
• Major Depressive Disorder
• Schizophrenia
• Epilepsy
• Major Depressive Disorder
• Bipolar Depression
• Dementia-Related Apathy
• Schizophrenia
• Dementia-Related Psychosis in
Alzheimer’s and Parkinson’s
Diseases
Psychosis
Mood Disorders
Cognition
Epileptiform Activity
Affect
Motor Function
Selectively Targeting the Receptor Subtype Related to Disease Physiology
M4: Muscarinic acetylcholine receptor M4; KOR: Kappa opioid receptor; PDE4B: Phosphodiesterase-4B;
GABAA
: Gamma-aminobutyric acid type A; D1/D5: Dopamine receptor subtypes D1, D5
• Panic Disorder |
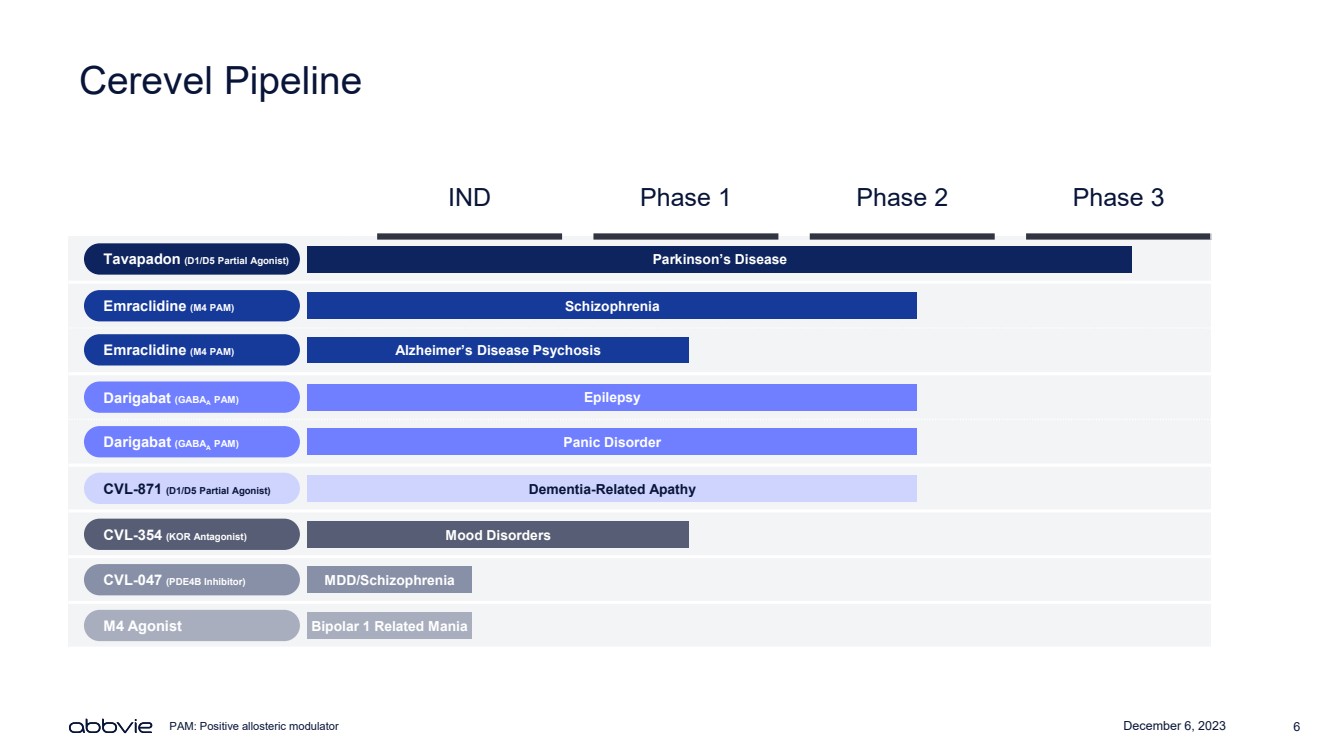
| December 6, 2023 6
Cerevel Pipeline
IND Phase 1 Phase 2 Phase 3
Tavapadon (D1/D5 Partial Agonist)
Emraclidine (M4 PAM)
Emraclidine (M4 PAM)
Darigabat (GABAA PAM)
Darigabat (GABAA PAM)
CVL-871 (D1/D5 Partial Agonist)
CVL-354 (KOR Antagonist)
CVL-047 (PDE4B Inhibitor)
M4 Agonist
Parkinson’s Disease
Schizophrenia
Alzheimer’s Disease Psychosis
Dementia-Related Apathy
Panic Disorder
Epilepsy
Mood Disorders
MDD/Schizophrenia
Bipolar 1 Related Mania
PAM: Positive allosteric modulator |

| December 6, 2023 7
Emraclidine
Selectively targeting the M4 muscarinic receptor to more effectively treat
psychosis related symptoms and improve tolerability
Emraclidine is a positive allosteric modulator of the muscarinic M4 receptor (M4 PAM), a new
mechanistic class that has the potential to provide significant efficacy, safety and tolerability
advantages compared to atypical antipsychotics
Emraclidine has shown a robust efficacy and safety profile in Phase 1b in schizophrenia patients;
Data demonstrate emraclidine’s potential to provide higher efficacy, differentiated safety/tolerability,
and more convenient dosing versus other muscarinic receptor modulators
Muscarinic receptor modulators as a class are demonstrating increasing potential
