Tenaya Therapeutics Launches Operations of New Genetic Medicines Manufacturing Center to Support the Development of Potentially First-In-Class Cardiovascular Therapeutics
June 16 2022 - 8:00AM
Business Wire
Facility to Provide Clinical Supply of Lead
Gene Therapy Programs TN-201 and TN-401 for Planned First-in-Human
Studies
94,000 sq. ft. Modular Facility has Initial
Production Capacity at the 1000L Scale
Tenaya Therapeutics, Inc. (NASDAQ: TNYA), a biotechnology
company with a mission to discover, develop and deliver curative
therapies that address the underlying causes of heart disease,
today announced that it has completed the build-out and operational
launch of its Genetic Medicines Manufacturing Center in Union City,
California. Tenaya is advancing a pipeline of therapeutic
candidates, including several adeno-associated virus (AAV) gene
therapies, for the potential treatment of both rare and prevalent
forms of heart disease.
This press release features multimedia. View
the full release here:
https://www.businesswire.com/news/home/20220616005336/en/
Tenaya’s Genetic Medicines Manufacturing
Center located in Union City, CA (Photo: Business Wire)
“Tenaya made an early, strategic commitment to internalize
several core capabilities to optimize the safety, efficacy, and
supply of our product candidates on behalf of patients. With
today’s announcement we have made a big leap forward on that
commitment by establishing end-to-end in-house manufacturing
capabilities for our pipeline of AAV-based gene therapies,” said
Faraz Ali, Chief Executive Officer of Tenaya. “The operational
launch of Tenaya’s Genetic Medicines Manufacturing Center
represents an important milestone as we prepare to advance our
robust pipeline of potentially first-in-class cardiovascular
therapies into initial clinical studies.”
Tenaya’s Genetic Medicines Manufacturing Center is designed to
meet regulatory requirements for production of AAV gene therapies
from discovery through commercialization under Current Good
Manufacturing Practice (cGMP) standards. Initial production efforts
will support first-in-human studies of Tenaya’s lead gene therapy,
TN-201. TN-201 is being developed for the treatment of genetic
hypertrophic cardiomyopathy (HCM) due to MYBPC3 gene mutations.
Tenaya plans to submit an Investigational New Drug (IND)
application for TN-201 to the U.S. Food and Drug Administration
(FDA) in the second half of this year. The facility will also
support cGMP production for TN-401, Tenaya’s gene therapy program
being developed for the treatment of genetic arrhythmogenic right
ventricular cardiomyopathy (ARVC) due to PKP2 gene mutations, for
which the company plans to submit an IND to the FDA in 2023.
“The investment in our own world-class manufacturing facility
provides Tenaya with greater control over product attributes,
quality, production timelines and costs, which we believe will
ultimately translate into better treatments for patients,” said
Kee-Hong Kim, Ph.D., Chief Technology Officer of Tenaya
Therapeutics. “Tenaya’s Genetic Medicines Manufacturing Center
complements our established internal genetic engineering and drug
discovery capabilities and is designed to meet our near- and
long-term needs such that we can readily scale and expand as our
pipeline matures and evolves.”
Tenaya completed customization of approximately half of the
94,000 square foot facility to incorporate manufacturing suites and
labs, office space and storage. Utilizing a modular design, the
state-of-the-art facility is now fully operational with initial
capacity to produce AAV-based gene therapies at the 1000L scale,
utilizing Tenaya’s proprietary baculovirus-based production
platform and suspension Sf9 cell culture system. The excess space
and modular design of the Genetic Medicines Manufacturing Center is
intended to provide Tenaya with considerable flexibility to expand
manufacturing capacity by increasing both the number and the scale
of bioreactors to meet future clinical and commercial production
needs.
The Union City location, approximately 30 miles from Tenaya’s
South San Francisco headquarters, is expected to enable the
seamless transition of Tenaya’s science from early research through
commercial manufacturing. The selection of this location is
intended to foster a culture of close collaboration across teams at
all stages of developing and testing novel AAV capsids, de-risk the
translation from research to process development and create
opportunities for improvements in production processes. The Genetic
Medicines Manufacturing Center is staffed by a growing in-house
team with expertise in all aspects of gene therapy manufacture,
including process development, analytical development, quality
assurance and quality control.
