NEWARK, Calif., Dec. 3, 2019 /PRNewswire/ -- Protagonist
Therapeutics, Inc. (Nasdaq:PTGX) today announced preliminary
results from patients with transfusion-dependent (TD)
beta-thalassemia in the ongoing TRANSCEND Phase 2 open-label study
of PTG-300. Dose-related drug exposure and reductions from baseline
serum iron and transferrin saturation (TSAT) levels were observed,
with significant reductions at the 40 mg and 80 mg weekly doses (p
< 0.02 for each assessment, see graphs below).
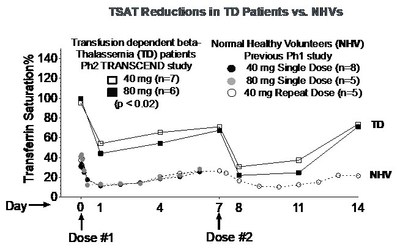
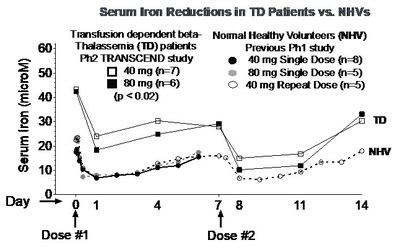
"The dose-related pharmacodynamic responses in serum iron and
TSAT levels observed in this preliminary analysis provide the first
evidence of the effects of PTG-300 in patients with
beta-thalassemia, who have highly elevated levels of iron in the
body," commented Samuel Saks, M.D.,
Protagonist Chief Medical Officer. "These early results suggest the
potential of finding an appropriate dose of PTG-300 for continued
development in the treatment of beta-thalassemia. While we have
observed clinical responders in the study based on the
pre-specified criteria of reductions in transfusion burden,
continued evaluation at higher doses will be required to evaluate
the rate and durability of these effects in order to reach
definitive conclusions. We look forward to the results from further
study with additional dose regimens and longer follow-up, with
clinical efficacy results expected in 2020."
"Treatment options for patients with beta-thalassemia are
limited and the complications associated with transfusion are
serious," commented Ashutosh Lal,
M.D., Program Director of the Comprehensive Thalassemia Center at
the UCSF Benioff Children's Hospital, Oakland, and PTG-300 beta-thalassemia study
investigator. "The TRANSCEND trial is examining whether
constraining iron availability improves endogenous hemoglobin
synthesis in patients with beta-thalassemia, an endpoint for which
there exists considerable preclinical evidence. These results
demonstrating the pharmacodynamic activity of PTG-300 in reducing
TSAT, though preliminary, warrant continued evaluation of PTG-300
for the potential treatment of beta-thalassemia."
"The consistent and significant effect on iron levels observed
in normal healthy volunteers in a previous study, and now in
patients with beta-thalassemia, provides strong rationale for
potential utility of PTG-300 in blood disorders directly dependent
on disruption of normal iron homeostasis in the body," commented
Dinesh V. Patel, Ph.D., Protagonist
President and Chief Executive Officer. "We are encouraged by these
findings and are continuing with our original plans of conducting
clinical proof-of-concept studies with PTG-300 in different blood
disorders such as beta-thalassemia, polycythemia vera, hereditary
hemochromatosis, and an investigator sponsored study in
myelodysplastic syndromes. We are well financed to conduct these
studies and our corporate objective is to make data-driven
decisions in 2020, with the intent of choosing our first clinical
indication for a potential pivotal study to begin in 2021."
In the study, PTG-300 was well-tolerated and systemic adverse
events were mild to moderate in severity and were typical of
patients with TD beta-thalassemia. These events were not
dose-related and did not prevent dose escalation. There was one
serious adverse event of vomiting and confusion, and the most
frequent treatment emergent adverse event observed was transient
erythema in 4 out of 33 patients (12 percent).
Conference Call and Webcast Information
Protagonist will host a conference call at 8:30 a.m.
EST today, Dec. 3, 2019. To access the live call, dial
1-844-515-9178 (U.S./Canada) or
1-614-999-9313 (international) and refer to conference ID number
1662899. The call will also be webcast and will be accessible from
"Events & Presentations" in the Investors section of the
Company's website at www.protagonist-inc.com. A replay will be
available on the Company's website following the call.
About the Phase 2 TRANSCEND Study
The global Phase 2 study is a single-arm, open label,
multiple-ascending dose design that will evaluate safety,
proof-of-concept and dose finding in adolescent and adult patients
with anemia associated with non-transfusion-dependent (NTD) or
transfusion-dependent (TD) beta-thalassemia. NTD patients will
receive 12 weeks treatment with PTG-300 in escalating dose cohorts.
