UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
WASHINGTON, D.C. 20549
FORM 8-K
CURRENT REPORT
Pursuant to Section 13 or 15(d) of the Securities Exchange Act of 1934
Date of Report (date of earliest event reported): June 2, 2019
Heat Biologics, Inc.
(Exact name of registrant as specified in charter)
Delaware
(State or other jurisdiction of incorporation)
| |
001-35994
| 26-2844103
|
(Commission File Number)
| (IRS Employer Identification No.)
|
801 Capitola Drive
Durham, NC 27713
(Address of principal executive offices and zip code)
(919) 240-7133
(Registrant’s telephone number including area code)
N/A
(Former Name and Former Address)
Check the appropriate box below if the Form 8-K filing is intended to simultaneously satisfy the filing obligation of registrant under any of the following provisions:
| | |
| ¨
| Written communications pursuant to Rule 425 under the Securities Act (17 CFR 230.425)
|
| ¨
| Soliciting material pursuant to Rule 14a-12 under the Exchange Act (17 CFR 240.14a-12)
|
| ¨
| Pre-commencement communications pursuant to Rule 14d-2(b) under the Exchange Act (17 CFR 240.14d-2(b))
|
| ¨
| Pre-commencement communications pursuant to Rule 13e-4(c) under the Exchange Act (17 CFR 240.13e-4(c))
|
Indicate by check mark whether the registrant is an emerging growth company as defined in Rule 405 of the Securities Act of 1933 (§230.405 of this chapter) or Rule 12b-2 of the Securities Exchange Act of 1934 (§240.12b-2 of this chapter).
| |
Emerging growth company ¨
|
|
If an emerging growth company, indicate by checkmark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act. ¨
Securities registered pursuant to Section 12(b) of the Act:
| | |
Title of each class
| Trading Symbol(s)
| Name of each exchange on which registered
|
Common Stock, $0.0002 par value per share
| HTBX
| The Nasdaq Capital Market
|
Item 7.01 Regulation FD Disclosure.
Members of the management team of Heat Biologics, Inc. (the “Company”) and certain clinical investigators in its ongoing Phase 2 study investigating HS-110 in combination with Bristol-Myers Squibb's anti-PD-1 checkpoint inhibitor, nivolumab (Opdivo®) presented the information in the presentation poster (the “Poster”) entitled “Viagenpumatucel-L (HS-110) plus Nivolumab In Patients With Advanced Non-Small Cell Lung Cancer After Checkpoint Inhibitor Treatment Failure” on June 2, 2019 at the American Society of Clinical Oncology’s 2019 Annual Meeting held in Chicago, Illinois. A copy of the Poster is attached to this Current Report on Form 8-K as Exhibit 99.1 and is incorporated herein by reference.
The furnishing of the attached Poster is not an admission as to the materiality of any information therein. The information contained in the Poster is summary information that is intended to be considered in the context of more complete information included in the Company’s filings with the Securities and Exchange Commission (the “SEC”) and other public announcements that the Company has made and may make from time to time by press release or otherwise. The Company undertakes no duty or obligation to update or revise the information contained in this report, although it may do so from time to time as its management believes is appropriate. Any such updating may be made through the filing of other reports or documents with the SEC, through press releases or through other public disclosures.
Item 8.01 Other Events.
On June 3, 2019, the Company issued a press release announcing that on June 2, 2019 members of the management team of the Company and certain clinical investigators in its ongoing Phase 2 study investigating HS-110 in combination with Bristol-Myers Squibb's anti-PD-1 checkpoint inhibitor, nivolumab (Opdivo®) presented the Poster at the American Society of Clinical Oncology’s 2019 Annual Meeting held in Chicago, Illinois. A copy of the press release regarding the Poster is attached as Exhibit 99.2 and incorporated herein by reference. The Poster provides new data (including additional interim data and 3 additional months of data since last data release) from the Company’s ongoing Phase 2 study investigating HS-110 in combination with Bristol-Myers Squibb's anti-PD-1 checkpoint inhibitor, nivolumab (Opdivo®). The updated results were obtained from Cohort B patients whose data has matured an additional 3 months since last reported at the ASCO-SITC Clinical Immuno-Oncology Symposium in February of this year.
A copy of the Company’s new corporate slide presentation that includes information from the Poster is also attached to this Current Report on Form 8-K as Exhibit 99.3 and is incorporated herein by reference.
The Company undertakes no duty or obligation to update or revise information included in this Current Report or the Exhibits.
Item 9.01. Financial Statements and Exhibits.
(d) Exhibits.
The following exhibits are filed with this Current Report on Form 8-K:
SIGNATURES
Pursuant to the requirements of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned hereunto duly authorized.
| | |
Dated: June 3, 2019
| HEAT BIOLOGICS, INC.
|
|
|
|
|
|
| By:
| /s/ Jeffrey Wolf
|
| Name:
| Jeffrey Wolf
|
| Title:
| Chairman, President and
Chief Executive Officer
|
EXHIBIT 99.1
![[htbx_ex99z1001.jpg]](http://www.sec.gov/Archives/edgar/data/1476963/000155335019000612/htbx_ex99z1001.jpg)
EXHIBIT 99.2
![[htbx_ex99z2001.jpg]](http://www.sec.gov/Archives/edgar/data/1476963/000155335019000612/htbx_ex99z2001.jpg)
Heat Biologics Announces Promising Interim Phase 2 Lung Cancer Data Suggesting that HS-110 Plus Nivolumab May Restore Clinical Benefit After Checkpoint Inhibitor Treatment Failure
Clinical benefit observed in 55% of patients receiving HS-110 plus nivolumab after checkpoint inhibitor treatment failure
HS-110 in combination with nivolumab demonstrates clinical activity in low CD8+ TIL “cold tumor” patients and PD-L1 negative tumors
The occurrence of dermal injection site reactions is associated with improved progression free survival (p=0.013) and overall survival (p=0.002)
Cohort B results presented yesterday at the 2019 ASCO Annual Meeting poster session
DURHAM, NC – – June 3, 2019 – Heat Biologics, Inc. (NASDAQ: HTBX), a biopharmaceutical company developing therapies designed to activate a patient’s immune system against cancer, today announced compelling new interim results from its ongoing Phase 2 study investigating HS-110 in combination with Bristol-Myers Squibb's anti-PD-1 checkpoint inhibitor, nivolumab (Opdivo®). The updated results were obtained from Cohort B patients whose data has matured an additional 3 months since last reported at the ASCO-SITC Clinical Immuno-Oncology Symposium in February of this year. This data may represent the first Phase 2 data showing clinical activity in non-small cell lung cancer (NSCLC) patients whose disease has progressed after prior treatment with a checkpoint inhibitor (CPI). The Cohort B results were presented yesterday at the 2019 American Society of Clinical Oncology (ASCO) Annual Meeting poster session.
