MEI Pharma to Present at the Stifel 2023 Healthcare Conference
November 08 2023 - 8:00AM
Business Wire
MEI Pharma, Inc. (Nasdaq: MEIP), a clinical-stage pharmaceutical
company focused on advancing new therapies for cancer, today
announced it will participate in the Stifel 2023 Healthcare
Conference. David Urso, president and chief executive officer, will
present a company overview and business update on Wednesday,
November 15 at 8:35 AM Eastern Time.
Investors and the general public are invited to listen to a live
webcast of the session through the "Investors and Media" section on
www.meipharma.com. A replay of the webcast will be made available
following the event.
About MEI Pharma
MEI Pharma, Inc. (Nasdaq: MEIP) is a clinical-stage
pharmaceutical company committed to developing novel and
differentiated cancer therapies. We build our pipeline by acquiring
promising cancer agents and creating value in programs through
development, strategic partnerships, out-licensing and
commercialization, as appropriate. Our approach to oncology drug
development is to evaluate our drug candidates in combinations with
standard-of-care therapies to overcome known resistance mechanisms
and address clear medical needs to provide improved patient
benefit. The drug candidate pipeline includes voruciclib, an oral
cyclin-dependent kinase 9 ("CDK9") inhibitor, and ME-344, an
intravenous small molecule mitochondrial inhibitor targeting the
oxidative phosphorylation pathway. For more information, please
visit www.meipharma.com. Follow us on X (formerly Twitter)
@MEI_Pharma and on LinkedIn.
Forward-Looking Statements
Certain information contained in this press release that are not
historical in nature are “forward-looking statements” within the
meaning of the “safe harbor” provisions of the Private Securities
Litigation Reform Act of 1995 including, without limitation,
statements regarding: the potential, safety, efficacy, and
regulatory and clinical progress of our product candidates,
including the anticipated timing for initiation of clinical trials
and release of clinical trial data and our expectations surrounding
potential regulatory submissions, approvals and timing thereof, our
business strategy and plans; the sufficiency of our cash, cash
equivalents and short-term investments to fund our operations; and
our ability to fund future capital returns. You should be aware
that our actual results could differ materially from those
contained in the forward-looking statements, which are based on
management’s current expectations and are subject to a number of
risks and uncertainties, including, but not limited to our failure
to successfully commercialize our product candidates; the
availability or appropriateness of utilizing the FDA’s accelerated
approval pathway for our product candidates; final data from our
pre-clinical studies and completed clinical trials may differ
materially from reported interim data from ongoing studies and
trials; costs and delays in the development and/ or FDA approval,
or the failure to obtain such approval, of our product candidates;
uncertainties or differences in interpretation in clinical trial
results; uncertainty regarding the impact of rising inflation and
the increase in interest rates as a result; potential economic
downturn; activist investors; our inability to maintain or enter
into, and the risks resulting from, our dependence upon
collaboration or contractual arrangements necessary for the
development, manufacture, commercialization, marketing, sales and
distribution of any products; competitive factors; our inability to
protect our patents or proprietary rights and obtain necessary
rights to third party patents and intellectual property to operate
our business; our inability to operate our business without
infringing the patents and proprietary rights of others; general
economic conditions; the failure of any products to gain market
acceptance; our inability to obtain any additional required
financing; technological changes; government regulation; changes in
industry practice; and one-time events. We do not intend to update
any of these factors or to publicly announce the results of any
revisions to these forward-looking statements. Under U.S. law, a
new drug cannot be marketed until it has been investigated in
clinical studies and approved by the FDA as being safe and
effective for the intended use.
View source
version on businesswire.com: https://www.businesswire.com/news/home/20231108897352/en/
David A. Walsey MEI Pharma Tel: 858-369-7104
investor@meipharma.com
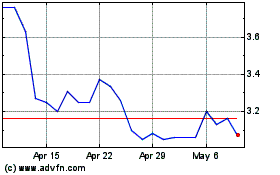
MEI Pharma (NASDAQ:MEIP)
Historical Stock Chart
From Mar 2024 to Apr 2024
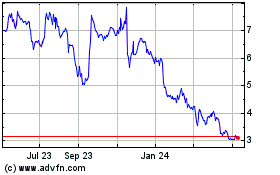
MEI Pharma (NASDAQ:MEIP)
Historical Stock Chart
From Apr 2023 to Apr 2024