in schizophrenia, with proof-of-mechanism established across several clinical trials
Two randomized, placebo-controlled Phase 2 trials ongoing that have the potential to support
approval in schizophrenia (data expected in 2H24); Plan to evaluate as a treatment
for dementia-related psychosis in Alzheimer’s and Parkinson’s diseases |
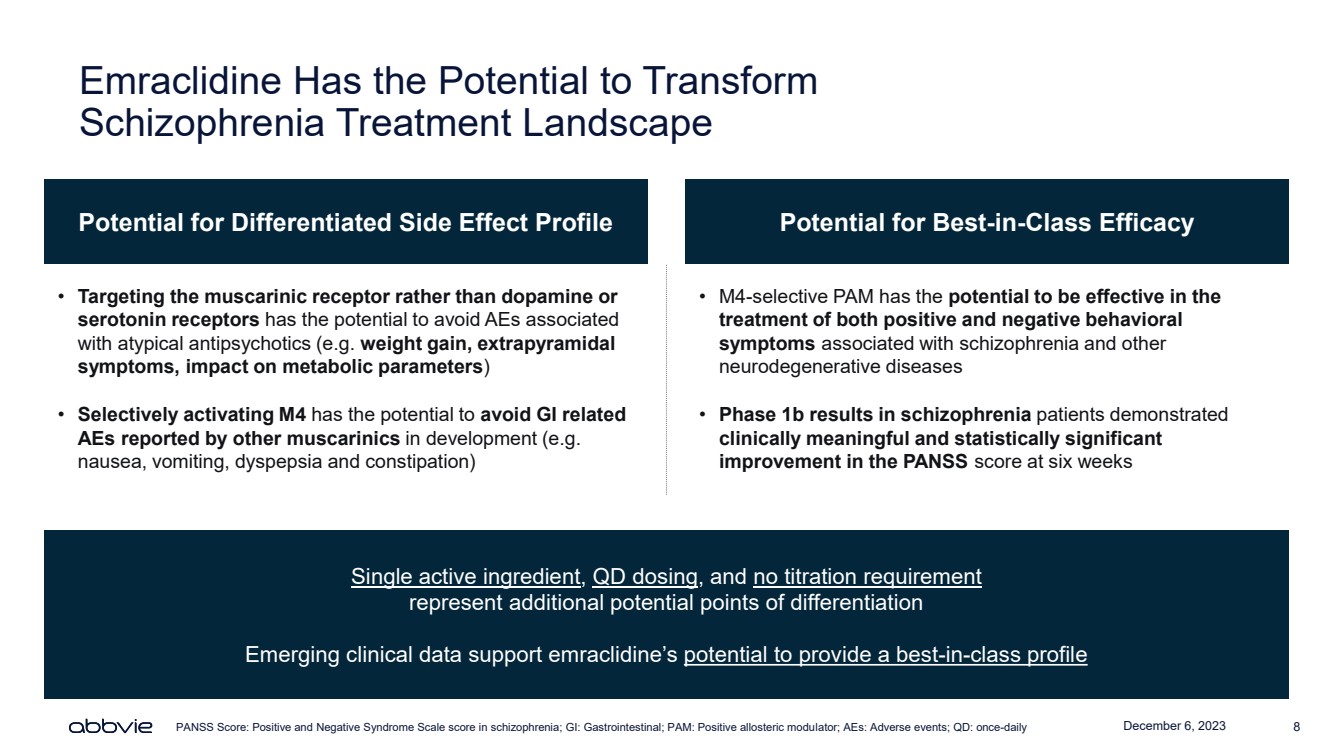
| December 6, 2023 8
Emraclidine Has the Potential to Transform
Schizophrenia Treatment Landscape
Potential for Differentiated Side Effect Profile Potential for Best-in-Class Efficacy
• Targeting the muscarinic receptor rather than dopamine or
serotonin receptors has the potential to avoid AEs associated
with atypical antipsychotics (e.g. weight gain, extrapyramidal
symptoms, impact on metabolic parameters)
• Selectively activating M4 has the potential to avoid GI related
AEs reported by other muscarinics in development (e.g.
nausea, vomiting, dyspepsia and constipation)
• M4-selective PAM has the potential to be effective in the
treatment of both positive and negative behavioral
symptoms associated with schizophrenia and other
neurodegenerative diseases
• Phase 1b results in schizophrenia patients demonstrated
clinically meaningful and statistically significant
improvement in the PANSS score at six weeks
Single active ingredient, QD dosing, and no titration requirement
represent additional potential points of differentiation
Emerging clinical data support emraclidine’s potential to provide a best-in-class profile
PANSS Score: Positive and Negative Syndrome Scale score in schizophrenia; GI: Gastrointestinal; PAM: Positive allosteric modulator; AEs: Adverse events; QD: once-daily |

| December 6, 2023 9
CVL-354
Potential Best-in-Class Kappa Opioid Receptor (KOR) Antagonist
KOR antagonism is a clinically validated mechanism
