false
0001476963
0001476963
2024-01-16
2024-01-16
0001476963
us-gaap:CommonStockMember
2024-01-16
2024-01-16
0001476963
NHWK:CommonStockPurchaseRightsMember
2024-01-16
2024-01-16
iso4217:USD
xbrli:shares
iso4217:USD
xbrli:shares
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
WASHINGTON, D.C. 20549
FORM 8-K
CURRENT REPORT
Pursuant to Section 13 or 15(d) of the Securities
Exchange Act of 1934
Date of Report (date of earliest event reported):
January 16, 2024
NightHawk Biosciences, Inc.
(Exact name of registrant as specified in
charter)
Delaware
(State or other jurisdiction of incorporation)
| 001-35994 |
26-2844103 |
| (Commission File Number) |
(IRS Employer Identification No.) |
627
Davis Drive, Suite
300
Morrisville, North Carolina 27560
(Address of principal executive offices and
zip code)
(919) 240-7133
(Registrant’s telephone number including
area code)
N/A
(Former Name and Former Address)
Check the appropriate box below if the Form
8-K filing is intended to simultaneously satisfy the filing obligation of registrant under any of the following provisions:
| |
¨ |
Written communications pursuant to Rule 425 under the Securities Act (17 CFR 230.425) |
| |
¨ |
Soliciting material pursuant to Rule 14a-12(b) under the Exchange Act (17 CFR 240.14a-12) |
| |
¨ |
Pre-commencement communications pursuant to Rule 14d-2(b) under the Exchange Act (17 CFR 240.14d-2(b)) |
| |
¨ |
Pre-commencement communications pursuant to Rule 13e-4(c) under the Exchange Act (17 CFR 240.13e-4(c)) |
Securities registered pursuant to Section 12(b) of the Act:
| Title of each class |
Trading Symbol(s) |
Name of each exchange on which registered |
| Common Stock, $0.0002 par value per share |
NHWK |
NYSE American LLC |
| Common Stock Purchase Rights |
None |
NYSE American LLC |
Indicate by check mark whether the registrant
is an emerging growth company as defined in Rule 405 of the Securities Act of 1933 (§230.405 of this chapter) or Rule 12b-2
of the Securities Exchange Act of 1934 (§240.12b-2 of this chapter).
| Emerging growth company ¨ |
|
If an emerging growth company, indicate by
checkmark if the registrant has elected not to use the extended transition period for complying with any new or revised financial
accounting standards provided pursuant to Section 13(a) of the Exchange Act. ¨
Item 7.01. Regulation
FD Disclosure.
NightHawk
Biosciences, Inc. (the “Company”) will be making several presentations to investors over
the next several weeks. In connection with the presentations, the Company intends to discuss the investor presentation, which is furnished
as Exhibit 99.1 to this Current Report on Form 8-K.
The information in this Item
7.01 and in the investor presentation furnished as Exhibit 99.1 to this Current Report on Form 8-K shall not be deemed to be “filed”
for purposes of Section 18 of the Securities Exchange Act of 1934, as amended, or otherwise subject to the liabilities of that section
or Sections 11 and 12(a)(2) of the Securities Act of 1933, as amended and shall not be incorporated by reference into any filing with
the U.S. Securities and Exchange Commission made by the Company, whether made before or after the date hereof, regardless of any general
incorporation language in such filing.
The investor presentation
furnished as Exhibit 99.1 to this Current Report on Form 8-K includes “safe harbor” language pursuant to the Private Securities
Litigation Reform Act of 1995, as amended, indicating that certain statements contained therein are “forward-looking” rather
than historical.
Item 8.01 Other Items
On January 16, 2024, the Company updated its
corporate presentation. The updated presentation highlights the Company’s focus on its contract development and manufacturing organization
(“CDMO”) business, which is operated by the Company’s subsidiary, Scorpius Biomanfacturing, Inc. (“Scorpius”).
Since inception, Scorpius has signed 12+ development and manufacturing contracts (representing over $20 million in potential revenue)
with customers that include premier biopharma and emerging biotech companies.
Item 9.01 Financial Statements and
Exhibits.
SIGNATURES
Pursuant to the requirements of the Securities
Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned hereunto duly authorized.
| Dated: January 16, 2024 |
NightHawk Biosciences, Inc. |
| |
|
| |
|
|
| |
By: |
/s/ Jeff Wolf |
| |
Name: |
Jeff Wolf |
| |
Title: |
Chief Executive Officer |
Exhibit 99.1