COL(ret) George E Peoples, MD, FACS, Heat’s Chief Medical Advisor, noted, “These latest Cohort B data provides us even greater confidence that the addition of HS-110 to nivolumab may restore anti-tumor activity in patients whose disease has progressed after treatment with a CPI. Of particular note, 4 out of 5 evaluable patients in Cohort B with PD-L1 negative tumors achieved disease stabilization and 4 out of 7 evaluable patients with low CD8+ TIL levels in their tumors achieved disease stabilization. We are encouraged by these positive results and look forward to reporting additional data later this year.”
Jeff Hutchins, Ph.D., Heat’s Chief Scientific and Operating Officer said, “The fact that we saw tumor shrinkage in 35% of patients and disease control in 55% of patients whose disease has progressed after treatment with a CPI supports our mechanistic hypothesis that the broad, T-cell mediated immune response activated by HS-110 may improve clinical outcomes for patients who have lost the benefit of treatment with a checkpoint inhibitor. It is also important to note that the occurrence of dermal injection site reactions is associated with statistically significant improved progression free survival and overall survival, providing further support for the mechanism of action of HS-110.”
Highlights for Cohort B patients are presented below:
·
HS-110 in combination with nivolumab demonstrates clinical activity in ‘difficult to treat’ low CD8+ TIL (≤10%) and PD-L1 negative (<1%) tumors:
·
4 out of 5 evaluable patients with PD-L1 negative tumors achieved disease stabilization, 1 of which was a RECIST partial response.
·
4 out of 7 evaluable patients with low CD8+ TIL tumors achieved disease stabilization, 2 of which were RECIST partial responses.
·
The addition of HS-110 to nivolumab may restore clinical benefit to patients whose disease has progressed after CPI treatment failure:
·
Tumor shrinkage observed in 35% of patients
·
Disease control rate of 55%
·
Median Progression-Free Survival (mPFS) of 2.7 months
·
Median Overall Survival (mOS) not yet reached
·
The occurrence of any grade dermal Injection Site Reaction during treatment (Y/N) is associated with improved Progression-Free Survival and Overall Survival:
·
mPFS: NR vs 1.8 months; HR 0.17 (95% CI, 0.03-0.84); p=0.013
·
mOS: NR vs 5 months; HR 0.13 (95% CI, 0.02-0.71); p=0.002
Treatment with HS-110 in combination with nivolumab was well tolerated, with no additional toxicities beyond those observed with single agent CPI therapy.
Trial results are summarized in the official 2019 ASCO Annual Meeting poster.
Trial Design
The Phase 2 trial is designed to evaluate the safety and efficacy of HS-110 combined with an immune checkpoint inhibitor for the treatment of advanced non-small cell lung cancer. Cohort B consists of patients who received a minimum of 4 months of treatment with a checkpoint inhibitor (CPI) as part of their prior therapy, but subsequently had documented progressive disease. Patients receive weekly HS-110 (1 x 107 cells) administered as 5 intradermal 0.1 mL injections for 18 weeks in combination with bi-weekly nivolumab 240 mg IV administered until confirmed disease progression or unacceptable toxicity, whichever occurs first. The primary endpoint is objective response rate (ORR); secondary endpoints include overall survival (OS), progression-free survival (PFS), disease control rate (DCR) and duration of response (DOR). Exploratory endpoints include correlation of clinical outcomes to baseline CD8+ TILs, PD-L1 expression, peripheral blood tumor mutation burden and ELISPOT analysis.
About Heat Biologics, Inc.
Heat Biologics is a biopharmaceutical company developing immunotherapies designed to activate a patient's immune system against cancer using of CD8+ "Killer" T-cells. Our T-Cell Activation Platform ("TCAP") produces therapies designed to turn "cold" tumors "hot" and be administered in combination with checkpoint therapies and other immuno-modulators to increase their effectiveness. HS-110 is our first biologic product candidate in a series of proprietary immunotherapies designed to stimulate a patient's own T-cells to attack cancer. Our ComPACT technology is the first potential, dual-acting immunotherapy designed to deliver T-cell activation and co-stimulation in a single product. We are currently enrolling patients in our Phase 2 clinical trial for advanced non-small cell lung cancer, in combination with Bristol-Myers Squibb's nivolumab (Opdivo®) and with Merck's pembrolizumab (Keytruda®). Pelican Therapeutics, Inc., a subsidiary of Heat, is focused on the development of co-stimulatory monoclonal antibody and fusion protein-based therapies designed to activate the immune system. For more information, please visit www.heatbio.com.
Forward Looking Statements
This press release includes forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995 on our current expectations and projections about future events. In some cases, forward-looking statements can be identified by terminology such as "may," "should," "potential," "continue," "expects," "anticipates," "intends," "plans," "believes," "estimates," and similar expressions. These statements are based upon current beliefs, expectation, and assumptions and include statements that the data may represent the first Phase 2 data showing clinical efficacy for non-small cell lung cancer patients whose disease has progressed after treatment with a checkpoint inhibitor (CPI), that the addition of HS-110 to nivolumab may restore anti-tumor activity in patients whose disease has progressed after treatment with a CPI, that the broad, T-cell mediated immune response activated by HS-110 may improve clinical outcomes for patients who have lost the benefit of treatment with a checkpoint inhibitor and the ability of Heat’s T-Cell Activation Platform to produce therapies designed to turn "cold" tumors "hot" and to increase their effectiveness of checkpoint therapies and other immuno-modulators. These statements are subject to a number of risks and uncertainties, many of which are difficult to predict, including the ability of Heat's therapies to perform as designed, to demonstrate safety and efficacy, as well as results that are consistent with prior results, the ability to enroll patients and complete the clinical trials on time and achieve desired results and benefits, Heat's ability to obtain regulatory approvals for commercialization of product candidates or to comply with ongoing regulatory requirements, regulatory limitations relating to Heat's ability to promote or commercialize its product candidates for specific indications, acceptance of its product candidates in the marketplace and the successful development, marketing or sale of products, Heat's ability to maintain its license agreements, the continued maintenance and growth of its patent estate, its ability to establish and maintain collaborations, its ability to obtain or maintain the capital or grants necessary to fund its research and development activities, and its ability to retain its key scientists or management personnel, and the other factors described in Heat's filings with the SEC. The information in this release is provided only as of the date of this release, and Heat undertakes no obligation to update any forward-looking statements contained in this release based on new information, future events, or otherwise, except as required by law.
Media and Investor Relations Contact
David Waldman
+1 919 289 4017
investorrelations@heatbio.com
EXHIBIT 99.3