The primary efficacy endpoint in NTD patients will be change in
hemoglobin from baseline. TD patients will receive 16 weeks
treatment with PTG-300 in escalating dose cohorts. The primary
efficacy endpoint in TD patients will be a change in transfusion
burden from baseline. All patients completing the trial will have
the opportunity to participate in an open-label extension for two
years. Additional information on the PTG-300 beta-thalassemia study
is available at
https://clinicaltrials.gov/ct2/show/NCT03802201.
About PTG-300
PTG-300 is an injectable hepcidin mimetic in clinical
development for the potential treatment of beta thalassemia and
polycythemia vera. Hepcidin is a natural peptide hormone that is a
critical regulator governing iron absorption, recycling and
utilization by the body. Iron plays an essential role in various
body functions, especially blood formation. Excess iron in the body
is toxic, resulting in tissue and organ damage over time.
Abnormally low hepcidin levels caused by genetic mutations or
secondary pathology can be addressed with a hepcidin mimetic to
restore iron homeostasis. PTG-300 has been granted Orphan Drug
designation in the U.S. and EU and has received Fast Track
designation by the FDA for development in the treatment of
beta-thalassemia. Myelodysplastic syndromes or hereditary
hemochromatosis represent additional opportunities for the
development of PTG-300.
About Protagonist Therapeutics, Inc.
Protagonist Therapeutics is a clinical stage biopharmaceutical
company that utilizes a proprietary technology platform to discover
and develop novel peptide-based drugs to transform existing
treatment paradigms for patients with significant unmet medical
needs. PTG-300 is an injectable hepcidin mimetic in development for
the potential treatment of iron overload anemia and related rare
blood diseases including beta-thalassemia and polycythemia vera.
PTG-200 is an oral, gut-restricted interleukin-23 receptor specific
antagonist peptide in Phase 2 clinical development for the
potential treatment of inflammatory bowel disease, with Crohn's
disease as the initial indication. The Company has a worldwide
license and collaboration agreement with Janssen Biotech for the
clinical development of PTG-200. PN-943 is an oral, gut-restricted
alpha-4-beta-7 integrin specific antagonist peptide in clinical
development for the potential treatment of inflammatory bowel
disease, with a Phase 2 ulcerative colitis study expected to
commence in the second quarter of 2020.
Protagonist is headquartered in Newark, California. For further information,
please visit http://www.protagonist-inc.com.
Cautionary Note on Forward-Looking Statements
This press release contains forward-looking statements for
purposes of the safe harbor provisions of the Private Securities
Litigation Reform Act of 1995. Forward-looking statements include
statements regarding our intentions or current expectations
concerning, among other things, the potential of PTG-300 as a
possible treatment for TD beta-thalassemia, the Company's success
at finding appropriate doses of PTG-300 for the treatment of
beta-thalassemia, the results of future studies for the treatment
of beta-thalassemia, the potential utility of PTG-300 in blood
disorders including beta-thalassemia, polycythemia vera, and
hereditary hemochromatosis, as well as myelodysplastic syndromes,
the Company's ability to fund its clinical trials, results of the
Phase 2 TRANSCEND Study,, the potential of PTG-200 and PN-943 as
possible treatments for inflammatory bowel disease, the initiation
of and enrollment of patients in our clinical trials, the results
of clinical trials and the outlook for our other programs. In some
cases, you can identify these statements by forward-looking words
such as "plan," "will," "expect," "potential," or the negative or
plural of these words or similar expressions. Forward-looking
statements are not guarantees of future performance and are subject
to risks and uncertainties that could cause actual results and
events to differ materially from those anticipated, including, but
not limited to, our ability to develop and commercialize our
product candidates, our ability to earn milestone payments under
our collaboration agreement with Janssen, our ability to use and
expand our programs to build a pipeline of product candidates, our
ability to obtain and maintain regulatory approval of our product
candidates. Additional information concerning these and other risk
factors affecting our business can be found in our periodic filings
with the Securities and Exchange Commission, including under the
heading "Risk Factors" contained in our Quarterly Report on Form
10-Q for the quarterly period ended September 30, 2019, filed with the Securities and
Exchange Commission. Forward-looking statements are not guarantees
of future performance, and our actual results of operations,
financial condition and liquidity, and the development of the
industry in which we operate, may differ materially from the
forward-looking statements contained in this press release. Any
forward-looking statements that we make in this press release speak
only as of the date of this press release. We assume no obligation
to update our forward-looking statements, whether as a result of
new information, future events or otherwise, after the date of this
press release.
 View original content to download
multimedia:http://www.prnewswire.com/news-releases/protagonist-therapeutics-announces-preliminary-phase-2-results-with-hepcidin-mimetic-ptg-300-in-the-treatment-of-transfusion-dependent-beta-thalassemia-300967872.html
View original content to download
multimedia:http://www.prnewswire.com/news-releases/protagonist-therapeutics-announces-preliminary-phase-2-results-with-hepcidin-mimetic-ptg-300-in-the-treatment-of-transfusion-dependent-beta-thalassemia-300967872.html
SOURCE Protagonist Therapeutics, Inc.