Sol-Gel Technologies Gets FDA Approval of Twyneo
July 27 2021 - 9:52AM
Dow Jones News
By Colin Kellaher
Sol-Gel Technologies Ltd. on Tuesday said the U.S. Food and Drug
Administration approved Twyneo, the company's first proprietary
drug product, for the treatment of acne vulgaris in patients ages
nine and older.
The Ness Ziona, Israel, dermatology company, which earlier this
year signed a licensing agreement with privately held Galderma to
commercialize Twyneo in the U.S., said it expects to receive a
regulatory milestone payment in conjunction with the approval.
Sol-Gel noted it retains the option to regain U.S.
commercialization rights five years after the first U.S. sales,
adding that Twyneo is patent-protected until 2038.
Trading in shares of Sol-Gel, which closed Monday at $12.11, was
halted premarket on Tuesday.
Write to Colin Kellaher at colin.kellaher@wsj.com
(END) Dow Jones Newswires
July 27, 2021 09:44 ET (13:44 GMT)
Copyright (c) 2021 Dow Jones & Company, Inc.
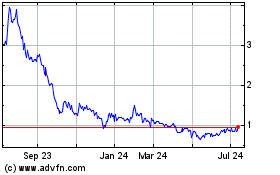
Sol Gel Technologies (NASDAQ:SLGL)
Historical Stock Chart
From Mar 2024 to Apr 2024
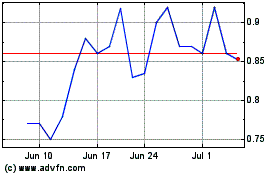
Sol Gel Technologies (NASDAQ:SLGL)
Historical Stock Chart
From Apr 2023 to Apr 2024