Heat Biologics NASDAQ: HTBX CORPORATE PRESENTATION June 2019

Forward Looking Statements This presentation includes statements that are, or may be deemed, ‘‘forward - looking statements’’ within the meaning of the Private Securities Litigation Reform Act of 1995 , as amended . In some cases, these forward - looking statements can be identified by the use of forward - looking terminology, including the terms “believes,” “estimates,” “anticipates,” “expects,” “plans,” “intends,” “may,” “could,” “might,” “will,” “should,” “approximately” or, in each case, their negative or other variations thereon or comparable terminology, although not all forward - looking statements contain these words . They appear in a number of places throughout this presentation and include statements regarding our intentions, beliefs, projections, outlook, analyses or current expectations concerning, among other things, our ongoing and planned discovery and development of drugs targeting cancer, the strength and breadth of our intellectual property, our ongoing and planned preclinical studies and clinical trials, the timing of and our ability to complete clinical trials and make regulatory filings and obtain and maintain regulatory approvals for our product candidates, our ability to partner our product development, the degree of clinical utility of our products, particularly in specific patient populations, expectations regarding clinical trial data, our results of operations, financial condition, liquidity, prospects, growth and strategies, the length of time that we will be able to continue to fund our operating expenses and capital expenditures, our expected financing needs and sources of financing, the industry in which we operate and the trends that may affect the industry or us . By their nature, forward - looking statements involve risks and uncertainties because they relate to events, competitive dynamics, and healthcare, regulatory and scientific developments and depend on the economic circumstances that may or may not occur in the future or may occur on longer or shorter timelines than anticipated . Although we believe that we have a reasonable basis for each forward - looking statement contained in this presentation, we caution you that forward - looking statements are not guarantees of future performance and that our actual results of operations, financial condition and liquidity, and the development of the industry in which we operate may differ materially from the forward - looking statements contained in this presentation as a result of, among other factors, the factors referenced in the “Risk Factors” section of our Annual Report on Form 10 - K/A for the year ended December 31 , 2018 , our quarterly reports on Form 10 - Q for the subsequent quarters and our other subsequent filings with the Securities and Exchange Commission (collectively, our “SEC Filings”) . In addition, even if our results of operations, financial condition and liquidity, and the development of the industry in which we operate are consistent with the forward - looking statements contained in this presentation, they may not be predictive of results or developments in future periods . Any forward - looking statements that we make in this presentation speak only as of the date of such statement, and we undertake no obligation to update such statements to reflect events or circumstances after the date of this presentation, except as required by law . 2

Our Mission To improve patient outcomes by developing more effective immunotherapies designed to Turn “COLD” tumors “HOT 3

T - cell Activation Co - stimulation Heat’s Core Focus Checkpoint Inhibition Effective Immuno - Oncology Therapy The three legs of an Immuno - Oncology Stool 4

Heat’s Combination Platforms Unlocking the Body’s Natural Defenses with a Broad Range of Combination Therapies Checkpoint Inhibitors PD1/PDL1 CTLA - 4 Lag - 3 TIM - 3 Plus others Combined with Co - Stimulation T - cell Activation Pelican PTX - 35 Monoclonal Antibody ImPACT® Therapy Cell - based Delivery of Multiple Antigens Activation of Patients’ CD8+ “Killer” T - cells ComPACT™ Therapy Cell - based Delivery of Multiple Antigens Activation of Patients’ CD8+ “Killer” T - cells Co - Stimulation to Enhance T - cell Activation and Expansion Heat Technologies 5

Product Pipeline Combination Therapies Designed to Activate CD8+ T - cells Against Cancer Combination Therapies Indication Comments ImPACT ® HS - 110 NSCLC ImPACT ™ activation technology in combination with nivolumab and pembrolizumab ComPACT ® HS - 130 Multiple Solid Tumors ComPACT™ activation technology in combination with checkpoint inhibitors Co - stimulators PTX - 35 Multiple Solid Tumors Humanized monoclonal antibody, functional agonist of human TNFRSF25 ($15.2M CPRIT grant) Pre - clinical Phase 1 Phase 2 Phase 3 6

ImPACT® “Off - the - shelf” Manufacturing Designed for Robust, Pan - antigen T - cell Activation • Frozen, fully - diluted single - dose vial • Final drug product: 10 million cells • Easily scaled manufacturing Low COG, off - the - shelf alternative to autologous therapies 7