of action in major depressive disorder
• CVL-354 shows high KOR
antagonism potency
• Potential to drive higher efficacy than other
KOR antagonists in development
KOR antagonists have the potential to provide clinically meaningful improvements in safety and
tolerability compared to existing treatments for MDD
• CVL-354 demonstrates high functional and
binding selectivity for KOR versus MOR
• Potential to provide tolerability improvement
compared to other KOR antagonists in
development (e.g. diarrhea, interactions with
pain medications)
MOR: Mu opioid receptor |

| December 6, 2023 10
Multiple Additional High-Potential Pipeline Assets
TAVAPADON
Partial agonist selectively targeting the
dopamine D1/D5 receptor
Potential to provide enhanced motor
control and improved tolerability
compared to standard of care
In Phase 3 development as a
monotherapy (early-stage PD) and
adjunctive therapy (late-stage PD), with
data anticipated in 2024
DARIGABAT
Alpha 2/3/5 selective GABAA
receptor PAM
Minimal activity against alpha-1 GABAA
receptor has the potential to minimize
sedation and addiction associated with
traditional non-selective GABAA
receptor
modulators, such as benzodiazepines
Phase 2 study in focal epilepsy intended
to establish proof-of-concept and
tolerability profile, with data anticipated
in 2024; Phase 2 study in panic disorder
initiated in 2023
CVL-871
Selective partial agonist of the dopamine
D1/D5 receptor subtypes designed to
achieve a modest level of partial
agonism
Exploratory Phase 2a study in
dementia-related apathy is ongoing |
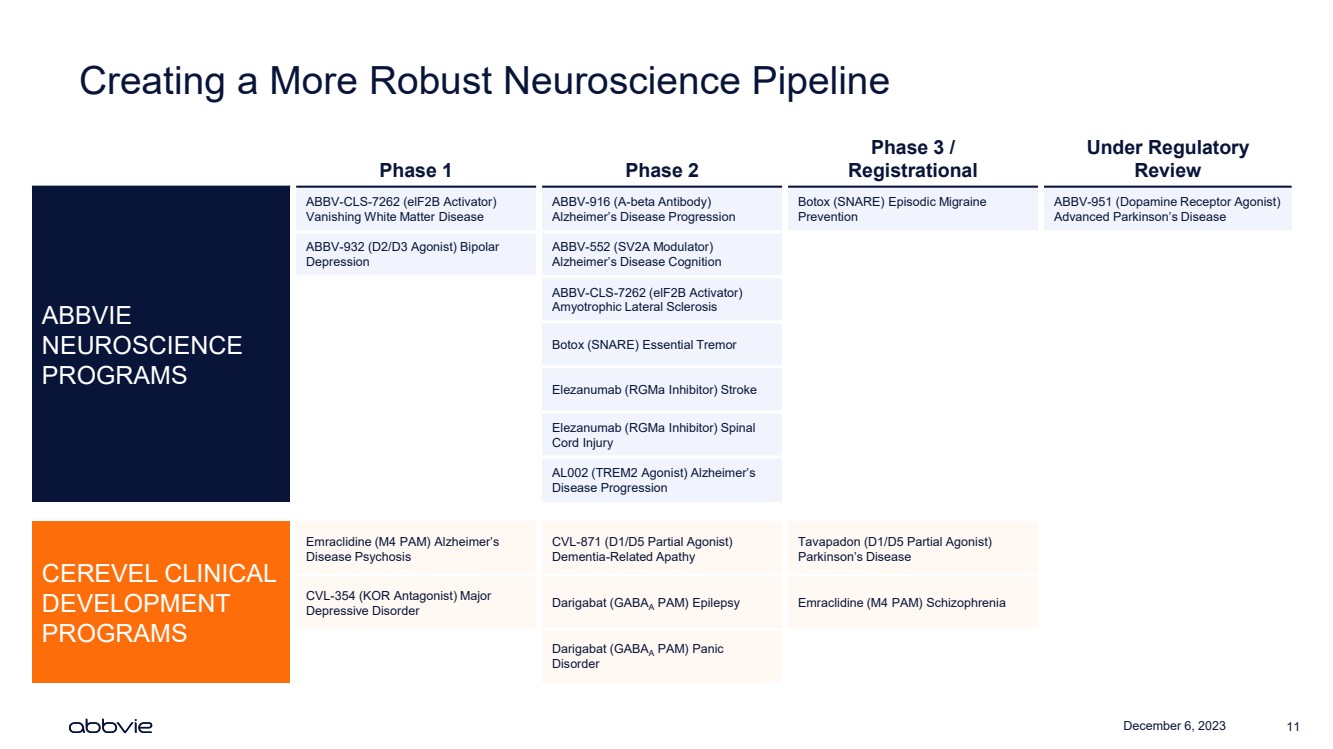
| December 6, 2023 11
Creating a More Robust Neuroscience Pipeline
Phase 1 Phase 2
Phase 3 /
Registrational
Under Regulatory
Review
ABBVIE
NEUROSCIENCE
PROGRAMS
ABBV-CLS-7262 (elF2B Activator)
Vanishing White Matter Disease
ABBV-916 (A-beta Antibody)
