Nemaura Initiates US FDA Studies, and Reports Positive Interim Data from Home-Use Portion of Study
July 31 2018 - 4:00AM

LOUGHBOROUGH, England. Nemaura Medical Inc. (NASDAQ: NMRD), a
medical technology company developing sugarBEAT® as a non-invasive
glucose trending device for use by persons with diabetes and
pre-diabetics today announced it has initiated the first of a
number of planned studies in support of a submission to the U.S
Food & Drug Administration (“FDA”) for approval of its
sugarBEAT® product, and further announced positive interim results
from the home-use portion of this initial study. The interim
results evaluated data from 25 patients, split approximately
equally between Type I and Type II diabetics, each wearing
sugarBEAT whilst going about their daily home/work routine for 1 or
2 non-consecutive days, for a total of 36 patient days. Each
patient recorded up to 5 finger prick readings per day, at
approximately equal intervals, over periods up to 14 hours.
Readings were measured using the Abbott Freestyle Optimum neo Blood
glucose meter (BGM). The results analysed 121 matched pair
points between the BGM and sugarBEAT®, and indicated 84.3% of the
data points had an overall MARD (Mean absolute relative deviation)
of 10.63%, and an overall nominal MARD of 16.3% (compared with
14.8%, 16.3% and 18% for Eversense, Dexcom G5 and Abbott Libre Pro
respectively*). It is the first time the company has published data
from a home use study, which replicates all the challenges of
real-world usage, and the data compares favorably to existing
CGMs. The initial U.S. study is expected to enroll a total of
75 patients wearing the device over a 7-day period, including 3
days in a clinical setting (525 total patient days; 225 total
in-clinic days). Initial U.S. study completion and FDA submission
is anticipated in Q1 2019, alongside poster presentation at ATTD in
Berlin in February 2019 for the in-clinic portion, which is
designed to record approximately 16,000 matched data
points. SugarBEAT® is expected to launch in the UK, followed
by other territories upon achieving CE approval, which is
anticipated in the coming months. *Further detailed summary of
the results are available on the publications page of the company’s
website. About Nemaura Medical, Inc. Nemaura Medical
Inc. (NASDAQ: NMRD), is a medical technology company developing
sugarBEAT® as a non-invasive, affordable and flexible glucose
trending device for use by persons with diabetes and
pre-diabetics.SugarBEAT® consists of a daily disposable adhesive
skin-patch connected to a rechargeable transmitter, with an app
displaying glucose readings at five minute intervals.SugarBEAT® can
additionally be used by insulin using persons with diabetes as an
adjunctive glucose monitoring device when calibrated by a finger
stick reading. For more information
visit: www.NemauraMedical.com www.SugarBEAT.com.Cautionary
Statement Regarding Forward Looking StatementsThe statements in
this press release that are not historical facts, and may
constitute forward-looking statements that are based on current
expectations and are subject to risks and uncertainties that could
cause actual future results to differ materially from those
expressed or implied by such statements. Those risks and
uncertainties include, but are not limited to, risks related to
regulatory approvals and the success of Nemaura Medical’s ongoing
studies, including the safety and efficacy of Nemaura Medical’s
sugarBEAT® system, the failure of future development and
preliminary marketing efforts, Nemaura Medical’s ability to secure
additional commercial partnering arrangements, risks and
uncertainties relating to Nemaura Medical and its partners’ ability
to develop, market and sell sugarBEAT®, the availability of
substantial additional equity or debt capital to support its
research, development and product commercialization activities, and
the success of its research, development, regulatory approval,
marketing and distribution plans and strategies, including those
plans and strategies related to sugarBEAT®. These and other risks
and uncertainties are identified and described in more detail in
Nemaura Medical’s filings with the Securities and Exchange
Commission, including, without limitation, its Annual Report on
Form 10-K for the current year, its Quarterly Reports on Form 10-Q,
and its Current Reports on Form 8-K. Nemaura Medical undertakes no
obligation to publicly update or revise any forward-looking
statements.ContactsNemaura MedicalBashir TimolDirector of Strategy
& Corporate Developmentbashir.timol@nemauramedical.comorThe
Ruth GroupLee Roth646-536-7012lroth@theruthgroup.com
Nemaura Medical (NASDAQ:NMRD)
Historical Stock Chart
From Jun 2024 to Jul 2024
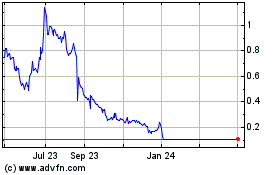
Nemaura Medical (NASDAQ:NMRD)
Historical Stock Chart
From Jul 2023 to Jul 2024