Aravive, Inc. (Nasdaq: ARAV), a clinical-stage oncology company
developing innovative therapeutics to treat life-threatening
diseases, today announced the appointments of John A. Hohneker,
M.D., Sigurd C. Kirk, and Peter T.C. Ho, M.D., Ph.D. to its Board
of Directors. Mr. Kirk was also appointed to serve on the Audit
Committee of the Board. Dr. Hohneker was also appointed to serve on
the Compensation Committee of the Board.
“We are pleased to welcome Dr. John Hohneker, Mr. Sigurd Kirk,
and Dr. Peter Ho to our Board of Directors. They will further
strengthen our Board, as they bring extensive drug development and
business development expertise to Aravive,” said Fred Eshelman,
Pharm.D., Chairman of the Board of Aravive. “Their experience in
biopharmaceutical and healthcare senior executive leadership
positions will be valuable to the Company as we continue to advance
our AVB-500 clinical development program to treat life-threatening
cancers. We look forward to their contributions to the future
success of Aravive.”
Dr. John Hohneker has more than 30 years of experience as an
innovative senior biopharmaceutical physician executive and leader
with significant drug development experience and a strong track
record of success. He currently serves on the Board of Directors of
Evelo Biosciences, a publicly traded company, and on the Board of
privately held Trishula Therapeutics, Inc.
Dr. Hohneker has advanced several drugs (biologics and small
molecules) from pre-clinical evaluation through Phases 1-4 and
market registration across multiple therapeutic areas, including
oncology and immunology. Previously, in various senior management
positions, including at Novartis and GlaxoSmithKline, he played a
critical role in numerous highly successful commercial product
launches.
Dr. Hohneker received his M.D. from the University of Medicine
and Dentistry of New Jersey, Robert Wood Johnson (previously known
as Rutgers) Medical School and completed his internship and
residency in internal medicine and his fellowship in medical
oncology at the University of North Carolina. He received his
bachelor’s degree in Chemistry from Gettysburg College.
Mr. Sigurd (Sig) C. Kirk is a senior corporate business
development executive with more than 15 years of pharmaceutical
leadership experience in the areas of branded biopharmaceutical,
medical device and generic products.
In his most recent position, Mr. Kirk was Executive Vice
President, Corporate Business Development at Allergan, where he was
a member of the Executive Leadership Team. He was an integral
member assessing development and commercial opportunities, leading
due diligence, as well as negotiating and transacting key legal and
financial terms.
Previously, Mr. Kirk was at Barr Pharmaceuticals, Inc., formerly
a $2.8B global specialty pharmaceutical company that was acquired
by Teva Pharmaceuticals. In his last role there, he was Senior Vice
President, Global Controller and Chief Accounting Officer.
Mr. Kirk started his career at Deloitte & Touche as an Audit
Manager, earning his CPA certification. He received his Bachelor of
Business Administration degree from Pace University.
Dr. Peter T.C. Ho has more than 25 years of biotechnology and
pharmaceutical industry experience in numerous operational roles
that have ranged from senior management in large pharmaceutical
companies, including leading the oncology discovery and early
development group at GlaxoSmithKline, to corporate officer roles in
small public biotech (Epizyme) and start-up private biotech
(BeiGene and Boston Pharmaceuticals) companies. He also currently
serves as Senior Scientific and Medical Advisor to Overland
Pharmaceuticals, D3 Bio, and M4K Pharma, and is a Scientific
Advisory Board member of Accent Therapeutics.
Dr. Ho has significant experience in senior executive leadership
roles in the areas of solid tumor and hematologic oncology. In
nearly 30 years in private industry and the federal government, he
has been directly responsible for the first-time-in-human dosing of
19 anticancer agents and has overseen the development of over 60
hematology and oncology compounds throughout all phases of clinical
trials. He has played a key role in the product approvals of
several new chemical entities (NCE) and biologics.
Dr. Ho is currently an Adjunct Associate Professor in the
Division of Chemical Biology and Medicinal Chemistry at the
Eshelman School of Pharmacy, University of North Carolina. Dr. Ho
received his M.D. and Ph.D. (pharmacology) degrees from Yale
University and then completed a pediatrics residency at The
Children's Hospital of Boston, followed by clinical fellowships in
pediatric hematology/oncology at the Dana-Farber Cancer Institute
and in clinical oncology and regulatory sciences jointly through
the U.S. FDA and the National Cancer Institute. He received his
bachelor’s degree in Biology at Johns Hopkins University.