Introducing gp96 – Its dual role The Immune System’s “Swiss Army Knife”* * Schild , H. & Rammensee , H. Gp - 96 – The Immune System’s Swiss Army Knife. Nature Immunology 2, 100 - 101 (2000) “Molecular Warning System” • A natural biological process to deliver proteins (antigens) & gp96 to our immune system • Gp96 “chaperones” newly - created proteins to the cell membrane where they are released and embedded • Gp96 chaperoned proteins are only naturally released via necrosis • Gp96, when present outside the cell, alerts the immune system and then becomes one of the most powerful adjuvants that show exclusive specificity to CD8+ (“killer”) T - cells Tethered to our cells with a “KDEL” leash gp96 antige n Leash Endoplasmic Reticulum Cell Membrane Antigen Antigen 8

ImPACT ® technology genetically modifies tumor cells by “severing the leash” that binds the gp96 to the endoplasmic reticulum, and replacing it with a secretory sequence that pumps gp96 out of the cell Mimics necrotic cell death by enabling fully - allogeneic “off - the - shelf” living cancer cells to “pump - out” their own antigens along with their gp96 chaperone Heat Biologics ImPACT® technology reprograms cancer cells to continuously secrete their own antigens ImPACT technology removes the leash that binds gp96 to the cell Heat’s T Cell Activation Platform - ImPACT ® “Severing the Leash” gp96 antige n Leas h Endoplasmic Reticulum Cell Membrane Antigen Designed to activate a powerful pan - antigen cytotoxic T - cell immune response 9

ImPACT® : I mmune P an - a ntigen C ytotoxic T herapy ImPACT Cell Cancer - killing T - cell ImPACT cells secrete antigens designed to specifically activate patient’s killer T - cells T - cells circulate to destroy patient’s tumor Cluster of five 0.1 mL intradermal injections Activated cells EXPRESS chaperoned antigens Activation Cell CD8+ T Cell Dendritic Cell Chaperoned antigens activate dendritic cells, which then ACTIVATE & PROLIFERATE CD8+ T - cells Dendritic Cell Chaperoned Antigen CD8+ T - cells locate and ELIMINATE cancer cells Tumor Cells CD8+ T Cells Heat’s unique cell - secreted gp96 firstly activates dendritic cells via TLR signaling and subsequently CD8+ T cells via antigen cross presentation 10

HS - 110 Generates an Adaptive Immune Response 1. Secretion of gp96 - Ig carrying tumor specific proteins represented on the patients tumor 2. Activation of APCs (TLR2/4) and cross - presentation of antigens (CD91) 3. Specific T - cell receptor engagement 4. Clonal Expansion of Tumor Antigen Specific T cells. 2 signals Delivered to APCs: x Antigen cross presentation to MHC class I via CD91 x Up regulation of co - stimulatory signals via TLR2/4 2 4 6 8 10 12 0 5 10 15 20 0 5 10 15 20 Day Post Transfer / Treatment % O T - I / C D 8 + T c e l l s % O T - 2 / C D 4 + T c e l l s P 11 HS - 110 (AD100 cell line)

HS - 110 + Opdivo Combination Therapy Potential to improve clinical responses and survival, without additional toxicity Source: BMS images 12

HS - 110 Trial Design A Phase 1b/2 study of HS - 110 in combination with multiple treatment regimens for patients with non - small cell lung cancer (The “DURGA” Trial) Primary Endpoints Phase 1b: Safety Phase 2: Cohort A&B: ORR Cohort C&D: PFS Secondary Endpoints OS, PFS, DCR, DOR Exploratory Endpoints Correlation of clinical outcomes to the following factors • Baseline CD8+ TILs (Low defined as ≤ 10% stromal CD8+ TILs) • Baseline PD - L1 expression (Negative defined as < 1% on tumor cells) • Peripheral blood tumor mutation burden (Low defined as < 10 mutations / Mb) • ELISPOT cytokine analysis Patient Treatment Setting Treatment Arm Enroll 2 nd Line or Greater N = 100 1 st Line Maintenance N = up to 20 Cohort HS110 + Nivolumab Cohort A: CPI naïve (N=40, including Phase 1b) Cohort B: CPI progressor (N=60) HS110 + Pembrolizumab +/ - Pemetrexed Cohort C: Maintenance pembrolizumab Cohort D: Maintenance pembrolizumab + pemetrexed 13

Cohort A: Patient Characteristics 14 Stage III or IV Advanced NSCLC Patients Cohort A (N = 42) EGFR or ALK positive 9 (22%) Prior lines of Tx 1 2 or more Unavailable 27 (64%) 13 (30%) 2 (5%) PD - L1 < 1% ≥ 1% Unevaluable 16 (38%) 13 (31%) 13 (31%) CD8+ TIL ≤ 10% > 10% Unevaluable 12 (29%) 8 (19%) 22 (52%) Cohort A (N = 42) Median age (range) 64 (37 - 87) Female gender 22 (52%) Caucasian 38 (90%) ECOG PS 1 26 (62%) Histology Adeno Squamous 39 (93%) 3 (7%) Smoking Status Current/past Never 37 (88%) 5 (12%) As of last data cut - off in January, 2019

