Terns Pharmaceuticals Appoints Jeffrey R. Jasper, Ph.D., as Senior Vice President, Head of Research and Promotes Diana Chung to Senior Vice President, Chief Development Officer
January 06 2022 - 7:05AM

Terns Pharmaceuticals, Inc. (“Terns” or the “Company”) (Nasdaq:
TERN), a clinical-stage biopharmaceutical company developing a
portfolio of small-molecule single-agent and combination therapy
candidates to address serious diseases such as non-alcoholic
steatohepatitis (NASH), today announced the appointment of Jeffrey
R. Jasper, Ph.D., as senior vice president, head of research. Dr.
Jasper joins Terns with more than 28 years of experience in the
biopharmaceutical industry. Terns also announced the promotion of
Diana Chung to senior vice president, chief development officer.
Both Dr. Jasper and Ms. Chung will report to Erin Quirk, M.D.,
president, head of research and development and chief medical
officer.
“Attracting top talent and developing our management team is
essential to our success,” said Senthil Sundaram, chief executive
officer at Terns. “We are pleased to have Jeff join our team as his
deep industry experience across a range of therapeutic areas will
be valuable as we advance our current and future pipeline
candidates. Diana is a thoughtful leader who is passionate about
Terns’ commitment to patients. Her clinical and scientific
expertise have been instrumental in our successes to date.
Together, these new appointments further strengthen our team, and
we are well positioned to advance our pipeline towards our first
combination trial for NASH in the first half of 2022.”
Prior to joining Terns, Dr. Jasper served as vice president,
drug discovery at Rubedo Life Sciences, overseeing drug discovery
and nonclinical development efforts on small molecule therapeutics
targeting diseases of aging. Before that, he served as executive
director, research science at Merck Research Laboratories, with
previous positions at Roche Bioscience, Theravance, Cytokinetics
and Revolution Medicines. He also co-founded Altos Therapeutics
(acquired by Takeda Pharmaceuticals) where he led the development
of gastrointestinal therapies. His academic activities include more
than 18 years as a member of the editorial board for the American
Journal of Physiology: Cell Physiology and 12 years instructing the
Molecular Pharmacology course at the University of California,
Berkeley Extension. Dr. Jasper has published more than 50
peer-reviewed journal articles and received his Ph.D. in
pharmacology from the University of California San Diego. He
completed his postdoctoral training at Stanford University and
received his B.S. in chemistry from California Polytechnic State
University.
“The overall goal for anyone in the biopharmaceutical industry
is to make a meaningful difference in the lives of patients
impacted by debilitating diseases and I believe that Terns’
approach to treating NASH, which leverages differentiated
approaches with well-validated, complementary mechanisms of action,
has significant potential to address the multi-faceted processes
involved in this disease. I am pleased to join this talented and
experienced team to continue to build our pipeline of drug
candidates to fulfill unmet needs for patients living with serious
diseases such as NASH,” said Dr. Jasper.
Ms. Chung was previously senior vice president, clinical
development and operations at Terns. She has more than 20 years of
experience in the pharmaceutical industry in drug discovery and
clinical development including management of Phase I-IV studies and
managed access programs.
“I am proud to be part of the Terns team and look forward to
continuing to provide my support and expertise in this new role as
we work towards our goal of bringing best-in-class therapies to
NASH patients,” said Ms. Chung. “The combination of positive
topline data readouts, ongoing and upcoming clinical trials and
anticipation of new drug candidates makes this an exciting time to
be at Terns.”
About Terns Pharmaceuticals
Terns Pharmaceuticals, Inc. is a clinical-stage
biopharmaceutical company developing a portfolio of small-molecule
single-agent and combination therapy candidates to address serious
diseases such as non-alcoholic steatohepatitis, or NASH. Terns’
pipeline includes three clinical stage development programs
including an FXR agonist, a VAP-1 inhibitor and a THR-β agonist,
and a preclinical small-molecule GLP-1 receptor agonist program.
For more information, please visit: www.ternspharma.com.