NightHawk Biosciences A rapidly growing, highly scalable biologics Contract Development & Manufacturing Organization (CDMO) Investor Presentation - January 2024 NYSE: NHWK

This presentation includes statements that are, or may be deemed, “forward - looking statements” within the meaning of the Private Securities Litigation Reform Act of 1995, as amended. In some cases, these forward - looking statements can be identified by the u se of forward - looking terminology, including the terms “believes”, “estimates”, “anticipates”, “expects”, “plans”, “intends”, “may” , “could”, “might”, “will”, “should”, “approximately” or, in each case, their negative or other variations thereon or comparable termino log y, although not all forward - looking statements contain these words. They appear in a number of places throughout this presentation and include statements regarding our intentions, beliefs, projections, outlook, analyses or current expectations, concerning, am ong other things, the demand for contract development & manufacturing organization services growing, our recognizing revenue in 2 024 from over $20M in Scorpius signed manufacturing contracts, expected high margins and long - term profitability, leveraging fixed costs as revenue continues to grow resulting in high margins and long - term profitability, the industry in which we operate and t he trends that may affect the industry or us. By their nature, forward - looking statements involve risks and uncertainties because they relate to events, competitive dynamics, and healthcare, regulatory and scientific developments and depend on the economic circumstances that may or may not occur in the future or may occur on longer or shorter timelines than anticipated. Although we believe that we have a reasonable basis for each forward - looking statement contained in this presentation, we caution you that forward - looking statements are not guarantees of future performance and that our actual results of operations, financial condition and liquidity, and the development of th e i ndustry in which we operate may differ materially from the forward - looking statements contained in this presentation as a result of, am ong other factors, the factors referenced in the “Risk Factors” section of our Annual Report on Form 10 - K for the year ended Decembe r 31, 2022, our quarterly reports on Form 10 - Q for the subsequent quarters and our other subsequent filings with the Securities an d Exchange Commission (collectively, our “SEC Filings”). In addition, even if results of operations, financial condition and li qui dity, and the development of the industry in which we operate are consistent with the forward - looking statements contained in this presentation, they may not be predictive of results or developments in future periods. Any forward - looking statements that we m ake in this presentation speak only as of the date of such statement, and we undertake no obligation to update such statements to reflect events or circumstances after the date of this presentation, except as required by law. Forward Looking Statements Forward Looking Statement

Full - Service CDMO Focused on Biologic Production for the Middle Market Scorpius seeks to differentiate itself from competitors with a comprehensive mix of service offerings and a nimble, “boutique” approach to project planning and execution

Investment Highlights x Growing demand for Contract Development & Manufacturing Organization (CDMO) services and significant shortage of dedicated clinical - scale manufacturing capacity within the industry x Pure - play biologics CDMO offering a comprehensive range of services from process and analytical development through clinical - stage and small - scale commercial cGMP manufacturing x Began 2023 with $3M signed contracts, which has grown to over $20M in signed contracts , with recognized revenue from a substantial number of these contracts expected in 2024 x Installed capacity to support a large and growing pipeline and backlog that is being driven by new customer acquisition and existing customer/program expansion x Customers include many premier biopharma and emerging biotech companies , as well as leading research institutions x Ability to leverage fixed costs as revenue continues to grow; expected to result in high margins and long - term profitability Investment Highlights

Refocused and Recharged Recently divested non - core assets and contractual obligations to focus on Scorpius BioManufacturing operations » Terminated major in - house research activities » Divested non - core assets » Slashed non - core liabilities and burn rate These efforts have eliminated approximately $40M of contractual obligations and reduced operating expenses by over $13M per year Now laser - focused on driving revenue and cash flow through our Scorpius operations

Increased Biologic Demand Driving Rapid Growth in CDMO Segment Large Molecule Drug Substance CDMO Market size is poised to grow from $10.86B in 2022 to $21B by 2030, growing at a CAGR of 8.6% in the forecast period (2023 - 2030)* * SkyQuest Large Molecule Industry Forecast, July 2023 Over the last decade, R&D spending has steadily shifted from its focus on small molecules to the growing biologics segment » This shift has resulted in significantly higher demand for pharma services providers to assist in advancing these large molecules through the clinic » An increasing number of FDA approvals for biologics - based drugs has further validated interest in the space » It takes years of investment to establish complex facilities with talented teams, strong quality management and operating procedures up to regulatory standards » Already - established CDMOs, like Scorpius, are well - positioned to develop relationships with biotech clients ahead of future competition **Coherent Market Insights, July 2021, Biologics Market Analysis.