Baseline (Cold) Week 10 (Hot) CD8+ TIL Infiltration Associated with Clinical Response Patient 1: Partial Response at Week 18 High CD8+ TIL ( > 10%) Patient 2: Partial Response at Week 9 High CD8+ TIL ( > 10%) Patient 3: Partial Response at Week 18 Low CD8+ TIL ( < 10%) High CD8+ TIL ( > 10%) Combination treatment drives “killer” CD8+ T - cells deep into tumors Low CD8+ TIL ( < 10%) Low CD8+ TIL ( < 10%) Clinical Support for HS - 110 + Nivolumab Mechanism of Action “Turning COLD Tumors HOT” 15
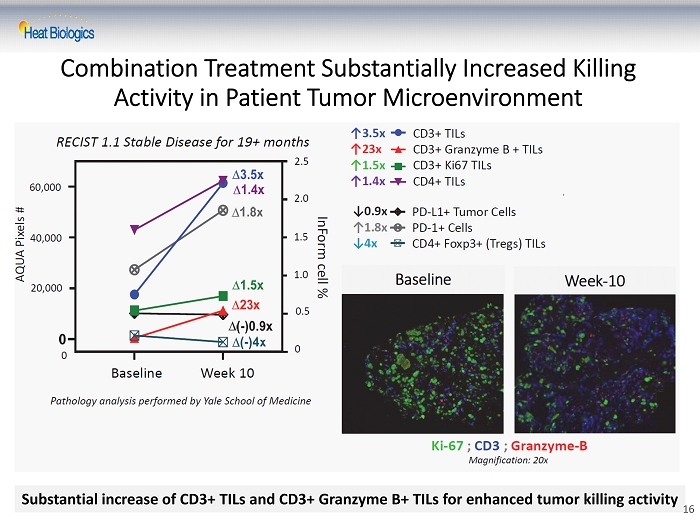
Combination Treatment Substantially Increased Killing Activity in Patient Tumor Microenvironment Substantial increase of CD3+ TILs and CD3+ Granzyme B+ TILs for enhanced tumor killing activity 16

0 6 12 18 24 30 0 25 50 75 100 Months P r o g r e s s i o n - F r e e S u r v i v a l ( % ) Progression - Free Survival (PFS) Cohort A: CPI Naïve pts treated by HS - 110 + Nivolumab at > 2L * Borghaei et al. 2015 Nivolumab versus Docetaxel in Advanced Nonsquamous Non – Small - Cell Lung Cancer. New England Journal of Medicine Median PFS 2.6 months (95% CI: 1.8 - 8 months) Months 0 6 12 18 24 30 25 50 75 100 Median PFS of nivolumab alone 2.3 months * (95% CI: 2.2 – 3.3 months) Progression Free Survival (%) # at risk 42 14 8 5 1 0 ITT Patient Population As of last data cut - off in January, 2019 17

0 6 12 18 24 30 36 42 0 25 50 75 100 Months S u r v i v a l ( % ) Overall Survival (%) Overall Survival (OS) Cohort A: CPI Naïve pts treated by HS - 110 + Nivolumab at > 2L Median OS of nivolumab alone 12.2 months* (95% CI: 9.7 – 15.0 months) * Borghaei et al. 2015 NEJM Median OS Not Reached (95% CI: 8.1 months - NR) 60% of patients still alive with median follow - up time of 14.4 months Months 25 50 75 100 0 6 12 18 24 30 36 42 # at risk 42 29 21 13 5 3 1 0 ITT Patient Population As of last data cut - off in January, 2019 18

Best Target Lesion Response PR 9 (21%) SD 12 (29%) Not evaluable 4 (10%) DCR 21 (50%) RECIST 1.1 Objective Response Rate = 21.4% (95% CI: 10.3 - 36.8%) -100 -75 -50 -25 0 25 50 75 100 C h a n g e f r o m B a s e l i n e ( % ) Change from Baseline (%) Best Target Lesion Response Best Overall Response Cohort A: CPI Naïve pts treated by HS - 110 + Nivolumab at > 2L Nivolumab alone in CPI naïve patients* ORR = 19% (95% CI: 15% - 24%) DCR = 44% * Borghaei et al 2015 NEJM Waterfall plot of best target lesion response using percent change from baseline of the SLD (sum of longest diameters) for all patients who received at least 1 post - baseline scan (n= 38 ) 19 ITT (N=42) Evaluable ITT (N=38) 22 out of 38 patients had no increase in tumor burden from baseline As of last data cut - off in January, 2019

Duration of Benefit Cohort A: CPI Naïve pts treated by HS - 110 + Nivolumab at > 2L 20 Responses and disease stabilization are durable and long - lasting 25 50 75 100 0 - 30 - 50 - 75 Change from Baseline (%) 0 3 6 9 12 15 18 21 24 27 Months from the First Dose of HS - 110 - 100 HS - 110 Dosing Completion As of last data cut - off in January, 2019

• Survival benefit observed in patients with low CD8+ TIL at baseline , as compared to high CD8+ TIL at baseline, HR = 0.39 (95% CI: 0.06 – 2.31) • Median overall survival not reached for both groups • The above benefit is contrary to what is expected based on current literature Gibney et al. Lancet Oncol. 2016 December ; 17(12): e542 – e551 Improved Survival in “Cold” Tumor Patients Overall Survival (OS) by Baseline CD8+ TIL Cohort A: CPI Naïve pts treated by HS - 110 + Nivolumab at > 2L 21

Cohort A: CPI Naïve pts treated by HS - 110 + Nivolumab at > 2L PD - L1 (<1%), N = 16 PD - L1 ( > 1%), N = 13 PD - L1 NE, N = 13 22 0 6 12 18 24 30 36 42 0 25 50 75 100 Months S u r v i v a l ( % ) PD-L1 (<1%), n = 16 PD-L1 (1%), n = 13 PD-L1 NE, n = 13 0 6 12 18 24 30 36 42 25 50 75 Overall Survival (%) Months 100 * Borghaei et al 2015 NEJM • Our therapy is agnostic to PD - L1 status • Similar survival observed in both PD - L1 positive and PD - L1 negative patients HR 0.85 (95% CI: 0.26 – 2.79) • For nivolumab alone, PD - L1 positive patients typically have better outcomes * Benefit Independent of PD - L1 Status Overall Survival (OS) by Baseline PD - L1 Status As of last data cut - off in January, 2019

