Aurinia Completes Licensing Deal with Merck Animal Health for Its Nanomicellar Formulation of Voclosporin for the Treatment o...
April 17 2017 - 4:02PM
Business Wire
Company retains rights to all human
usesNovel topical formulation expands indication potential
for voclosporin
Aurinia Pharmaceuticals Inc. (NASDAQ:AUPH/TSX:AUP) (“Aurinia” or
the “Company”) today announced that it has signed a Definitive
Agreement (“the Agreement”) granting Merck Animal Health, known as
MSD Animal Health outside of the United States and Canada,
worldwide rights to develop and commercialize Aurinia’s patented
nanomicellar voclosporin ophthalmic solution (VOS) for the
treatment of dry eye syndrome in dogs.
Under the terms of this agreement Aurinia will receive an
upfront payment and is eligible to receive further payments based
on certain development and sales milestones. Furthermore, Aurinia
will receive royalties based on global product sales. Merck Animal
Health will be responsible for the remaining clinical development
and commercialization of VOS for use in the animal health field,
while Aurinia retains all human health rights. The companies will
share in the final work product and any technical knowledge that
may be generated during the collaboration.
“This partnership with Merck Animal Health underscores our
long-standing belief that voclosporin has the potential to be
effectively used across a range of therapeutic areas, in addition
to its primary potential indication for the treatment of lupus
nephritis,” said Richard Glickman, Aurinia’s CEO and Chairman of
the Board. “In addition to enhancing dry eye treatment options in
the animal health field, VOS has a differentiated product profile
with long patent life that has the potential to compete in the
multi-billion dollar human prescription dry eye market. While this
ophthalmology project will continue to be advanced by Merck Animal
Health, the Aurinia clinical team will remain focused on our lupus
nephritis program, which is on track to begin enrollment for the
AURORA Phase III trial this quarter.”
Throughout the past year, Merck Animal Health has conducted
proof of concept research in dogs suffering from dry eye syndrome,
which affects one out of every 22 dogs. The early symptoms can be
easily missed by pet owners, leading to irreversible damage to a
dog’s vision and eventually resulting in blindness.
“VOS has the potential to address significant unmet medical
needs in ophthalmology, and the preliminary results of our canine
dry eye trial are very promising,” says Holger Lehmann, DVM, PhD ,
Associate Vice President, Drug Discovery, Merck Animal Health. “We
look forward to continuing our collaboration with Aurinia and
believe this partnership is a prime example of the synergies that
exist between human and animal health drug development for the
benefit of all of our patients.”
“Completed preclinical and human Phase Ib studies using
Aurinia’s nanomicellar VOS formulation have shown encouraging
results in terms of delivery of active drug to the target tissues
of the eye,” said Neil Solomons, MD, Chief Medical Officer of
Aurinia. “The nanomicellar formulation enables high concentrations
of voclosporin to be incorporated into a preservative-free solution
for local delivery to the ocular surface. This has been shown to
potentially improve efficacy, dosing frequency and tolerability
versus the current treatments for dry eye syndrome. We are excited
about the potential for topical ocular administration of
voclosporin utilizing this unique and proprietary nanomicellar drug
delivery technology.”
Aurinia is exploring all options to create value with its
proprietary nanomicellar ocular formulation of voclosporin in the
human health field including, but not limited to, further
development, out-licensing or divestiture while remaining focused
on the Phase III lupus nephritis program.
About Dry Eye in DogsKeratoconjunctivitis sicca (KCS or
dry eye) is a common eye disease of dogs. This disease is
characterized by inflammation of the lacrimal glands, resulting in
reduced or absent tear formation and secondary
bacterial conjunctivitis. If left untreated, damage to the
tear glands is irreversible, leading to painful dry eye, and often
leads to permanent blindness.