Alzheimer’s Disease Progression
Botox (SNARE) Episodic Migraine
Prevention
ABBV-951 (Dopamine Receptor Agonist)
Advanced Parkinson’s Disease
ABBV-932 (D2/D3 Agonist) Bipolar
Depression
ABBV-552 (SV2A Modulator)
Alzheimer’s Disease Cognition
ABBV-CLS-7262 (elF2B Activator)
Amyotrophic Lateral Sclerosis
Botox (SNARE) Essential Tremor
Elezanumab (RGMa Inhibitor) Stroke
Elezanumab (RGMa Inhibitor) Spinal
Cord Injury
AL002 (TREM2 Agonist) Alzheimer’s
Disease Progression
CEREVEL CLINICAL
DEVELOPMENT
PROGRAMS
Emraclidine (M4 PAM) Alzheimer’s
Disease Psychosis
CVL-871 (D1/D5 Partial Agonist)
Dementia-Related Apathy
Tavapadon (D1/D5 Partial Agonist)
Parkinson’s Disease
CVL-354 (KOR Antagonist) Major
Depressive Disorder Darigabat (GABAA PAM) Epilepsy Emraclidine (M4 PAM) Schizophrenia
Darigabat (GABAA PAM) Panic
Disorder |

| December 6, 2023 12
Transaction and Financial Overview
• AbbVie has agreed to acquire all outstanding shares of Cerevel for a purchase price of $45.00 per share in all-cash transaction
• Premium of approximately 73% to the unaffected closing share price on December 1, 2023
• Purchase price of $8.7B; Implied transaction value of ~$8.4B net of estimated cash acquired
• Will fund the transaction with a combination of cash and debt
PURCHASE
PRICE
• Emraclidine and CVL-354 both represent multibillion-dollar peak sales opportunities
• Emraclidine represents most substantial component of deal value
• Modest value ascribed to CVL-354 given early stage of development
DEAL
VALUE
CAPITAL
ALLOCATION
PRIORITIES
FINANCIAL
IMPACT
• Closing expected in the middle of 2024, subject to Cerevel shareholder approval, regulatory approvals and other customary closing conditions
• Expected to negatively impact adjusted diluted EPS by approximately $0.19 in 2024 (partial year) and approximately $0.41 in 2025 based on