23 Cohort A: CPI Naïve pts treated by HS - 110 + Nivolumab at > 2L OS by Injection Site Reaction (ISR) Survival is significantly better in patients who experienced dermal injection site reaction, thus supporting the HS - 110 mechanism of action 0 6 12 18 24 30 36 42 0 25 50 75 100 Months S u r v i v a l ( % ) ISR Yes (n = 24) ISR No (n = 18) ISR = No (N=18) Months 0 6 12 18 24 30 36 42 25 50 100 75 # at risk 24 18 22 9 19 4 13 2 6 0 4 0 2 0 0 0 Overall Survival (%) ISR = Yes (N = 24) Typical Injection Site Reaction mOS : NR vs. 7.2 months HR: 0.15 (95% CI: 0.05 - 0.45) p = 0.0001 As of last data cut - off in January, 2019

0 12 24 36 Low High OS (months) H S - 1 1 0 G e n e r a t e d E L I S P O T s Median OS (months) 17.3 23.5 Survival Benefit with Increased Immune Activity Cohort A: CPI Naïve pts treated by HS - 110 + Nivolumab at > 2L OS (Months) 0 12 24 36 High Low HS - 110 Generated ELISPOTs High = Patients with absolute ELISPOT increases above the group median Low = Patients with absolute ELISPOT increases below the group median Over the course of treatment, patients* with higher levels of HS - 110 elicited immune response from peripheral blood demonstrated a survival benefit of approximately 6 months 24 * Only patients with blood samples available at baseline and during treatment were included in this analysis As of last data cut - off in January, 2019

Cohort B: Patient Characteristics 25 As of last data cut - off in January, 2019 • Stage III or IV advanced NSCLC patients • Patients were heavily pretreated (80% with 2 + lines of prior therapy) Cohort B (N = 20) PD - L1 < 1% ≥ 1% Unevaluable 7 (35%) 8 (40%) 5 (25%) CD8+ TIL ≤ 10% > 10% Unevaluable 7 (35%) 6 (30%) 7 (35%) Time (months) on prior CPI: Median (range) 10.2 (6, 19) Time (months) between last CPI dose and study entry: Median(range) 1.7 (1, 21) Cohort B (N = 20) Median age (range) 65 (56 - 84) Female gender 14 (70%) Caucasian 15 (75%) ECOG PS 1 10 (50%) EGFR or ALK positive 2 (10%) Histology Adeno Squamous 17 (85%) 3 (15%) Smoking Status Current/past Never 17 (85%) 3 (15%) Prior lines of Tx 1 2 ≥ 3 3 (15%) 9 (45%) 8 (40%)
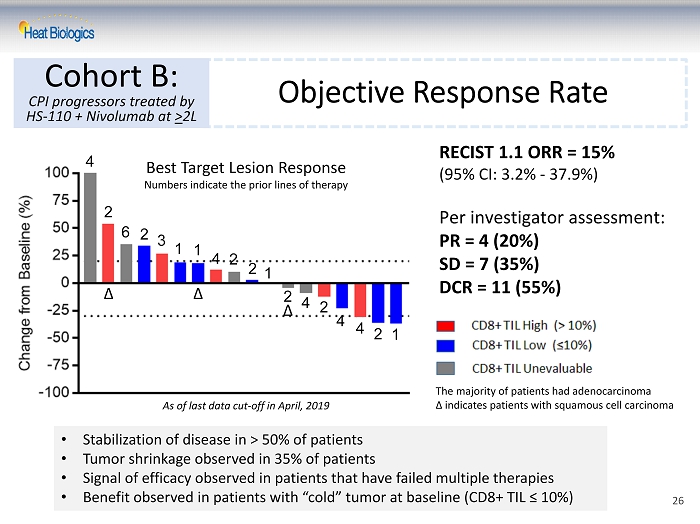
Objective Response Rate Cohort B: CPI progressors treated by HS - 110 + Nivolumab at > 2L As of last data cut - off in April, 2019 4 2 6 2 3 1 1 4 2 2 1 2 4 2 4 4 2 1 RECIST 1.1 ORR = 15% (95% CI: 3.2% - 37.9%) Per investigator assessment: PR = 4 (20%) SD = 7 (35%) DCR = 11 (55%) • Stabilization of disease in > 50% of patients • Tumor shrinkage observed in 35% of patients • Signal of efficacy observed in patients that have failed multiple therapies • Benefit observed in patients with “cold” tumor at baseline (CD8+ TIL ≤ 10%) Δ Δ Δ The majority of patients had adenocarcinoma Δ indicates patients with squamous cell carcinoma Best Target Lesion Response Numbers indicate the prior lines of therapy 26

PFS by Injection Site Reaction (ISR) Cohort B: CPI progressors treated by HS - 110 + Nivolumab at > 2L As of last data cut - off in April, 2019 Progression - Free Survival (%) As of last data cut - off in April, 2019 ISR = Yes (N = 12) ISR = No (N = 8) mPFS: NR vs. 1.8 months HR = 0.17 (95% CI, 0.03 - 0.84) p = 0.013 PFS is significantly better in patients who experienced dermal injection site reaction, thus supporting the HS - 110 mechanism of action 27
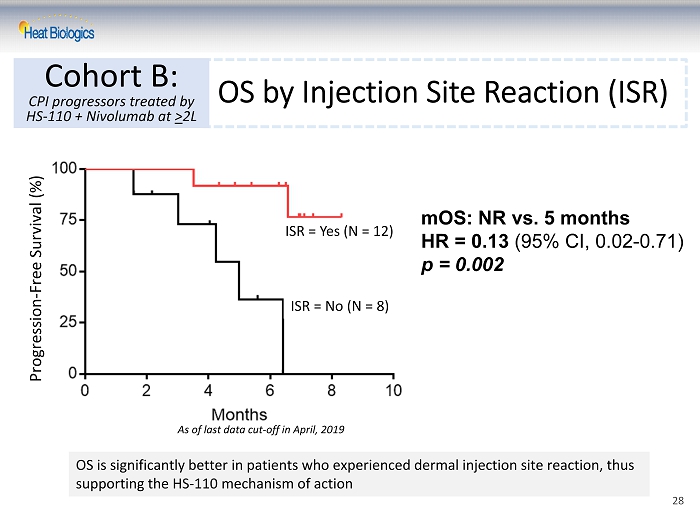
OS by Injection Site Reaction (ISR) Cohort B: CPI progressors treated by HS - 110 + Nivolumab at > 2L Progression - Free Survival (%) As of last data cut - off in April, 2019 ISR = Yes (N = 12) ISR = No (N = 8) mOS : NR vs. 5 months HR = 0.13 (95% CI, 0.02 - 0.71) p = 0.002 O S is significantly better in patients who experienced dermal injection site reaction, thus supporting the HS - 110 mechanism of action 28