About Dry Eye in HumansDry Eye Syndrome (DES), also known
as keratoconjunctivitis sicca (KCS), is a multifactorial,
heterogeneous condition wherein the eyes have insufficient amount
of tears, which are necessary for normal eye function. Prevalence
estimates range from 14% to 33% of the general population, with
between 30 and 40 million people believed to be affected in the
U.S. It is one of the most common reasons for visits to
ophthalmologists. DES prevalence is likely to continue to grow as a
result of the ageing population, (risk of DES increases 35% each
decade after age 40), higher physician and patient awareness,
increasing visual tasking and worsening environmental
conditions.
About AuriniaAurinia is a clinical stage
biopharmaceutical company focused on developing and commercializing
therapies to treat targeted patient populations that are suffering
from serious diseases with a high unmet medical need. The company
is currently developing voclosporin, an investigational drug, for
the treatment of lupus nephritis (LN). The company is headquartered
in Victoria, BC and focuses its development efforts globally.
www.auriniapharma.com
About Merck Animal HealthFor over a century, Merck has
been a global health care leader working to help the world be well.
Merck Animal Health, known as MSD Animal Health outside the United
States and Canada, is the global animal health business unit of
Merck. Through its commitment to the Science of Healthier Animals™,
Merck Animal Health offers veterinarians, farmers, pet owners and
governments one of the widest range of veterinary pharmaceuticals,
vaccines and health management solutions and services. Merck Animal
Health is dedicated to preserving and improving the health,
well-being and performance of animals. It invests extensively in
dynamic and comprehensive R&D resources and a modern, global
supply chain. Merck Animal Health is present in more than 50
countries, while its products are available in some 150 markets.
For more information, visit www.merck-animal-health.com or
connect with us on LinkedIn, Facebook and Twitter at @MerckAH.
We seek safe harbor.
Forward Looking StatementsThis press release contains
forward-looking statements, including statements related to
Aurinia's nanomicellar formulation of voclosporin (VOS) for the
treatment of canine dry eye syndrome and for potential application
in human health. It is possible that such results or conclusions
may change based on further analyses of these data. Words such as
"plans," "intends," “may,” "will," "believe," and similar
expressions are intended to identify forward-looking statements.
These forward-looking statements are based upon Aurinia’s current
expectations. Forward-looking statements involve risks and
uncertainties. Aurinia’s actual results and the timing of events
could differ materially from those anticipated in such
forward-looking statements as a result of these risks and
uncertainties, which include, without limitation, the risk that
Aurinia’s analyses, assessment and conclusions regarding VOS set
forth in this release may change based on further analyses of such
data, and the risk that clinical studies for VOS may not lead to
regulatory approval. These and other risk factors are discussed
under "Risk Factors" and elsewhere in Aurinia’s Annual Information
Form for the year ended December 31, 2016 filed with Canadian
securities authorities and available at www.sedar.com and on Form
40-F with the U.S. Securities Exchange Commission and available at
www.sec.gov, each as updated by subsequent filings, including
filings on Form 6-K. Aurinia expressly disclaims any obligation or
undertaking to release publicly any updates or revisions to any
forward-looking statements contained herein to reflect any change
in Aurinia's expectations with regard thereto or any change in
events, conditions or circumstances on which any such statements
are based.
View source
version on businesswire.com: http://www.businesswire.com/news/home/20170417005838/en/
Aurinia PharmaceuticalsInvestors:Celia EconomidesHead of
IR &
Communicationsceconomides@auriniapharma.comorMedia:Christopher
HippolyteChristopher.hippolyte@inventivhealth.com
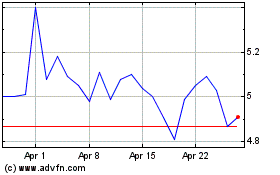
Aurinia Pharmaceuticals (NASDAQ:AUPH)
Historical Stock Chart
From Mar 2024 to Apr 2024
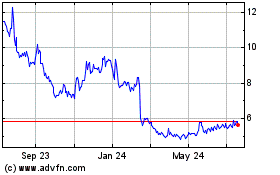
Aurinia Pharmaceuticals (NASDAQ:AUPH)
Historical Stock Chart
From Apr 2023 to Apr 2024