increased R&D, operating and interest expenses; Expected to have positive operating margin in 2028, with EPS accretion beginning in 2030
• AbbVie maintains adjusted diluted EPS floor of $11.00 in 2024, inclusive of negative impact from both Cerevel and ImmunoGen transactions;
Will provide formal 2024 EPS guidance on 4Q23 earnings call
• No change to AbbVie’s capital allocation priorities
• Remain committed to a strong growing dividend
• Committed to achieving net leverage ratio of 2x by the end of 2026; Expect to maintain A3/A- credit rating |

| December 6, 2023 13
Key Takeaways
A strong strategic fit for AbbVie that represents a unique opportunity to acquire a pipeline of
potentially best-in-class assets focused on treating neurological and psychiatric diseases
• Provides AbbVie with promising discovery programs and clinical-stage assets that are highly complementary to our
neuroscience portfolio
• Emraclidine is a late-stage asset with the potential to provide significant efficacy, safety and tolerability advantages
compared to approved atypical antipsychotics and other muscarinic receptor modulators in development
• Multiple assets advancing in clinical development with best-in-class potential in respective indications
Substantial shareholder value creation with multibillion dollar sales potential across the portfolio of
assets
• AbbVie will leverage its commercial capabilities, international infrastructure, and regulatory and clinical expertise to
maximize Cerevel’s high-value assets
• Cerevel’s deep scientific expertise augments AbbVie’s discovery capabilities in psychiatry |
Cover
|
Dec. 06, 2023 |
| Document Information [Line Items] |
|
| Document Type |
8-K
|
| Amendment Flag |
false
|
| Document Period End Date |
Dec. 06, 2023
|
| Entity File Number |
001-35565
|
| Entity Registrant Name |
ABBVIE INC.
