Biocept Names Michael Dugan, M.D. Chief Medical Officer and Medical Director
August 24 2020 - 8:00AM

Biocept, Inc. (Nasdaq: BIOC), a leading commercial provider of
liquid biopsy tests designed to provide physicians with clinically
actionable information to improve the outcomes of patients,
announces the appointment of Michael C. Dugan, M.D. as Chief
Medical Officer and Medical Director. Dr. Dugan has served in
leading medical positions at Exact Sciences, Quest Diagnostics,
Nichols Institute and Roche Molecular Systems. At Biocept he
will be responsible for medical policy decision-making and the
operations of the Company’s CLIA-certified, CAP-accredited,
high-complexity molecular laboratory.
“Mike is highly respected in the molecular diagnostics industry,
and we are thrilled to welcome him to our executive team,” said
Michael Nall, Biocept's President and CEO. “Mike is
particularly known within our industry for his outstanding
leadership capability and commercial vision. He has proven
experience in developing clinical strategies and designing and
overseeing studies that validate clinical utility. He brings
established relationships with key thought-leaders in molecular
diagnostics, has served as a senior clinical spokesperson at
medical and scientific conferences, liaised with regulatory
agencies and supervised CLIA lab operations. We expect to
benefit significantly from his clinical and strategic insights as
we grow by expanding our portfolio of clinically actionable assays
and provide further evidence of their clinical validity and
utility.”
“I’m excited to join Biocept to further develop and
commercialize leading-edge assays that profile and monitor
molecular signatures of cancer to help improve patient treatment
choices and outcomes,” Dr. Dugan said. “I look forward to
further evaluating our clinical market opportunities and working
with our Biocept team to develop strategic markets for our
products. This will include further clinical studies and
clinical education related to our molecular assays and work to
ensure we gain medical policy coverage and reimbursement for our
new clinical diagnostic tests.”
Dr. Dugan brings to Biocept more than 25 years of industry
experience, most recently as President and CEO of MCDXI Medical
Diagnostics International, a professional advisory group practice
where he consulted with biotechnology companies in oncology
diagnostics, medical device development, and clinically related
software applications. Before that, he was Senior Medical
Advisor and Chief Medical Officer of Clinical Genomics, Inc., a
biotechnology company developing an innovative product for
colorectal cancer recurrence monitoring using circulating tumor DNA
(ctDNA) in plasma. He previously was Senior Vice President of
Clinical Development and Medical Affairs at Exact Sciences
Corporation, Medical Director at Quest Diagnostics Nichols
Institute (and Specialty Laboratories which they acquired), Chief
Medical Officer at bioTheranostics, Vice President and Chief
Medical Officer at Roche Molecular Systems, Inc. and Vice President
of Pathology Services at Genzyme Genetics (acquired by
LabCorp).
Dr. Dugan received a BS in biology from the University of Notre
Dame and a medical degree from the University of Arizona College of
Medicine. He completed his post-graduate training in the Department
of Pathology at Yale-New Haven Hospital, and in the Departments of
Pathology and Laboratory Medicine at Childrens Hospital Los Angeles
and at the University of California, Los Angeles.
About Biocept Biocept, Inc. is a
molecular diagnostics company with commercialized assays for lung,
breast, gastric, colorectal and prostate cancers, and melanoma. The
Company uses its proprietary liquid biopsy technology to provide
physicians with clinically actionable information for treating and
monitoring patients diagnosed with cancer. The Company’s patented
Target Selector™ liquid biopsy technology platform captures and
analyzes tumor-associated molecular markers in both circulating
tumor cells (CTCs) and in circulating tumor DNA (ctDNA). With
thousands of tests performed, the platform has demonstrated the
ability to identify cancer mutations and alterations to inform
physicians about a patient’s disease and therapeutic options. In
addition, Biocept recently added COVID-19 testing to support
efforts to fight the pandemic. For additional information, please
visit www.biocept.com.
Forward-Looking Statements Disclaimer Statement
This release contains forward-looking statements that are based
upon current expectations or beliefs, as well as a number of
assumptions about future events. Although we believe that the
expectations reflected in the forward-looking statements and the
assumptions upon which they are based are reasonable, we can give
no assurance that such expectations and assumptions will prove to
have been correct. Forward-looking statements are generally
identifiable by the use of words like "may," "will," "should,"
"could," "expect," "anticipate," "estimate," "believe," "intend,"
or "project" or the negative of these words or other variations on
these words or comparable terminology. To the extent that
statements in this release are not strictly historical, including
without limitation statements as to our ability to improve the
outcomes of patients diagnosed with cancer, our ability to benefit
from the addition of Dr. Dugan to the Biocept team, our ability to
expand our portfolio of clinically actionable assays and provide
further evidence of their clinical validity and utility, and our
ability to gain medical policy coverage and reimbursement for our
new clinical diagnostic tests, such statements are forward-looking,
and are made pursuant to the safe harbor provisions of the Private
Securities Litigation Reform Act of 1995. The reader is cautioned
not to put undue reliance on these forward-looking statements, as
these statements are subject to numerous risk factors as set forth
in our Securities and Exchange Commission (SEC) filings. The
effects of such risks and uncertainties could cause actual results
to differ materially from the forward-looking statements contained
in this release. We do not plan to update any such forward-looking
statements and expressly disclaim any duty to update the
information contained in this press release except as required by
law. Readers are advised to review our filings with the SEC, which
can be accessed over the Internet at the SEC's website located at
http://www.sec.gov/.
Investor Contact: LHA Investor
Relations Jody Cain
Jcain@lhai.com 310-691-7100
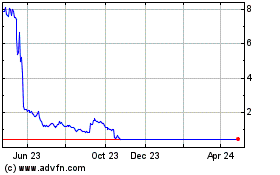
Biocept (NASDAQ:BIOC)
Historical Stock Chart
From Aug 2024 to Sep 2024
Biocept (NASDAQ:BIOC)
Historical Stock Chart
From Sep 2023 to Sep 2024