STAAR Surgical Begins Commercialization of EVO Viva™ Presbyopia Correcting Lens
November 04 2020 - 4:03PM
Business Wire
Innovative EVO Viva Implantable Collamer® Lens
Corrects Near, Intermediate and Distance Vision
STAAR Surgical Company (NASDAQ: STAA), a leading developer,
manufacturer and marketer of implantable lenses and companion
delivery systems for the eye, today announced the initial
commercialization of its EVO Viva™ presbyopia correcting
lens including the first patient implant.
This press release features multimedia. View
the full release here:
https://www.businesswire.com/news/home/20201104005615/en/
Erik L. Mertens, MD, FEBOphth, Medical
Director and Eye Surgeon, Mediopolis, Antwerp Private Clinic, Holds
a Packaged EVO Viva™ lens kit. (Photo: Business Wire)
“The first patient received his EVO Viva lenses on
October 23 in Belgium from Dr. Erik Mertens, and we are delighted
to hear from Dr. Mertens that the patient is quite happy with his
lenses,” stated Caren Mason, President and CEO of STAAR Surgical.
“Our presbyopia-correcting EVO Viva lens is aimed at
patients in their 40s and 50s who struggle with near vision and the
burdens associated with wearing reading glasses. EVO Viva
consumer websites are now live in multiple languages, the first
clinical paper has been published and a video clinical summary is
now available.”
Erik Mertens, MD, Medical Director and Eye Surgeon, Mediopolis,
stated, “My experience during the international study and the first
worldwide implantation of the commercially available EVO
Viva ICL made me realize how important it is to make
patients very happy when they get rid of reading glasses. Not being
able to read up close makes people realize they’re getting older
and this frustrates them enormously. With the EVO Viva ICL,
I can rejuvenate the eye without changing the integrity of it
because the crystalline lens and the cornea are preserved. If in
the future an alternative procedure is needed the EVO Viva
ICL can be easily removed without any drawbacks. This innovative
procedure will definitely change the way eye surgeons treat
presbyopia in the near future.”
French, Dutch and Spanish language EVO Viva consumer
websites are now live. Additional local language websites will be
rolled out in the coming months. The websites have been designed to
evoke memories of a time when the patient’s vision was clear –
prior to presbyopia blurring near vision. The nostalgic lookback is
supported by the EVO Viva “Rewind Your Vision” and
“See Young Again!” primary messaging. Visitors to the EVO
Viva websites can learn more about product benefits by
browsing various pages, including Life with Viva, Why Viva,
Procedures and FAQs. The EVO Viva websites include STAAR’s
latest Doc Finder with images of clinics and doctors.
Principal investigating surgeons and Dr. Mark Packer have
published the first clinical paper discussing the EVO Viva
clinical data in the peer-reviewed journal, Clinical
Ophthalmology. The paper outlines the improvements in
uncorrected near, intermediate and distance visual acuity for
patients who desire spectacle independence. The average age of
patients in the trial was 49 with patients who required between
1.00 and 2.50 diopters of reading add with an average of 1.66
diopters. Patients were bilaterally implanted with emmetropia
targeted in all eyes. Objective measures of visual acuity, contrast
sensitivity and defocus curves were measured as well as patient
reported outcomes to understand how patients feel about their
experience. The paper concludes that contrast sensitivity was
preserved with the EVO Viva lens and that patients
demonstrated a high level of satisfaction, improved QoL, and
spectacle independence.
Ms. Mason added, “In the coming months we will work closely with
a select group of implanting surgeons to develop an EVO Viva
playbook of best practices. The playbook, which we expect to
broadly introduce to EVO surgeons at our Experts Summit ahead of
the 39th Congress of the European Society of Cataract and
Refractive Surgeons in Barcelona in September 2021, will be
squarely focused on helping clinicians properly add the Viva
lens to their current armamentarium and optimize the objective and
subjective factors that will delight patients.”
The EVO Viva™ lens is a new treatment option for
potential future consideration by the 1.7 billion people globally
with presbyopia who want to get rid of their reading glasses. The
lens is intended to give a continuous range of focus from near,
through intermediate and to far distance.
1 Fricke, Fricke, Global Prevalence of Presbyopia and Vision
Impairment from Uncorrected Presbyopia, 2018.
About STAAR Surgical
STAAR, which has been dedicated solely to ophthalmic surgery for
over 30 years, designs, develops, manufactures and markets
implantable lenses for the eye with companion delivery systems.
These lenses are intended to provide visual freedom for patients,
lessening or eliminating the reliance on glasses or contact lenses.
All of these lenses are foldable, which permits the surgeon to
insert them through a small incision. STAAR’s lens used in
refractive surgery is called an Implantable Collamer® Lens or
“ICL,” which includes the EVO Visian ICL™ product line. More than
1,000,000 Visian® ICLs have been implanted to date and STAAR
markets these lenses in over 75 countries. To learn more about the
ICL go to: www.discovericl.com. Headquartered in Lake Forest, CA,
the company operates manufacturing and packaging facilities in
Aliso Viejo, CA; Monrovia, CA; and Nidau, Switzerland. For more
information, please visit the Company’s website at
www.staar.com.
Safe Harbor
All statements that are not statements of historical fact are
forward-looking statements, including statements about any of the
following: any financial projections, expectations for sales,
revenue, or earnings relating to the EVO Viva family of lenses, and
any statements of assumptions underlying any of the foregoing,
including those relating to product launch, product success/market
adoption, and market expansion activities. Important factors that
could cause actual results to differ materially from those
indicated by such forward-looking statements include risks and
uncertainties related to the COVID-19 pandemic and related public
health measures, as well as the factors set forth in the Company’s
Quarterly Report on Form 10-Q for the quarter ended April 3, 2020,
and Annual Report on Form 10-K for the year ended January 3, 2020
under the caption “Risk Factors,” which is on file with the
Securities and Exchange Commission and available in the “Investor
Information” section of the company’s website under the heading
“SEC Filings.” We disclaim any intention or obligation to update or
revise any financial projections or forward-looking statement due
to new information or events. These statements are based on
expectations and assumptions as of the date of this press release
and are subject to numerous risks and uncertainties, which could
cause actual results to differ materially from those described in
the forward-looking statements. The risks and uncertainties include
the following: global economic conditions; and the willingness of
surgeons and patients to adopt a new or improved product and
procedure such as the EVO Viva lens. The EVO and EVO Viva versions
of our ICL lens are not yet approved for sale in the United
States.
View source
version on businesswire.com: https://www.businesswire.com/news/home/20201104005615/en/
Investors & Media Brian Moore Vice President,
Investor, Media Relations and Corporate Development (626) 303-7902,
Ext. 3023 bmoore@staar.com
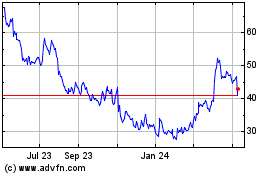
STAAR Surgical (NASDAQ:STAA)
Historical Stock Chart
From Mar 2024 to Apr 2024
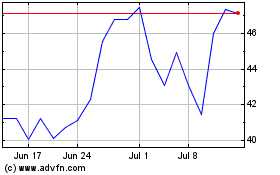
STAAR Surgical (NASDAQ:STAA)
Historical Stock Chart
From Apr 2023 to Apr 2024