About Tenaya Therapeutics
Tenaya Therapeutics is a biotechnology company committed to a
bold mission: to discover, develop and deliver curative therapies
that address the underlying drivers of heart disease. Founded by
leading cardiovascular scientists from Gladstone Institutes and the
University of Texas Southwestern Medical Center, Tenaya is
developing therapies for rare genetic cardiovascular disorders, as
well as for more prevalent heart conditions, through three distinct
but interrelated product platforms: Gene Therapy, Cellular
Regeneration and Precision Medicine. For more information, visit
www.tenayatherapeutics.com.
Forward Looking Statements
This press release contains forward-looking statements as that
term is defined in Section 27A of the Securities Act of 1933 and
Section 21E of the Securities Exchange Act of 1934. Statements in
this press release that are not purely historical are
forward-looking statements. Words such as “potential,” “will,”
“plans,” “believe,” “expected,” and similar expressions are
intended to identify forward-looking statements. Such
forward-looking statements include, among other things, statements
regarding the therapeutic potential of Tenaya’s pipeline of
therapeutic candidates; Tenaya’s plan to use the cGMP manufacturing
facility for the production of TN-201 and TN-401; Tenaya’s belief
that it’s cGMP manufacturing facility will enable seamless
transition from early research through commercial manufacturing and
translate into better treatments for patients; the expected timing
for submission of IND applications for TN-201 and TN-401; and
statements by Tenaya’s chief executive officer and chief technology
officer. The forward-looking statements contained herein are based
upon Tenaya’s current expectations and involve assumptions that may
never materialize or may prove to be incorrect. These
forward-looking statements are neither promises nor guarantees and
are subject to a variety of risks and uncertainties, including but
not limited to: risks associated with the process of discovering,
developing and commercializing drugs that are safe and effective
for use as human therapeutics and operating as an early stage
company; Tenaya’s ability to successfully manufacture product
candidates in a timely and sufficient manner that is compliant with
regulatory requirements; Tenaya’s ability to develop, initiate or
complete preclinical studies and clinical trials, and obtain
approvals, for any of its product candidates; the timing, progress
and results of preclinical studies for TN-201, TN-401 and Tenaya’s
other programs; Tenaya’s ability to raise any additional funding it
will need to continue to pursue its business and product
development plans; negative impacts of the COVID-19 pandemic on
Tenaya’s manufacturing and operations, including preclinical
studies and planned clinical trials; the timing, scope and
likelihood of regulatory filings and approvals; the potential for
any clinical trial results to differ from preclinical, interim,
preliminary, topline or expected results; Tenaya’s manufacturing,
commercialization and marketing capabilities and strategy; the loss
of key scientific or management personnel; competition in the
industry in which Tenaya operates; Tenaya’s reliance on third
parties; Tenaya’s ability to obtain and maintain intellectual
property protection for its product candidates; general economic
and market conditions; and other risks. Information regarding the
foregoing and additional risks may be found in the section entitled
“Risk Factors” in documents that Tenaya files from time to time
with the Securities and Exchange Commission. These forward-looking
statements are made as of the date of this press release, and
Tenaya assumes no obligation to update or revise any
forward-looking statements, whether as a result of new information,
future events or otherwise, except as required by law.
View source
version on businesswire.com: https://www.businesswire.com/news/home/20220616005336/en/
Investors Michelle Corral Vice President, Investor
Relationship and Corporate Communications Tenaya Therapeutics
IR@tenayathera.com Media Wendy Ryan Ten Bridge
Communications Wendy@tenbridgecommunications.com
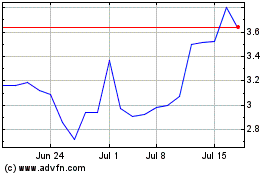
Tenaya Therapeutics (NASDAQ:TNYA)
Historical Stock Chart
From Aug 2024 to Sep 2024
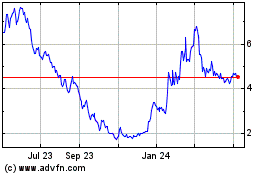
Tenaya Therapeutics (NASDAQ:TNYA)
Historical Stock Chart
From Sep 2023 to Sep 2024