Business History Nov ’20: Scorpius BioManufacturing is founded Aug ’21: Broke ground on San Antonio, TX facility 2H ’22: Onboarded key execs and industry experts to build out management team Q4 ’22: Onboarded first client for microbial commercial contract Oct ’22: Opened San Antonio facility 2H ‘21 1H ‘22 2H ‘22 1H ‘23 2H ‘23 2H ‘20 1H ‘21 In the two years since breaking ground, Scorpius has generated revenue and is currently executing on a diverse pipeline of CDMO contracts Q1’24: Substantial pipeline with preparation for first GMP batches underway Worked with industry - leading advisors to design and build state - of - the - art, co - located mammalian and microbial facilities 1H ‘24 Q1’24: Over $20M in signed contracts with substantial revenue from these contracts anticipated in 2024 Since inception, Scorpius has signed 12+ development and manufacturing agreements with various pharma and biotech clients

Scorpius Snapshot Clinical - scale biomanufacturing in San Antonio, TX located in the heart of a thriving innovation district Highly skilled scientific and manufacturing leadership team US - sourced supply chain with US and international customers In - house process development and analytical services including Microbial fermentation, MAbs , Cell Therapy products

San Antonio Facility Overview Expertise in Mammalian and Microbial Manufacturing 40,000+ sq. ft. footprint includes – Brand new, state - of - the - art 40,000 sq. ft. facility with: • ~5,400 sq. ft. Mammalian / Cell Therapy manufacturing space • ~3,200 sq. ft. Microbial manufacturing space • ~2,000 sq. ft. of analytical development / QC space • ~2,000 sq. ft. of process development space – Additional 20,000 sq. ft. warehouse space – Large molecule manufacturing capabilities span preclinical through mid - late clinical scale and small - scale commercial: • Mammalian (250 mL development – 500 L manufacturing) • Microbial (250 mL development – 200 L manufacturing) – Clinical - scale development for third - party clients started in Oct ’22 – First client GMP batches planned for Q1 ’2 4 – 80+ employees, including a veteran management team with deep experience and tenure in the CDMO space
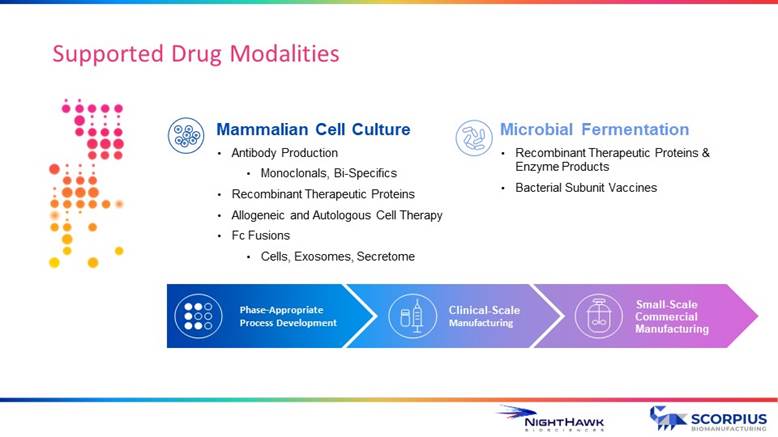
Supported Drug Modalities Phase - Appropriate Process Development Clinical - Scale Manufacturing Small - Scale Commercial Manufacturing Mammalian Cell Culture • Antibody Production • Monoclonals, Bi - Specifics • Recombinant Therapeutic Proteins • Allogeneic and Autologous Cell Therapy • Fc Fusions • Cells, Exosomes, Secretome Microbial Fermentation • Recombinant Therapeutic Proteins & Enzyme Products • Bacterial Subunit Vaccines

Financial Profile Robust Pipeline Supported by Streamlined Marketing Approach » Since inception, Scorpius has signed 12+ development and manufacturing agreements , establishing strong brand recognition » Over $20M in signed contracts with substantial revenue from these contracts anticipated in 2024 » Rapidly expanding pipeline with several near - term opportunities » ~$32M of direct facility investment (PP&E, etc.) » $50M+ revenue/ yr capacity in current San Antonio facility configuration, with substantial expansion possibilities

