Alaunos Therapeutics Appoints Dr. Robert J. Hofmeister to Board of Directors
March 30 2023 - 7:00AM

Alaunos Therapeutics, Inc. (“Alaunos” or the “Company”) (Nasdaq:
TCRT), a leading T-cell receptor (TCR) cell therapy company
advancing a clinical-stage pipeline of therapeutics for solid
tumors, today announced the appointment of Robert J. Hofmeister,
PhD, MS, Chief Scientific Officer of a stealth early stage
biotechnology company, to the Company’s Board of Directors,
effective immediately. Dr. Hofmeister will replace Chris Bowden,
MD.
“We extend our gratitude to Chris for his
invaluable contributions to Alaunos during his tenure, and welcome
Robert to the Alaunos Board,” said Kevin S. Boyle, Sr., Chief
Executive Officer of Alaunos. “Robert brings deep expertise in the
discovery and development of engineered T-cell receptor therapies
and a background in cellular immunology and translational science.
We look forward to leveraging his insights and experience to help
accelerate expanding our library of TCRs and advancing our TCR-T
Library Phase 1/2 trial.”
Prior to joining his current company, Dr.
Hofmeister spent almost seven years in scientific leadership at
TCR2 Therapeutics, a company developing novel therapies leveraging
the TCR complex to fight both solid tumors and hematologic
malignancies, culminating in the role of Chief Scientific Officer.
As the first TCR2 employee, he was instrumental in building and
leading the R&D function and driving the development of its
proprietary TRuC®-T cell platform and its TC-210 program from
concept to first cleared Investigational New Drug (IND)
application. As part of the executive leadership team, he also
contributed to successfully taking the company public and securing
growth capital.
“Alaunos is at the forefront of the science and
development of T-cell therapies, an exciting space I am passionate
about and in which I have spent decades working,” said Dr.
Hofmeister. “With proprietary platforms, an industry-leading
library of TCRs, and the first TCR-T clinical trial in solid tumors
targeting driver mutations with promising early clinical data,
Alaunos has made significant progress. I look forward to working
alongside the executive team and my fellow Board members to help
Alaunos realize its mission to revolutionize the treatment of solid
tumors.”
Earlier in his career, Dr. Hofmeister held
various roles at EMD Serono where he was involved in the
development of now approved Bavencio (avelumab) and building the
company’s immuno-oncology platform. He began his biotech career at
Micromet AG, now Amgen Research Munich, where he helped shape the
development of Blincyto, the first FDA-approved bispecific antibody
for the treatment of refractory ALL. Dr. Hofmeister received his
Ph.D. from the University of Regensburg in Germany and completed a
postdoctoral fellowship at the National Cancer Institute.
About Alaunos Therapeutics,
Inc.Alaunos Therapeutics is a leader in the science of
T-cell receptor (TCR) cell therapy working to revolutionize solid
cancer treatment and outcomes. The clinical-stage company’s TCR
T-cell therapy (TCR-T) is one of the most advanced TCR programs
targeting driver mutations in solid tumors with an ongoing Phase
1/2 trial of its TCR-T product candidates across six solid cancers.
Alaunos is powered by two proprietary platforms: its elegantly
efficient non-viral Sleeping Beauty cell engineering platform; and
its hunTR® discovery platform, which is expanding its
industry-leading library of TCRs against high-frequency driver
mutations. Alaunos is a part of an ongoing collaboration with the
National Cancer Institute (NCI), part of the National Institutes of
Health (NIH), working to advance the science of TCR therapy. For
more information, visit www.alaunos.com.
Forward-Looking Statements Disclaimer
This press release contains forward-looking
statements as defined in the Private Securities Litigation Reform
Act of 1995, as amended. Forward-looking statements are statements
that are not historical facts, and in some cases can be identified
by terms such as “may,” “will,” “could,” “expects,” “plans,”
“anticipates,” “believes” or other words or terms of similar
meaning. These statements include, but are not limited to,
statements regarding the Company's business and strategic plans,
the anticipated outcome of preclinical and clinical studies by the
Company or its third-party collaborators, the Company’s
manufacturing capabilities and the timing of the Company's research
and development programs, including the expected timeline for
enrolling and dosing patients, submitting and receiving approvals
on INDs and similar regulatory submissions and the timing and
forums for announcing data from the Company's clinical trials.
Although the management team of Alaunos believes that the
expectations reflected in such forward-looking statements are
reasonable, investors are cautioned that forward-looking
information and statements are subject to various risks and
uncertainties, many of which are difficult to predict and generally
beyond the control of Alaunos, that could cause actual results and
developments to differ materially from those expressed in, or
implied or projected by, the forward-looking information and
statements. These risks and uncertainties include, among other
things, changes in the Company’s operating plans that may impact
its cash expenditures; the uncertainties inherent in research and
development, future clinical data and analysis, including whether
any of Alaunos’ product candidates will advance further in the
preclinical research or clinical trial process, including receiving
clearance from the U.S. Food and Drug Administration or equivalent
foreign regulatory agencies to conduct clinical trials and whether
and when, if at all, they will receive final approval from the U.S.
Food and Drug Administration or equivalent foreign regulatory
agencies and for which indication; the strength and enforceability
of Alaunos’ intellectual property rights; and competition from
other pharmaceutical and biotechnology companies as well as risk
factors discussed or identified in the public filings with the
Securities and Exchange Commission made by Alaunos, including those
risks and uncertainties listed in the most recent periodic report
filed by Alaunos with the Securities and Exchange Commission.
Alaunos is providing this information as of the date of this press
release, and Alaunos does not undertake any obligation to update or
revise the information contained in this press release whether as a
result of new information, future events, or any other reason.
Investor Contact: Alex
LoboStern Investor RelationsAlex.lobo@sternir.com
Media Contact:Heather Anderson6
Degrees PRhanderson@6degreespr.com
Alaunos Therapeutics (NASDAQ:TCRT)
Historical Stock Chart
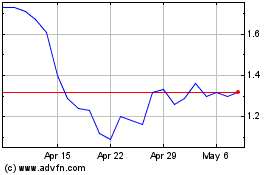
From Mar 2024 to Apr 2024
Alaunos Therapeutics (NASDAQ:TCRT)
Historical Stock Chart
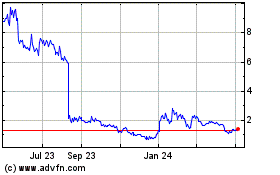
From Apr 2023 to Apr 2024