Finch Therapeutics Appoints Joseph Vittiglio, JD, as Chief Business and Legal Officer
December 20 2021 - 7:00AM

Finch Therapeutics Group, Inc. (“Finch” or “Finch Therapeutics”)
(Nasdaq: FNCH), a clinical-stage microbiome therapeutics company
leveraging its Human-First Discovery® platform to develop a novel
class of orally administered biological drugs, today announced the
promotion of Joseph Vittiglio, JD, to Chief Business and Legal
Officer. Mr. Vittiglio has more than 25 years of experience leading
strategic transactions within the life sciences industry.
“Joe’s breadth of experience building successful biotech
companies and catalyzing strategic partnerships is invaluable to
Finch as we continue to advance our platform and expand our
pipeline of microbiome product candidates,” said Mark Smith, PhD,
Chief Executive Officer of Finch Therapeutics. “As a platform
company, we believe that collaborations will play an important role
in unlocking the full potential of this technology and Joe’s
experience and demonstrated leadership at Finch position him well
to take on these additional responsibilities.”
“As Finch enters a transformational period, with a Phase 3 trial
underway in recurrent C. difficile and additional programs
positioned to enter the clinic in 2022, I am pleased to take on
this new role and leverage my legal and corporate development
experience to continue building on our progress,” said Mr.
Vittiglio. “I look forward to continuing to work with the talented
Finch team with the goal of building value for our stakeholders and
executing upon our mission to bring microbiome therapeutics to
patients in need.”
Mr. Vittiglio joined Finch Therapeutics in December 2020 as
General Counsel and Corporate Secretary. In his previous role with
Finch, he guided the company’s initial public offering in 2021,
raising $130.8 million in capital, and led the company’s legal and
quality teams. Prior to Finch, Mr. Vittiglio was the General
Counsel and Chief Business Officer for AMAG Pharmaceuticals, where
he led the company’s legal and business development initiatives,
including multiple out-licensing and partnership collaborations,
and provided oversight for multiple therapeutic product launches.
Prior to AMAG Pharmaceuticals, he held leadership roles at Flexion
Therapeutics, AVEO Pharmaceuticals, and Oscient Pharmaceuticals.
Mr. Vittiglio earned his Juris Doctor degree from Northeastern
University School of Law and his Bachelor of Arts degree from Tufts
University.
About Finch Therapeutics
Finch Therapeutics is a clinical-stage microbiome
therapeutics company leveraging its Human-First
Discovery® platform to develop a novel class of orally
administered biological drugs. With the capabilities to develop
both complete and targeted microbiome therapeutics, Finch is
advancing a rich pipeline of candidates designed to address a wide
range of unmet medical needs. Finch’s lead candidate, CP101, is in
late-stage clinical development for the prevention of
recurrent C. difficile infection (CDI), and has received
Breakthrough Therapy and Fast Track designations from the U.S. Food
and Drug Administration. In June 2020, Finch announced that CP101
met its primary efficacy endpoint in PRISM3, the first of two
pivotal trials to support the development of CP101 for the
prevention of recurrent CDI. PRISM4, a Phase 3 trial, is designed
to serve as the second pivotal trial of CP101 for recurrent CDI.
Finch is also developing CP101 for the treatment of chronic
hepatitis B virus, and FIN-211 for the treatment of the
gastrointestinal and behavioral symptoms of autism spectrum
disorder. Finch has a partnership with Takeda focused on the
development of targeted microbiome therapeutics for inflammatory
bowel disease.
Human-First Discovery® is a registered trademark of Finch
Therapeutics Group, Inc.
Forward-Looking Statements
Statements contained in this press release regarding matters
that are not historical facts are “forward-looking statements”
within the meaning of the Private Securities Litigation Reform Act
of 1995, as amended. Words such as “anticipates”, “believes”,
“expects”, “intends”, “plans”, “potential”, "projects”, “would” and
"future" or similar expressions are intended to identify
forward-looking statements. These forward-looking statements
include, but are not limited to, statements regarding: Finch’s
ability to advance its platform and expand its pipeline of
microbiome product candidates; and the timing of Finch’s clinical
trials, including programs positioned to enter the clinic in 2022.
Because such statements are subject to risks and uncertainties,
actual results may differ materially from those expressed or
implied by such forward-looking statements. These risks and
uncertainties include, among others: Finch’s limited operating
history and historical losses; Finch’s ability to raise additional
funding to complete the development and any commercialization of
its product candidates; Finch’s dependence on the success of its
lead product candidate, CP101; the possibility that Finch may be
delayed in initiating, enrolling or completing any clinical trials;
results of clinical trials may not be indicative of final or future
results from later stage or larger clinical trials (or in broader
patient populations once the product is approved for use by
regulatory agencies) or may not be favorable or may not support
further development; Finch’s product candidates may not generate
the benefits to patients that are anticipated; anticipated
regulatory approvals may be delayed or refused; competition from
third parties that are developing products for similar uses;
Finch’s ability to maintain patent and other intellectual property
protection and the possibility that Finch’s intellectual property
rights may be infringed, invalid or unenforceable or will be
threatened by third parties; Finch’s ability to qualify and scale
its manufacturing capabilities to support multiple global clinical
trials; Finch’s lack of experience in selling, marketing and
distributing its product candidates; Finch’s dependence on third
parties in connection with manufacturing, clinical trials and
preclinical studies; and risks relating to the impact and duration
of the COVID-19 pandemic on Finch’s business. These and other risks
are described more fully in Finch’s filings with the Securities and
Exchange Commission (“SEC”), including the section titled “Risk
Factors” in Finch’s Quarterly Report on Form 10-Q filed with the
SEC on November 10, 2021, as well as discussions of potential
risks, uncertainties, and other important factors in Finch’s other
filings with the SEC. All forward-looking statements contained in
this press release speak only as of the date on which they were
made. Except to the extent required by law, Finch undertakes no
obligation to update such statements to reflect events that occur
or circumstances that exist after the date on which they were
made.
Investor Contact:Stephen JasperGilmartin
Group(858) 525-2047stephen@gilmartinir.com
Media Contact:Jenna UrbanBerry & Company
Public Relations(212) 253-8881jurban@berrypr.com
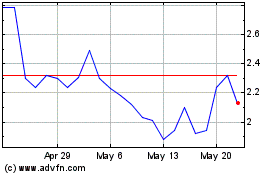
Finch Therapeutics (NASDAQ:FNCH)
Historical Stock Chart
From Jun 2024 to Jul 2024
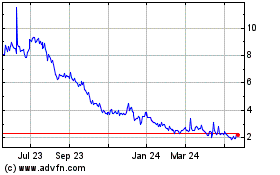
Finch Therapeutics (NASDAQ:FNCH)
Historical Stock Chart
From Jul 2023 to Jul 2024