Duration of Clinical Benefit Cohort B: CPI progressors treated by HS - 110 + Nivolumab at > 2L Median Progression - Free Survival (PFS) 2.7 months 5 out of 20 patients have not progressed on therapy As of last data cut - off in A pril , 2019 On study Disease Progression Alive Deceased 0 1 2 3 4 5 6 7 8 9 Months 29

Safety Profile to Date 1,000+ Doses - No Serious Adverse Reactions • Over 1,000 doses administered to 12 0 + patients • Only one patient ended treatment due to a non - serious adverse reaction* • No systemic use of steroids required to treat reactions • No treatment - related serious adverse reactions Favorable Safety Profile To Date *Represents the only patient of ~200 patients dosed who discontinued treatment for a vaccine - related adverse event Immune Reaction* ≤ Grade 3 toxicity Injection Reactions Week 1 Week 2 No additive toxicities to standard of care 30

Summary of HS - 110 Phase 2 Interim Data • HS - 110 in combination with nivolumab appears safe and well tolerated • Preliminary results demonstrate the ability of HS - 110 plus nivolumab to induce CD8+ T - cell tumor infiltration in previously “cold” tumors • The occurrence of dermal injection site reactions is associated with improved overall survival in Cohort A • Data in Cohort B suggests that the addition of HS - 110 to nivolumab may restore responsiveness after tumor progression on prior checkpoint inhibitors • The occurrence of dermal injection site reactions (any grade) is associated with improved progression - free survival and overall survival in Cohort B 31

» Heat acquired 80% controlling interest in Pelican in May 2017 » Pre - clinical synergy with Heat’s ImPACT® and checkpoint therapy » $15.2M grant award from the Cancer Prevention Research Institute of Texas (CPRIT) propels PTX - 35 through a 70 - patient, first - in - human clinical program » PTX - 35 is a potential best - in - class, T - cell co - stimulator specific to “killer” CD8+ “memory” T - cells TNFRSF25 represents an emerging target in immuno - oncology Heat Biologics Acquires Pelican Therapeutics Unlocking the Body’s Natural Defenses with a Broad Range of Combination Therapies 32

TNFRSF25 as a Novel I/O Combination Target • TNFRSF25 is the most recently discovered T cell costimulator, and is a rapidly emerging target • Pelican is the only company that has publicly announced developing TNFRSF25 agonist antibodies for I/O Mahoney K. et al . Nat Rev Drug Discov . 2015 “Although TNFRSF25 is chromosomally localized with 4 - 1BB, OX40, GITR and CD27, sequence analysis suggests that TNFRSF25 is relatively divergent or perhaps ancestral…Thus we might expect that therapeutics directed against this pathway will have unique activity.” - Gordon Freeman et al 33 33

T Cell APC TL1a TNFRSF25 CD8 Expand activated CD8+ CD8 Expand but delay activation of existing Treg Treg Treg Inhibits Tconv to iTreg conversion CD4 iTreg X One molecule – three types of T - cells TNFRSF25: Why So Complicated? 34 TNFRSF25’s evolutionary origin Potentially a mechanism that preserves needed tissue and friendly bacteria during an immune response Example: gut invasion occurs in the context of friendly microbiome bacteria. How does the body protect what is needed while weeding out invaders? Time and context are everything… 34

TNFRSF25 is Primarily Expressed on CD8+ and CD4+ T - Cells compared to other T - cell co - stimulators Genomics Institute of the Novartis Research Foundation Su et al . PNAS 2004: 101 (16);6062 - 7 ( red = high expression) 35

TNFRSF25 is preferentially expressed on CD8+ T - cells compared to other T - cell co - stimulators • Co - stimulation occurs only in the context of TCR recognition of antigen • Drives the development of antigen - specific CD8+ T - cells (mimics TL1A, the specific ligand of TNFRSF25) Compared to agonists OX40, GITR, 4 - 1BB: • TNFRSF25 shows superior activity in stimulating memory CD8+ cells in mice • TNFRSF25 agonism plus ImPACT results in improved survival in mouse melanoma models PTX - 35 Data Highlights CD8+ T - cell Specificity Antigen - specific T - cell proliferation Increased effector cytokine production Increased effector immune function Increased survival in mice cancer models In mice, TNFRSF25 agonists increases 36

Preferential CD8+ T - cell Induction with TNFRSF25 in Mice Pre - clinical studies with murine agonist antibody shows TNFRSF25 preferentially ‘boosts’ CD8+ T - cells, whereas OX40 is preferential to CD4+ T - cells The frequency of antigen - specific memory CD4+ or memory CD8+ T - cells following treatment of mice with ImPACT™ alone, or in combination with OX40 or TNFRSF25 antibodies Schreiber et al. J Immunol 2012:189(7);3311 - 8 % Antigen Specific Memory CD8+ T - cells TNFRS25 OX40 ImPACT + + + + 37

TNFRSF25 Agonist + ImPACT Significantly Increases Survival GITR 4 - 1BB TNFRSF25 Schreiber T. et al. SITC 2014 Control 4 - 1BB ImPACT ImPACT + 4 - 1BB Control GITR ImPACT ImPACT + GITR Control TNFRSF25 ImPACT ImPACT + TNFRSF25 Established (nine - day) B16 - F10 melanoma mouse model 38