|
| Entity Central Index Key |
0001551152
|
| Entity Tax Identification Number |
32-0375147
|
| Entity Incorporation, State or Country Code |
DE
|
| Entity Address, Address Line One |
1 North Waukegan Road
|
| Entity Address, City or Town |
North Chicago
|
| Entity Address, State or Province |
IL
|
| Entity Address, Postal Zip Code |
60064-6400
|
| City Area Code |
847
|
| Local Phone Number |
932-7900
|
| Written Communications |
false
|
| Soliciting Material |
false
|
| Pre-commencement Tender Offer |
false
|
| Pre-commencement Issuer Tender Offer |
false
|
| Entity Emerging Growth Company |
false
|
| Common Stock [Member] | NEW YORK STOCK EXCHANGE, INC. [Member] |
|
| Document Information [Line Items] |
|
| Title of 12(b) Security |
Common Stock, $0.01 Par Value
|
| Trading Symbol |
ABBV
|
| Security Exchange Name |
NYSE
|
| Common Stock [Member] | NYSE CHICAGO, INC. [Member] |
|
| Document Information [Line Items] |
|
| Title of 12(b) Security |
Common Stock, $0.01 Par Value
|
| Trading Symbol |
ABBV
|
| Security Exchange Name |
CHX
|
| Senior Notes 1.375 Percent due 2024 [Member] | NEW YORK STOCK EXCHANGE, INC. [Member] |
|
| Document Information [Line Items] |
|
| Title of 12(b) Security |
1.375% Senior Notes due 2024
|
| Trading Symbol |
ABBV24
|
| Security Exchange Name |
NYSE
|
| Senior Notes 1.250 Percent due 2024 [Member] | NEW YORK STOCK EXCHANGE, INC. [Member] |
|
| Document Information [Line Items] |
|
| Title of 12(b) Security |
1.250% Senior Notes due 2024
|
| Trading Symbol |
ABBV24B
|
| Security Exchange Name |
NYSE
|
| Senior Notes 0.750 Percent due 2027 [Member] | NEW YORK STOCK EXCHANGE, INC. [Member] |
|
| Document Information [Line Items] |
|
| Title of 12(b) Security |
0.750% Senior Notes due 2027
|
| Trading Symbol |
ABBV27
|
| Security Exchange Name |
NYSE
|
| Senior Notes 2.125 Percent due 2028 [Member] | NEW YORK STOCK EXCHANGE, INC. [Member] |
|
| Document Information [Line Items] |
|
| Title of 12(b) Security |
2.125% Senior Notes due 2028
|
| Trading Symbol |
ABBV28
|
| Security Exchange Name |
NYSE
|
| Senior Notes 2.625 Percent due 2028 [Member] | NEW YORK STOCK EXCHANGE, INC. [Member] |
|
| Document Information [Line Items] |
|
| Title of 12(b) Security |
2.625% Senior Notes due 2028
|
| Trading Symbol |
ABBV28B
|
| Security Exchange Name |
NYSE
|
| Senior Notes 2.125 Percent due 2029 [Member] | NEW YORK STOCK EXCHANGE, INC. [Member] |
|
| Document Information [Line Items] |
|
| Title of 12(b) Security |
2.125% Senior Notes due 2029
|
| Trading Symbol |
ABBV29
|
| Security Exchange Name |
NYSE
|
| Senior Notes 1.250 Percent due 2031 [Member] | NEW YORK STOCK EXCHANGE, INC. [Member] |
|
| Document Information [Line Items] |
|
| Title of 12(b) Security |
1.250% Senior Notes due 2031
|
| Trading Symbol |
ABBV31
|
| Security Exchange Name |
NYSE
|
| X |
- DefinitionBoolean flag that is true when the XBRL content amends previously-filed or accepted submission.
| Name: |
dei_AmendmentFlag |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionFor the EDGAR submission types of Form 8-K: the date of the report, the date of the earliest event reported; for the EDGAR submission types of Form N-1A: the filing date; for all other submission types: the end of the reporting or transition period. The format of the date is YYYY-MM-DD.
| Name: |
dei_DocumentPeriodEndDate |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:dateItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe type of document being provided (such as 10-K, 10-Q, 485BPOS, etc). The document type is limited to the same value as the supporting SEC submission type, or the word 'Other'.
| Name: |
dei_DocumentType |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:submissionTypeItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionAddress Line 1 such as Attn, Building Name, Street Name
| Name: |
dei_EntityAddressAddressLine1 |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- Definition
+ References
+ Details
| Name: |
dei_EntityAddressCityOrTown |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionCode for the postal or zip code
| Name: |
dei_EntityAddressPostalZipCode |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionName of the state or province.