About AraviveAravive, Inc. is a
clinical-stage oncology company developing innovative therapeutics
to treat life-threatening diseases. Aravive’s lead therapeutic,
AVB-500, is a first-in-class ultra-high affinity decoy protein that
targets the GAS6-AXL signaling pathway associated with tumor cell
growth, tumor metastasis, resistance to treatment and decreased
survival. AVB-500 has the potential to be combined with multiple
anti-cancer therapies across several tumor types, due to its novel
mechanism of action and favorable safety profile. AVB-500 has been
granted Fast Track Designation by the U.S. Food and Drug
Administration in platinum resistant recurrent ovarian cancer. The
Company is currently evaluating AVB-500 in a registrational Phase 3
trial in platinum resistant ovarian cancer and a Phase 1b/2 trial
in clear cell renal cell carcinoma. Aravive plans to initiate a
Phase 1b/2 trial evaluating AVB-500 in first-line treatment of
pancreatic cancer in the second half of 2021. The Company is based
in Houston, Texas and received a Product Development Award from the
Cancer Prevention & Research Institute of Texas (CPRIT) in
2016. For more information, please visit www.aravive.com.
Forward-Looking StatementsThis communication
contains forward-looking statements within the meaning of the
Private Securities Litigation Reform Act of 1995. In some cases,
forward-looking statements can be identified by terminology such as
"may," "should," "potential," "continue," "expects," "anticipates,"
"intends," "plans," "believes," "estimates," and similar
expressions and includes statements regarding the new directors
further strengthening the Company’s Board of Directors, the
contributions to be made by the new members of the Board of
Directors, plans to investigate AVB-500 in a Phase 1b/2 clinical
trial as a first-line treatment for pancreatic cancer, and the
potential of AVB-500 to be combined with multiple anti-cancer
therapies across several tumor types, due to its novel mechanism of
action and favorable safety profile. Forward-looking statements are
based on current beliefs and assumptions, are not guarantees of
future performance and are subject to risks and uncertainties that
could cause actual results to differ materially from those
contained in any forward-looking statement as a result of various
factors, including, but not limited to, risks and uncertainties
related to: the contributions to be derived from the new directors,
the ability of the new directors and management team to execute the
Company’s business plan, the Company’s ability to recruit for and
enroll the expected number of patients into the Phase 3 trial of
AVB-500 in PROC and its other trials as planned and its ability to
report data as planned, the ability to initiate a Phase 1b/2 trial
evaluating AVB-500 in first-line treatment of pancreatic cancer in
the second half of 2021, the impact of COVID-19 on the Company's
clinical strategy, clinical trials, supply chain and fundraising,
the Company's ability to expand development into pancreatic cancer
and other additional oncology indications, the Company's dependence
upon AVB-500, AVB-500's ability to have favorable results in
clinical trials and ISTs, the clinical trials of AVB-500 having
results that are as favorable as those of preclinical and clinical
trials, the ability to receive regulatory approval, potential
delays in the Company's clinical trials due to regulatory
requirements or difficulty identifying qualified investigators or
enrolling patients especially in light of the COVID-19 pandemic;
the risk that AVB-500 may cause serious side effects or have
properties that delay or prevent regulatory approval or limit its
commercial potential; the risk that the Company may encounter
difficulties in manufacturing AVB-500; if AVB-500 is approved,
risks associated with its market acceptance, including pricing and
reimbursement; potential difficulties enforcing the Company's
intellectual property rights; the Company's reliance on its
licensor of intellectual property and financing needs. The
foregoing review of important factors that could cause actual
events to differ from expectations should not be construed as
exhaustive and should be read in conjunction with statements that
are included herein and elsewhere, including the risk factors
included in the Company's Annual Report on Form 10-K for the fiscal
year ended December 31, 2020, recent Current Reports on Form 8-K
and subsequent filings with the SEC. Except as required by
applicable law, the Company undertakes no obligation to revise or
update any forward-looking statement, or to make any other
forward-looking statements, whether as a result of new information,
future events or otherwise.
Contact:Joseph T. SchepersVP, Investor
Relations, Aravive, Inc.jschepers@aravive.com (770) 558-5517
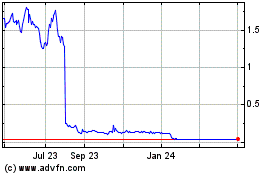
Aravive (NASDAQ:ARAV)
Historical Stock Chart
From Aug 2024 to Sep 2024
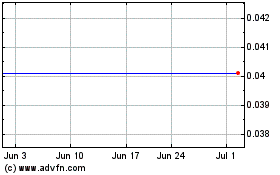
Aravive (NASDAQ:ARAV)
Historical Stock Chart
From Sep 2023 to Sep 2024