Cautionary Note Regarding Forward-Looking
Statements
This press release contains forward-looking statements about
Terns Pharmaceuticals, Inc. (the “Company,” “we,” “us,” or “our”)
within the meaning of the federal securities laws, including those
related to the Company’s expectations of timing and potential
results of the Company’s clinical trials and other development
activities, such as the combination trial of TERN-101 and TERN-501
and the AVIATION Trial of TERN-201 in NASH; the therapeutic
potential of the Company’s single-agent and combination therapy
candidates; the potential for the mechanisms of action of the
Company’s product candidates to be therapeutic targets for NASH;
the potential utility and progress of the Company’s product
candidates in NASH, including the clinical utility of the data from
and the endpoints used in the Company’s clinical trials; the
Company’s clinical development plans and activities; the Company’s
expectations regarding the profile of its product candidates,
including tolerability, safety, metabolic stability and
pharmacokinetic profile; the Company’s ability to continue to
execute on its clinical strategy and plans; and the Company’s
expectations with regard to its cash runway. All statements other
than statements of historical facts contained in this press
release, including statements regarding the Company’s strategy,
future financial condition, future operations, future trial
results, projected costs, prospects, plans, objectives of
management and expected market growth, are forward-looking
statements. In some cases, you can identify forward-looking
statements by terminology such as “aim,” “anticipate,” “assume,”
“believe,” “contemplate,” “continue,” “could,” “design,” “due,”
“estimate,” “expect,” “goal,” “intend,” “may,” “objective,” “plan,”
“positioned,” “potential,” “predict,” “seek,” “should,” “target,”
“will,” “would” and other similar expressions that are predictions
of or indicate future events and future trends, or the negative of
these terms or other comparable terminology. The Company has based
these forward-looking statements largely on its current
expectations, estimates, forecasts and projections about future
events and financial trends that it believes may affect its
financial condition, results of operations, business strategy and
financial needs. In light of the significant uncertainties in these
forward-looking statements, you should not rely upon
forward-looking statements as predictions of future events. These
statements are subject to risks and uncertainties that could cause
the actual results and the implementation of the Company’s plans to
vary materially, including the risks associated with the
initiation, cost, timing, progress, results and utility of the
Company’s current and future research and development activities
and preclinical studies and clinical trials. In particular, the
impact of the COVID-19 pandemic on the Company’s ability to
progress with its research, development, manufacturing and
regulatory efforts, including the Company’s clinical trials for its
product candidates, will depend on future developments that are
highly uncertain and cannot be predicted with confidence at this
time, such as the ultimate duration of the pandemic, travel
restrictions, quarantines, social distancing and business closure
requirements in the United States and in other countries, and the
effectiveness of actions taken globally to contain and treat the
disease. These risks are not exhaustive. For a detailed discussion
of the risk factors that could affect the Company’s actual results,
please refer to the risk factors identified in the Company’s SEC
reports, including but not limited to its Annual Report on Form
10-K for the year ended December 31, 2020 and its Quarterly Reports
on Form 10-Q for the periods ended March 31, 2021, June 30, 2021
and September 30, 2021. Except as required by law, the Company
undertakes no obligation to update publicly any forward-looking
statements for any reason.
Contacts for Terns
InvestorsJustin Nginvestors@ternspharma.com
MediaJenna UrbanBerry & Company Public
Relationsmedia@ternspharma.com
Terns Pharmaceuticals (NASDAQ:TERN)
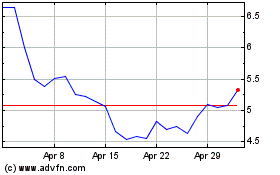
Historical Stock Chart
From Mar 2024 to Apr 2024
Terns Pharmaceuticals (NASDAQ:TERN)
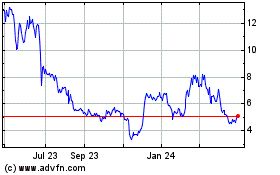
Historical Stock Chart
From Apr 2023 to Apr 2024