Harpoon Therapeutics Announces First Patients with Small Cell Lung Cancer Dosed in HPN328 Combination Cohort
September 15 2023 - 7:15AM

Harpoon Therapeutics, Inc. (Nasdaq: HARP), a clinical-stage
immuno-oncology company developing novel T cell engagers, today
announced dosing of the first patients with small cell lung cancer
(SCLC) in an ongoing Phase 1/2 trial of HPN328, a DLL3 targeting
TriTAC®, in combination with atezolizumab (Tecentriq®). Harpoon
previously entered a Master Clinical Supply Agreement with F.
Hoffmann-La Roche for the supply of atezolizumab. Under this
agreement, Harpoon is the sponsor of the trial and Roche will
supply atezolizumab. This announcement is being made in conjunction
with Harpoon’s investor event, “DLL3 Market Opportunity and KOL
Discussion of HPN328,” held virtually and in person today in New
York beginning at 8 a.m. ET.
“Dosing the first patients in these combination cohorts for
HPN328 with atezolizumab in patients with SCLC marks a significant
milestone for this clinical program,” said Luke Walker, M.D., Chief
Medical Officer for Harpoon Therapeutics. “Building on the strength
of our Phase 1 data, we remain committed to realizing the full
potential of HPN328 as an important treatment option for patients
with SCLC and other neuroendocrine tumors across early and late
lines of therapy.”
About the HPN328 + Atezolizumab Combination
Cohort Previously treated extensive stage SCLC patients
enrolled in these combination cohorts will be dosed with HPN328
administered once every 2 weeks (Q2W) by IV infusion during each
28-day cycle. Atezolizumab will be administered once every 4 weeks
(Q4W) by IV infusion on day 1 of each 28-day cycle. Primary outcome
measures will include frequency and severity of treatment emergent
adverse events (TEAEs), number and severity of dose limiting
toxicities (DLTs), and pharmacokinetic parameters. Secondary
outcome measures will include objective response rate (ORR),
progression-free survival (PFS), overall survival (OS), duration of
response (DOR), and anti-drug antibody (ADA) formation. Enrollment
in the combination cohorts has been initiated at the 12 mg Q2W
HPN328 dose level, and escalation is planned per protocol dependent
on data, with initial results from these combination cohorts
expected in 2024. Separately, HPN328 interim Phase 1 monotherapy
data will be presented at the European Society for Medical Oncology
(ESMO) held on October 20-24, 2023.
Webcast Information for “DLL3 Market Opportunity and KOL
Discussion of HPN328”
- The webcast will begin at 8 a.m. ET and can be accessed using
this link:
https://viavid.webcasts.com/starthere.jsp?ei=1629744&tp_key=667a2aef59
- A live webcast and archived replay of the event will be
accessible on the Investor Relations page of the Harpoon website
at https://ir.harpoontx.com/events-and-presentations.
About Harpoon TherapeuticsHarpoon
Therapeutics is a clinical-stage immuno-oncology company
developing a novel class of T cell engagers that harness the power
of the body’s immune system to treat patients suffering from cancer
and other diseases. T cell engagers are engineered proteins that
direct a patient’s own T cells to kill target cells that express
specific proteins, or antigens, carried by the target cells. Using
its proprietary Tri-specific T cell Activating Construct (TriTAC®)
platform, Harpoon is developing a pipeline of novel
TriTACs initially focused on the treatment of solid tumors and
hematologic malignancies. Harpoon has also developed a proprietary
ProTriTAC™ platform, which applies a prodrug concept to its
TriTAC platform to create a therapeutic T cell engager that
remains inactive until it reaches the tumor. Harpoon’s third
proprietary technology platform, extended release TriTAC-XR, is
designed to mitigate cytokine release syndrome. For additional
information about Harpoon Therapeutics, please visit
www.harpoontx.com and follow us on Twitter and LinkedIn.
Tecentriq® (atezolizumab) is a registered trademark
of F. Hoffmann La-Roche Ltd.
Cautionary Note on Forward-looking
StatementsThis press release contains forward-looking
statements within the meaning of the Private Securities Litigation
Reform Act of 1995. Words such as “continue,” “will” and similar
expressions (as well as other words or expressions referencing
future events, conditions, or circumstances) are intended to
identify forward-looking statements. These forward-looking
statements are based on Harpoon Therapeutics’ expectations and
assumptions as of the date of this press release. These
forward-looking statements involve risks and uncertainties that
could cause Harpoon Therapeutics’ clinical development programs,
future results, or performance to differ significantly from those
expressed or implied by the forward-looking statements.
Forward-looking statements in this press release include, but are
not limited to, statements relating to the effects of advancing
Harpoon Therapeutics’ platforms, the company’s future business
growth and partnerships, expectations regarding strengthening the
company’s business team, and other statements that are not
historical fact. These and other factors that may cause Harpoon
Therapeutics’ actual results to differ from those expressed or
implied in the forward-looking statements in this press release are
discussed in Harpoon Therapeutics’ filings with the U.S.
Securities and Exchange Commission, including under “Risk Factors”
in Harpoon Therapeutics’ quarterly report on Form 10-Q for the
quarter ended June 30, 2023, and future filings
by Harpoon Therapeutics. Except as required by
law, Harpoon Therapeutics assumes no obligation to update
any forward-looking statements contained herein to reflect any
change in expectations, even as new information becomes
available.
Contact:ICR WestwickeRobert H. UhlManaging
Director858-356-5932robert.uhl@westwicke.com
Harpoon Therapeutics (NASDAQ:HARP)
Historical Stock Chart
From Apr 2024 to May 2024
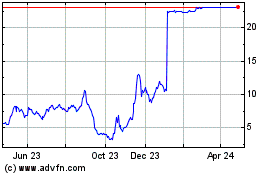
Harpoon Therapeutics (NASDAQ:HARP)
Historical Stock Chart
From May 2023 to May 2024