| Name: |
dei_EntityAddressStateOrProvince |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:stateOrProvinceItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionA unique 10-digit SEC-issued value to identify entities that have filed disclosures with the SEC. It is commonly abbreviated as CIK. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b-2
| Name: |
dei_EntityCentralIndexKey |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:centralIndexKeyItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionIndicate if registrant meets the emerging growth company criteria. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b-2
| Name: |
dei_EntityEmergingGrowthCompany |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionCommission file number. The field allows up to 17 characters. The prefix may contain 1-3 digits, the sequence number may contain 1-8 digits, the optional suffix may contain 1-4 characters, and the fields are separated with a hyphen.
| Name: |
dei_EntityFileNumber |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:fileNumberItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionTwo-character EDGAR code representing the state or country of incorporation.
| Name: |
dei_EntityIncorporationStateCountryCode |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:edgarStateCountryItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe exact name of the entity filing the report as specified in its charter, which is required by forms filed with the SEC. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b-2
| Name: |
dei_EntityRegistrantName |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe Tax Identification Number (TIN), also known as an Employer Identification Number (EIN), is a unique 9-digit value assigned by the IRS. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b-2
| Name: |
dei_EntityTaxIdentificationNumber |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:employerIdItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionLocal phone number for entity.
| Name: |
dei_LocalPhoneNumber |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionBoolean flag that is true when the Form 8-K filing is intended to satisfy the filing obligation of the registrant as pre-commencement communications pursuant to Rule 13e-4(c) under the Exchange Act. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 13e
-Subsection 4c
| Name: |
dei_PreCommencementIssuerTenderOffer |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionBoolean flag that is true when the Form 8-K filing is intended to satisfy the filing obligation of the registrant as pre-commencement communications pursuant to Rule 14d-2(b) under the Exchange Act. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 14d
-Subsection 2b
| Name: |
dei_PreCommencementTenderOffer |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionTitle of a 12(b) registered security. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b
| Name: |
dei_Security12bTitle |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:securityTitleItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionName of the Exchange on which a security is registered. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection d1-1
| Name: |
dei_SecurityExchangeName |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:edgarExchangeCodeItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionBoolean flag that is true when the Form 8-K filing is intended to satisfy the filing obligation of the registrant as soliciting material pursuant to Rule 14a-12 under the Exchange Act. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Section 14a
-Number 240
-Subsection 12
| Name: |
dei_SolicitingMaterial |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionTrading symbol of an instrument as listed on an exchange.
| Name: |
dei_TradingSymbol |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:tradingSymbolItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionBoolean flag that is true when the Form 8-K filing is intended to satisfy the filing obligation of the registrant as written communications pursuant to Rule 425 under the Securities Act. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Securities Act
-Number 230
-Section 425
| Name: |
dei_WrittenCommunications |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- Details
| Name: |
us-gaap_StatementClassOfStockAxis=us-gaap_CommonStockMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
dei_EntityListingsExchangeAxis=exch_XNYS |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
dei_EntityListingsExchangeAxis=exch_XCHI |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
us-gaap_StatementClassOfStockAxis=abbv_SeniorNotes1.375Percentdue2024Member |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
us-gaap_StatementClassOfStockAxis=abbv_SeniorNotes1.250Percentdue2024Member |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
us-gaap_StatementClassOfStockAxis=abbv_SeniorNotes0.750Percentdue2027Member |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
us-gaap_StatementClassOfStockAxis=abbv_SeniorNotes2.125Percentdue2028Member |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
us-gaap_StatementClassOfStockAxis=abbv_SeniorNotes2.625Percentdue2028Member |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
us-gaap_StatementClassOfStockAxis=abbv_SeniorNotes2.125Percentdue2029Member |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
us-gaap_StatementClassOfStockAxis=abbv_SeniorNotes1.250Percentdue2031Member |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
AbbVie (NYSE:ABBV)
Historical Stock Chart
From Jun 2024 to Jul 2024

AbbVie (NYSE:ABBV)
Historical Stock Chart
From Jul 2023 to Jul 2024