Unpublished George Fromm et al; Heat Biologics, 2014 TNFRSF25 Causes No Intra - tumoral T - reg Proliferation in a Lung - tumor Model 39

Development of Agonist Antibody: PTX - 35 • The lead mAb, PTX - 35, was affinity - matured and selected based on functional activation of TNFRSF25 across species. • The functional cross - reactivity of PTX - 35 was further validated by demonstrating that PTX - 35 binds a unique epitope conserved in placental mammals. • Humanized and affinity matured PTX - 35 is now in IND enabling development. • IND filing slated for 2Q2019. 40

2019 Corporate Milestones 41 Q1 Q2 Q3 Q4 Phase 2 interim data readout Complete Phase 2 enrollment Phase 2 interim data readout HS - 110 File IND Initial data readout PTX - 35 File IND Initial data readout HS - 130

Corporate Highlights Potential Best in Class Oncology Treatment - T - cell activating platform (TCAP) produces allogeneic, off - the - shelf therapies designed to activate the immune system to turn immunologically “cold” tumors “hot” Combination Effect - Our therapies are designed to be administered with checkpoint inhibitors and other immuno - modulators to enhance immune response Off - the - shelf Therapies - We can administer drug immediately without extracting patient material, simplifying treatment and substantially lowering cost of goods Clinical Data with Checkpoint Inhibitors (CPI) - Positive interim data from ongoing Phase 2 trial of HS - 110 + CPI in non - small cell lung cancer (NSCLC) patients (both CPI naïve and CPI progressors) Diverse Technology Platforms - Multiple complementary platform technologies Strong Management Team - Senior team with broad experience in biotech, pharma, clinical development and research 42

Appendix 43

Management and Advisors Management Scientific Advisors Robert Levy, Ph.D. University of Miami Roger Cohen, MD University of Pennsylvania Robert Negrin , MD Stanford University Llew Keltner, MD, Ph.D. Epistat Anthony Tolcher , MD Next Oncology Gary Acton, MD Advisor Board of Directors Jeff Wolf Founder, Chairman and CEO John Prendergast, Ph.D. Lead Independent Director John Monahan, Ph.D. Director Edward Smith Director 44

C o n t r o l V a c c i n e O X 4 0 T N F R S F 2 5 4 - 1 B B G I T R P D - L 1 P D - 1 C T L A - 4 L A G - 3 V a c c i n e + O X 4 0 V a c c i n e + T N F R S F 2 5 V a c c i n e + 4 - 1 B B V a c c i n e + G I T R V a c c i n e + P D - L 1 V a c c i n e + P D - 1 V a c c i n e + C T L A - 4 V a c c i n e + L A G - 3 0 5 10 15 P e a k O T - I / C D 8 F r e q u e n c y ** *** *** Preclinical Data of CD8+ T cell Activation ImPACT ® alone and in combination with co - stimulator agonists: OX40, TNFRSF25, PD - 1 Source: Fromm et al. Society for Immunotherapy of Cancer Annual Meeting, 2016 • Higher T - cell responses observed in mice treated with ImPACT alone • ImPACT® boosted CD8+ T - cells to even higher levels when combined with co - stimulator agonist antibodies: OX40, TNFRSF25, PD - 1 • Findings suggest synergies when combining ImPACT with Pelican’s TNFRSF25 antibody CD8+ T - cell Activation 45

ComPACT™ Platform Technology gp96 - Ig OX40L - Fc The first potential dual - acting immunotherapy designed to deliver T - cell activation and co - stimulation in a single product – combination therapy without additive costs 46

ComPACT™ Outperforms OX40 Monoclonal Antibodies in Pre - clinical Models ComPACT generates ~50% complete tumor rejection compared to ~16% with OX40 agonist antibody combinations Superior primary and memory T - cell response in mouse model Translates into increased overall survival and tumor reduction in a mouse tumor model 47

TNFRSF25 - An Emerging Target for T - cell Co - stimulation Target Companies Co - stimulator Combinations 4 - 1BB Pfizer, ISA Pharma, Gilead, Bristol Myers, Inhibrx , Immatics , Pieris, NCI Phase 1/2 Combinations: Chemotherapy, epigenetic modulator, CD20 mAb , HPV vaccine, CART, PD - L1, BTK, Herceptin, modified CD8+ T cells, IL - 2, CSF1R, LAG - 3, bispe c ific w/ HER - 2, two co - stimulators (w/ OX40) OX40 MedImmune , GSK, Incyte, Genentech, Pfizer, AbbVie, Bristol Myers, Alligator Bio Phase 1/2 OX - 40 prior to surgery. Other combinations: PD - 1, PD - L1, CTLA4, axitinib , 41BB, smoothened inhibitor Anti - CD33, azacitidine , TLRs, bi - specifics of OX40 and CTLA - 4 GITR Novartis, Incyte - Agenus, MedImmune , Leap Therapeutics, BMS, OncoMed Phase 1 Combinations: PD - 1, Fc disabled co - stimulation, hexameric GITR ligand, IDO inhibitor, CTLA - 4, GITR ligand - fusion CD27 Celldex - BMS, Merck - Aduro Phase 1/2 Combinations: PD - 1/L - 1, sunitinib, CD20, melanoma vaccine, TL3 agonist ICOS Celgene - Jounce, GSK Phase 1/2 Combinations: PD - 1, CTLA4, docetaxel TNFRSF25 Heat (under Pelican) Filing IND in Q2 2019 in advanced solid tumors. Combination studies being planned • TNFRSF25 is a potential best - in - class T cell co - stimulator due to its preferential specificity to ‘memory’ CD8+ T cells • Pelican is the only company with a disclosed program targeting TNFRSF25 48
This regulatory filing also includes additional resources:
htbx_ex99z1.pdf
Heat Biologics (NASDAQ:HTBX)
Historical Stock Chart
From Mar 2024 to Apr 2024
Heat Biologics (NASDAQ:HTBX)
Historical Stock Chart
From Apr 2023 to Apr 2024