Balance Sheet Highlights » Over $20 million of PP&E acquired without term debt (as of 09/30/23) » No term debt » ROU assets and liabilities represent all Scorpius facilities ($22M) and leased specialized equipment ($2.5M) (as of 09/30/23) » Capital investment into facility and equipment completed in Q3, 2023 » Clean capital structure » ~26.1 million common shares outstanding (as of 01/15/24) » No warrants outstanding (as of 01/15/24)

2024 Goals Plans to grow market share through several initiatives in 2024: » Deliver seamless execution on signed manufacturing contracts » Expand pipeline with target biotech, pharma, and research customers » Streamline operations to deliver at scale » Bolster and strengthen in - house development and analytical services » Develop talent and culture, creating a team that can win long - term Focused on driving revenue and cash flow

Leadership Team Joe Payne VP, Quality & Regulatory 20+ Years Of Experience Alcami , Tergus , Teva Brian O’Mara VP, Process Sciences 20+ Years Of Experience Ambrx , Bristol - Myers Squibb, Genencor, Wyeth Stephan Kutzer Senior Advisor 25+ Years Of Experience Alcami , Lonza Matt LeClair VP, Manufacturing Operations 30+ Years Of Experience Abzena, Takeda Steve Lavezoli VP, Business Development 20+ Years Of Experience Catalent, W.L. Gore Bill Ostrander Chief Financial Officer 20+ Years Of Experience KBI Jeff Wolf Founder & CEO 30+ Years Of Experience Avigen , TyRx Pharma, EluSys Tx Ania Szymanska Site Quality Head 25+ Years Of Experience Opex Tx, Bellicum , Marker Tx

Summary x Growing demand for Contract Development & Manufacturing Organization (CDMO) services and significant shortage of dedicated clinical - scale manufacturing capacity within the industry x Scorpius began 2023 with $3M of signed manufacturing contracts, which has grown to over $20M in signed manufacturing contracts, with recognized revenue from a substantial number of these contracts expected in 2024 x Customers include many premier pharma and biotech companies, as well as leading research institutions x Recently divested non - core assets to focus on CDMO operations; eliminated approximately $40M of contractual obligations and reduced operating expenses by over $13M per year x Ability to leverage fixed costs as revenue continues to grow; expected to result in higher margins and long - term profitability

