AstraZeneca's Orpathys Conditionally Approved by China for Lung Cancer Patients
June 23 2021 - 2:36AM
Dow Jones News
By Anthony O. Goriainoff
AstraZeneca PLC said Wednesday that China has conditionally
approved Orpathys for patients with non-small cell lung cancer and
mesenchymal-to-epithelial transition gene alterations.
The pharmaceutical company said that continued approval is
contingent upon the successful completion of a confirmatory trial,
and that it was based on positive results from a Phase 2 trial
conducted in the country.
"Orpathys demonstrated robust anti-tumor activity based on an
independent review of objective response rate in the trial's
primary endpoint and its disease control rate," the company
said.
AstraZeneca said the approval of Orpathys follows a priority
review designation by China's Center for Drug Evaluation of the
National Medical Products Administration, and represents the first
global regulatory approval for the MET tyrosine kinase
inhibitor.
Write to Anthony O. Goriainoff at
anthony.orunagoriainoff@dowjones.com
(END) Dow Jones Newswires
June 23, 2021 02:36 ET (06:36 GMT)
Copyright (c) 2021 Dow Jones & Company, Inc.
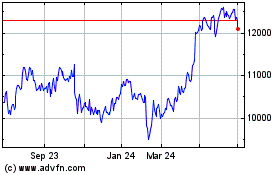
Astrazeneca (LSE:AZN)
Historical Stock Chart
From Apr 2024 to May 2024
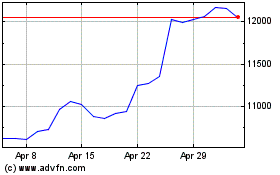
Astrazeneca (LSE:AZN)
Historical Stock Chart
From May 2023 to May 2024