Microneedles Breakthrough Brings 3M One Step Closer to Introducing New Alternative Drug Delivery Method
January 28 2015 - 11:09AM
Business Wire
3M hollow microneedle device now available for
clinical trials*; holds the potential to replace traditional
injections
Getting an injection at the doctor’s office typically does not
rank high on many people’s wish list. Thanks to 3M scientists’
know-how, the day is getting closer when patients may be receiving
their prescription medications at home via microscopic needles.
Pharmaceutical and biotechnology companies can now partner with 3M
on development and conducting clinical trials using its 3M™ Hollow
Microstructured Transdermal System (hMTS). Patient-friendly and
easy to use, 3M hMTS is designed to open new opportunities for
pharmaceutical companies and patients.
3M Hollow Microstructured Transdermal
System (hMTS) (Photo: 3M).
The device’s availability for clinical trials comes after
conducting a number of studies and design verification tests. Based
on 3M microreplication technology, pharmaceutical and biotech
companies can take advantage of this patient-friendly hollow
microneedle device for difficult-to-deliver biologics.
To reach this current stage of clinical readiness with the hMTS
device, 3M has undertaken a rigorous process, including finalizing
the device design, manufacturing critical components from medical
grade materials, establishing GMP array manufacturing and device
assembly, as well as filing documentation with FDA.
3M conducted a human tolerability study with the goal of
selecting the appropriate microneedle array for use in clinical
studies. The outcome of this study found very good delivery times
for 2 mL (less than 2 minutes on average). These results provide
foundational data in assessing the safety of the device. Clinical
supplies are now available for assessment in potential development
partners' trials.
“From the foundation laid by our recent human study, we are
excited to extend our hollow microneedle device and expertise to
companies who are ready for clinical studies. Pharma companies can
now evaluate 3M hMTS in their clinical trials as a delivery system
for a new drug product or a product line extension,” said Ingrid
Blair, Vice President, Business and Marketing, 3M Drug Delivery
Systems. “Keeping patient preference top of mind is key and with
this new system, pharmaceutical companies have more options to
satisfy patients.”
“3M™ Hollow Microstructured Transdermal System continues to
demonstrate a number of unique benefits, including reproducible
intradermal delivery, a proven ability to deliver formulations up
to 2 mL with various viscosities, and API-dependent PK profile
benefits,” continued Blair. “Its patient-friendly features and the
ability for patients to easily self-administer open new
opportunities to move treatments out of the clinic and into the
patient’s own home. We are looking forward to working with
pharmaceutical partners to provide this microneedle drug delivery
alternative.”
For more information, visit 3M.com/dds or contact
1-800-643-8086.
*Disclaimer: Initiation of clinical studies may require a
submission for regulatory review.
About 3M Drug Delivery Systems3M Drug Delivery Systems
partners with pharmaceutical and biotech companies to develop and
manufacture pharmaceutical products using 3M's inhalation,
transdermal or microneedle drug delivery technology. 3M offers a
full range of feasibility, development and manufacturing
capabilities to help bring products to market. Regulatory
expertise, quality assurance, operations, marketed product support
and other in-house resources are available for each step of the
development and commercialization process. For more information,
please visit www.3M.com/dds or call 1-800-643-8086.
About 3M3M captures the spark of new ideas and transforms
them into thousands of ingenious products. Our culture of creative
collaboration inspires a never-ending stream of powerful
technologies that make life better. 3M is the innovation company
that never stops inventing. With $31 billion in sales, 3M employs
89,000 people worldwide and has operations in more than 70
countries. For more information, visit www.3M.com or follow @3MNews
on Twitter.
3M is a trademark of 3M Company.
Photos/Multimedia Gallery Available:
http://www.businesswire.com/multimedia/home/20150128005967/en/
Karwoski & CourageMichael Gugala,
612-342-9604m.gugala@creativepr.com
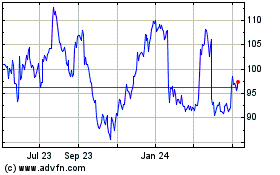
3M (NYSE:MMM)
Historical Stock Chart
From Apr 2024 to May 2024
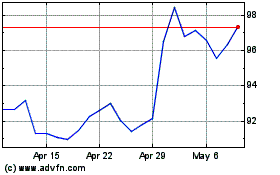
3M (NYSE:MMM)
Historical Stock Chart
From May 2023 to May 2024