A Full - Service CDMO Focused on Biologic Production for the Middle Market
v3.23.4
Cover
|
Jan. 16, 2024 |
| Document Type |
8-K
|
| Amendment Flag |
false
|
| Document Period End Date |
Jan. 16, 2024
|
| Entity File Number |
001-35994
|
| Entity Registrant Name |
NightHawk Biosciences, Inc.
|
| Entity Central Index Key |
0001476963
|
| Entity Tax Identification Number |
26-2844103
|
| Entity Incorporation, State or Country Code |
DE
|
| Entity Address, Address Line One |
627
Davis Drive
|
| Entity Address, Address Line Two |
Suite
300
|
| Entity Address, City or Town |
Morrisville
|
| Entity Address, State or Province |
NC
|
| Entity Address, Postal Zip Code |
27560
|
| City Area Code |
(919)
|
| Local Phone Number |
240-7133
|
| Written Communications |
false
|
| Soliciting Material |
false
|
| Pre-commencement Tender Offer |
false
|
| Pre-commencement Issuer Tender Offer |
false
|
| Entity Emerging Growth Company |
false
|
| Common Stock [Member] |
|
| Common Stock |
Common Stock, $0.0002 par value per share
|
| Trading Symbol |
NHWK
|
| Security Exchange Name |
NYSEAMER
|
| Common Stock Purchase Rights [Member] |
|
| Common Stock |
Common Stock Purchase Rights
|
| Trading Symbol |
None
|
| Security Exchange Name |
NYSEAMER
|
| X |
- DefinitionBoolean flag that is true when the XBRL content amends previously-filed or accepted submission.
| Name: |
dei_AmendmentFlag |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionFor the EDGAR submission types of Form 8-K: the date of the report, the date of the earliest event reported; for the EDGAR submission types of Form N-1A: the filing date; for all other submission types: the end of the reporting or transition period. The format of the date is YYYY-MM-DD.
| Name: |
dei_DocumentPeriodEndDate |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:dateItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe type of document being provided (such as 10-K, 10-Q, 485BPOS, etc). The document type is limited to the same value as the supporting SEC submission type, or the word 'Other'.
| Name: |
dei_DocumentType |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:submissionTypeItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionAddress Line 1 such as Attn, Building Name, Street Name
| Name: |
dei_EntityAddressAddressLine1 |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionAddress Line 2 such as Street or Suite number
| Name: |
dei_EntityAddressAddressLine2 |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- Definition
+ References
+ Details
| Name: |
dei_EntityAddressCityOrTown |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionCode for the postal or zip code
| Name: |
dei_EntityAddressPostalZipCode |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionName of the state or province.
| Name: |
dei_EntityAddressStateOrProvince |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:stateOrProvinceItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionA unique 10-digit SEC-issued value to identify entities that have filed disclosures with the SEC. It is commonly abbreviated as CIK. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b-2
| Name: |
dei_EntityCentralIndexKey |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:centralIndexKeyItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionIndicate if registrant meets the emerging growth company criteria. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b-2
| Name: |
dei_EntityEmergingGrowthCompany |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionCommission file number. The field allows up to 17 characters. The prefix may contain 1-3 digits, the sequence number may contain 1-8 digits, the optional suffix may contain 1-4 characters, and the fields are separated with a hyphen.
| Name: |
dei_EntityFileNumber |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:fileNumberItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionTwo-character EDGAR code representing the state or country of incorporation.
| Name: |
dei_EntityIncorporationStateCountryCode |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:edgarStateCountryItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe exact name of the entity filing the report as specified in its charter, which is required by forms filed with the SEC. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b-2
| Name: |
dei_EntityRegistrantName |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionThe Tax Identification Number (TIN), also known as an Employer Identification Number (EIN), is a unique 9-digit value assigned by the IRS. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b-2
| Name: |
dei_EntityTaxIdentificationNumber |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:employerIdItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionLocal phone number for entity.
| Name: |
dei_LocalPhoneNumber |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:normalizedStringItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionBoolean flag that is true when the Form 8-K filing is intended to satisfy the filing obligation of the registrant as pre-commencement communications pursuant to Rule 13e-4(c) under the Exchange Act. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 13e
-Subsection 4c
| Name: |
dei_PreCommencementIssuerTenderOffer |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionBoolean flag that is true when the Form 8-K filing is intended to satisfy the filing obligation of the registrant as pre-commencement communications pursuant to Rule 14d-2(b) under the Exchange Act. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 14d
-Subsection 2b
| Name: |
dei_PreCommencementTenderOffer |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionTitle of a 12(b) registered security. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection b
| Name: |
dei_Security12bTitle |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:securityTitleItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionName of the Exchange on which a security is registered. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Number 240
-Section 12
-Subsection d1-1
| Name: |
dei_SecurityExchangeName |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:edgarExchangeCodeItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionBoolean flag that is true when the Form 8-K filing is intended to satisfy the filing obligation of the registrant as soliciting material pursuant to Rule 14a-12 under the Exchange Act. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Exchange Act
-Section 14a
-Number 240
-Subsection 12
| Name: |
dei_SolicitingMaterial |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionTrading symbol of an instrument as listed on an exchange.
| Name: |
dei_TradingSymbol |
| Namespace Prefix: |
dei_ |
| Data Type: |
dei:tradingSymbolItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- DefinitionBoolean flag that is true when the Form 8-K filing is intended to satisfy the filing obligation of the registrant as written communications pursuant to Rule 425 under the Securities Act. Reference 1: http://www.xbrl.org/2003/role/presentationRef
-Publisher SEC
-Name Securities Act
-Number 230
-Section 425
| Name: |
dei_WrittenCommunications |
| Namespace Prefix: |
dei_ |
| Data Type: |
xbrli:booleanItemType |
| Balance Type: |
na |
| Period Type: |
duration |
|
| X |
- Details
| Name: |
us-gaap_StatementClassOfStockAxis=us-gaap_CommonStockMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
| X |
- Details
| Name: |
us-gaap_StatementClassOfStockAxis=NHWK_CommonStockPurchaseRightsMember |
| Namespace Prefix: |
|
| Data Type: |
na |
| Balance Type: |
|
| Period Type: |
|
|
Nighthawk Biosciences (AMEX:NHWK)
Historical Stock Chart
From Mar 2024 to Apr 2024
Nighthawk Biosciences (AMEX:NHWK)
Historical Stock Chart
From Apr 2023 to Apr 2024